Introduction
Gastric cancer (GC) is the fourth most common
malignant disease globally and is the second highest mortality rate
due to cancer globally (1). Owing to
the poor response to treatment observed in patients with
advanced-stage GC, the 5-year overall survival (OS) rate is in the
range of 25–30% worldwide (2).
Therefore, it is necessary to diagnose GC at an early stage.
Understanding the molecular mechanisms underlying GC development is
also essential for determining methods to inhibit tumor
progression.
The homeobox B13 (HOXB13) gene belongs to the HOX
family, which are known to function in encoding nuclear
transcription factors involved in establishing and maintaining
patterns of differentiation during development (3). The HOXB13 gene, which is located in
chromosomal region 17q21.2, encodes a 31-kDa protein (4). The 5′end of HOXB13 contains two CpG
islands, one in the promoter/exon1 and the other 4.5 kb upstream of
the transcription start site, suggesting that HOXB13 gene
expression may be controlled by DNA methylation (5). Mice with loss-of-function mutations in
HOXB13 exhibited an overgrown-tail phenotype, with increased cell
proliferation and decreased apoptosis in the tail; this evidence
indicated that HOXB13 inhibited proliferation and activated
programmed cell death (6).
Overexpression of HOXB13 inhibited prostate cell proliferation,
inducing G1-phase cell cycle arrest as mediated by downregulation
of T-cell factor-4 (TCF-4) expression (7). HOXB13 also downregulated the expression
of TCF-4 at the protein level, suppressing the growth of colorectal
cancer cells (8). HOXB13 also serves
diverse biological functions in embryonic development and
terminally differentiated tissue (9,10).
Many studies have identified HOXB13 as a candidate
tumor suppressor gene in several types of cancer, including
colorectal cancer (8), renal cancer
(11), melanoma (12) and breast cancer (13). Additionally, hypermethylation of the
HOXB13 gene promoter was a potential mechanism for decrease
expression (8,13–15). Other
studies, however, revealed that HOXB13 was overexpressed in
numerous types of tumor, potentially contributing to carcinogenesis
and tumor progression in prostate (16), breast (17), ovarian (18,19),
cervical (20) and oral cancer
(21). The HOXB13 germline variant
G84E mutation may be involved in promoting tumor progression, with
the function of HOXB13 depending on the tissue in which expressed
(22,23). Although previous studies have
demonstrated that HOXB13 is a contrasting biomarker for tumor
development in numerous types of cancer, the function of HOXB13 in
gastric cancer is unclear (8,11–21).
To understand the function of the HOXB13 gene in GC,
the present study detected the expression levels of HOXB13 mRNA in
gastric cancer tissues and corresponding non-malignant gastric
tissues and then evaluated the association between HOXB13 mRNA
expression and survival time. The methylation status of HOXB13 in
GC cell lines and GC tissues was also detected, and then the
association between its expression and DNA methylation was
assessed.
Materials and methods
Patients and tissue samples
The present study included 85 patients in total,
which included 45 males and 40 females. The average age was 61
years old and the age range was 35–75. All patients underwent
gastrectomy between September 2007 and August 2012 at the Fourth
Affiliated Hospital of China Medical University (Shenyang, China).
Gastric tissue specimens and corresponding nonmalignant gastric
tissues were collected following tumor excision during gastrectomy
and were diagnosed by at least two pathologists. Tumor
classification was performed according to the Tumor-Node-Metastasis
(TNM) grading system, seventh edition (24). Following this, patients were monitored
periodically and their tumor marker levels, including
carcinoembryonic antigen and carbohydrate antigen 19-9 assessed,
their blood tested, chest and abdominal computed tomography images
captured and gastroscopy performed. OS rates were defined as the
time from surgery to mortality or the last follow-up. The final
follow-up date was February 2016. The median duration of follow-up
was 26 months (range 5–90 months). The present study was approved
by the Research Ethics Committee of China Medical University
(Shenyang, China). All patients provided written informed consent
to participate in this research.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted using TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) from
tissues or cultured cells. cDNA was synthesized from RNA by using
an Expanding Reverse Transcriptase kit (Takara Bio, Inc., Otsu,
Japan) according to the manufacturer's protocol. qPCR was used to
detect the expression of HOXB13 mRNA in a reaction volume of 25 µl,
including 12.5 µl SYBR Green (Takara Bio, Inc.), 2 µl cDNA, 1 µl of
each primer and 8.5 µl diethyl pyrocarbonate water. The mixture was
incubated by the following program: 95°C for 30 sec, 40 cycles of
95°C for 5 sec, 60°C for 32 sec. The primers used were: HOXB13
forward, 5′-TGTTGCCAGGGAGAACAGAAC-3′ and reverse,
5′-CGCTGGAGTCTGCAAATGCT-3′ (25); and
β-actin (ACTB) forward, 5′-TGGCACCCAGCACAATGAA-3′ and reverse,
5′-CTAAGTCATAGTCCGCCTAGAAGCA-3′. For each PCR, diethyl
pyrocarbonate water was used as a negative control. The expression
level of HOXB13 mRNA was standardized to the ACTB mRNA expression
level, and data was quantified using the 2−ΔΔCq method
(26).
Gastric cells and culture
The SGC-7901, BGC-823, MGC-803, MKN-45 and AGS GC
cell lines, and the immortalized normal gastric GES-1 cell line (as
the control), were obtained from the Institute of Biochemistry and
Cell Biology, Chinese Academy of Sciences (Shanghai, China). Cells
were cultured in RPMI 1640 (Invitrogen; Thermo Fisher Scientific,
Inc.). Cell lines were cultured with 10% fetal bovine serum
(Thermo Fisher Scientific, Inc.) at 37°C in a humidified
incubator with 5% CO2.
