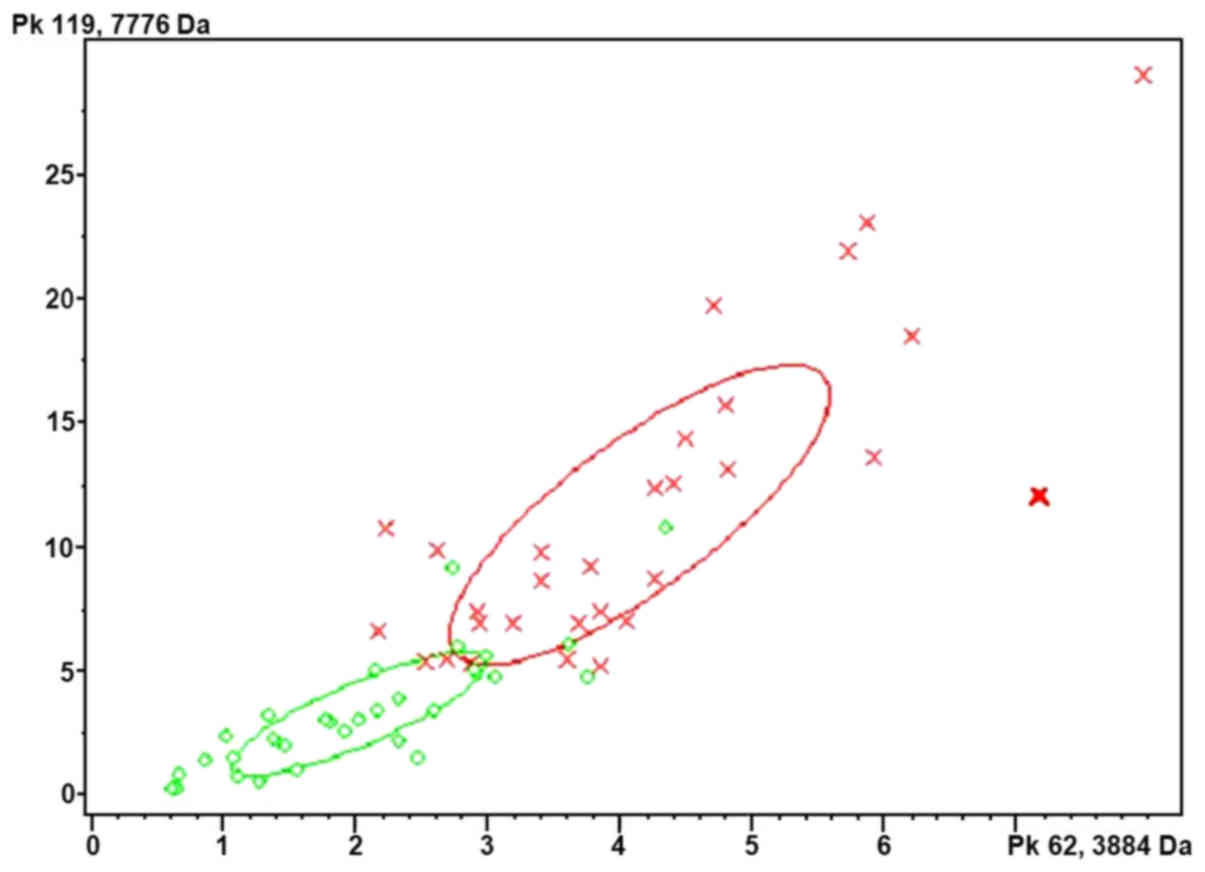
