Introduction
At present, surgical resection serves as the
preferred modality in cancer treatment, and cancer prognosis is
largely dictated by the stage of tumor metastasis (1). However, despite surgery by an
experienced surgeon, the diffusion of tumor cells into blood and
lymph circulation is inevitable (2).
It is accepted that tumor metastasis is determined by the
metastatic ability of tumor cells, and anti-metastatic actions,
such as surgical resection (3).
Numerous previous studies have demonstrated that tumor metastasis
is affected by various factors during surgery, including anesthesia
(4), acute pain (5) and stress reactions (6). Among these factors, the effect of
anesthetic drugs on tumor metastasis has attracted interest.
Certain previous studies (7,8) have
indicated that the drugs used in anesthesia may directly inhibit
cellular and humoral immunity, which may be associated with tumor
recurrence. However in other studies (9,10), the
effect of propofol treatment in suppressing the adhesion and
metastasis of breast cancer cells was revealed. Melamed et
al (11) compared the effects of
different general anesthetics on tumor-bearing rats via intravenous
injection. The results indicated that the number of tumor cells in
the lungs of the rats was increased 5.5 and 2-folds in the rats of
ketamine- and thiopentone-treated groups, respectively, but neither
propofol nor diazepam treatment demonstrated these results.
Nonetheless, the mechanism of propofol in the inhibition of tumor
metastasis and proliferation remains unknown.
It is well established that angiogenesis serves a
prerequisite role in cancer infiltration and metastasis, and that
cyclooxygenase-2 (COX-2) overexpression is closely associated with
angiogenesis in tumors (12). As the
rate-limiting enzyme involved in the conversion of arachidonic acid
into prostaglandin (PGs), COX-2 has been identified to be
overexpressed in multiple tumors, including gastrointestinal,
prostatic, lung and breast cancer (13–15). The
meta-analysis of 90 studies conducted by Harris (16) indicated that regular administration of
selective COX-2 inhibitors significantly reduced the incidence of
colon, breast, lung and prostatic cancer. As breast cancer has been
studied more extensively in recent years, the effects of COX-2 have
been increasingly highlighted (17,18).
Animal experiments have demonstrated that the overexpression of
COX-2 induced the genesis of breast cancer in transgenic mice
(19). Selective inhibition of COX-2
effectively reduced tumorigenesis in rats (20), which indicates the direct involvement
of COX-2 in the genesis of breast cancer.
In the present study, the inhibitory effects of two
anesthetics, propofol and ketamine, on MCF-7 cells were compared
and the effects of these two general anesthetics on cytokine
production in mice bearing MCF-7 tumors were investigated.
Materials and methods
Reagents and antibodies
Propofol, ketamine, microculture tetrazolium (MTT)
and celecoxib, which is a high selectively COX-2 inhibitor, used in
the present study were analytically or chemically pure, and were
purchased from Shanghai Sangon Biotechnology Co., Ltd. (Shanghai,
China). The human breast cancer MCF-7 cell line was obtained from
the American Type Culture Collection (Manassas, VA, USA). Fetal
bovine serum (FBS) and RPMI-1640 medium were provided by Gibco,
Thermo Fisher Scientific, Inc. (Waltham, MA, USA). The ELISA kit
was purchased from Shanghai BlueGene Biotech Co., Ltd. (Shanghai,
China). Rabbit anti-COX2 (catalog no. 10034) and mouse monoclonal
anti-PGE2 antibody (catalog no. 18219) were purchased from Cayman
Chemical Company (Ann Arbor, MI, USA) and were used as 1:300. The
mouse anti-β-actin (catalog no. A1978, 1:2,000 dilution), the mouse
monoclonal antibodies anti-VEGF (catalog no. V4758, 1:100 dilution)
and the anti-rabbit horseradish peroxidase (catalog no. P7899,
1:100,000 dilution) were provided by Sigma-Aldrich; Merck KGaA
(Darmstadt, Germany).
Cell culture and MTT assay
A 2×105/ml MCF-7 cell suspension was
prepared with RPMI-1640 culture medium containing 10% FBS, and was
transferred to a 96-well plate with 100 µl in each well. Then, the
cells were incubated at 37°C with 5% CO2 for 24 h to
obtain monolayer cells. Propofol, ketamine and celecoxib were
diluted in RPMI-1640 medium containing 10% FBS to concentrations of
10, 20, 40, 60, 80, 100, 120, 140, 160 and 180 µmol/l, and 100 µl
drug solution was added to each well. A total of 5 parallel wells
were set for each, and the medium containing no drugs was added in
the control group. Cell incubation for an additional 24 h was
performed at 37°C with 5% CO2. The culture medium was
subsequently removed and, the cells were washed three times with
cold PBS solution by centrifugation at 340 × g for 5 min at room
temperature. Following this, 180 µl fresh culture medium and 20 µl
of MTT solution (5 mg/ml) were added to the cells and incubated at
37°C with 5% CO2 for another 4 h. The crystallization
product was dissolved in 150 µl dimethyl sulfoxide subsequent to
the removal of the supernatant by centrifugation at 700 × g for 10
min at room temperature, and the cell survival rate was calculated
using the optical density (OD) value as determined by enzyme-linked
analyzer (Omega Bio-Tek, Inc., Norcross, GA, USA) at 490 nm. All
test samples were assayed in quadruplicate, and cell viability was
calculated using the following formula: Cell viability=(mean
absorbance of test wells-mean absorbance of medium control
wells)/(mean absorbance of untreated wells-mean absorbance of
medium control well) ×100%.
