Introduction
Malignant mesothelioma (MM) is a relatively rare but
highly aggressive malignant neoplasm arising from mesothelial cells
of the pleura, peritoneum, pericardium, and tunica vaginalis. It is
well-correlated with occupational and environmental asbestos
exposure. (1,2) The incidence of MM has increased in many
countries; (3) in Japan, mortality
due to MM has increased since the 1990s, and is predicted to peak
in the 2030s (4).
Epithelioid mesothelioma (EM) must be differentiated
from reactive mesothelial hyperplasia (RMH), which is a
non-neoplastic condition frequently caused by pleuritis,
peritonitis, or serosal invasion of other cancers. Due to the close
resemblance of EM to RMH, differentiation by routine histological
observation alone can be challenging.
Various established and novel immunohistochemical
markers have been utilized to distinguish EM from other
malignancies (5–8) and RMH (6,9–17) Multiple potential immunohistochemical
markers, including Ki-67, desmin, epithelial membrane antigen
(EMA), p53, glucose transporter 1, insulin-like growth factor 2
messenger RNA binding protein-3 and BRCA1-associated protein 1
(BAP1) have been evaluated. However, despite the use of these
immunohistochemical markers, the distinction between EM and RMH
remains challenging in some cases.
Recently, detection of p16 (CDKN2A)
homozygous deletion (p16 HD) using fluorescence in
situ hybridization (FISH) has been used to differentiate MM
from RMH, with 100% specificity. However, the sensitivity of this
marker for pleural EM varies between 45 and 86%, while its
sensitivity for peritoneal EM ranges from 14 to 41% in different
laboratories (10,18–20). In
our unpublished experience, p16 HD (detected by FISH) was
present in 63.2% (12/19) of EM cases, but absent in all RMH cases
(0/20). Although the detection of p16 HD using FISH may be
considered highly specific, its sensitivity in differentiating EM
from RMH is not very high. In addition, FISH analysis cannot be
applied in all cases or in all pathology laboratories, given its
high cost and stringent experimental requirements.
We recently reported that phorbol
12-myristate-13-acetate-induced protein-1 (PMAIP-1; Noxa) and
baculoviral IAP repeat-containing 5 (BIRC5; Survivin) mRNA
expression levels are significantly higher in EM than in
non-neoplastic pleural tissue, and discussed the utility of
anti-Noxa antibody for the distinction between EM and RMH (21). However, the utility of Survivin IHC
for the differentiation of benign and malignant mesothelial
proliferation has not yet been assessed.
Here, we studied the utility of Survivin and Ki-67
expressions along with the loss of BAP1 expression in
distinguishing benign from malignant mesothelial proliferation.
Materials and methods
Patients and histological samples
We used formalin-fixed, paraffin-embedded (FFPE)
specimens from 78 patients with a definite histological diagnosis
of EM who had undergone thoracoscopic pleural biopsy,
pleurectomy/decortication, extrapleural pneumonectomy, or autopsy
between 2000 and 2016. FFPE histological samples from surgical
specimens obtained from 80 patients with a histological diagnosis
of RMH were obtained via thoracoscopic biopsy, laparoscopic biopsy,
or surgical resection between 2005 and 2016. These samples were
retrieved from the archives of the Department of Pathology at
Hiroshima University (Hiroshima, Japan). Each of the tumour
specimens was independently reviewed by three pathologists (K.K.,
V.J.A, and Y.T.), and all cases of mesothelioma were diagnosed
according to currently accepted World Health Organization
Histological Criteria (6,22).
The tissue samples were retrieved from the archive
of the Department of Pathology at Hiroshima University's Institute
of Biomedical and Health Sciences. The collection of tissue
specimens for this study was carried out in accordance with the
‘Ethics Guidelines for Human Genome/Gene Research’ enacted by the
Japanese Government. Ethical approval was obtained from the
institutional ethics review committee (Hiroshima University E-974).
All experimental procedures were in accordance with the with
ethical guidelines.
Immunohistochemical procedures
Immunohistochemical staining of sections from the
FFPE tissue samples was performed using Ventana BenchMark GX (Roche
Diagnostics, Basel, Switzerland). In brief, after deparaffinization
using EZ-Prep (Roche Diagnostics) and antigen retrieval using Cell
Conditioning 1 buffer at 95°C for 32 min, sections were incubated
with primary antibodies. The primary antibodies were anti-Survivin
(cat. no. AF886, polyclonal, dilution of 1:200; R&D systems,
Minneapolis, MN, USA), anti-BAP1 (C-4, dilution of 1:50; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), and anti-Ki-67 (MIB-1,
dilution of 1:25; Dako, Glostrup, Denmark). Incubation with
secondary antibodies and detection was performed using the Ventana
UltraView Universal DAB Detection kit.
Nuclear staining of Survivin, BAP1, and Ki-67 in EM
or RMH cells with the same or higher intensity than internal
positive controls was regarded as positive staining. Negative
staining of BAP1 was defined as completely absent nuclear staining
in the target cells in the presence of a positive internal control
such as lymphocytes or stromal cells. Although some cases had weak
cytoplasmic positivity for Survivin and BAP1, we have not included
cases with only cytoplasmic positivity for Survivin and BAP1 for
evaluation in this study. Immunoreactivity of Survivin and Ki-67
was evaluated using a labelling index (% of positive cells) in the
‘hot spot’ exhibiting the highest number of positive cells compared
to the rest of the lesion. We evaluated at least 100 (maximum 500)
EM or RMH cells in high power fields (×400). Counting of labelling
indices of Survivin and Ki-67 was performed by three pathologists
(K.K., V.J.A, and Y.T.) independently; the mean of three numbers
was then calculated.
Statistical analysis
Receiver operating characteristic (ROC) curve
analysis was performed to establish the cut-off values for the
Survivin and Ki-67 labelling indices. The cut-off points were
determined based on the Youden index. All statistical analyses were
performed using EZR (Saitama Medical Center, Jichi Medical
University, Saitama, Japan), a graphical user interface for R (The
R Foundation for Statistical Computing, Vienna, Austria). More
precisely, it is a modified version of R commander designed to add
statistical functions frequently used in biostatistics (23).
Sensitivity, specificity, positive predictive
values, negative predictive values, and diagnostic accuracies were
calculated for each marker and combinations of two markers.
