Introduction
Liver cancer is one of the most common digestive
system malignancies in clinical practice. The occurrence and
development of liver cancer is a complex process with various
factors and pathways involved. With the characteristics of occult
onset, fast progress and high mortality, 5-year survival rate of
patients with this disease is <10% (1,2). Liver
cancer mainly affects elderly population, and incidence is higher
in men than in women. In recent years, age of onset is becoming
younger and younger (3). There are no
typical clinical symptoms of early stages of liver cancer, and most
patients were diagnosed at advanced stages, so to improve the
diagnosis of early liver cancer is particularly important (4). Clinical diagnostic methods for liver
cancer include clinical manifestations, laboratory tests and
imaging studies. Imaging studies include ultrasound, CT and MRI,
within which the diagnosis rate of CT and MRI is high (5,6). With the
improvements in contrast-enhanced ultrasound (CEUS), CEUS now can
be used to observe blood perfusion of liver tissue in real-time,
with high sensitivity and accuracy. Therefore, CEUS has become one
of the most commonly used methods for the diagnosis of liver cancer
(7). Radiofrequency ablation (RFA) is
a non-invasive treatment that has been widely used in clinical
practices (8). Enhanced CT can be
used to rapidly scan images without blind spots, so it is generally
used to evaluate the efficacy of RFA. CEUS can also timely and
accurately evaluate the efficacy of RFA (9). In this study, CEUS and enhanced CT were
used in the diagnosis of liver cancer and evaluation of efficacy of
RFA. Consistency between the two methods was analyzed.
Materials and methods
General information
A total of 60 patients with liver cancer were
selected from April 2016 to May 2017 in Dongying People's Hospital
(Dongying, China). All patients were subjected to CEUS and enhanced
CT, and finally diagnosed by pathological examination. All patients
underwent EFA, and CEUS and CT were used to evaluate curative
effect. The general information of these patients are presented in
Table I. The study was approved by
the Ethics Committee of Dongying People's Hospital (Dongying,
China). Signed informed consents were obtained from the
patients.
 | Table I.General information of patients. |
Table I.
General information of patients.
| Items | Patients (n=60) |
|---|
| Male (n, %) | 38 (63.33) |
| Female (n, %) | 22 (36.67) |
| Age (years) | 45–79 |
| Average age
(years) | 58.56±9.43 |
| Category (n, %) |
|
| Primary | 52 (86.67) |
| Metastatic | 8
(13.33) |
| Lesions
classification (n, %) |
|
| Single lesion | 55 (91.67) |
| Multiple lesions | 5 (8.33) |
Method
Preparation before examination
Patients were fasted for 6 h before CT scan. All
patients were informed with all details in the whole procedure.
Breathing training (breathe slowly) was performed to avoid the
effects of changes in respiratory rate on image quality.
Enhanced CT examination
Patients were fixed in supine position and both
hands were placed on a headrest. Dual Source CT scanner (Siemens
AG, Munich, Germany) was used for scanning. Non-ionic iodine
contrast agent iohexol (300 mg I/ml; Bayer Schering Pharma AG,
Berlin, Germany) was injected (3.0 ml/sec) into the elbow vein of
patients as a dose of 1.5 ml/kg. Dynamic enhanced scanning was
performed at sub-arterial phase (20–30 sec after injection of
iohexol), intravenous phase (60–70 sec after injection of iohexol)
and balance phase (150–240 sec after injection of iohexol). All
images were sent to the workstation.
CEUS
CEUS was performed using Philips iU22 color Doppler
system (Philips Healthcare, Amsterdam, The Netherlands). Probe
frequency, 3.0 MHz; harmonic frequency, 3.5 MHz; speed range,
0.06–0.12 m/sec. Contrast agent SonoVue (registration no.
H20080059; Bracco, Milan, Italy) was used. One dose of SonoVue was
mixed with 5 ml of 9% sodium chloride solution to make a suspension
(density of sulfur hexafluoride microbubbles was
2×108/ml). Ultrasonic probe section was fixed in the
target area. Contrast agent was injected into peripheral vein. When
contrast agent reached the target, lesions were scanned through
fan-shaped approach, and contrast agent enhancement and perfusion
were timely observed.
Treatment
After local anesthesia, cool-tip RFA needles were
inserted through the marked skin. The number of needles was
determined by the size of the lesion: 2 needles were used if the
tumor diameter was >3.0 cm, otherwise only 1 needle was used.