Western blot analysis
Total protein in cultured cells and tissue specimens
was extracted using the Qproteome Mammalian Protein Prep kit
(Qiagen GmbH, Hilden, German) and protein concentration was
determined with a BCA Protein Assay kit (Bio-Rad, Milan, Italy).
Protein samples were denatured by boiling for 5 min and 30 µg
protein samples were electrophoresed by 12% SDS-PAGE. The protein
samples were then transferred to polyvinylidene fluoride membranes.
Following blocking with blocking buffer (5% skim milk in 50 mM
Tris-HCl, 200 mM NaCl and 0.05% Tween-20, pH 7.5) for 2 h at room
temperature, membranes were incubated overnight at 4°C with the
HOXB13 antibody (1:1,000; cat. no. ab28575) or ACTB antibody (cat.
no. ab6276; both Abcam, Cambridge, MA, USA). The next day, those
membranes were washed three times with PBS and incubated for 120
min at room temperature with a horseradish peroxidase conjugated
goat anti-rabbit immunoglobulin G secondary antibody (cat. no.
A0545; 1:1,000; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany).
Following washing, the immunoreactive protein bands were visualized
using an Electrochemiluminescence Detection kit (cat. no. P0018;
Beyotime Institute of Biotechnology, Haimen, China). Each
experiment was repeated at least three times.
DNA extraction and
methylation-specific PCR (MSP)
Total DNA was extracted from MKN-45 and tissue
specimens using the Takara Universal Genomic DNA Extraction kit
Ver.3.0 (Takara Bio, Inc.), according to the manufacturer's
protocol. The EZ DNA Methylation-Gold kit (Zymo Research, Irvine,
CA, USA) was used to perform bisulfite conversion for subsequent
methylation analysis. The primers used for MSP were complementary
to the promoter region of the HOXB13 gene. The primers were as
follows: Methylated HOXB13 CpG islands forward,
5′-TATTTTGGATGGAGTTAAGGATATC-3′ and reverse,
5′-ATAATTAACAACAAACATCAACGTA-3′; and unmethylated HOXB13 CpG
islands forward, 5′-ATTTTGGATGGAGTTAAGGATATTG-3′ and reverse,
5′-CATAATTAACAACAAACATCAACATA-3′ (27). The reaction mixture was in a volume of
50 µl containing 1 µl DNA, 2X GC buffer I, 1 µl of each primer, 2.5
mM dNTP Mix and 2.5 U LA Taq polymerase (Takara Bio, Inc.). The MSP
conditions were as follows: 94°C for 5 min, 40 cycles of 94°C for
30 sec, 54°C for 30 sec, 72°C for 45 sec and 72°C for 10 min. The
GES-1 cell line and peripheral blood cell-derived DNAs treated with
the CpG methlytransferase SssI (New England Biolabs, Ipswich, MA,
USA) were used as negative and positive controls respectively. All
procedures were repeated at least three times. The PCR products
were subjected to 2% agarose gel electrophoresis at 120 V for 40
min and quantified using the Fluor Chen 2.0 system.
5-Aza-cytidine treatment
MKN-45 cells were plated at a density of
5×105 cells per well into 6-well cell culture plates and
incubated at 37°C in a humidified incubator with 5% CO2.
Following culturing overnight, 0, 5 or 10 µmol/l 5-Aza-dC
(Sigma-Aldrich; Merck KGaA) was added and cells were incubated for
3 days, with 1.0 µmol/l trichostatin A (TSA; Beyotime Institute of
Biotechnology) added on the final day at room temperature. Next,
total RNA and DNA was extracted for detecting HOXB13 mRNA and
protein expression levels as aforementioned.
Statistical analysis
All statistical analysis was performed using SPSS
19.0 software (IBM Corp., Armonk, NY, USA). Paired Student's t-test
was used to analyze the association between HOXB13 mRNA expression
and clinicopathological features. Kaplan-Meier curves and log-rank
tests were used to estimate the influence of HOXB13 mRNA expression
on OS. Cox's proportional hazard model was used to assess hazard
ratios and corresponding 95% confidence intervals. One-way analysis
of variance (ANOVA) was used to assess the associations between
HOXB13 expression and methylation status. P<0.05 was considered
to indicate a statistically significant difference.
Results
Downregulation of HOXB13 mRNA
expression in gastric cell lines and tissues
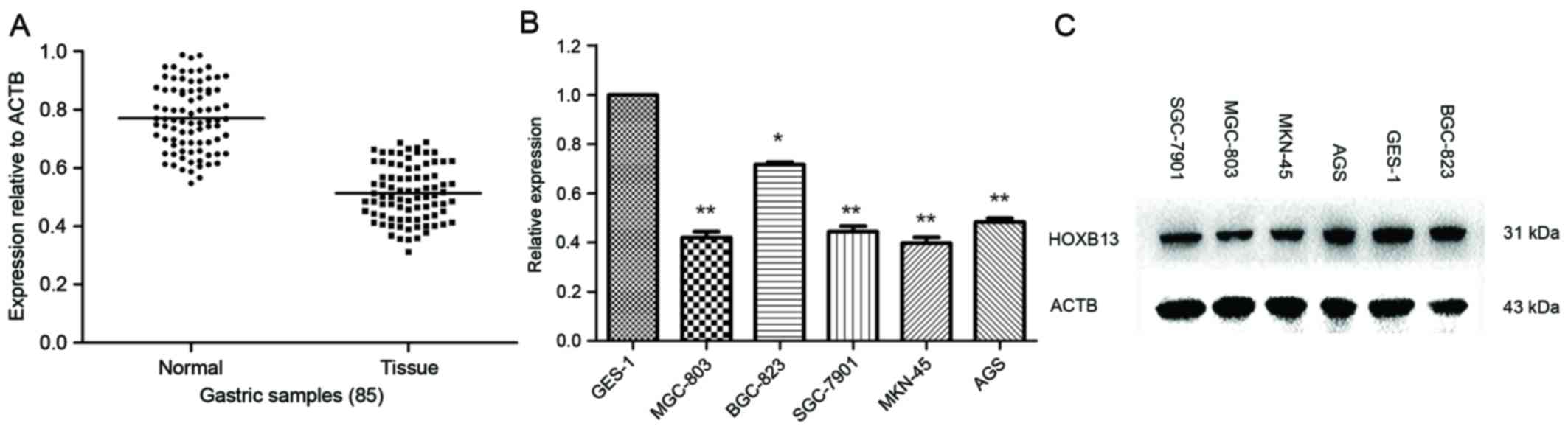
HOXB13 mRNA expression levels were analyzed in 85 GC
tissues and corresponding non-malignant tissues. RT-qPCR revealed
that the expression of HOXB13 mRNA in GC tissues was distinctly
lower than those in corresponding non-malignant tissues (Fig. 1A). The expression of HOXB13 mRNA and
protein levels were further examined by RT-qPCR and western
blotting in six gastric cell lines (five cancer, one non-cancer).
