Introduction
Clear cell tumor, also known as ‘sugar’ tumor,
belongs to the family of perivascular epithelial cell tumors
(PEComas) and can originate from various organs in the body, such
as the rectum, pancreas, heart, salivary gland, trachea and lung
(1,2).
Clear cell tumor of the lung (CCTL) was first described by Liebow
and Castleman (3) in 1963 and is
generally regarded as an extremely rare benign mesenchymal lung
tumor. It is usually incidentally detected on routine check-up
chest radiograph as a solitary pulmonary nodule. There are no
significant findings in physical examinations and laboratory
studies although only a few patients displayed symptoms (headaches,
weakness, cough, bloody sputum, hemoptysis, chest pain, essential
thrombocytosis and unexplained high fever) (4). Only a few sporadic cases have been
reported and study of the interval growth of this tumor is
extremely limited. The present study reports a CCTL that exhibited
growth pattern change on follow-up imaging studies over 7 years
along with a review of the literature. The present case study was
approved by the Kyung Hee University Hospital at Gangdong
Institutional Review Board (Seoul, Korea) and the patient provided
informed written consent for the publication of the present
study.
Case report
A 58-year-old man was admitted to our hospital for
further evaluation of an incidentally detected nodular opacity on
routine check-up chest radiograph. Physical and laboratory
examinations did not exhibit any abnormalities. He was a