LDRF-120S multipole RFA instrument (Mianyang Lide Electronic
Technology Co., Ltd., Mianyang, China) was used to treat the lesion
after the tip of the needle reached the bottom of the tumor.
Treatment was monitored under ultrasound, and the whole process was
between 10 and 20 min.
Evaluation method
Examination results were analyzed by two senior
diagnostic imaging physicians with >10 years of work experience
using double-blind method. Another imaging physician was included
in case of inconsistence. With pathological examination results as
gold standard, accuracies of CEUS and enhanced CT were compared.
Maximum section area was measured by CEUS and enhanced CT at 1 and
3 months after treatment. Measurement was performed 3 times and the
average value was calculated. Cross-sectional area was calculated
according to the following formula: S = l/4πXY (X, left and right
diameter; Y, front and back diameter).
Evaluation standard of RFA efficacy was as follows
(10): i) Completely effective:
Fibroid volume reduction rate of ≥50%; ⅱ) partially effective:
Fibroid volume reduction rate of <50%; and ⅲ) invalid: Fibroid
recurrence or fibroid volume increased. Fibroid volume reduction
rate = (fibroid volume before treatment - fibroid volume after
treatment) / fibroid volume before treatment −100%. Treatment
effective rate = partially effective rate + partial effective
rate.
Statistical analysis
Statistical software SPSS 19.0 (SPSS, Inc., Chicago,
IL, USA) was used. Measurement data were expressed as mean ±
standard deviation (mean ± SD) and processed using t-test.
Enumeration data were expressed as number or (%) and processed
using χ2 test. ANOVA was used for comparison between