Results
Survivin expression and cut-off
value
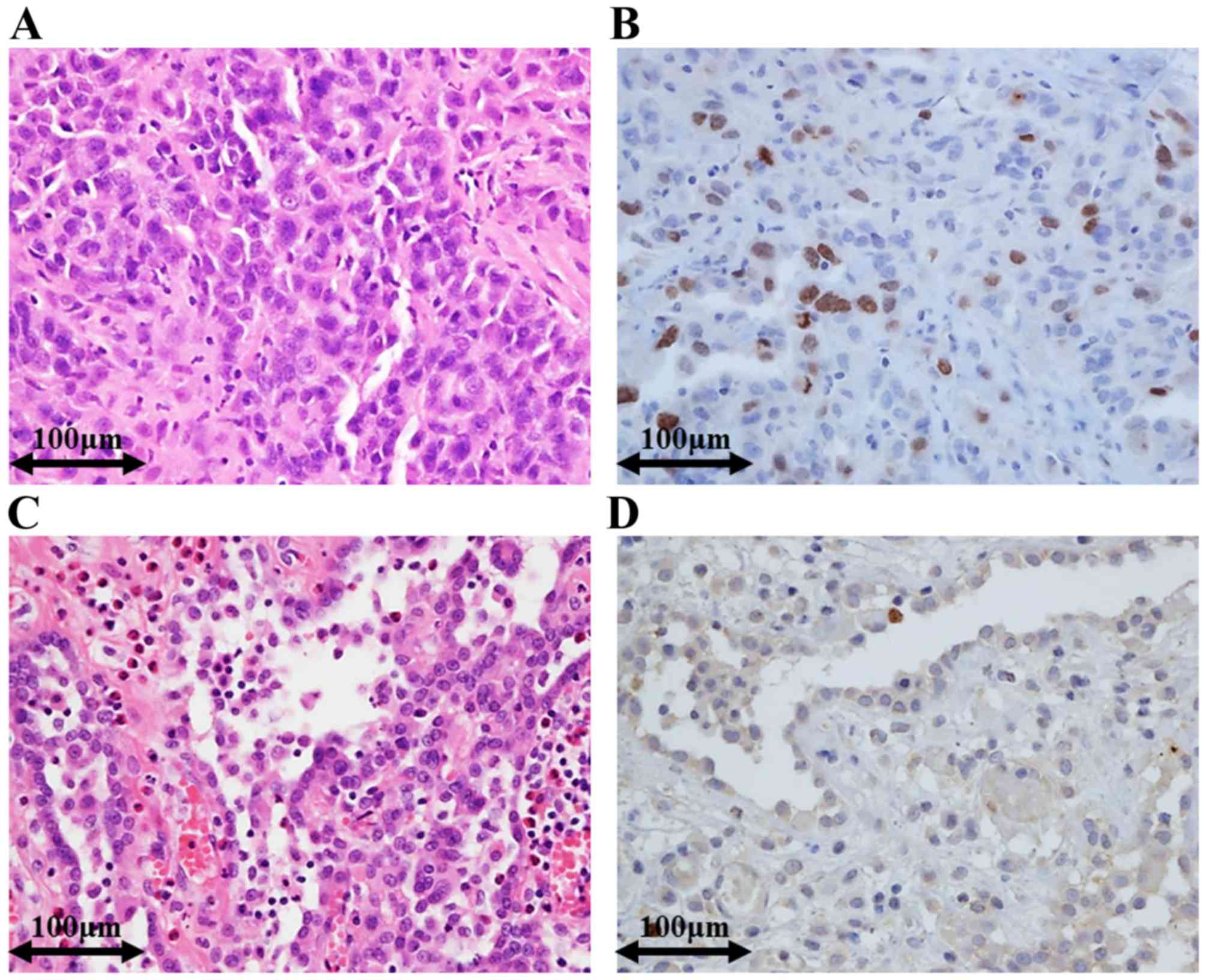
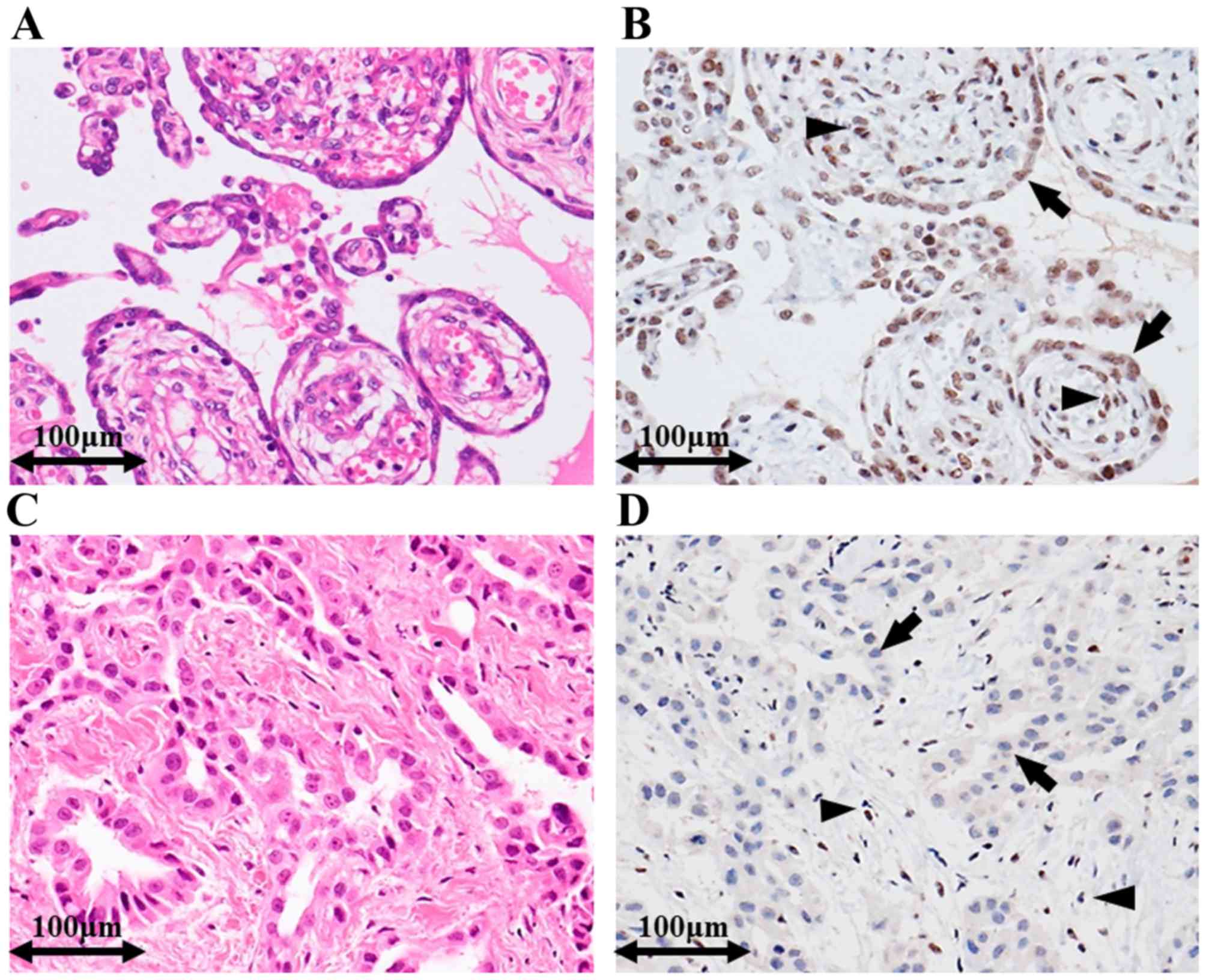
Representative immunohistochemical staining images
for EM and RMH are shown in Fig. 1.