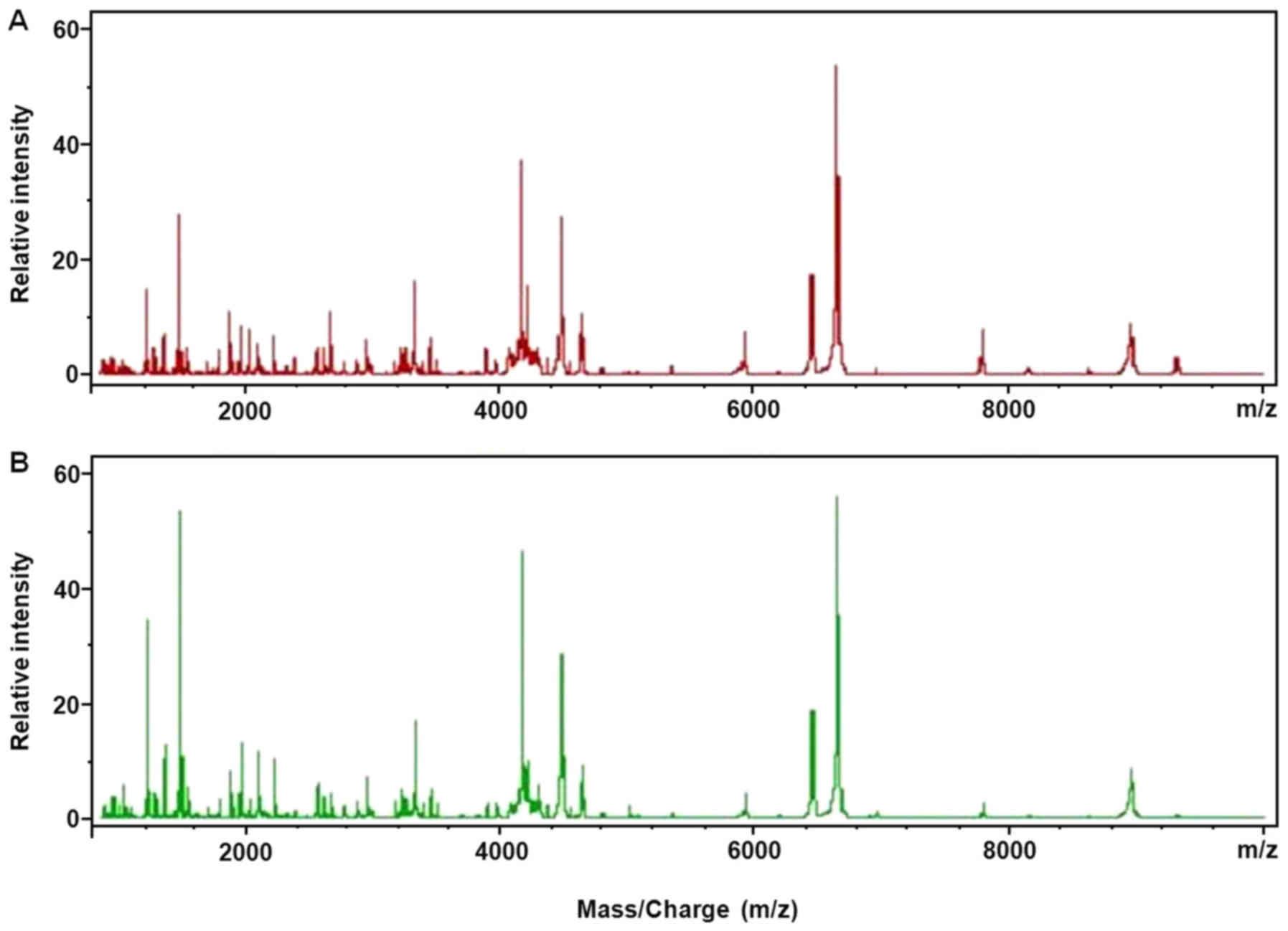
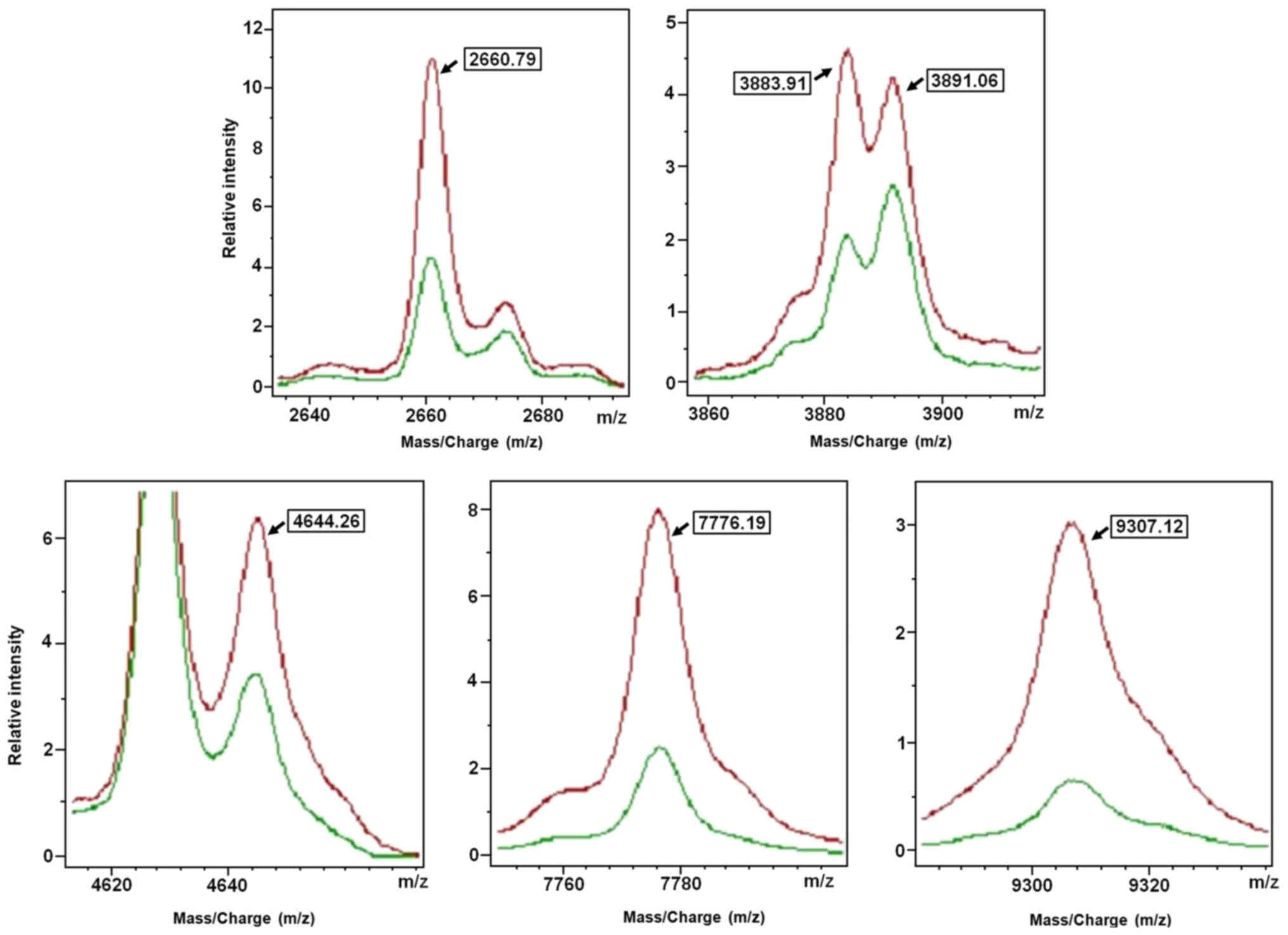
|
1
|
World Health Organization: Cancer.