multiple groups and the post hoc test was Dunnett's test.
Consistency of efficacy evaluation was analyzed using Bland-Altman
test. Correlation between the two different examination methods was
analyzed by Linear regression analysis, and the closer the value of
r2 was to 1, the higher the correlation value between
the two methods was. Area under the ROC curve, and sensitivity and
specificity of two methods were analyzed, and the significance
level of test was α=0.05.
Results
Comparison of the accuracy of two
methods for the diagnosis of liver cancer
No significant differences in the accuracy of the
two methods for the diagnosis of liver cancer were found
(p>0.05; Table II).
 | Table II.Comparison of the accuracy of CEUS and
enhanced CT for the diagnosis of liver cancer (n, %). |
Table II.
Comparison of the accuracy of CEUS and
enhanced CT for the diagnosis of liver cancer (n, %).
| Methods | Confirmed
diagnosis | Misdiagnosis | Erroneous
diagnosis | Diagnostic
accuracy |
|---|
| CEUS | 53 (88.33) | 3 (5.00) | 4 (6.67) | 53 (88.33) |
| Enhanced CT | 55 (91.67) | 2 (3.33) | 3 (5.00) | 55 (91.67) |
| χ2 |
|
|
| 0.093 |
| P-value |
|
|
| 0.761 |
Comparison of area under the ROC curve
of CEUS and enhanced CT
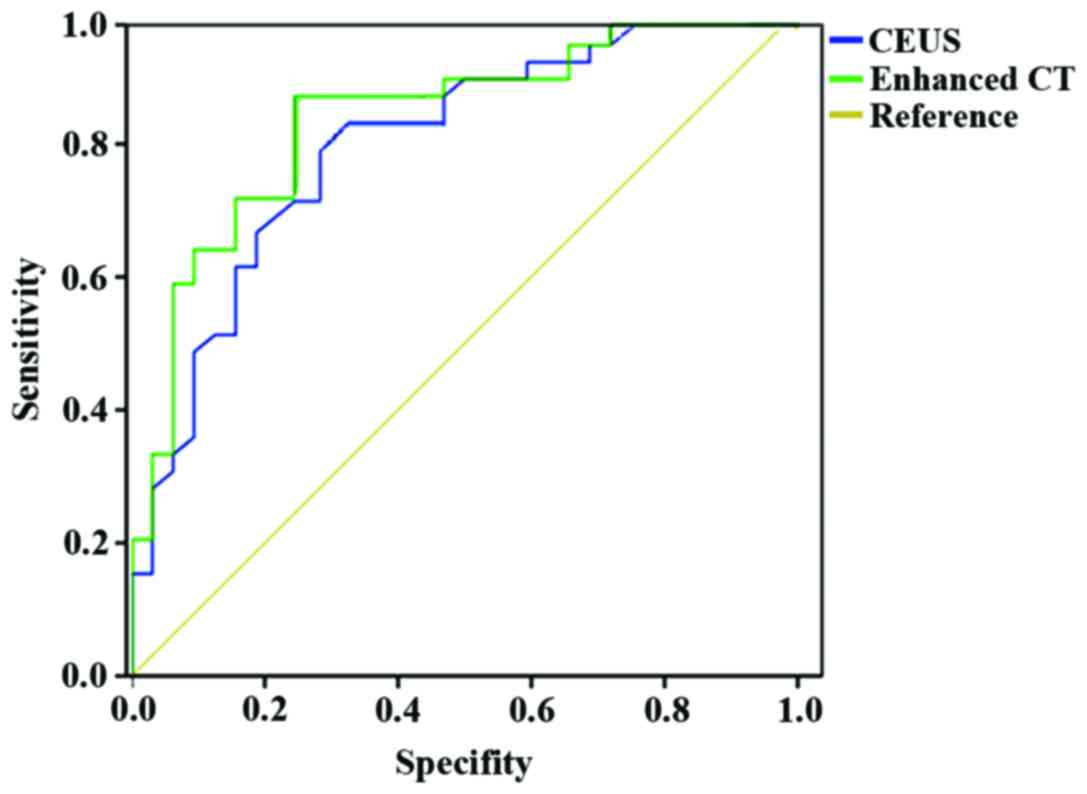
Area under the ROC curve of CEUS was 0.896 with a
sensitivity of 90.2% and a specificity of 88.7%. The area under the
ROC curve for enhanced CT diagnosis was 0.907 with a sensitivity of
91.8% and a specificity of 89.7%. Detailed results are presented in
Fig. 1.
Comparison of maximum tumor
cross-sectional area measured by two methods
There was no significant difference in maximum tumor