Survivin expression was significantly higher in EM than in RMH. The
mean of the Survivin labelling indices in EM [mean, 9.3; range,
0–24.5, standard deviation (SD), 6.5] was significantly higher than
that in RMH (mean, 1.2; range, 0–4.0, SD, 1.2) (t-test, P-value
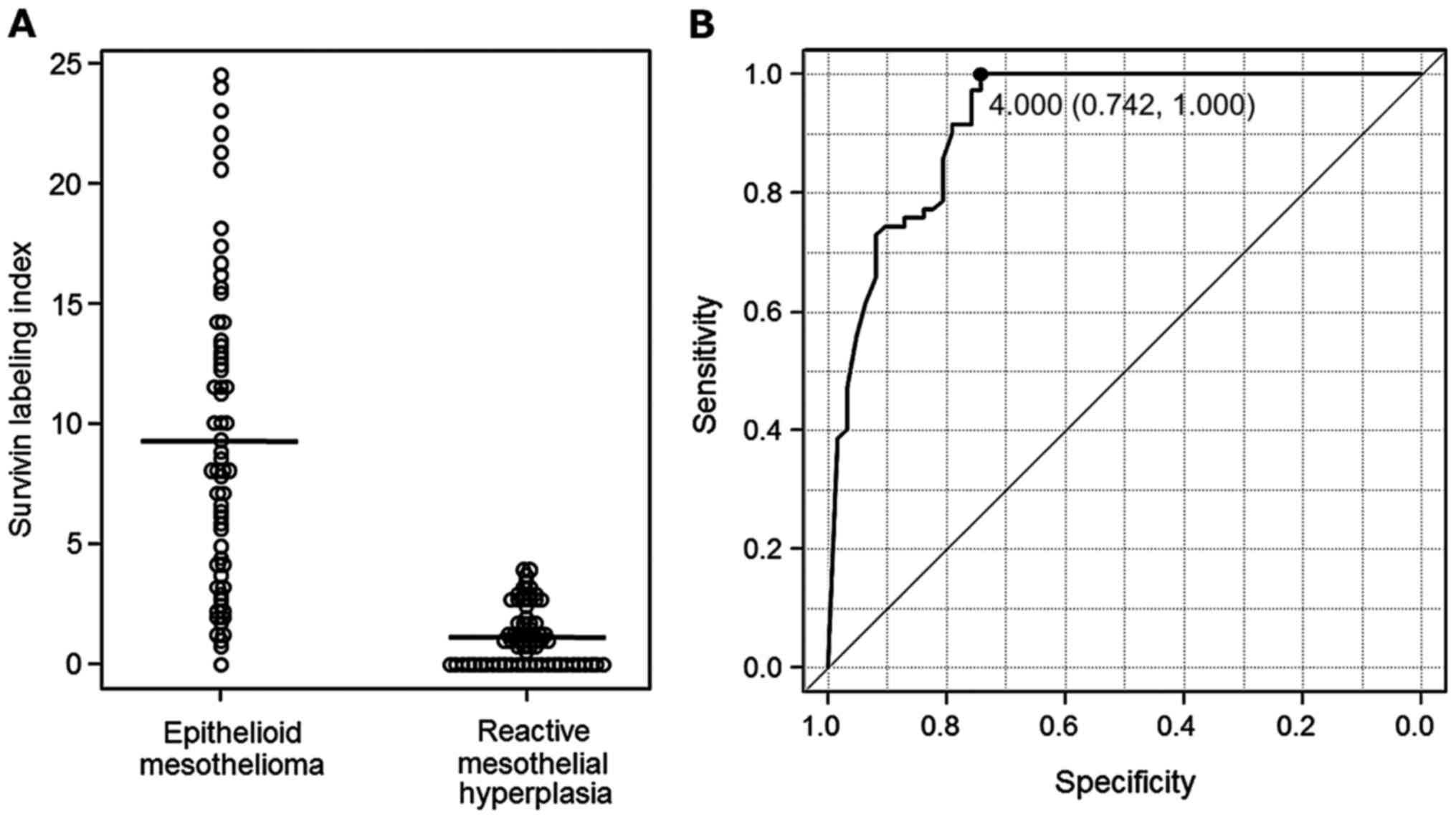
<0.001). Distributions of the Survivin labelling indices in EM
and RMH are shown in Fig. 2A.
The cut-off value for the Survivin IHC assay led by
the result of ROC analysis was 4.000 (Fig. 2B). Based on the ROC analysis, and in
consideration of convenience in practical pathological diagnosis,
we set the cut-off value for the Survivin IHC assay at 5%.
Immunoreactivity of Survivin was classified as negative (positivity
of less than 5% of the mesothelioma cells or non-neoplastic
mesothelial cells) or positive (positivity of over 5% of the
mesothelioma or mesothelial cells).