MCF-7 tumor-bearing nude mice
model
All animal experiments were performed in accordance
with the Animal Management Rules of the Ministry of Health of the
People's Republic of China and the guidelines of the Animal Care
and Use Committee of Jilin University (Changchun, China). Ethical
approval was obtained from the Ethics Committee of the China-Japan
Union Hospital of Jilin University. The nude mice were purchased
from Experimental Animal Center of Changchun Biological Institute
(Changchun, China), and kept under specific pathogen-free
conditions. The room temperature was maintained at 27±1°C and the
relative humidity was 40–60. The mice were fed three times a day
and were exposed to 10 h light per day.
A total of 48 female BALB/c nude mice (18–20 g, 4–5
weeks old) were equally divided into two groups: A propofol
treatment group and a ketamine treatment group. The mice in each
group were additionally divided into three subgroups and a control
group, and each mouse was inoculated with 5×106 living
MCF-7 cells subcutaneously in the left axilla. The suspension of
single MCF-7 cells was obtained following trypsin digestion of the
cells in the exponential growth phase. Intra-peritoneal
administration of the drugs at doses of 30, 60 and 80 mg/kg for
each subgroup was performed subsequent to the diameter of tumor
tissues reaching 0.5 cm. After 4 h of propofol and ketamine
administration, blood was collected through decapitation for 30
min. Then, the levels of IL-1, IL-6, IL-8 and tumor necrosis factor
(TNF)-α were determined using an ELISA kit according to the
protocol of the manufacturer.
ELISA assay
MCF-7 cells in the logarithmic growth phase were
obtained and seeded at 1~2×106 cells/well to a 6-well
plate. Cells were observed under a Leica DM6000B digital light
microscope (Leica Microsystems, Wetzlar, Germany) at 200×
magnification and viewed four fields (top left, bottom left, top
right, bottom right). Then, the cells were incubated for 24 h at
37°C, 5% CO2 and saturated humidity. The culture medium
was changed after 24 h, and then propofol and ketamine were added
at final concentrations of 0, 60, 120 and 180 µmol/l. After a 24
h-incubation at 37°C, the culture medium was removed and the
supernatant was collected by centrifugation at 700 × g for 10 min
at room temperature following trypsin digestion. Then, 0.5 ml
supernatant by centrifugation at 700 × g for 10 min was added into
0.1 ml HCl, and the mixture was centrifuged at 700 × g for 10 min
at room temperature to obtain the supernatant. Following this, 0.1
ml NaOH was added to neutralize the acidulated sample. A total of
100 µl standard substance or sample was added into the
corresponding wells according to manufacturer's instructions. OD
values at 490 nm were determined by using enzyme-linked analyzer
(Omega Bio-Tek, Inc.), and zero adjustment was performed in the
sample-free well, with 3 parallel wells set for each standard
substance and sample. This experiment was repeated three times.
Western blot assay
MCF-7 cells in the logarithmic growth phase were
obtained and seeded at a density of 1–2×106 cells/well
into a 6-well plate. Then, the cells were incubated for 24 h at
37°C and 5% CO2 and saturated humidity. The culture
medium was changed after 24 h, and then propofol, celecoxib and
ketamine were added at final concentrations of 180 µmol/l.
Following 24 h incubation at 37°C with 5% CO2, the
culture medium was removed and the supernatant was collected by
centrifugation at 700 × g for 5 min at room temperature following
trypsin digestion. The cells were pelleted at 700 × g for 5 min at
4°C and lysed in 50 µl cell lysis buffer (20 mM Tris, pH 7.4, 100
mM NaCl, 1% Triton, 1 mM phenylmethylsulfonyl fluoride, 10 µg/ml
leupeptin, 10 µg/ml aprotinin). The protein concentration was
determined by Bradford assay (Bio-Rad Laboratories, Inc., Hercules,
CA, USA). Equivalent amounts (50 µg) of protein were analyzed using
10% SDS-PAGE gels. The gels were then electro-blotted onto
polyvinylidene fluoride membranes. Following blocking with 5% milk
at 37°C for 2 h, membranes were incubated at 4°C with primary
antibody overnight as described previously in the reagents and
antibodies section. Finally, the relevant proteins were visualized
following incubation with the appropriate secondary horseradish
peroxidase-labeled antibody at 37°C for 1 h followed by
visualization using enhanced chemiluminescent agent (Fuzhou Maixin
Biotech Co., Ltd., Fuzhou, China). Densitometric scanning analysis
was performed using ImageJ software version 1.62 (National
Institutes of Health, Bethesda, MD, USA).