35-pack-year current smoker and had a history of diabetes mellitus.
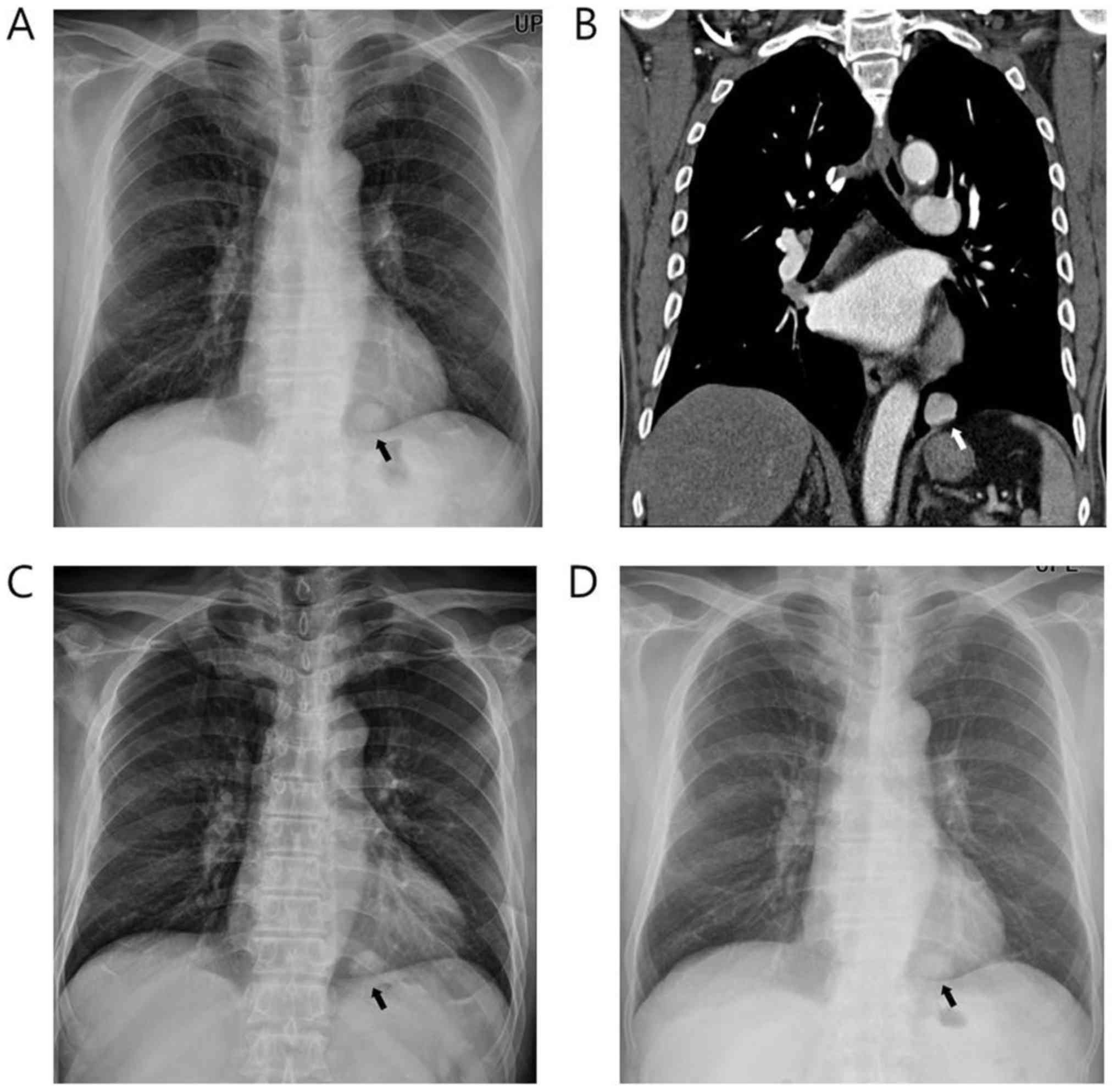
On chest radiograph, a round and well-defined nodular opacity was
identified in the left lower lung field (Fig. 1A). Contrast-enhanced chest computed
tomography (CT) demonstrated a 2.7 cm nodule in the left lower
lobe, and the nodule showed strong homogeneous contrast enhancement
(Fig. 1B). There was no definite
cavitation or necrosis. The nodule was retrospectively traced on
whole spine radiography that was performed to evaluate back pain 18
months ago (Fig. 1C). The maximal
diameter of the nodule was 2.6 cm, and there was no significant
interval change in size over 18 months. Because the patient was
unwilling to proceed with further evaluation of the nodule and the
nodule was stable, the patient was decided to undergo serial
observation.
One year later, the patient underwent follow-up
evaluation. The nodule showed no significant interval change in
size on chest radiographs (Fig. 1D).
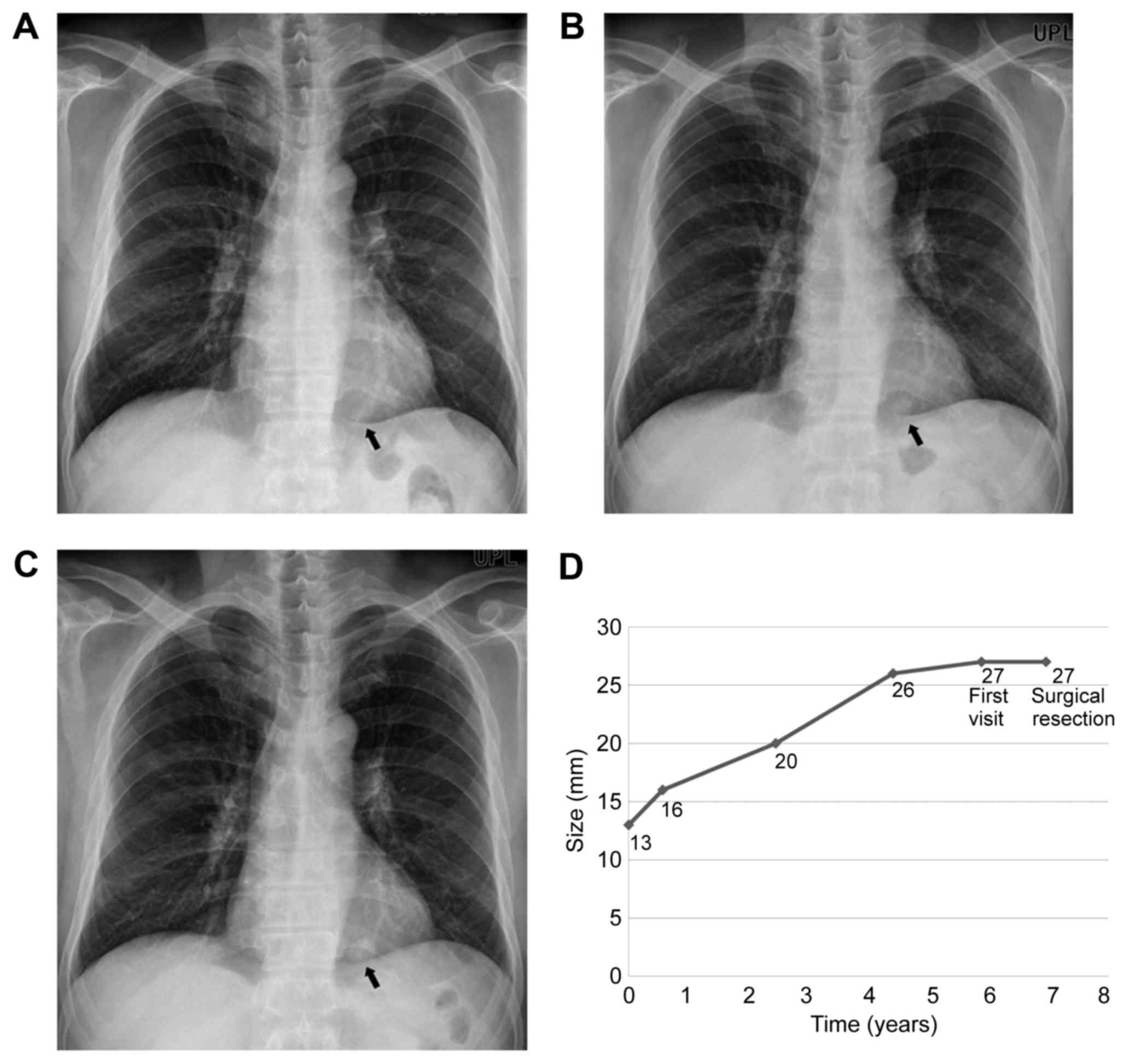
However, he brought his old chest radiographs from an outside
hospital, in which the nodule had showed slow interval growth from
1.3 to 2.6 cm over 4.5 years (Fig.
2A-D).
Although there was no significant interval change in
size over the most recent 2.5 years, the malignant potential of the
nodule was unknown because of previous interval growth. Therefore,
the nodule was resected by video-assisted thoracoscopic wedge
resection.
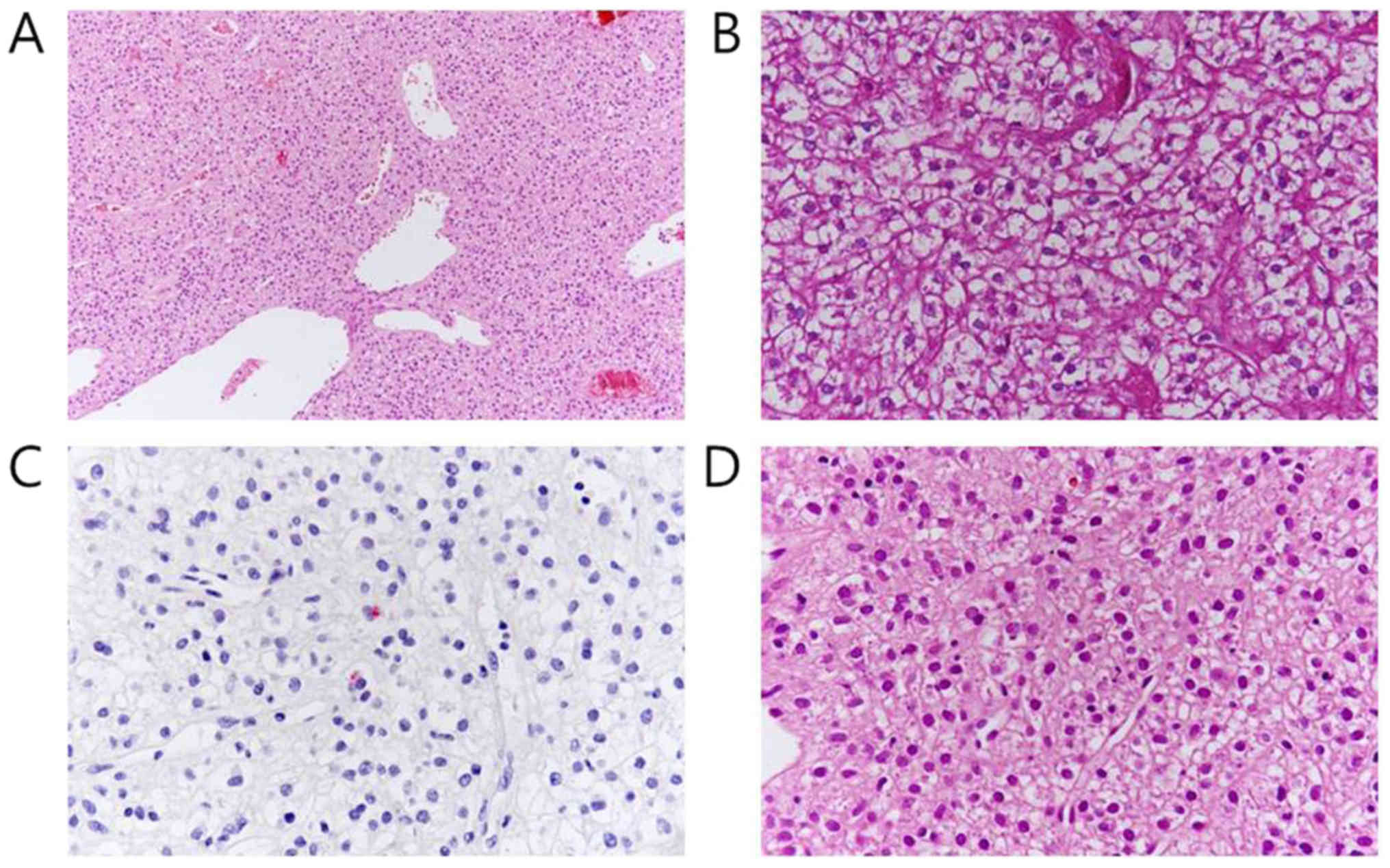