Forty-two of 62 (67.7%) EM cases were positive for
Survivin. In contrast, none of the RMH cases were positive for
Survivin (Table I).
 | Table I.Immunohistochemical findings of
Survivin, Ki-67, and BAP1 in epithelioid mesothelioma and reactive
mesothelial hyperplasia. |
Table I.
Immunohistochemical findings of
Survivin, Ki-67, and BAP1 in epithelioid mesothelioma and reactive
mesothelial hyperplasia.
|
| Epithelioid
mesothelioma | Reactive
mesothelial hyperplasia |
|---|
|
|
|
|
|---|
| Immunohistochemical
data | n (%) | Negative | Positive | n (%) | Negative | Positive |
|---|
| Survivin
expression | 42/62 (67.7) | 20 | 42 | 0/70 | 70 | 0 |
| Ki-67
expression | 57/67 (85.1) | 10 | 57 | 7/56 (12.5) | 49 | 7 |
| BAP1-loss | 49/74 (66.2) | 25 | 49 | 0/78 | 78 | 0 |
Ki-67 expression and cut-off
value
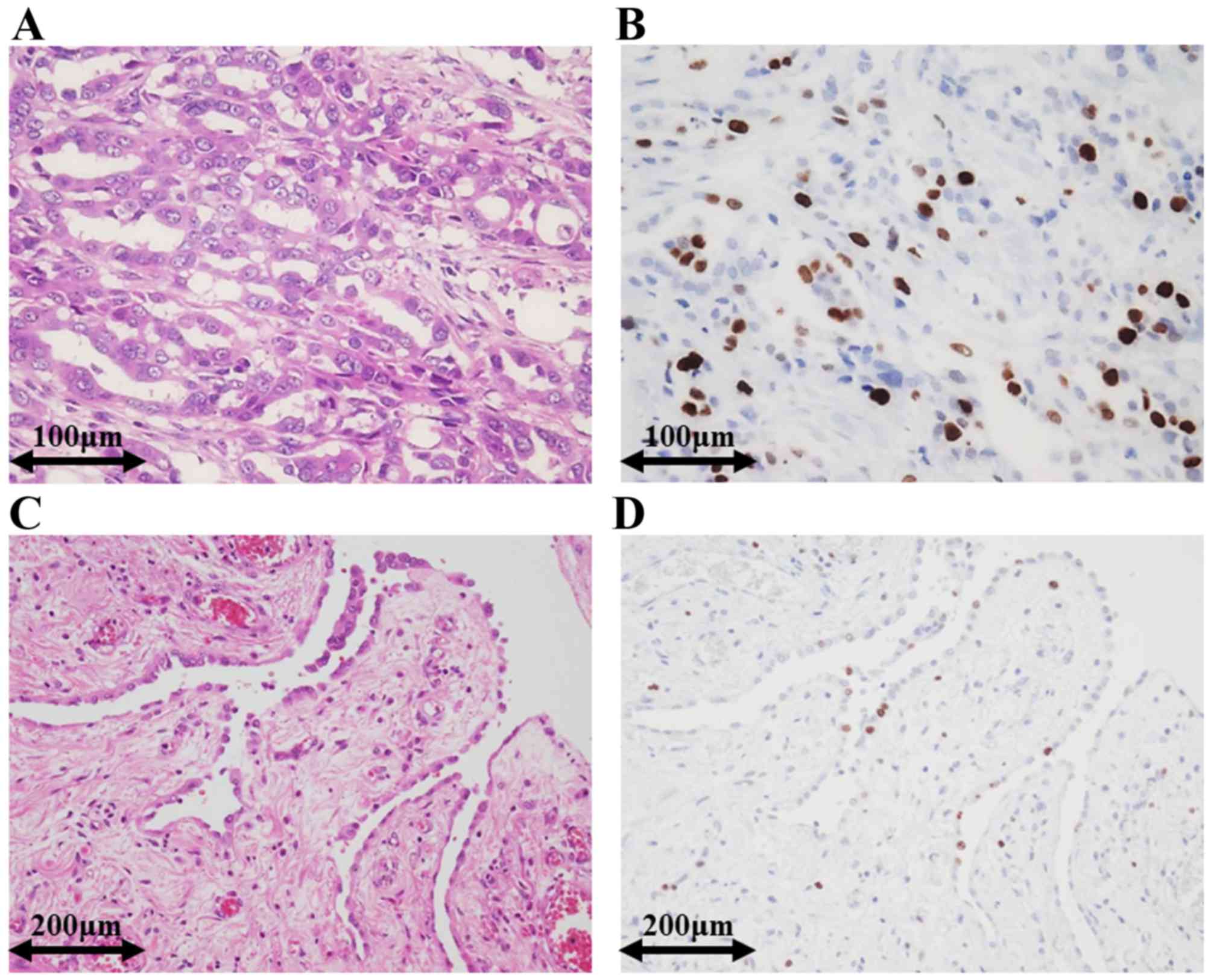
Representative immunohistochemical staining images
for EM and RMH are shown in Fig. 3.
Ki-67 expression was also significantly higher in EM than in RMH.
The mean of the Ki-67 labelling indices in EM (mean, 32.6; range,
1.0–90.0; SD, 22.1) was significantly higher than that in RMH
(mean, 3.5; range, 0–20.0, SD, 4.2) (t-test, P-value <0.001).