The results which used Student's t-test revealed that HOXB13 mRNA
and protein expression levels were significantly lower in three of
the five GC cells lines compared with GES-1 (the non-cancer
control; Fig. 1B and C). It was worth
noting that HOXB13 protein expression levels were lower in MGC-803
and MKN-45 cells compared with GES-1 cells in Fig. 1C. As there may exist translation level
adjustment, protein expression levels are not necessarily in
accordance with mRNA expression levels.
Association between expression of
HOXB13 mRNA and clinicopathological features
The associations between HOXB13 mRNA expression and
the clinicopathological factors of 85 patients with GC were
assessed. As presented in Table I,
HOXB13 mRNA expression was significantly associated with tumor
differentiation (P=0.008), tumor invasion depth (P=0.027), the
presence of lymph node metastases (P=0.016) and TNM stage
(P=0.007). However, HOXB13 mRNA expression was not associated with
the clinicopathological features tested, including age, sex, tumor
location, tumor size and Borrmann type (Table I) (28).
 | Table I.Associations between the
clinicopathological features and HOXB13 mRNA expression. |
Table I.
Associations between the
clinicopathological features and HOXB13 mRNA expression.
| Variable | Patients (n) | HOXB13 mRNA
levela | P-value |
|---|
| Non-malignant
tissues | 85 | 0.771±0.114 |
<0.001b |
| Tumor tissues | 85 | 0.514±0.095 |
|
| Age, years |
|
| 0.200 |
| ≥65 | 42 | 0.535±0.088 |
|
|
<65 | 43 | 0.508±0.102 |
|
| Sex |
|
| 0.165 |
|
Male | 45 | 0.508±0.095 |
|
|
Female | 40 | 0.537±0.095 |
|
| Location |
|
| 0.738 |
|
Upper/middle | 38 | 0.517±0.100 |
|
|
Lower | 47 | 0.524±0.092 |
|
| Size, cm |
|
| 0.216 |
|
<5 | 41 | 0.508±0.930 |
|
| ≥5 | 44 | 0.534±0.098 |
|
| Borrmann type |
|
| 0.077 |
|
I+II | 30 | 0.546±0.095 |
|
|
III+IV | 55 | 0.508±0.094 |
|
|
Differentiation |
|
| 0.008b |
|
Well/moderate | 39 | 0.551±0.087 |
|
|
Poor | 46 | 0.496±0.097 |
|
| Invasion depth |
|
| 0.027b |
|
T1+T2 | 36 | 0.548±0.091 |
|
|
T3+T4 | 49 | 0.502±0.095 |
|
| TNM stage |
|
| 0.007b |
|
I+II | 34 | 0.556±0.095 |
|
|
III+IV | 51 | 0.499±0.090 |
|
| Lymph node
metastasis |
|
| 0.016b |
| No | 32 | 0.554±0.083 |
|
|
Yes | 53 | 0.502±0.099 |
|
Expression of HOXB13 mRNA relative to
prognosis
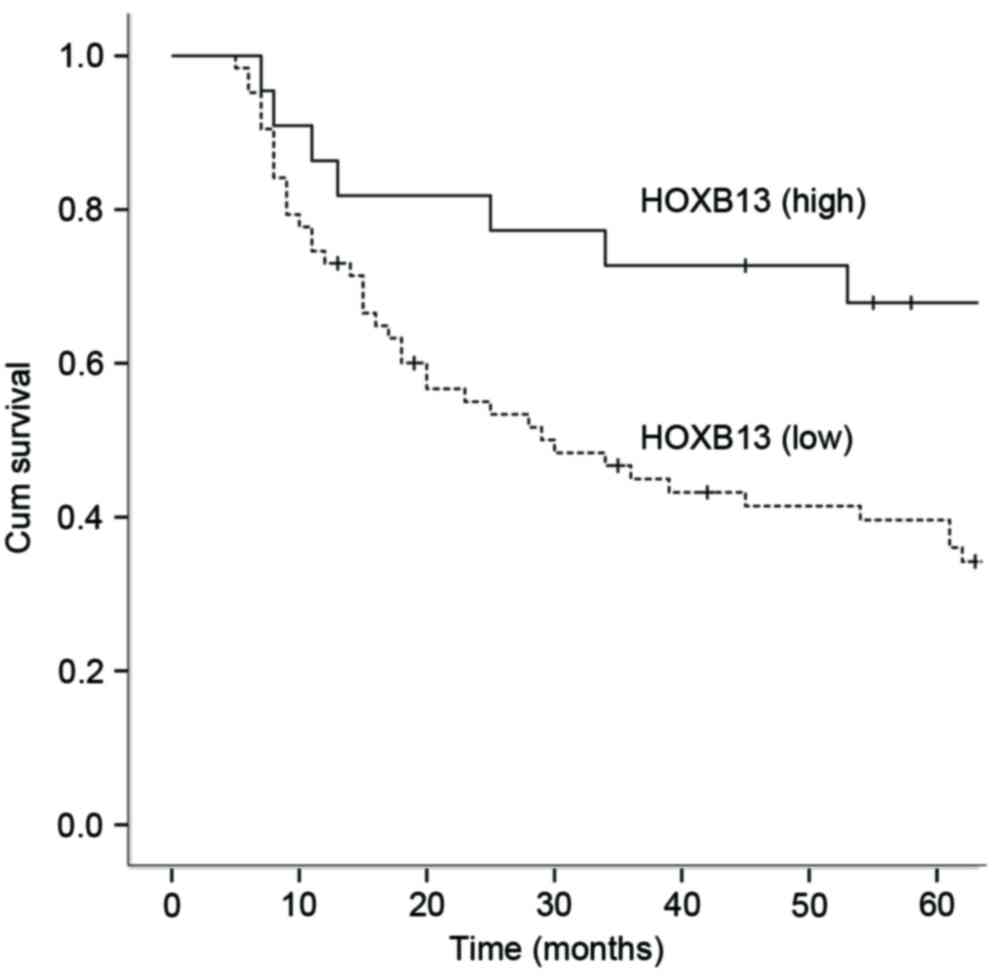
Kaplan-Meier survival curves were constructed to
assess the prognostic significance of HOXB13 mRNA expression and
various clinicopathological features (Table II). mRNA expression levels in GC
tissues were distinguished by comparing with the mean expression
quantity (0.771) of HOXB13 mRNA in 85 non-malignant gastric
tissues. A total of 22 patients expressed higher quantities and 63
patients expressed lower quantities of HOXB13. HOXB13 mRNA
expression was inversely associated with 5-year OS. In patients