http://www.who.int/mediacentre/factsheets/fs297/en/April
23–2015
|
|
2
|
Stewart EL, Tan SZ, Liu G and Tsao MS:
Known and putative mechanisms of resistance to EGFR targeted
therapies in NSCLC patients with EGFR mutations-a review. Transl
Lung Cancer Res. 4:67–81. 2015.PubMed/NCBI
|
|
3
|
Pao W and Chmielecki J: Rational,
biologically based treatment of EGFR-mutant non-small-cell lung
cancer. Nat Rev Cancer. 10:760–774. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sequist LV, Joshi VA, Jänne PA, Muzikansky
A, Fidias P, Meyerson M, Haber DA, Kucherlapati R, Johnson BE and
Lynch TJ: Response to treatment and survival of patients with
non-small cell lung cancer undergoing somatic EGFR mutation
testing. Oncologist. 12:90–98. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tsao MS, Sakurada A, Cutz JC, Zhu CQ,
Kamel-Reid S, Squire J, Lorimer I, Zhang T, Liu N, Daneshmand M, et
al: Erlotinib in lung cancer-molecular and clinical predictors of
outcome. N Engl J Med. 353:133–144. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Taniguchi Y, Tamiya A, Nakahama K, Naoki
Y, Kanazu M, Omachi N, Okishio K, Kasai T and Atagi S: Impact of
metastatic status on the prognosis of EGFR mutation-positive
non-small cell lung cancer patients treated with first-generation
EGFR-tyrosine kinase inhibitors. Oncol Lett. 14:7589–7596.
2017.PubMed/NCBI
|
|
7
|
Jackman D, Pao W, Riely GJ, Engelman JA,
Kris MG, Jänne PA, Lynch T, Johnson BE and Miller VA: Clinical
definition of acquired resistance to epidermal growth factor
receptor tyrosine kinase inhibitors in non-small-cell lung cancer.
J Clin Oncol. 28:357–360. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Laurila N and Koivunen JP: EGFR inhibitor
and chemotherapy combinations for acquired TKI resistance in
EGFR-mutant NSCLC models. Med Oncol. 32:2052015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hirsch FR, Varella-Garcia M, Bunn PA Jr,
Franklin WA, Dziadziuszko R, Thatcher N, Chang A, Parikh P, Pereira
JR, Ciuleanu T, et al: Molecular predictors of outcome with
gefitinib in a phase III placebo-controlled study in advanced
non-small-cell lung cancer. J Clin Oncol. 24:5034–5042. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lynch TJ, Bell DW, Sordella R,
Gurubhagavatula S, Okimoto RA, Brannigan BW, Harris PL, Haserlat
SM, Supko JG, Haluska FG, et al: Activating mutations in the
epidermal growth factor receptor underlying responsiveness of
non-small-cell lung cancer to gefitinib. N Engl J Med.
350:2129–2139. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Paez JG, Jänne PA, Lee JC, Tracy S,
Greulich H, Gabriel S, Herman P, Kaye FJ, Lindeman N, Boggon TJ, et
al: EGFR mutations in lung cancer: Correlation with clinical
response to gefitinib therapy. Science. 304:1497–1500. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sharma SV, Bell DW, Settleman J and Haber
DA: Epidermal growth factor receptor mutations in lung cancer. Nat
Rev Cancer. 7:169–181. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ellison G, Zhu G, Moulis A, Dearden S,
Speake G and McCormack R: EGFR mutation testing in lung cancer: A
reivew of available methods and their use for analysis of tumor
tissue and cytology samples. J Clin Pathol. 66:79–89. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kitamura A, Hosoda W, Sasaki E, Mitsudomi
T and Yatabe Y: Immunohistochemical detection of EGFR mutation
using mutation-specific antibodies in lung cancer. Clin Cancer Res.
16:3349–3355. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sandanayake NS, Camuzeaux S, Sinclair J,
Blyuss O, Andreola F, Chapman MH, Webster GJ, Smith RC, Timms JF
and Pereira SP: Identification of potential serum peptide
biomarkers of biliary tract cancer using MALDI MS profiling. BMC
Clin Pathol. 14:72014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Callesen AK, Madsen JS, Vach W, Kruse TA,
Mogensen O and Jensen ON: Serum protein profiling by solid phase
extraction and mass spectromety: A future diagnostics tools?