The gross specimen reveals a well-defined nodule
measuring 2.7 cm in diameter without evidence of cavitation,
hemorrhage, or calcification. Microscopically, the tumor consisted
of rounded cells with clear to pale eosinophilic cytoplasm rich in
periodic acid-Schiff (PAS)-positive glycogen granules (Fig. 3A, B). Immunohistochemial staining
showed human melanoma black (HMB)-45, vimentin, and CD34
expression, but failed to express cytokeratin AE1/AE3, and
epithelial membrane antigen (Fig.
3C). The tumor cells showed mild variation in nuclear size, but
mitosis was not identified (Fig. 3D).
On the basis of morphology and immunohistochemistry results, a
diagnosis of CCTL was established. After surgical resection, the
patient remained well with no evidence of local recurrence.
Discussion
CCTL is a very rare benign mesenchymal tumor with
less than 60 cases reported in the English literature (5). The tumor can occur in any age group,
although it affects elderly patients more often, and it has equal
sex predilection with slight female predominance (1). According to the 2004 World Health
Organization classification of lung tumors, CCTL belongs to the
family of PEComas (1). The tumor
consists of clear cells with a large amount of cytoplasmic periodic
acid-Schiff (PAS)-positive glycogen; this is why the CCTL is also
called sugar tumor. CCTL has distinguishable immunohistochemical
features that include positivity for S-100 protein and human
melanoma black (HMB)-45 and no reactivity for cytokeratin, an
epithelial marker (6).