expressing low amounts of HOXB13 mRNA, OS rate was 42.9%, which was
lower compared with the 5-year OS rate of 68.2% (Log-rank test,
P<0.05) in patients expressing high levels of HOXB13. Similarly,
tumor differentiation, invasion depth, Borrmann type and TNM stage
were revealed to be associated with OS rates. Cox multivariate
analysis was performed on expression of HOXB13 mRNA, invasion
depth, differentiation, Borrmann type and TNM stage. This analysis
revealed that HOXB13 mRNA expression, invasion depth,
differentiation and Borrmann type all acted as independent
prognostic factors for OS (Table
III and Fig. 2).
 | Table II.Univariate analysis of survival in
gastric cancer cases, assessed using the log rank test. |
Table II.
Univariate analysis of survival in
gastric cancer cases, assessed using the log rank test.
| Variable | 5-year OS | P-value |
|---|
| Age, years |
| 0.499 |
|
≥65 | 41.9 |
|
|
<65 | 38.1 |
|
| Sex |
| 0.409 |
|
Male | 40.0 |
|
|
Female | 40.0 |
|
| Location |
| 0.701 |
|
Upper/middle | 44.7 |
|
|
Lower | 42.5 |
|
| Size, cm |
| 0.763 |
|
<5 | 38.6 |
|
| ≥5 | 41.5 |
|
| Borrmann type |
|
<0.001a |
|
I+II | 73.3 |
|
|
III+IV | 21.8 |
|
|
Differentiation |
|
<0.001a |
|
Well/moderate | 66.7 |
|
|
Poor | 17.4 |
|
| Invasion depth |
|
<0.001a |
|
T1+T2 | 66.7 |
|
|
T3+T4 | 20.4 |
|
| Lymph node
metastasis |
| 0.072 |
| No | 53.1 |
|
|
Yes | 32.1 |
|
| TNM stage |
| 0.021a |
|
I+II | 50.0 |
|
|
III+IV | 33.3 |
|
| HOXB13 mRNA
expression |
| 0.010a |
|
High | 68.2 |
|
|
Low | 42.9 |
|
 | Table III.Multivariate analysis of survival in
gastric cancer cases, assessed using the log rang test. |
Table III.
Multivariate analysis of survival in
gastric cancer cases, assessed using the log rang test.
| Variable | B | SE | P-value | HR (95% CI) |
|---|
| Borrmann type | 1.068 | 0.267 |
<0.001a | 2.901
(1.723–4.914) |
|
Differentiation | 0.918 | 0.259 |
<0.001a | 2.504
(1.506–4.163) |
| Invasion depth | 0.930 | 0.274 | 0.001a | 2.535
(1.480–4.341) |
| TNM stage | 0.062 | 0.147 | 0.675 | 1.063
(0.798–1.418) |
| HOXB13 mRNA
expression | −0.699 | 0.312 | 0.025a | 0.497
(0.270–0.915) |
Promoter methylation status of HOXB13
gene in gastric cell lines and tissues
The methylation status of HOXB13 gene promoter CpG
islands was assessed by MSP. The data revealed that the HOXB13 gene
promoter was hypermethylated in BGC823, AGS, MKN45 and SGC-7901
cells, partially methylated in MGC-803 cells, and unmethylated in
GES1 cells (Fig. 3A).
Hypermethylation of the HOXB13 gene promoter was observed in 60%
(51/85) of gastric cancer tissue specimens (Fig. 3B), but in only 11.8% (10/85) of
corresponding non-malignant gastric tissues. The 10 hypermethylated
adjacent non-malignant gastric tissues had postoperative
pathological diagnoses of intestinal metaplasia or hyperplasia.