Statistical analysis
The experiments were performed in triplicate, and
the data are presented as the mean ± standard deviation. Data
between ≥3 groups were compared using one-way analysis of variance
followed by a post-hoc test (Dunnett's test) using SPSS software
package (version 13.0; SPSS, Inc., Chicago, IL, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
Inhibition of MCF-7 cells by propofol
and ketamine treatment as determined by MTT assay
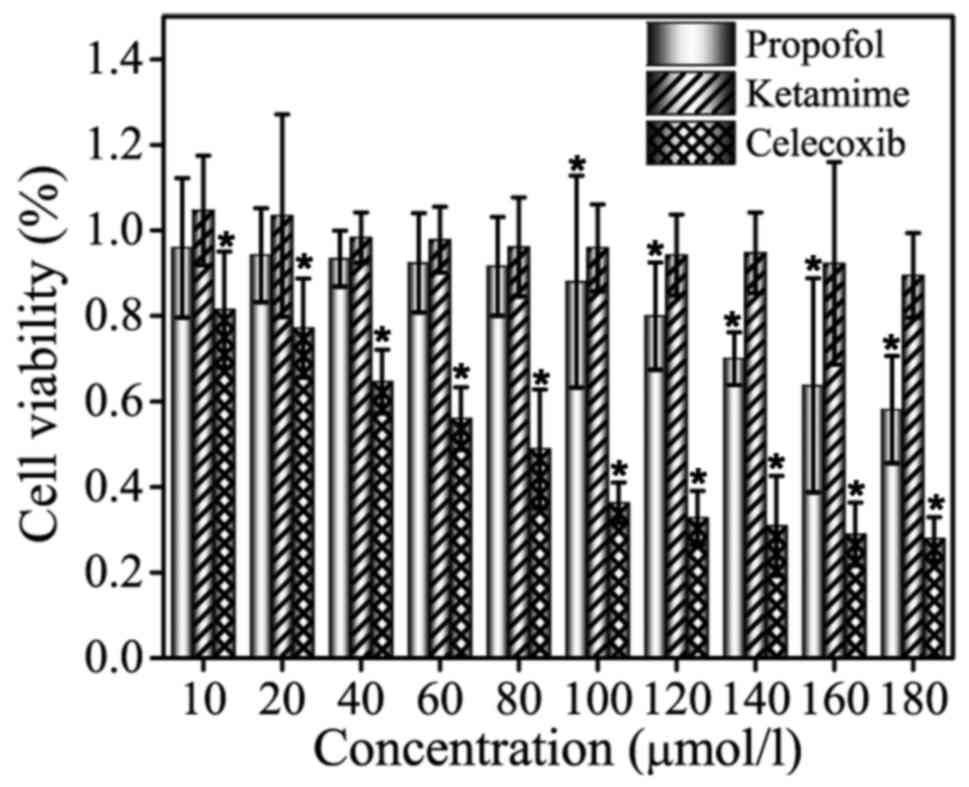
Celecoxib, propofol and ketamine were selected for
MTT assay at screening concentrations of 10, 20, 40, 60, 80, 100,
120, 140, 160 and 180 µmol/l, and the results are shown in Fig. 1. As indicated in the figure, no
significant inhibitory effect of ketamine treatment on cell
viability was observed in MCF-7 cells. Compared with group
ketamine, treatment with celecoxib exhibited a significant
inhibitory effect on MCF-7 cell viability (P<0.05). For
propofol, the inhibition was not evident at low drug concentrations
(10–80 µmol/l), whereas higher concentrations (100–180 µmol/l)
exhibited an inhibitory effect that was weaker compared with
celecoxib treatment. MCF-7 cells were inhibited by celecoxib and
propofol treatment in a dose-dependent manner, and the cell
survival rate decreased with increasing drug concentration. In
summary, the results validated that ketamine does not exhibit an
inhibitory effect on the proliferation of MCF-7 cells, and propofol
inhibits MCF-7 cells in a dose-dependent manner with the efficient
concentrations ranging between 100–180 µmol/l.
Analysis of changes in levels of
cytokines in MCF-7 tumor-bearing mice by ELISA assay
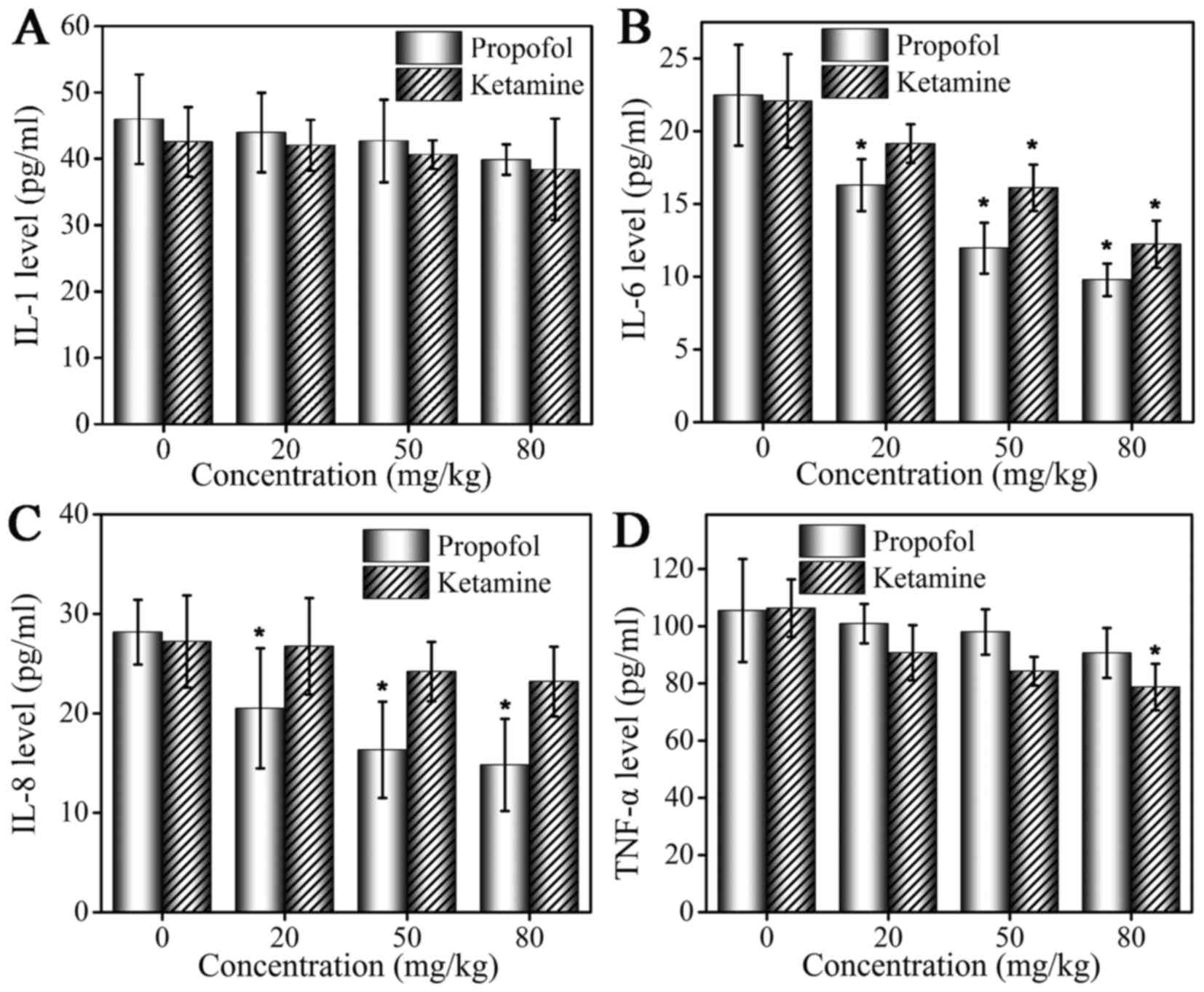
Serum levels of IL-1, IL-6, IL-8 and TNF-α following
administration of propofol and ketamine are demonstrated in
Fig. 2. At 4 h following anesthetic
administration, serum cytokine levels were altered in the mice in
the two groups. An ELISA assay was performed to detect levels of