Proteomics. 9:1428–1441. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lopez MF, Mikulskis A, Kuzdzal S, Golenko
E, Petricoin EF III, Liotta LA, Patton WF, Whiteley GR, Rosenblatt
K, Gurnani P, et al: A novel, high-throughput workflow for
discovery and identification of serum carrier protein-bound peptide
biomarker candidates in ovarian cancer samples. Clin Chem.
53:1067–1074. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Han KQ, Huang G, Gao CF, Wang XL, Ma B,
Sun LQ and Wei ZJ: Identification of lung cancer patients by serum
protein profiling using surface-enhanced laser
desorption/ionization time-of-flight mass spectrometry. Am J Clin
Oncol. 31:133–139. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Koopmann J, Zhang Z, White N, Rosenzweig
J, Fedarko N, Jagannath S, Canto MI, Yeo CJ, Chan DW and Goggins M:
Serum diagnosis of pancreatic adenocarcinoma using surface-enhanced
laser desorption and ionization mass spectrometry. Clin Cancer Res.
10:860–868. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li J, Zhang Z, Rosenzweig J, Wang YY and
Chan DW: Proteomics and bioinformatics approaches for
identification of serum biomarkers to detect breast cancer. Clin
Chem. 48:1296–1304. 2002.PubMed/NCBI
|
|
21
|
Taguchi F, Solomon B, Gregorc V, Roder H,
Gray R, Kasahara K, Nishio M, Brahmer J, Spreafico A, Ludovini V,
et al: Mass spectrometry to classify non-small-cell lung cancer
patients for clinical outcome after treatment with epidermal growth
factor receptor tyrosine kinase inhibitors: A multicohort
cross-institutional study. J Natl Cancer Inst. 99:838–846. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lazzari C, Spreafico A, Bachi A, Roder H,
Floriani I, Garavaglia D, Cattaneo A, Grigorieva J, Viganò MG,
Sorlini C, et al: Changes in plasma mass-spectral profile in course
of treatment of non-small cell lung cancer patients with epidermal
growth factor receptor tyrosine kinase inhibitors. J Thorac Oncol.
7:40–48. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hirsch FR and Bunn PA Jr: EGFR testing in
lung cancer is ready for prime time. Lancet Oncol. 10:432–433.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mitsudomi T, Kosaka T and Yatabe Y:
Biological and clinical implications of EGFR mutations in lung
cancer. Int J Clin Oncol. 11:190–198. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schiller JH, Harrington D, Belani CP,
Langer C, Sandler A, Krook J, Zhu J and Johnson DH: Eastern
Cooperative Oncology Group: Comparison of four chemotherapy
regimens for advanced non-small-cell lung cancer. N Engl J Med.
346:92–98. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sandler A, Gray R, Perry MC, Brahmer J,
Schiller JH, Dowlati A, Lilenbaum R and Johnson DH:
Paclitaxel-carboplatn alone or with bevacizumab for non-small-cell
lung cancer. N Engl J Med. 355:2542–2550. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumors:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carlson RV, Boyd KM and Webb DJ: The
revision of the Declaration of Helsinki: Past, present and future.
Br J Clin Pharmacol. 57:695–713. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu X, Lu Y, Zhu G, Lei Y, Zheng L, Qin H,
Tang C, Ellison G, McCormack R and Ji Q: The diagnostic accuracy of
pleural effusion and plasma samples versus tumour tissue for
detection of EGFR mutation in patients with advanced non-small cell
lung cancer: Comparison of methodologies. J Clin Pathol.
66:1065–1069. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim CH, Kim SH, Park SY, Yoo J, Kim SK and
Kim HK: Identification of EGFR mutations by immunohistochemistry
with EGFR Mutation-specific antibodies in biopsy and resection
specimens from pulmonary adenocarcinoma. Cancer Res Threat.