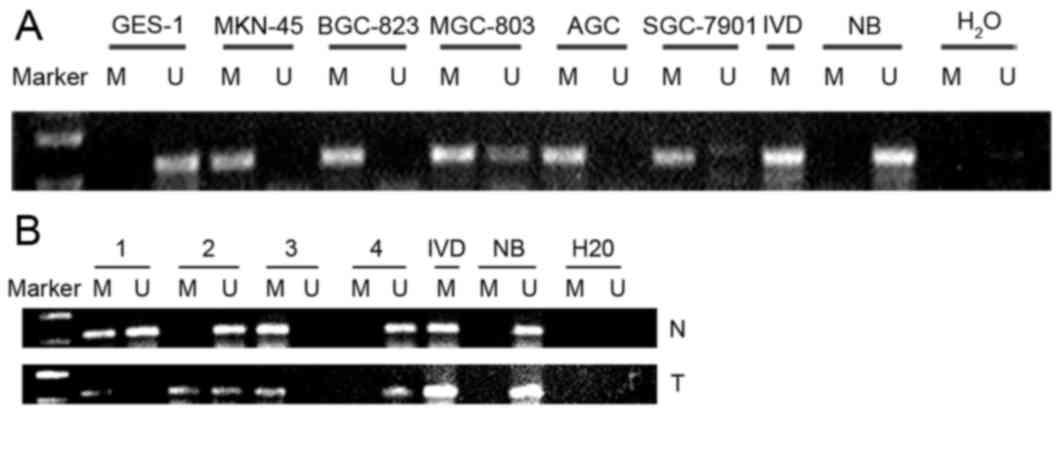 | Figure 3.Methylation status of the HOXB13 gene
in GC cell lines and GC tissues. (A) HOXB13 gene promoter was
hypermethylated in BGC-823, AGS, MKN-45 and SGC-7901 cells,
partially methylated in MGC-803 cells and unmethylated in GES1
cell. (B) HOXB13 gene promoter methylation status in GC tissues and
corresponding nonmalignant gastric tissues. IVD was used as a
positive control for methylation status, and NB samples were used
as non-methylation positive control. GC, gastric cancer; HOXB13,
homeobox B13; IVD, in vitro methylation DNA; NB, normal
blood; M, methylated; U, unmethylated. |
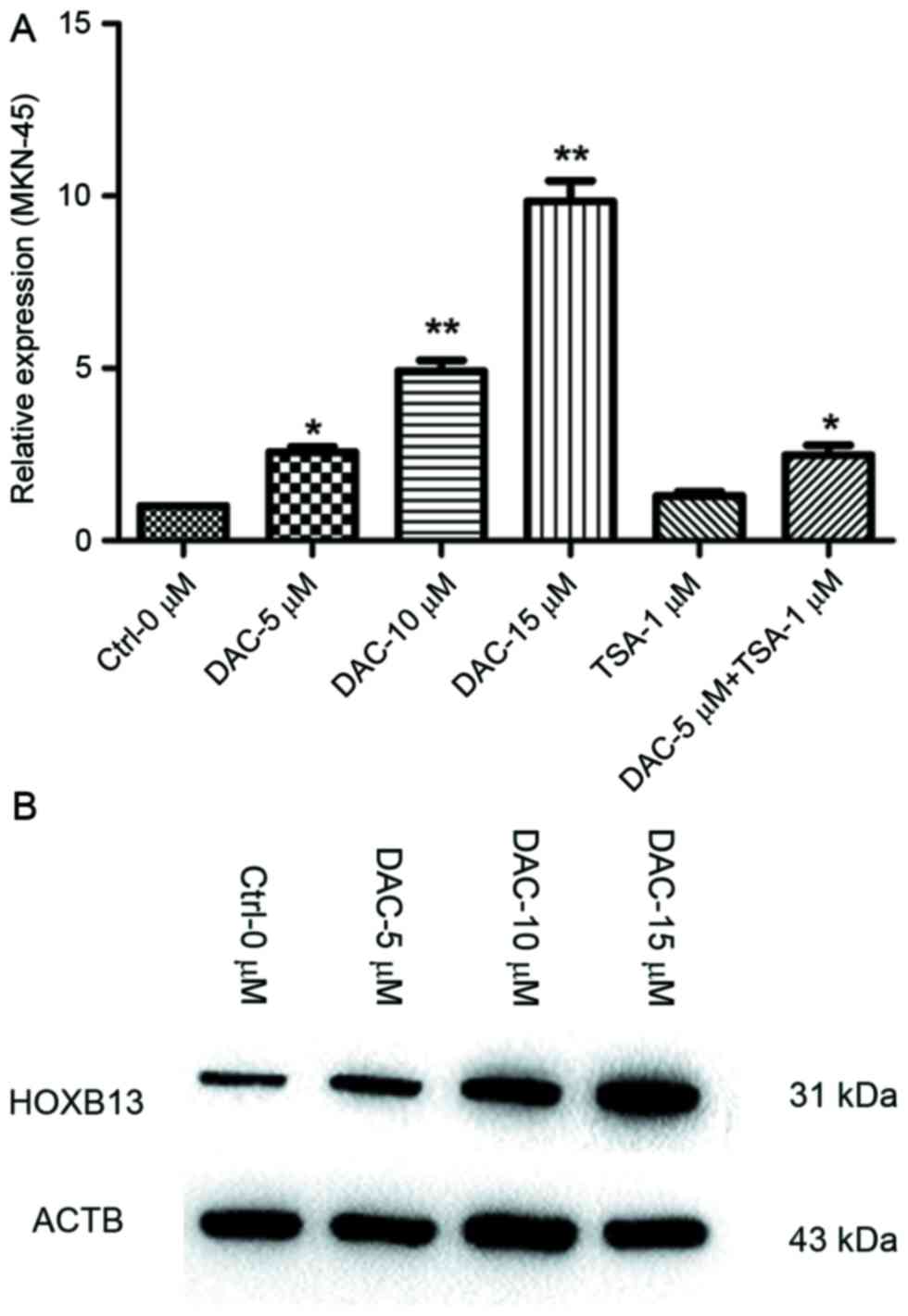
5-Aza-dC treatment induces upregulated
expression of HOXB13
To assess whether methylation of the HOXB13 gene
promoter was responsible for downregulation of HOXB13 expression,
the cell line with the lowest expression (MKN-45) was treated with
the DNA methylation inhibitor 5-Aza-dC and the histone deacetylase
inhibitor TSA. The results which were assessed by one-way ANOVA
demonstrated that 5-Aza-dC upregulated HOXB13 expression, with the
highest expression occurring at a concentration of 15 µM 5-Aza-dC.