cytokines. Compared with the control group, mice treated with
propofol and ketamine exhibited decreased serum levels of IL-1, but
the inhibition was weak and no significant difference was observed
(Fig. 2A). The level of IL-6 was
inhibited by propofol and ketamine, and a more marked inhibitory
effect was observed with increased drug concentrations. In
addition, propofol demonstrated a significantly stronger inhibition
effect compared with that of ketamine (Fig. 2B). The level of IL-8 was inhibited by
propofol at a low concentration (20 mg/kg), while ketamine did not
demonstrate a marked inhibitory effect on the level of IL-8
(Fig. 2C). The inhibition of TNF-α
production was observed in the ketamine and propofol groups but at
different levels. A marked inhibitory effect was observed in the
ketamine group, and a weaker inhibitory effect was observed in the
propofol group (Fig. 2D).
Inhibitory effects of propofol and
ketamine on COX-2 of MCF-7 cells determined by ELISA assay
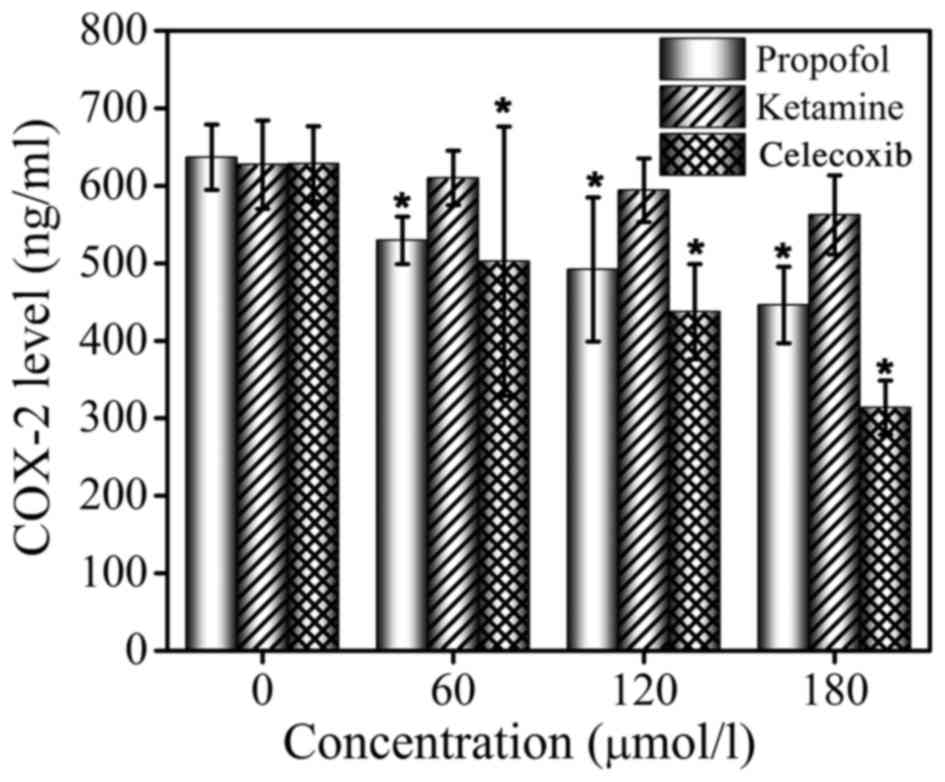
ELISA assays were performed to determine the
inhibition of COX-2 in MCF-7 cells by propofol, ketamine and
celecoxib. Celecoxib, a known COX-2 inhibitor, was used as the
positive control. The ELISA assay results are presented in Fig. 3. It was demonstrated that ketamine
treatment did not exhibit an inhibitory effect on COX-2 levels at
concentrations 0, 60, 120 or 180 µmol/l, while celecoxib and
propofol treatment significantly inhibited the production of COX-2
in a dose-dependent manner. It was identified that the release of
COX-2 was effectively inhibited by propofol, even though this
effect was less marked compared with that of celecoxib. The ELISA
assay confirmed that the expression of COX-2 protein was
significantly downregulated by propofol, but treatment with
ketamine did not demonstrate this effect, indicating that the
production of COX-2 may be inhibited by propofol.