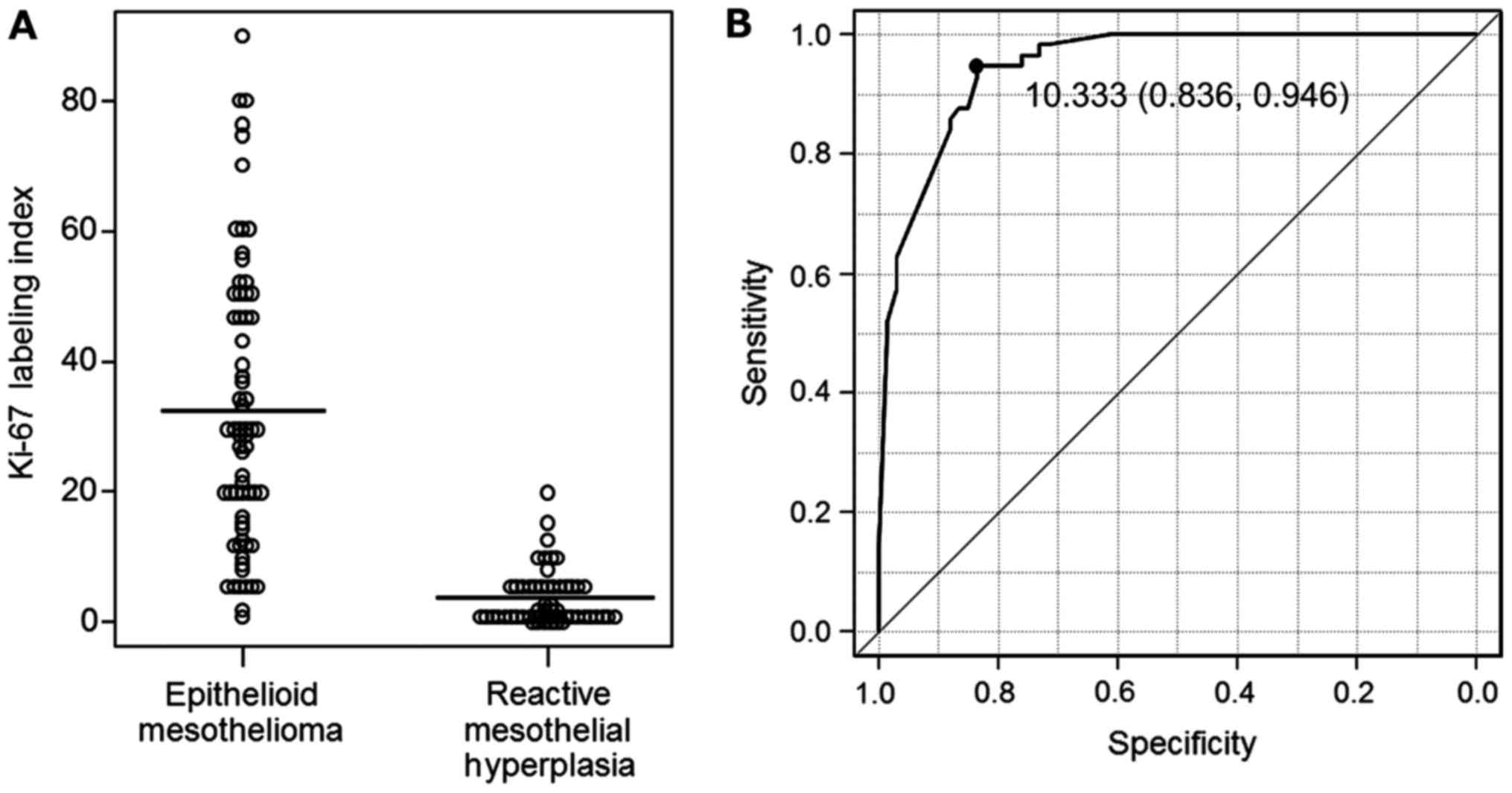
Distributions of the Ki-67 labelling indices in EM and RMH are
shown in Fig. 4A.
The cut-off value for the Ki-67 IHC assay led by the
result of ROC analysis was 10.333 (Fig.
4B). Based on the ROC analysis, and in consideration of
convenience in practical pathological diagnosis, we set the cut-off
value for the Ki-67 IHC assay at 10%. Immunoreactivity of Ki-67 was
classified as negative (positivity of less than 10% of the
mesothelioma cells or non-neoplastic mesothelial cells) or positive
(positivity of over 10% of the mesothelioma or mesothelial
cells).
Fifty-seven of 67 (85.1%) EM cases and 7 of 56
(12.5%) RMH cases were positive for Ki-67 (Table I).
BAP1 expression. Loss of nuclear BAP1 expression was
observed in 49 of 74 (66.2%) cases of EM (Table I). Almost all cases without BAP1
expression had a homogenous expression loss pattern. No
heterogeneous loss patterns were observed. In contrast, nuclear
BAP1 expression was preserved in all 78 RMH cases (Table I). Representative immunohistochemical
staining images for EM and RMH are shown in Fig. 5.
Utilities of each marker and
combinations of two markers
The sensitivity and specificity of each marker and
combinations of two markers for the distinction between EM and RMH
are shown in Table II. Among three
single markers and six combination patterns of two markers,
‘Survivin-positive and/or BAP1-loss’ finding showed the highest
diagnostic accuracy (95.3%).
 | Table II.Sensitivity, specificity, PPVs, NPVs
and diagnostic accuracies of each marker and combinations of two
markers for the differential diagnosis between epithelioid
mesothelioma and reactive mesothelial hyperplasia. |
Table II.
Sensitivity, specificity, PPVs, NPVs
and diagnostic accuracies of each marker and combinations of two
markers for the differential diagnosis between epithelioid
mesothelioma and reactive mesothelial hyperplasia.
| Immunohistochemical
findings | Sensitivity
(%) | Specificity
(%) | PPV (%) | NPV (%) | Accuracy (%) |
|---|
|
Survivin-positive | 67.7 | 100.0 | 100.0 | 77.8 | 84.8 |
| BAP1-loss | 66.2 | 100.0 | 100.0 | 75.7 | 83.6 |
| Ki-67-positive | 85.1 |
87.5 |
89.1 | 83.1 | 86.2 |
| Survivin-positive
and/or BAP1-loss | 89.8 | 100.0 | 100.0 | 92 | 95.3 |
| Both
Survivin-positive and BAP1-loss | 39.0 | 100 | 100.0 | 65.7 | 71.9 |
| Survivin-positive
and/or Ki-67-positive | 91.1 |
86.3 |
87.9 | 89.8 | 88.8 |
| Both
Survivin-positive and Ki-67-positive | 66.1 | 100.0 | 100.0 | 72.9 | 82.2 |
| BAP1-loss and/or
Ki-67-positive | 96.9 |
92.1 |
94.3 | 95.9 | 94.8 |
| Both BAP1-loss and
Ki-67-positive | 53.8 | 100 | 100.0 | 64.3 | 74.8 |
Discussion
Accurate histopathological differentiation between
MM and RMH is extremely important, not only for clinical
management, but also for the appropriate operation of the public
compensation system for victims of environmental and occupational
asbestos exposure and their dependents. To obtain a better marker
for EM, we evaluated the diagnostic utilities of Survivin, BAP1,
and Ki-67 in differentiating EM from RMH. We found that the
sensitivity and specificity of the nuclear Survivin labelling index
following the use of a properly determined cut-off value was
appropriate in distinguishing EM from RMH. The utility of Survivin
IHC for the differentiation between benign and malignant
mesothelial proliferation has not been reported to date. To the
best of our knowledge, this is the first report evaluating the
utility of Survivin IHC in differentiating EM from RMH.