HOXB13 expression was also restored with a combination treatment of
5-Aza-dC and TSA, but this was only slightly higher than that
induced by 5-Aza-dC alone (P<0.05; Fig. 4A). HOXB13 protein expression was also
increased in MKN-45 cells following 5-Aza-dC treatment (Fig. 4B).
Association between expression of
HOXB13 mRNA and methylation status
One-way ANOVA was used to assess the association
between HOXB13 mRNA expression and DNA methylation status in 85 GC
tissues: mRNA expression of HOXB13 was significantly lower in
tissues with methylated promoter regions than in tissues with
partial or no methylation. However, there was no difference in
expression of HOXB13 between tissues with partial HOXB13 promoter
methylation and no HOXB13 promoter methylation. Therefore, low
HOXB13 expression was associated with DNA hypermethylation.
Discussion
DNA methylation is a heritable epigenetic alteration
that does not alter the DNA nucleotide sequence but is involved in
transcriptional repression (29).
Methylation of DNA at promoter CpG islands where transcription is
initiated leads to the silencing of tumor suppressor genes, which
contributes directly to cancer development (30,31).
Previous studies have revealed that numerous tumor suppressor genes
undergo aberrant DNA methylation in GC (32,33).
HOXB13 gene expression was observed during the late
development of the tailbud and posterior of mouse embryos and
HOXB13 expressed in the spinal, digestive and urogenital system in
humans (34). A number of studies
have revealed a decrease in HOXB13 expression in colorectal cancer
(5), renal cancer (11), melanoma cancer (12) and breast cancer (13), making it a candidate tumor suppressor
gene. Promoter hypermethylation of the HOXB13 gene is considered to
be a potential mechanism for decreased expression (13–15,34).
In the present study, HOXB13 mRNA expression was
revealed to be decreased in five GC cell lines, compared with the
normal gastric cell line GES-1; HOXB13 mRNA expression was also
significantly lower in GC tissues than in non-malignant gastric
tissues. These data demonstrated that HOXB13 may act as a candidate
tumor suppressor in GC. In the 22 GC cases where HOXB13 mRNA
expression was higher than the mean expression quantity in adjacent
nonmalignant gastric tissues, the HOXB13 promoter was unmethylated
in 12 cases and partially methylated in 10 cases (none were
hypermethylated). This result indicated that the expression of
HOXB13 mRNA in GC was markedly affected by DNA methylation. HOXB13
mRNA expression was associated with tumor differentiation, invasion
depth, lymph node metastasis and TNM stage. Notably, the expression
of HOXB13 mRNA in poorly differentiated GC tissues was lower than
that in well/moderately differentiated GC tissues. Poorly
differentiated GC cells possess higher malignancy than
well/moderate differentiated cells, indicating that the decrease in
HOXB13 mRNA expression occurs in more malignant cases of GC. HOXB13
mRNA expression is lower in GC tissues than adjacent non-malignant
gastric tissues, which, along with later TNM stage and deeper
invasion depth, means greater disease progression of GC. The
expression of HOXB13 mRNA in GC with lymph node metastasis is
significantly lower than GC without lymph node metastasis. Marra
et al (35) reported that the
loss of HOXB13 expression in non-muscle-invasive bladder
transitional cancer is significantly associated with shorter
disease-free survival. The results of a log-rank test revealed that
there were significant differences in OS between patients of HOXB13
mRNA low expression and patients of HOXB13 mRNA high expression.
The results of Cox proportional hazards model analysis demonstrated
that expression of HOXB13 mRNA was an independent prognostic marker
for OS. Decreased HOXB13 mRNA expression may therefore act as a
potential predictor of the degree of GC malignancy and overall
disease prognosis, results that are also supported by Okuda et
al (14).
A previous study revealed that epigenetic
mechanisms, including DNA methylation, were associated with tumor
development and progression (36).
Promoter hypermethylation was the mechanism underlying the
decreased expression of HOXB13. In the present study, the HOXB13
gene was hypermethylated in all but one GC cell lines, and
partially methylated in MGC-803. Aberrant methylation of the HOXB13
promoter was also observed in GC tissue samples. Hypermethylation
of the HOXB13 gene promoter was identified in 51 (60.0%) of the 85
gastric tissues, while methylation in was only detected in 10
non-malignant adjacent gastric tissues (11.8%). Additionally,
adjacent nonmalignant gastric tissues in hypermethylation status
may be associated with tissue accompanied with precancerous
lesions. To examine the association between the methylation status
of the HOXB13 promoter and HOXB13 expression, a one-way ANOVA was
used. The results of this analysis demonstrated that expression of
HOXB13 in GC tissues with a hypermethylated HOXB13 promoter was
markedly lower compared with the expression of HOXB13 in GC tissues
with partially methylated or unmethylated HOXB13 promoters.