Expression levels of PGE2 and VEGF in
MCF-7 cells determined by ELISA and western blot assays
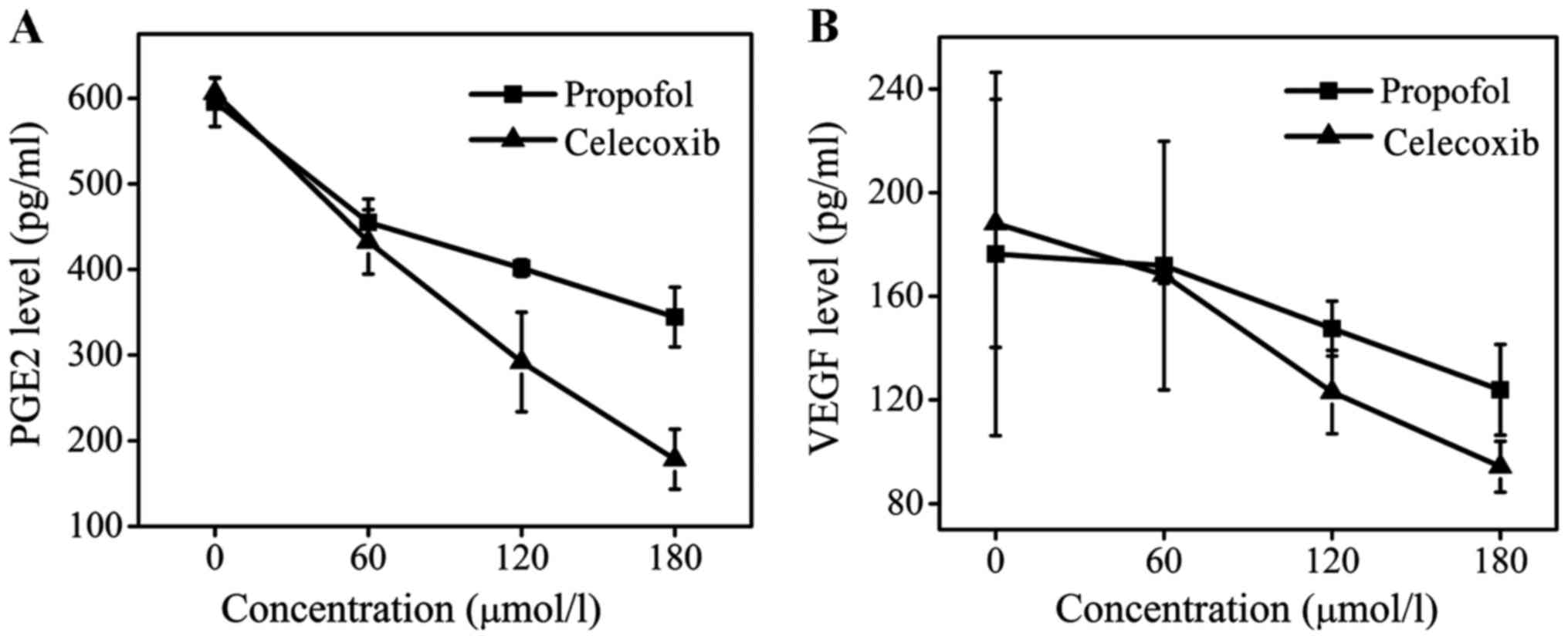
PGE2 is an important downstream protein of COX-2 and
is involved in the promotion of tumor cell growth and angiogenesis
(21–24). VEGF is also an important cytokine in
the genesis and development of tumors (25). The levels of PEG2 and VEGF following
celecoxib treatment were determined, and it was identified that
celecoxib treatment may effectively downregulate the expression of
PEG2 and VEGF (Fig. 4). At a dose of
180 µmol/l, celecoxib reduced the concentration of PEG2 ~3-fold
compared with the control group and decreased the level of VEGF to
100 pg/ml, whereas the level of VEGF in the propofol group was 130
pg/ml. The inhibitory effect of propofol treatment on the levels of
PEG2 and VEGF was less marked compared with that of celecoxib, but
remained evident. Compared with the control group, the levels of
PGE2 and VEGF in the supernatant of MCF-7 cells was decreased
following propofol treatment in a dose-dependent manner. Therefore,
it may be concluded that the effect of propofol on tumor growth and
development may be attributed to its ability to suppress the enzyme
activity of COX-2 and inhibit the release of PGE2 and VEGF.