Most CCTLs have no symptoms and are usually
incidentally discovered on routine chest radiography or CT scan
(4). On chest radiographs, CCTLs
present as a coin lesion or peripheral solitary nodule with
well-defined smooth margins (5,7). Because
of the rich vascular stroma, most CCTLs show homogenous intense
enhancement on CT scans. Kim et al (8), reported an early wash-in and washout
pattern of CCTL on dynamic CT. CCTLs usually show no
18F-2-deoxy-D-glucose (FDG) uptake, but a 2.8-cm benign
CCTL that showed extensive FDG uptake has been reported (9).
Although CCTL is generally regarded as benign, there
are some reports of CCTLs that exhibit malignant behavior in terms
of local invasion or vascularity (10). Necrosis, mitotic index of 1 per 50
high-power fields, marked pleomorphism, and nuclear atypia also
raise the possibility of malignant potential (11). It is difficult to definitively
evaluate these pathological features by cytologic study such as
fine needle aspiration or biopsy. Therefore, surgical resection is
recommended for diagnosis and curative treatment (8).
The rarity of CCTL and the paucity of published
literature on CCTL makes it difficult to identify the interval
growth pattern of CCTL. Only few cases have reported the interval
growth of CCTLs. However, previous reports only stated an interval
growth without mentioning the exact change in size (9,12) or
suggested a maximum volume doubling time estimated from initial
normal chest radiograph (13). No
previous reports remarked on serial imaging follow up of CCTL.