However, there was no marked difference between the expression of
HOXB13 in GC tissues with partially methylated promoter and the
expression of HOXB13 in GC tissues with unmethylated promoters.
These data revealed that low HOXB13 expression was caused by DNA
hypermethylation and indicated that the methylation of HOXB13 may
enhance the degree of malignancy of GC.
To verify the association between methylation of
HOXB13 and HOXB13 expression further, 5-Aza-dC and TSA were used to
treat the MKN-45 GC cell line, which had the lowest HOXB13 of all
tested cell lines. The results demonstrated that 5-Aza-dC enhanced
HOXB13 RNA and protein expression, but TSA had a weaker effect on
HOXB13 expression. Additionally, the expression of HOXB13 was
positively associated with 5-Aza-dC concentration. This result
further demonstrated that expression of HOXB13 was regulated by DNA
hypermethylation.
In conclusion, the present study indicated that
HOXB13 was downregulated in GC, which was caused by DNA
hypermethylation. HOXB13 mRNA expression was associated with tumor
differentiation, invasion depth, lymph node metastases and TNM
stage, and may be a potential prognostic factor for GC. The present
study demonstrated that HOXB13 is a tumor suppressor gene in GC and
has potential as a prognostic biomarker and as a target for
pharmacological intervention. Further studies in vivo are
necessary to determine the involvement of HOXB13 in GC
progression.
Acknowledgements
The authors would like to thank the researchers in
the Laboratory of the Fourth Affiliated Hospital of China Medical
University for their technical assistance.
Funding
This study was funded by the Natural Science
Foundation of Liaoning (grant no. 201602817).
Availability of data and materials
The data and materials used analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
DQD was responsible for the design of the experiment
and analysis and interpretation of data. BQS, CDZ, LW and JCL
carried out the acquisition of data. BQS and CDZ were involved in
drafting the manuscript and revising it critically for important
intellectual content. All authors provided final approval of the
version to be published.
Ethics approval and consent to
participate
Research involving human subjects, human material or
human data had be performed in accordance with the Declaration of
Helsinki and had be approved by Ethics committee of the Fourth
Affiliated Hospital of China Medical University. All participants
consented to participate in this study.
Consent for publication
The patient, or parent, guardian or next of kin (in
case of deceased patients) had provided written informed consent
for the publication of any associated data and accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shen L, Shan YS, Hu HM, Price TJ, Sirohi
B, Yeh KH, Yang YH, Sano T, Yang HK, Zhang X, et al: Management of
gastric cancer in Asia: Resource-stratified guidelines. Lancet
Oncol. 14:e535–e547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Allemani C, Weir HK, Carreira H, Harewood
R, Spika D, Wang XS, Bannon F, Ahn JV, Johnson CJ, Bonaventure A,
et al: Global surveillance of cancer survival 1995–2009: Analysis
of individual data for 25,676,887 patients from 279
population-based registries in 67 countries (CONCORD-2). Lancet.
385:977–1010. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Soubeyran P, Haglund K, Garcia S, Barth
BU, Iovanna J and Dikic I: Homeobox gene cdx1 regulates ras, rho
and pi3 kinase pathways leading to transformation and tumorigenesis
of intestinal epithelial cells. Oncogene. 20:4180–4187. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhu JY, Sun QK, Wang W and Jia WD:
High-level expression of HOXB13 is closely associated with tumor
angiogenesis and poor prognosis of hepatocellular carcinoma. Int J
Clin Exp Pathol. 7:2925–2933. 2014.PubMed/NCBI
|
|
5
|
Ghoshal K, Motiwala T, Claus R, Yan P,
Kutay H, Datta J, Majumder S, Bai S, Majumder A, Huang T, et al:
HOXB13, a target of DNMT3B, is methylated at an upstream CpG
island, and functions as a tumor suppressor in primary colorectal
tumors. PLoS One. 5:e103382010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Economides KD, Zeltser L and Capecchi MR:
Hoxb13 mutations cause overgrowth of caudal spinal cord and tail
vertebrae. Dev Biol. 256:317–330. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jung C, Kim RS, Lee SJ, Wang C and Jeng
MH: HOXB13 homeodomain protein suppresses the growth of prostate
cancer cells by the negative regulation of T-cell factor 4. Cancer
Res. 64:3046–3051. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jung C, Kim RS, Zhang H, Lee SJ, Sheng H,
Loehrer PJ, Gardner TA, Jeng MH and Kao C: HOXB13 is downregulated
in colorectal cancer to confer TCF4-mediated transactivation. Br J
Cancer. 92:2233–2239. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Decker B and Ostrander EA: Dysregulation
of the homeobox transcription factor gene HOXB13: Role in prostate
cancer. Pharmgenomics Pers Med. 7:193–201. 2014.PubMed/NCBI
|
|
10
|
Chung N, Jee BK, Chae SW, Jeon YW, Lee KH
and Rha HK: HOX gene analysis of endothelial cell differentiation
in human bone marrow-derived mesenchymal stem cells. Mol Biol Rep.
36:227–235. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang Q, Jin J and Tao Q: Aberrant
methylation of tumor suppressor genes in renal cell carcinoma. Ai
Zheng. 26:1276–1280. 2007.(In Chinese). PubMed/NCBI
|
|
12
|
Muthusamy V, Duraisamy S, Bradbury CM,
Hobbs C, Curley DP, Nelson B and Bosenberg M: Epigenetic silencing
of novel tumor suppressors in malignant melanoma. Cancer Res.