47:653–660. 2015. View Article : Google Scholar
|
|
31
|
Yang L, Tang C, Xu B, Wang W, Li J, Li X,
Qin H, Gao H, He K, Song S and Liu X: Classification of epidermal
growth factor receptor gene mutation status using serum proteomic
profiling predicts tumor response in patients with stage IIIB or IV
Non-small-cell lung cancer. PLoS One. 10:e01289702015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou J, Wang J, Zeng Y, Zhang X, Hu Q,
Zheng J, Chen B, Xie B and Zhang WM: Implication of
epithelial-mesenchymal transition in IGF1R-induced resistance to
EGFR-TKIs in advanced non-small cell lung cancer. Oncotarget.
6:44332–44345. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xu J, Wang J and Zhang S: Mechanisms of
resistance to irreversible epidermal growth factor receptor
tyrosine kinase inhibitors and therapeutic strategies in non-small
cell lung cancer. Oncotarget. 8:90557–90578. 2017.PubMed/NCBI
|
|
34
|
Mansuet-Lupo A, Zouiti F, Alifano M,
Tallet A, Charpentier MC, Ducruit V, Devez F, Lemaitre F,
Laurent-Puig P, Damotte D and Blons H: Intratumoral distribution of
EGFR mutations and copy number in metastatic lung cancer, what
impact on the initial molecular diagnosis? J Transl Med.
12:1312014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gregorc V, Novello S, Lazzari C, Barni S,
Aieta M, Mencoboni M, Grossi F, De Pas T, de Marinis F, Bearz A, et
al: Predictive value of a proteomic signature in patients with
non-small-cell lung cancer treated with second-line erlotinib or
chemotherapy (PROSE): A biomarker-stratified, randomised phase 3
trial. Lancet Oncol. 15:713–721. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lazzari C, Novello S, Barni S, Aieta M, De
Marinis F, De Pas T, Grossi F, Mencoboni M, Bearz A, Floriani I, et
al: Randomized proteomic stratified phase III study of second-line
erlotinib (E) versus chemotherapy (CT) in patients with inoperable
non-small cell lung cancer (PROSE). J Clin Oncol. 31:2013.DOI:
10.1200/jco.2013.31. View Article : Google Scholar
|
|
37
|
Kuiper JL, Lind JS, Groen HJ, Roder J,
Grigorieva J, Roder H, Dingemans AM and Smit EF:
VeriStrat® has prognostic value in advanced stage NSCLC
patients treated with erlotinib and sorafenib. Br J Cancer.
107:1820–1825. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Carbone DP, Ding K, Roder H, Grigorieva J,
Roder J, Tsao MS, Seymour L and Shepherd FA: Prognostic and
predictive role of the VeriStrat plasma test in patients with
advanced non-small-cell lung cancer treated with erlotinib or
placebo in the NCIC clinical trials group BR.21 tiral. J Thorac
Oncol. 7:1653–1660. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Milan E, Lazzari C, Anand S, Floriani I,
Torri V, Sorlini C, Gregorc V and Bachi A: SAA1 is over-expressed
in plasma of non small cell lung cancer patients with poor outcome
after treatment with epidermal growth factor receptor
tyrosine-kinase inhibitors. J Proteomics. 76:91–101. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Amann JM, Lee JW, Roder H, Brahmer J,
Gonzalez A, Schiller JH and Carbone DP: Genetic and proteomic
features associated with survival after treatment with erlotinib in
first-line therapy of non-small cell lung cancer in Eastern
Cooperative Oncology Group 3503. J Thorac Oncol. 5:169–178. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chung CH, Seeley EH, Roder H, Grigorieva
J, Tsypin M, Roder J, Burtness BA, Argiris A, Forastiere AA,
Gilbert J, et al: Detection of tumor epidermal growth factor
receptor pathway dependence by serum mass spectrometry in cancer
patients. Cancer Epidemiol Biomarkers Prev. 19:358–365. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bai H, Mao L, Wang HS, Zhao J, Yang L, An
TT, Wang X, Duan CJ, Wu NM, Guo ZQ, et al: Epidermal growth factor
receptor mutations in plasma DNA samples predict tumor response in
Chinese patients with stages IIIB to IV non-small-cell lung cancer.