Survivin is the smallest member of the inhibitor of
apoptosis (IAP) family, and is expressed highly in most human
foetal tissues and cancers. However, it is completely absent in
terminally-differentiated tissues. Survivin functions as a
regulator of both cell division and apoptosis. The function of
Survivin differs according to cellular localization. Cytosolic
Survivin is believed to function as an apoptotic suppressor, while
nuclear Survivin is postulated to regulate cell division (24). Overexpression of Survivin is
associated with tumour progression and poor prognosis in many types
of human malignancies, including MM (25,26). In
fact, several reports indicate that Survivin is a promising marker
for the diagnosis of malignant pleural effusion (27). Survivin has also been reported to be
associated with anti-tumour activity and outcomes of chemotherapy
in MM, and is a new therapeutic target for the treatment of MM
(28–30).
While the Survivin labelling indices of the EM cases
in our study were similar to those reported by Meerang et al
(25), they were significantly lower
than those reported by Hmeljak et al (median, 67; mean, 63;
range, 9.7–94.9; SD, 20.8) (26).
This discrepancy in Survivin expression may be due to differences
in staining technique, source of antibodies used for analysis, and
the quantification technique. In our study, we used fully automated
immunohistochemical staining utilising equipment from Roche for
reproducible results. We also used commercially available
antibodies from reputable sources and evaluated nuclear reactivity
alone. Evaluation of nuclear reactivity was reproducible and was
independently confirmed by 3 pathologists.
Several studies have determined that germline
mutations in the gene for BAP1 predispose individuals to
developing various tumours, including MM, cutaneous melanocytic
tumours, uveal melanoma, lung adenocarcinoma, and meningioma
(31). These studies suggest that
germline mutations in BAP1 result in a ‘tumour
predisposition syndrome’ linking BAP1 to many other cancers.
Somatic mutations in the BAP1 gene have also been relatively
frequently reported in MMs, uveal melanomas, and renal cell
carcinomas (31). BAP1 is encoded by
the BAP1 gene, which is located on the short arm of
chromosome 3 (3p21). BAP1 is a deubiquitinase targeting histones
and the host cell factor-1 transcriptional co-factor, and plays a
role in transcriptional regulation, chromatin modulation, cell
cycle regulation, and DNA repair (31,32).
Several different alterations in the BAP1 gene have been
described, including large deletions of exons leading to loss of
the N-terminal region, or to premature protein termination, focal
deletions, frameshift mutations due to insertions or deletions,
splice site mutations, and base substitutions leading to nonsense
and missense mutations. Frameshift mutations and missense and
nonsense substitutions are the most common sequence alterations.
Truncating mutations frequently result in loss of the nuclear
localization signal and/or the C-terminal protein-binding domain,
while missense mutations interfere with the ubiquitin hydrolase
function of BAP1 (31). As the
detection of these alterations in BAP1 has been made
possible in recent years using immunohistochemistry (IHC),
immunohistochemical detection of BAP1 loss has also been reported
to be useful in distinguishing MM from RMH. However, the
sensitivity of this assay in differentiating MM from RMH does not
exceed 70% (10–13). Several studies indicate that the loss
of nuclear BAP1 expression as assessed by IHC is closely correlated
with genetic alterations in BAP1 (33–35).
In the present study, the frequency of BAP1 loss in
EM was 66.2% (49/74), similar to those found in previous reports
(10–13). Recently, Hida et al reported a
focal heterogeneous BAP1 staining pattern in mesothelioma cases
(10). However, in our study, almost
all EM cases had either a uniform positive staining pattern or
completely negative staining for BAP1. There were some EM cases
that appeared to have focal staining for BAP1; however, careful
observation of these cases under high power magnification confirmed
that these focal positive cells were in fact inflammatory cells
infiltrating into the mesothelioma or stromal cells. We classified
such cases as cases with no loss of BAP1 expression. This may be
the reason for the observed heterogeneous BAP1 staining pattern in
mesothelioma. However, other reasons, such as differences in
staining techniques and improper processing of the tumour, may also
contribute to apparent differences between studies.
The specificity of a Survivin labelling index of
over 5% and a loss of BAP1 expression was 100%. However,
sensitivity of Survivin labelling index (67.7%) and loss of BAP1
expression (66.2%) alone are not sufficient for differential
diagnosis. Although diagnostic accuracies of Survivin (84.8%) and
BAP1 (83.6%) as single markers were inferior to that of EMA
(95.5%), (21) the diagnostic
accuracy of the combination of Survivin and BAP1 (Survivin-positive
and/or BAP1-loss) was 95.3%, which was almost similar to EMA.
Recently, Shinozaki-Ushiku et al proposed using a
combination of BAP1 and enhancer of zeste homolog 2 (EZH2)
expression to differentiate between MM from RMH; the sensitivity of
this combination was 90%, while the specificity was absolute
(36). The sensitivity (89.8%) and
specificity (100%) of the combination of Survivin and BAP1 IHC in
this study was comparable to those of previous reports (36).
A positive correlation between nuclear Survivin and
Ki-67 labelling indices was previously reported by Meerang et
al (25). We observed a similar
correlation between Survivin and Ki-67 labelling indices in our
study (data not shown). Although this correlation was present in
both EM and RMH, it was more conspicuous in EM. Ki-67 protein is
present during all active phases of the cell cycle (G1, S, G2, and
mitosis), but is absent in resting cells (G0). Therefore, Ki-67 is
well known as a so-called ‘proliferation marker’, and the Ki-67
labelling index is often correlated with the clinical course of
cancer (37,38). On the other hand, nuclear Survivin
plays important roles in the regulation of mitosis. Survivin
expression is found to be dominant only in the G2/M phase, and
Survivin is known to localize to components of the mitotic spindle
during the metaphase and anaphase of mitosis (39,40).