66:11187–11193. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tommasi S, Karm DL, Wu X, Yen Y and
Pfeifer GP: Methylation of homeobox genes is a frequent and early
epigenetic event in breast cancer. Breast Cancer Res. 11:R142009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Okuda H, Toyota M, Ishida W, Furihata M,
Tsuchiya M, Kamada M, Tokino T and Shuin T: Epigenetic inactivation
of the candidate tumor suppressor gene HOXB13 in human renal cell
carcinoma. Oncogene. 25:1733–1742. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rauch T, Li H, Wu X and Pfeifer GP:
MIRA-assisted microarray analysis, a new technology for the
determination of DNA methylation patterns, identifies frequent
methylation of homeodomain-containing genes in lung cancer cells.
Cancer Res. 66:7939–7947. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ewing CM, Ray AM, Lange EM, Zuhlke KA,
Robbins CM, Tembe WD, Wiley KE, Isaacs SD, Johnq D, Wang Y, et al:
Germline mutations in HOXB13 and prostate-cancer risk. N Engl J
Med. 366:141–149. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ang MK, Ooi AS, Thike AA, Tan P, Zhang Z,
Dykema K, Furge K, Teh BT and Tan PH: Molecular classification of
breast phyllodes tumors: Validation of the histologic grading
scheme and insights into malignant progression. Breast Cancer Res
Treat. 129:319–329. 2012. View Article : Google Scholar
|
|
18
|
Miao J, Wang Z, Provencher H, Muir B,
Dahiya S, Carney E, Leong CO, Sgroi DC and Orsulic S: HOXB13
promotes ovarian cancer progression. Proc Natl Acad Sci USA.
104:17093–17098. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yuan H, Kajiyama H, Ito S, Chen D, Shibata
K, Hamaguchi M, Kikkawa F and Senga T: HOXB13 and ALX4 induce SLUG
expression for the promotion of EMT and cell invasion in ovarian
cancer cells. Oncotarget. 6:13359–13370. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lopez R, Garrido E, Piña P, Hidalgo A,
Lazos M, Ochoa R and Salcedo M: HOXB homeobox gene expression in
cervical carcinoma. Int J Gynecol Cancer. 16:329–335. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
De Souza Setubal Destro MF, Bitu CC,
Zecchin KG, Graner E, Lopes MA, Kowalski LP and Coletta RD:
Overexpression of HOXB7 homeobox gene in oral cancer induces
cellular proliferation and is associated with poor prognosis. Int J
Oncol. 8:141–149. 2010.
|
|
22
|
Maia S, Cardoso M, Pinto P, Pinheiro M,
Santos C, Peixoto A, Bento MJ, Oliveira J, Henrique R, Jeronimo C
and Teixeira MR: Identification of two novel HOXB13 germline
mutations in portuguese prostate cancer patients. PLoS One.
10:e01327282015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Beebe-Dimmer J, Hathcock M, Yee C, Okoth
LA, Ewing CM, Isaacs WB, Cooney KA and Thibodeau SN: The HOXB13
G84E mutation is associated with an increased risk for prostate
cancer and other malignancies. Cancer Epidemiol Biomarkers Prev.
24:1366–1372. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Brierley JD, Gospodarowicz MK and
Wittekind C: TNM classification of malignant tumours.
Wiley-Blackwell. 80:1803–1804. 2010.
|
|
25
|
Wang Z, Dahiya S, Provencher H, Muir B,
Carney E, Coser K, Shioda T, Ma XJ and Sgroi DC: The prognostic
biomarkers HOXB13, IL17BR, and CHDH are regulated by estrogen in
breast cancer. Clin Cancer Res. 13:6327–6334. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang F, Yang Y, Fu Z, Xu N, Chen F, Yin H,
Lu X, Shen R and Lu C: Differential DNA methylation status between
breast carcinomatous and normal tissues. Biomed Pharmacother.
68:699–707. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Japanese Gastric Cancer Association:
Japanese classification of gastric carcinoma-2nd English edition.
Gastric Cancer. 1:10–24. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gopalakrishnan S, Van Emburgh BO and
Robertson KD: DNA methylation in development and human disease.
Mutat Res. 647:30–38. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kanwal R and Gupta S: Epigenetics and
cancer. J Appl Physiol (1985). 109:598–605. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chang X, Zhang S, Ma J, Li Z, Zhi Y, Chen
J, Lu Y and Dai D: Association of NDRG1 gene promoter methylation
with reduced NDRG1 expression in gastric cancer cells and tissue
specimens. Cell Biochem Biophys. 66:93–101. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu X, Chang X, Li Z, Wang J, Deng P, Zhu
X, Liu J, Zhang C, Chen S and Dai D: Aberrant SOX11, promoter
methylation is associated with poor prognosis in gastric cancer.
Cell Oncol (Dordr). 38:183–194. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang J, Li Y, Liu J, Zhang C and Dai D:
Epigenetic regulation of microrna-335 and its clinical significance
in gastric cancer. J Modern Oncol. 2016.
|
|
34
|
Zeltser L, Desplan C and Heintz N:
Hoxb-13: A new Hox gene in a distant region of the HOXB cluster
maintains colinearity. Development. 122:2475–2484. 1996.PubMed/NCBI
|
|
35
|
Marra L, Cantile M, Scognamiglio G,
Perdonà S, La Mantia E, Cerrone M, Giqantino V, Cillo C, Caraqlia
M, Piqnata S, et al: Deregulation of HOX B13 expression in urinary
bladder cancer progression. Curr Med Chem. 20:833–839. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Varinot J, Cussenot O, Roupret M, Conort
P, Bitker MO, Chartier-kastler E, Cheng L and Compérat E: HOXB13 is
a sensitive and specific marker of prostate cells, useful in
distinguishing between carcinomas of prostatic and urothelial
origin. Virchows Arch. 463:803–809. 2013. View Article : Google Scholar : PubMed/NCBI
|