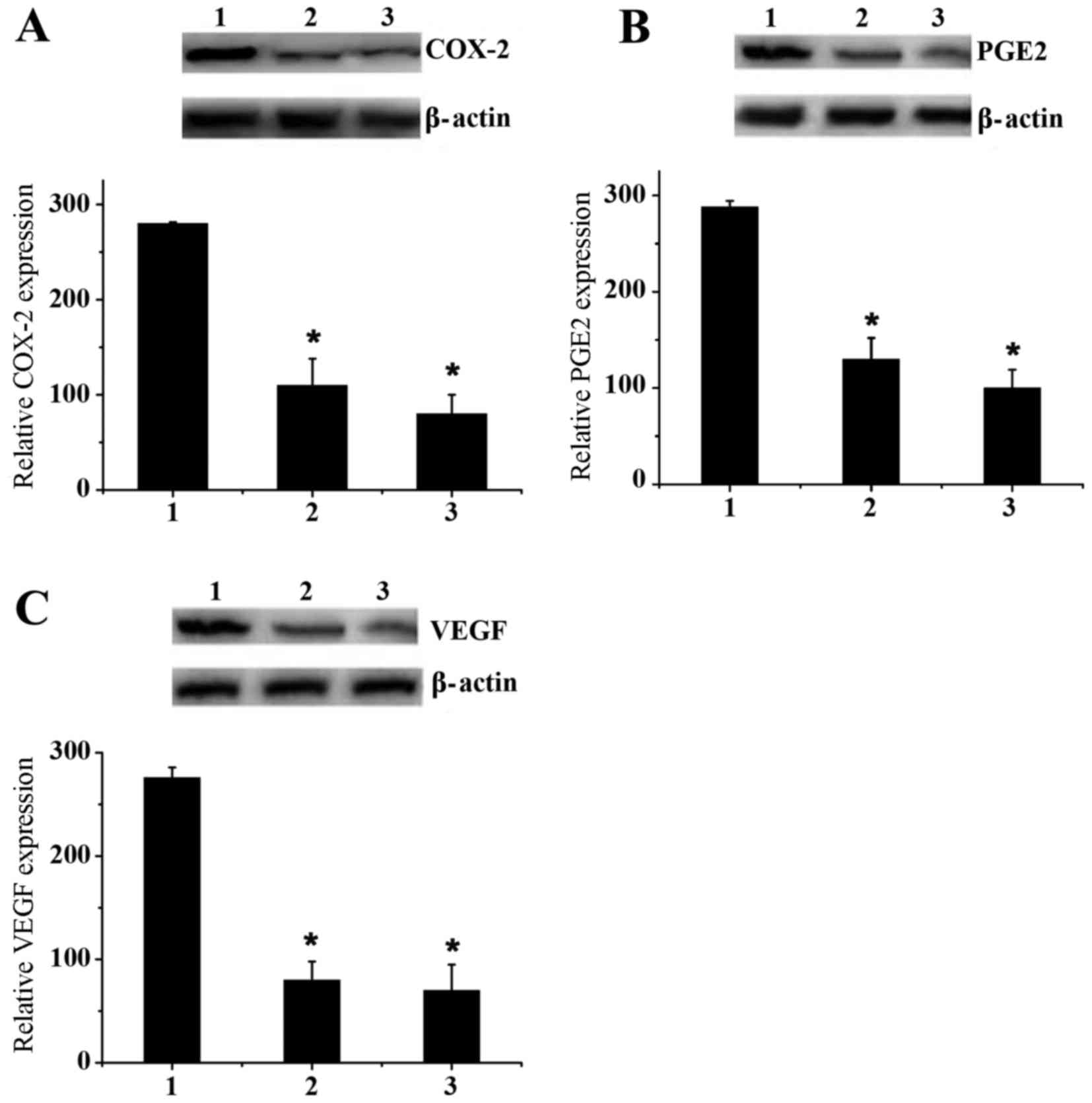
Western blot assays were performed to verify the
ELISA assay results (Fig. 5). MCF-7
cells were cultured ex vivo in 6-well plates with a dose of
180 µmol/l ketamine, propofol and celecoxib. Fig. 5A indicated that propofol effectively
downregulated the expression of COX-2 compared with ketamine,
although this effect was less marked than with celecoxib. Similar
results were observed for PGE2 and VEGF expression levels (Fig. 5B and C). These results effectively
confirmed the previous conclusion drawn by ELISA assay.
Discussion
Propofol (2,6-diisopropylphenol) is one of a number
of extensively used intravenous anesthetic agents that also exhibit
antitumor effects (26,27). Propofol, at clinically relevant
concentrations, may inhibit tumor invasion and result in apoptosis
of human cancer cells (28). For
example, Tsuchiya et al (29)
demonstrated that propofol treatment may induce the apoptosis of
human promyelocytic leukemia cells. Miao et al (30) has also shown that propofol is able to
significantly decrease the invasive activity of human colon
carcinoma cells. In animal studies (31,32),
propofol exerted antitumor activities by modulating immune
reaction. However, it is unclear whether propofol exhibits
inhibitory effects on tumor metastasis and proliferation. To the
best of our knowledge, this is the first study investigating the
mechanism of propofol in inhibiting MCF-7 tumor cell growth. It was
demonstrated that propofol inhibited MCF-7 cells in a
dose-dependent manner by inhibiting the expression of IL-6 and IL-8
and by downregulating COX-2 protein.
IL-6 is one of the cytokines with the most extensive
range of functions. IL-6 facilitates the genesis and development of
tumors primarily by regulating genes, which control cell cycle,
accelerate tumor angiogenesis, promote local inflammatory response
to tumor and enhance self-renewal of tumor stem cells (33). In the propofol treatment group, the
serum level of IL-6 was lower compared with that of the ketamine
treatment group, which may suggest a potential tumor inhibition
mechanism of propofol.