Therefore, both nuclear Survivin and Ki-67 may be considered
proliferation markers. We can thus explain both the high expression
of Survivin and Ki-67 in EM compared to RMH, and the positive
correlation between the nuclear Survivin and Ki-67 labelling
indices.
Although various studies have reported the
usefulness of Ki-67 IHC in differentiating EM from RMH, (14–17) it is
not routinely utilized for the confirmation of mesothelioma due to
its low sensitivity and specificity.
The sensitivity, specificity, and diagnostic
accuracy of Ki-67 (85.1, 87.5, and 86.2%, respectively) in this
study were almost the same or slightly higher compared with
previous reports (14,15,17). These
values were relatively high but not sufficient for differential
diagnosis by single marker. However, the diagnostic accuracy of the
combination of Ki-67 and BAP1 was 94.8%, which was almost the same
as that of the combination of Survivin and BAP1.
We evaluated the utility of Survivin, BAP1, and
Ki-67 IHC in distinguishing EM from RMH. Based on our results,
‘Survivin-positive and/or BAP1-loss’ finding strongly suggest EM,
therefore we recommend the use of a combination of Survivin and
BAP1. In addition, further evaluation of the Ki-67 labelling index
may be useful for accurate differential diagnosis.
Acknowledgements
The authors would like to thank the Technical Centre
of Hiroshima University for technical assistance. This study was
funded in part by the Japanese Ministry of Health, Labour, and
Welfare.
References
|
1
|
Robinson BW, Musk AW and Lake RA:
Malignant mesothelioma. Lancet. 366:397–408. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roggli VL, Sharma A, Butnor KJ, Sporn T
and Vollmer RT: Malignant mesothelioma and occupational exposure to
asbestos: A clinicopathological correlation of 1445 cases.
Ultrastruct Pathol. 26:55–65. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Delgermaa V, Takahashi K, Park EK, Le GV,
Hara T and Sorahan T: Global mesothelioma deaths reported to the
World Health Organization between 1994 and 2008. Bull World Health
Organ. 89:716–724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Murayama T, Takahashi K, Natori Y and
Kurumatani N: Estimation of future mortality from pleural malignant
mesothelioma in Japan based on an age-cohort model. Am J Ind Med.
49:1–7. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Churg A, Roggli V, Galateau-Salle F, Cagle
PhT, Gibbs AR, Hasleton PhS, Henderson DW, Vignaud JM, Inai K,
Praet M, et al: Tumours of the pleuraWHO Classification of Tumours
of the Lung, Pleura, Thymus and Heart. Travis WD, Brambilla E,
Burke AP, Marx A and Nicholson AG: IARC Press; Lyon: pp. 153–181.
2015
|
|
6
|
Husain AN, Colby T, Ordonez N, Krausz T,
Attanoos R, Beasley MB, Borczuk AC, Butnor K, Cagle PT, Chirieac
LR, et al: Guidelines for pathologic diagnosis of malignant
mesothelioma: 2012 update of the consensus statement from the
International Mesothelioma Interest Group. Arch Pathol Lab Med.
137:647–667. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ordóñez NG: Application of
immunohistochemistry in the diagnosis of epithelioid mesothelioma:
A review and update. Hum Pathol. 44:1–19. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kushitani K, Amatya VJ, Okada Y, Katayama
Y, Mawas AS, Miyata Y, Okada M, Inai K, Kishimoto T and Takeshima
Y: Utility and pitfall of immunohistochemistry in the differential
diagnosis between epithelioid mesothelioma and poorly
differentiated lung squamous cell carcinoma. Histopathology.
70:375–384. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Minato H, Kurose N, Fukushima M, Nojima T,
Usuda K, Sagawa M, Sakuma T, Ooi A, Matsumoto I, Oda M, et al:
Comparative immunohistochemical analysis of IMP3, GLUT1, EMA,
CD146, and desmin for distinguishing malignant mesothelioma from
reactive mesothelial cells. Am J Clin Pathol. 141:85–93. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hida T, Hamasaki M, Matsumoto S, Sato A,
Tsujimura T, Kawahara K, Iwasaki A, Okamoto T, Oda Y, Honda H and
Nabeshima K: BAP1 immunohistochemistry and p16 FISH results in
combination provide higher confidence in malignant pleural
mesothelioma diagnosis: ROC analysis of the two tests. Pathol Int.
66:563–570. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hwang HC, Sheffield BS, Rodriguez S,
Thompson K, Tse CH, Gown AM and Churg A: Utility of BAP1
immunohistochemistry and p16 (CDKN2A) FISH in the diagnosis of
malignant mesothelioma in effusion cytology specimens. Am J Surg
Pathol. 40:120–126. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McGregor SM, Dunning R, Hyjek E,
Vigneswaran W, Husain AN and Krausz T: BAP1 facilitates diagnostic
objectivity, classification, and prognostication in malignant
pleural mesothelioma. Hum Pathol. 46:1670–1678. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cigognetti M, Lonardi S, Fisogni S,
Balzarini P, Pellegrini V, Tironi A, Bercich L, Bugatti M, Rossi G,
Murer B, et al: BAP1 (BRCA1-associated protein 1) is a highly
specific marker for differentiating mesothelioma from reactive
mesothelial proliferations. Mod Pathol. 28:1043–1057. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kimura F, Okayasu I, Kakinuma H, Satoh Y,
Kuwao S, Saegusa M and Watanabe J: Differential diagnosis of
reactive mesothelial cells and malignant mesothelioma cells using
the cell proliferation markers minichromosome maintenance protein
7, geminin, topoisomerase II alpha and Ki-67. Acta Cytol.
57:384–390. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kimura F, Kawamura J, Watanabe J,
Kamoshida S, Kawai K, Okayasu I and Kuwao S: Significance of cell
proliferation markers (Minichromosome maintenance protein 7,
topoisomerase IIalpha and Ki-67) in cavital fluid cytology: Can we
differentiate reactive mesothelial cells from malignant cells?