J Clin Oncol. 27:2653–2659. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
He C, Liu M, Zhou C, Zhang J, Ouyang M,
Zhong N and Xu J: Detection of epidermal growth factor receptor
mutations in plasma by mutant-enriched PCR assay for prediction of
the response to gefitinib in patients with non-small-cell lung
cancer. Int J Cancer. 125:2393–2399. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jian G, Songwen Z, Ling Z, Qinfang D, Jie
Z, Liang T and Caicun Z: Prediction of epidermal growth factor
receptor mutations in the plasma/pleural effusion to efficacy of
gefitinib treatment in advanced non-small cell lung cancer. J
Cancer Res Clin Oncol. 136:1341–1347. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Mack PC, Holland WS, Burich RA, Sangha R,
Solis LJ, Li Y, Beckett LA, Lara PN Jr, Davies AM and Gandara DR:
EGFR mutations detected in plasma are associated with patient
outcomes in erlotinib plus docetaxel-treated non-small cell lung
cancer. J Thorac Oncol. 4:1466–1472. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kimura H, Kasahara K, Shibata K, Sone T,
Yoshimoto A, Kita T, Ichikawa Y, Waseda Y, Watanabe K, Shiarasaki
H, et al: EGFR mutation of tumor and serum in gefitinib-treated
patients with chemotherapy-naive non-small cell lung cancer. J
Thorac Oncol. 1:260–267. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kimura H, Suminoe M, Kasahara K, Sone T,
Araya T, Tamori S, Koizumi F, Nishio K, Miyamoto K, Fujimura M and
Nakao S: Evaluation of epidermal growth factor receptor mutation
status in serum DNA as a predictor of response to gefitinib
(IRESSA). Br J Cancer. 97:778–784. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kuang Y, Rogers A, Yeap BY, Wang L,
Makrigiorgos M, Vetrand K, Thiede S, Distel RJ and Jänne PA:
Noninvasive detection of EGFR T790M in gefitinib or erlotinib
resistant non-small cell lung cancer. Clin Cancer Res.
15:2630–2636. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen YM: Update of epidermal growth factor
receptor-tyrosine kinase inhibitors in non-small-cell lung cancer.
J Chin Med Assoc. 76:249–257. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shiau CJ, Babwah JP, da Cunha Santos G,
Sykes JR, Boerner SL, Geddie WR, Leighl NB, Wei C, Kamel-Reid S,
Hwang DM and Tsao MS: Sample features associated with success rates
in population-based EGFR mutation testing. J Thorac Oncol.
9:947–956. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Socinski MA, Villaruz LC and Ross J:
Understanding mechanisms of resistance in the epithelial growth
factor receptor in non-small cell lung cancer and the role of
biopsy at progression. Oncologist. 22:3–11. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Doebele RC, Pilling AB, Aisner DL,
Kutateladze TG, Le AT, Weickhardt AJ, Kondo KL, Linderman DJ,
Heasley LE, Franklin WA, et al: Mechanisms of resistance to
crizotinib in patients with AKL gene rearranged non-small cell lung
cancer. Clin Cancer Res. 18:1472–1482. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Travis WD, Brambilla E, Noguchi M,
Nicholson AG, Geisinger K, Yatabe Y, Powell CA, Beer D, Riely G,
Garg K, et al: International association for the study of lung
Cancer/American Thoracic Society/European respiratory society:
International multidisciplinary classification of lung
adenocarcinoma: Executive summary. Proc Am Thorac Soc. 8:381–385.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Ou SH, Bartlett CH, Mino-Kenudson M, Cui J
and Iafrate AJ: Crizotinib for the treatment of ALK-rearranged
non-small cell lung cancer: A success story to usher in the second
decade of molecular targeted therapy in oncology. Oncologist.
17:1351–1375. 2012. View Article : Google Scholar : PubMed/NCBI
|