It was also noted from the present study that the
inhibitory effect of propofol treatment on the levels of IL-8 was
more marked compared with that of ketamine. This observation
suggested that the different inhibitory effects of propofol and
ketamine on tumor metastasis may be associated with differences in
IL-8 levels. Singh et al (34)
demonstrated the close interaction between IL-8 and COX-2 in bone
metastasis of breast cancer. The correlation between the expression
of IL-8 and COX-2 in breast cancer has also been highlighted by
numerous studies (34,35), and it has been suggested that IL-8 and
COX-2 are mutually regulated to promote the genesis and development
of tumor (36). Therefore, the
present study hypothesized that propofol may inhibit COX-2. The
ELISA assay results confirmed that the production of COX-2 may be
downregulated by propofol. As PGE2 is an important downstream
protein of COX-2 that promotes tumor cell growth and angiogenesis,
and VEGF is also an important cytokine in the genesis and
development of tumors, the present study also investigated whether
propofol exhibits inhibitory effects on PGE2 and VEGF expression by
ELISA and western blot assays. The results confirmed the
hypothesis.
In conclusion, the present study suggested that
propofol may suppress the proliferation of MCF-7 cells and
identified that propofol inhibited the expression of IL-6 and IL-8.
Subsequent to treatment with propofol, downregulated COX-2 protein
expression was observed in MCF-7 cells, and the levels of VEGF and
PGE2 in the supernatant were also decreased. Therefore, a potential
mechanism of propofol in inhibiting tumor development and
metastasis is the inhibition of the expression of IL-6, IL-8 and
COX-2. The present study provides original data, and hypothesizes
the antitumor mechanisms of propofol in MCF-7 cells. However, as
the number of tumor samples and the tumor grade were limited,
additional large-scale studies are required to explore the
antitumor mechanism of propofol.
Acknowledgements
The authors would like to thank other members in
Departments of Anesthesiology and Radiology, China-Japan Union
Hospital of Jilin University for valuable suggestions and
writing.
References
|
1
|
Jemal A, Siegel R, Ward E, Murray T, Xu J
and Thun MJ: Cancer statistic, 2007. CA Cancer J Clin. 57:43–66.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Eschwège P, Dumas F, Blanchet P, Le Maire
V, Benoit G, Jardin A, Lacour B and Loric S: Haematogenous
dissemination of prostatic epithelial cells during radical
prostatectomy. Lancet. 346:1528–1530. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Holmgren L, O'Reilly MS and Folkman J:
Dormancy of micrometastases: Balanced proliferation and apoptosis
in the presence of angiogenesis suppression. Nat Med. 1:149–153.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shapiro J, Jersky J, Katzav S, Feldman M
and Segal S: Anesthetic drugs accelerate the progression of
postoperative metastases of mouse tumors. J Clin Invest.
68:678–685. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Page GG, Blakely WP and Ben-Eliyahu S:
Evidence that post-operative pain is a mediator of the
tumor-promoting effects of surgery in rats. Pain. 90:191–199. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ben-Eliyahu S: The price of anticancer
intervention. Does surgery promote metastasis? Lancet Oncol.
3:578–579. 2002.PubMed/NCBI
|
|
7
|
Biki B, Mascha E, Moriarty DC, Fitzpatrick
JM, Sessler DI and Buggy DJ: Anesthetic technique for radical
prostatectomy surgery affects cancer recurrence: A retrospective
analysis. Anesthesiology. 109:180–187. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tsui BC, Rashiq S, Schopflocher D, Murtha
A, Broemling S, Pillay J and Finucane BT: Epidural anesthesia and
cancer recurrence rates after radical prostatectomy. Can J Anaesth.
57:107–112. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Forget P, Vandenhende J, Berliere M,
Machiels JP, Nussbaum B, Legrand C and De Kock M: Do intraoperative
analgesics influence breast cancer recurrence after mastectomy? A
retrospective analysis. Anesth Analg. 110:1163–1635. 2010.