Interestingly, CCTL showed a growth pattern change during 7 years
of follow up in this case. On serial radiographs, the nodule
initially showed linear gradual interval growth, but it remained
2.7 cm in diameter for the next 2.5 years. Although the limit of
detectable changes in size on standard radiography has been
estimated to be 3.0 to 5.0 mm (14),
a reasonable assumption is that the nodule growth rate was
extremely low during the past 2.5 years. We should keep in mind
CCTL can show stable or aggressive behavior depending on the
detection time and observation period. Growth pattern change and
malignant potential of CCTL underlines the need of constant and
close observation of these tumors.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JIK made substantial contributions to the conception
and design of the study. EKY and HNL analyzed and interpreted the
patient data regarding the follow up simple radiograph images and
CT images and EKY was a major contributor in writing the
manuscript. KYW performed the histological examination of the lung.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present case study was approved by the Kyung Hee
University Hospital at Gangdong Institutional Review Board (Seoul,
Korea).
Consent for publication
The patient provided written informed consent for
the publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Travis WD, Brambilla E, Nicholson AG,
Yatabe Y, Austin JHM, Beasley MB, Chirieac LR, Dacic S, Duhig E,
Flieder DB, et al: The 2015 World Health Organization
classification of lung tumors: Impact of genetic, clinical and
radiologic advances since the 2004 classification. J Thorac Oncol.
10:1243–1260. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tazelaar HD, Batts KP and Srigley JR:
Primary extrapulmonary sugar tumor (PEST): A report of four cases.
Mod Pathol. 14:615–622. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liebow A and Castleman B: Benign clear
cell (‘sugar’) tumors of the lung. Yale J Biol Med. 43:213–222.
1971.PubMed/NCBI
|
|
4
|
Chen YB, Guo LC, Huang JA, Ji C and Ling
CH: Clear cell tumor of the lung: A retrospective analysis. Am J
Med Sci. 347:50–53. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang GX, Zhang D, Diao XW and Wen L: Clear
cell tumor of the lung: A case report and literature review. World
J Surg Oncol. 11:2472013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mizobuchi T, Masahiro N, Iwai N, Kohno H,
Okada N and Nakada S: Clear cell tumor of the lung: Surgical and
immunohistochemical findings. Gen Thorac Cardiovasc Surg.
58:243–247. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Das S, Cherian SV, Das N, Hamarneh WA,
Garcha Singh A, Singh Preet P and Lenox R: A 52-Year-Old smoker
with an incidental pulmonary nodule. Chest. 141:1346–1350. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kim WJ, Kim SR, Choe YH, Lee KY, Park SJ,
Lee HB, Chung MJ, Jin GY and Lee YC: Clear cell ‘sugar’ tumor of
the lung: A well-enhanced mass with an early washout pattern on
dynamic contrast-enhanced computed tomography. J Korean Med Sci.
23:1121–1124. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zarbis N, Barth TF, Blumstein NM and
Schelzig H: Pecoma of the lung: A benign tumor with extensive
18F-2-deoxy-D-glucose uptake. Interact Cardiovasc Thorac Surg.
6:676–678. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kavunkal AM, Pandiyan MS, Philip MA,
Parimelazhagan KN, Manipadam MT and Cherian VK: Large clear cell
tumor of the lung mimicking malignant behavior. Ann Thorac Surg.
83:310–312. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hornick JL and Fletcher CD: PEComa: What
do we know so far? Histopathology. 48:75–82. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mishina T, Suzuki I, Fujino M, Watanabe N,
Akita H, Narita Y and Kawakami Y: A benign clear cell tumor of the
lung that grew gradually over five years. Nihon Kyobu Shikkan
Gakkai Zasshi. 33:765–770. 1995.PubMed/NCBI
|
|
13
|
Kalkanis A, Trianti M, Psathakis K,
Mermigkis C, Kalkanis D, Karagkiouzis G, Razou A and Tsintiris K: A
clear cell tumor of the lung presenting as a rapidly growing coin
lesion: Is it really a benign tumor? Ann Thorac Surg. 91:588–591.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ost D, Fein AM and Feinsilver SH: Clinical
practice. The solitary pulmonary nodule. N Engl J Med.
348:2535–2542. 2003. View Article : Google Scholar : PubMed/NCBI
|