Diagn Cytopathol. 38:161–167. 2010.PubMed/NCBI
|
|
16
|
Hasteh F, Lin GY, Weidner N and Michael
CW: The use of immunohistochemistry to distinguish reactive
mesothelial cells from malignant mesothelioma in cytologic
effusions. Cancer Cytopathol. 118:90–96. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Taheri ZM, Mehrafza M, Mohammadi F,
Khoddami M, Bahadori M and Masjedi MR: The diagnostic value of
Ki-67 and repp86 in distinguishing between benign and malignant
mesothelial proliferations. Arch Pathol Lab Med. 132:694–697.
2008.PubMed/NCBI
|
|
18
|
Churg A, Sheffield BS and Galateau-Salle
F: New markers for separating benign from malignant mesothelial
proliferations: Are we there yet? Arch Pathol Lab Med. 140:318–321.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hiroshima K, Wu D, Hasegawa M, Koh E,
Sekine Y, Ozaki D, Yusa T, Walts AE, Marchevsky AM, Nabeshima K, et
al: Cytologic differential diagnosis of malignant mesothelioma and
reactive mesothelial cells with fish analysis of p16. Diagn
Cytopathol. 44:591–598. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Walts AE, Hiroshima K, McGregor SM, Wu D,
Husain AN and Marchevsky AM: BAP1 immunostain and CDKN2A (p16) FISH
analysis: Clinical applicability for the diagnosis of malignant
mesothelioma in effusions. Diagn Cytopathol. 44:599–606. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kushitani K, Amatya VJ, Mawas AS, Miyata
Y, Okada M and Takeshima Y: Use of anti-noxa antibody for
differential diagnosis between epithelioid mesothelioma and
reactive mesothelial hyperplasia. Pathobiology. 83:33–40. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Travis WD, Brambilla E, Nicholson AG,
Yatabe Y, Austin JHM, Beasley MB, Chirieac LR, Dacic S, Duhig E,
Flieder DB, et al: The 2015 World Health Organization
Classification of Lung Tumors: Impact of genetic, clinical and
radiologic advances since the 2004 classification. J Thorac Oncol.
10:1243–1260. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kanda Y: Investigation of the freely
available easy-to-use software ‘EZR’ for medical statistics. Bone
Marrow Transplant. 48:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garg H, Suri P, Gupta JC, Talwar GP and
Dubey S: Survivin: A unique target for tumor therapy. Cancer Cell
Int. 16:492016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Meerang M, Bérard K, Friess M, Bitanihirwe
BK, Soltermann A, Vrugt B, Felley-Bosco E, Bueno R, Richards WG,
Seifert B, et al: Low merlin expression and high survivin labeling
index are indicators for poor prognosis in patients with malignant
pleural mesothelioma. Mol Oncol. 10:1255–1265. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hmeljak J, Erčulj N, Dolžan V, Pižem J,
Kern I, Kovač V, Cemažar M and Cör A: Is survivin expression
prognostic or predictive in malignant pleural mesothelioma?
Virchows Arch. 462:315–321. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen S, Wang Y, An L, Fei ZT and Li T: The
diagnostic value of survivin in malignant pleural effusion: A
meta-analysis. Clin Chim Acta. 441:142–147. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bertino P, Panigada M, Soprana E, Bianchi
V, Bertilaccio S, Sanvito F, Rose AH, Yang H, Gaudino G, Hoffmann
PR, et al: Fowlpox-based survivin vaccination for malignant
mesothelioma therapy. Int J Cancer. 133:612–623. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
De Cesare M, Cominetti D, Doldi V,
Lopergolo A, Deraco M, Gandellini P, Friedlander S, Landesman Y,
Kauffman MG, Shacham S, et al: Anti-tumor activity of selective
inhibitors of XPO1/CRM1-mediated nuclear export in diffuse
malignant peritoneal mesothelioma: The role of survivin.
Oncotarget. 6:13119–13132. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Goričar K, Kovač V, Franko A, Dodič-Fikfak
M and Dolžan V: Serum survivin levels and outcome of chemotherapy
in patients with malignant mesothelioma. Dis Markers.
2015:3167392015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Murali R, Wiesner T and Scolyer RA:
Tumours associated with BAP1 mutations. Pathology. 45:116–126.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Scheuermann JC, de Ayala Alonso AG, Oktaba
K, Ly-Hartig N, McGinty RK, Fraterman S, Wilm M, Muir TW and Müller
J: Histone H2A deubiquitinase activity of the Polycomb repressive
complex PR-DUB. Nature. 465:243–247. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bott M, Brevet M, Taylor BS, Shimizu S,
Ito T, Wang L, Creaney J, Lake RA, Zakowski MF, Reva B, et al: The
nuclear deubiquitinase BAP1 is commonly inactivated by somatic
mutations and 3p21.1 losses in malignant pleural mesothelioma. Nat
Genet. 43:668–672. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Testa JR, Cheung M, Pei J, Below JE, Tan
Y, Sementino E, Cox NJ, Dogan AU, Pass HI, Trusa S, et al: Germline
BAP1 mutations predispose to malignant mesothelioma. Nat Genet.
43:1022–1025. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yoshikawa Y, Sato A, Tsujimura T, Emi M,
Morinaga T, Fukuoka K, Yamada S, Murakami A, Kondo N, Matsumoto S,
et al: Frequent inactivation of the BAP1 gene in epithelioid-type
malignant mesothelioma. Cancer Sci. 103:868–874. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shinozaki-Ushiku A, Ushiku T, Morita S,
Anraku M, Nakajima J and Fukayama M: Diagnostic utility of BAP1 and
EZH2 expression in malignant mesothelioma. Histopathology.
70:722–733. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Scholzen T and Gerdes J: The Ki-67
protein: From the known and the unknown. J Cell Physiol.
182:311–322. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Brown DC and Gatter KC: Monoclonal
antibody Ki-67: Its use in histopathology. Histopathology.
17:489–503. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Altieri DC: Validating survivin as a
cancer therapeutic target. Nat Rev Cancer. 3:46–54. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kim JY, Chung JY, Lee SG, Kim YJ, Park JE,
Yoo KS, Yoo YH, Park YC, Kim BG and Kim JM: Nuclear interaction of
Smac/DIABLO with survivin at G2/M arrest prompts docetaxel-induced
apoptosis in DU145 prostate cancer cells. Biochem Biophys Res
Commun. 350:949–954. 2006. View Article : Google Scholar : PubMed/NCBI
|