View Article : Google Scholar
|
|
10
|
Chen X, Lu P, Chen L, Yang SJ, Shen HY, Yu
DD, Zhang XH, Zhong SL, Zhao JH and Tang JH: Perioperative
propofol-paravertebral anesthesia decreases the metastasis and
progression of breast cancer. Tumour Biol. 36:8259–8266. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Melamed R, Bar-Yosef S, Shakhar G, Shakhar
K and Ben-Eliyahu S: Suppression of natural killer cell activity
and promotion of tumor metastasis by ketamine, thiopental, and
halothane, but not by propofol: Mediating mechanisms and
prophylactic measures. Anesth Analg. 97:1331–1339. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kirkpatrick K, Ogunkolade W, Elkak A,
Bustin S, Jenkins P, Ghilchik M and Mokbel K: The mRNA expression
of cyclo-oxygenase-2 (COX-2) and vascular endothelia growth factor
(VEGF) in human breast cancer. Curr Med Res Opin. 18:237–241. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang SH, Liu CH, Conway R, Han DK,
Nithipatikom K, Trifan OC, Lane TF and Hla T: Role of prostaglandin
E2-dependent angiogenic switch in cyclooxygenase 2-induced breast
cancer progression. Proc Natl Acad Sci USA. 101:591–596. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Simeone AM, Li YJ, Broemeling LD, Johnson
MM, Tuna M and Tari AM: Cyclooxygenase-2 is essential for HER-2/neu
to suppress N-(4-hydroxyphenyl)retinamide apoptotic effects in
breast cancer cells. Cancer Res. 64:1224–1228. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Barnes N, Haywood P, Flint P, Knox WF and
Bundred NJ: Survivin expression in in situ and invasive breast
cancer relates to COX-2 expression and DCIS recurrence. Br J
Cancer. 94:253–258. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Harris RE: Cyclooxygenase-2 (cox-2)
blockade in the chemoprevention of cancers of the colon, breast,
prostate, and lung. Inflammopharmacology. 17:55–67. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Qin G, Xu F, Qin T, Zheng Q, Shi D, Xia W,
Tian Y, Tang Y, Wang J, Xiao X, et al: Palbociclib inhibits
epithelial-mesenchymal transition and metastasis in breast cancer
via c-Jun/COX-2 signaling pathway. Oncotarget. 6:41794–41808. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hugo HJ, Saunders C, Ramsay RG and
Thompson EW: New insights on COX-2 in chronic inflammation driving
breast cancer growth and metastasis. J Mammary Gland Biol
Neoplasia. 20:109–119. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu CH, Chang SH, Narko K, Trifan OC, Wu
MT, Smith E, Haudenschild C, Lane TF and Hla T: Overexpression of
cyclooxygenase-2 is sufficient to induce tumorigenesis in
transgenic mice. J Biol Chem. 267:18563–18569. 2001. View Article : Google Scholar
|
|
20
|
Harris RE, Kasbari S and Farrar WB:
Prospective study of nonsteroidal anti-inflammatory drugs and
breast cancer. Oncol Rep. 6:71–73. 1999.PubMed/NCBI
|
|
21
|
Joo YE, Rew JS, Seo YH, Choi SK, Kim YJ,
Park CS and Kim SJ: Cyclooxygenase-2 overexpression correlates with
vascular endothelial growth factor expression and tumor
angiogenesis in gastric cancer. J Clin Gastroenterol. 37:28–33.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wise H: Lack of interaction between
prostaglandin E2 receptor subtypes in regulating adenylyl cyclase
activity in cultured rat dorsal root ganglion cells. Eur J
Pharmacol. 535:69–77. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Komuro M, Kamiyama M, Furuya Y, Takihana
Y, Araki I and Takeda M: Gene and protein expression profiles of
prostaglandin E2 receptor subtypes in the human corpus cavernosum.
Int J Impot Res. 18:275–281. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Deasy BM, O'Sullivan-Coyne G, O'Donovan
TR, McKenna SL and O'Sullivan GC: Cyclooxygenase-2 inhibitors
demonstrate anti-proliferative effects in oesophageal cancer cells
by prostaglandin E(2)-independent mechanisms. Cancer Lett.
256:246–258. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kellesarian SV, Al-Kheraif AA, Vohra F,
Ghanem A, Malmstrom H, Romanos GE and Javed F: Cytokine profile in
the synovial fluid of patients with temporomandibular joint
disorders: A systematic review. Cytokine. 77:98–106. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ren XF, Li WZ, Meng FY and Lin CF:
Differential effects of propofol and isoflurane on the activation
of T-helper cells in lung cancer patients. Anaesthesia. 65:478–482.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Miyata T, Kodama T, Honma R, Nezu Y,
Harada Y, Yogo T, Hara Y and Tagawa M: Influence of general
anesthesia with isoflurane following propofol-induction on natural
killer cell cytotoxic activities of peripheral blood lymphocytes in
dogs. J Vet Med Sci. 75:917–921. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mammoto T, Mukai M, Mammoto A, Yamanaka Y,
Hayashi Y, Mashimo T, Kishi Y and Nakamura H: Intravenous
anesthetic, propofol inhibits invasion of cancer cells. Cancer
Lett. 184:165–170. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tsuchiya M, Asada A, Arita K, Utsumi T,
Yoshida T, Sato EF, Utsumi K and Inoue M: Induction and mechanism
of apoptotic cell death by propofol in HL-60 cells. Acta
Anaesthesiol Scand. 46:1068–1074. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Miao Y, Zhang Y, Wan H, Chen L and Wang F:
GABA-receptor agonist, propofol inhibits invasion of colon
carcinoma cells. Biomed Pharmacother. 64:583–588. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kushida A, Inada T and Shingu K:
Enhancement of antitumor immunity after propofol treatment in mice.
Immunopharmacol Immunotoxicol. 29:477–486. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Inada T, Kubo K and Shingu K: Possible
link between cyclooxygenase-inhibiting and antitumor properties of
propofol. J Anesth. 25:569–575. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tchirkov A, Khalil T, Chautard E, Mokhtari
K, Véronèse L, Irthum B, Vago P, Kémény JL and Verrelle P:
Interleukin-6 gene amplification and shortened survival in
glioblastoma patients. Br J Cancer. 96:474–476. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Singh B, Berry JA, Vincent LE and Lucci A:
Involvement of IL-8 in COX-2-mediated bone metastases from breast
cancer. J Surg Res. 134:44–51. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chou WY, Chuang KH, Sun D, Lee YH, Kao PH,
Lin YY, Wang HW and Wu YL: Inhibition of PKC-Induced COX-2 and IL-8
expression in human breast cancer cells by glucosamine. J Cell
Physiol. 230:2240–2251. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Simeone AM, Nieves-Alicea R, McMurtry VC,
Colella S, Krahe R and Tari AM: Cyclooxygenase-2 uses the protein
kinase C/interleukin-8/urokinase-type plasminogen activator pathway
to increase the invasiveness of breast cancer cells. Int J Oncol.
30:785–792. 2007.PubMed/NCBI
|