cross-sectional area measured by the two methods at 1 and 3 months
after treatment (p>0.05). As shown in Table III.
 | Table III.Comparison of the maximum tumor
cross-sectional area measured by two methods (mean±SD,
cm2). |
Table III.
Comparison of the maximum tumor
cross-sectional area measured by two methods (mean±SD,
cm2).
| Methods | Cases | Before treatment | 1 month after
treatment | 3 months after
treatment |
|---|
| CEUS | 60 | 5.56±1.36 | 3.47±1.07 | 1.15±0.52 |
| Enhanced CT | 60 | 5.68±1.74 | 3.51±1.09 | 1.13±0.54 |
| t-test |
| 0.421 | 0.203 | 0.517 |
| P-value |
| 0.675 | 0.839 | 0.606 |
Comparison of the evaluation of RFA
efficacy by two methods
No significant differences were found in evaluation
of RFA efficacy by two methods at 1 and 3 months after treatment
(p>0.05). As shown in Table
IV.
 | Table IV.Comparison of the evaluation of RFA
efficacy by two methods (n, %). |
Table IV.
Comparison of the evaluation of RFA
efficacy by two methods (n, %).
| Methods | Cases | 1 month after
treatment | 3 months after
treatment |
|---|
| CEUS | 60 | 49 (81.67) | 56 (93.33) |
| Enhanced CT | 60 | 47 (78.33) | 59 (98.33) |
| t-test |
| 0.052 | 0.835 |
| P-value |
| 0.819 | 0.361 |
Analysis of the consistency of CEUS
and enhanced CT in measuring maximum tumor cross-sectional
area
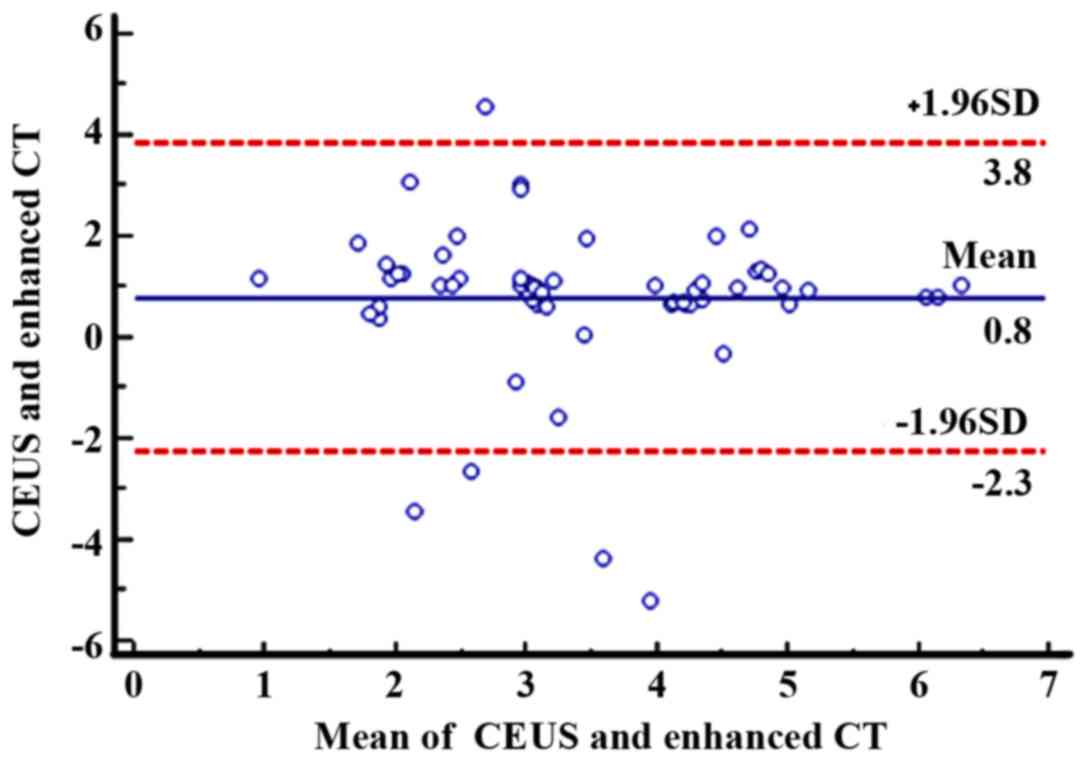
Bland-Altman test showed that, at 1 month after
treatment, as can be seen from Fig.
2, the mean of the difference between the paired data of 60
patients was 0.8 cm2, and the 95% agreement margin was
3.8 to −2.3 cm2, and 8.33% (5/60) points are outside the
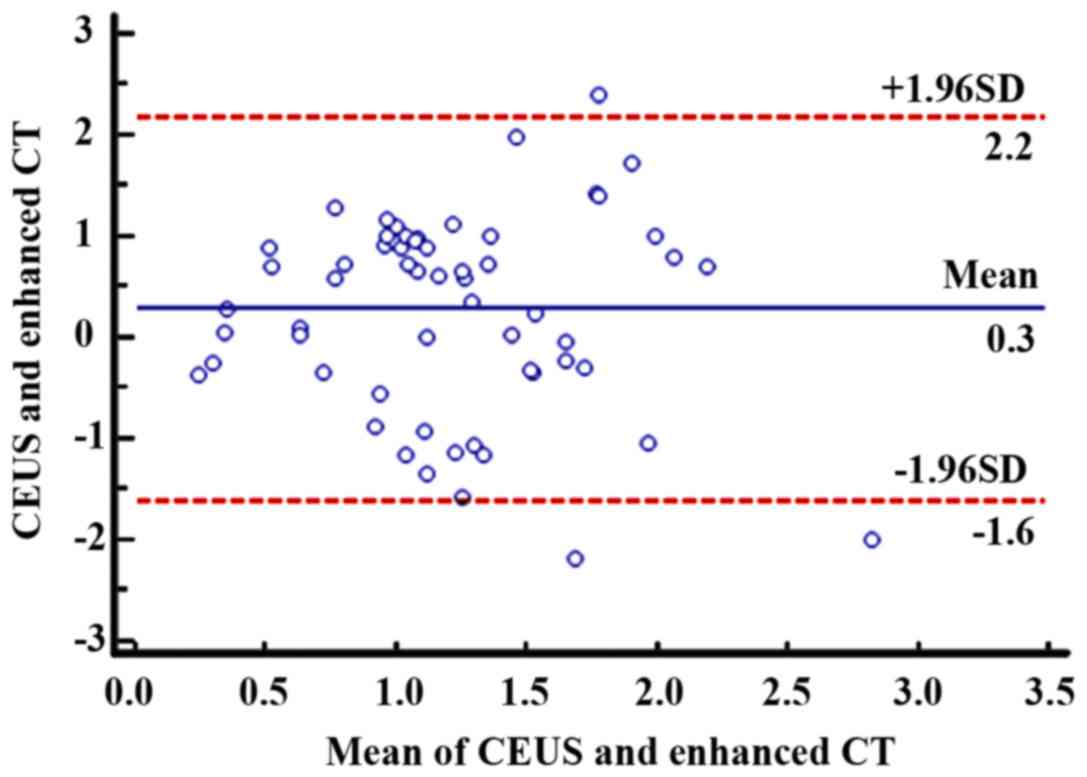
95% consistency limit. At 3 months after treatment, as can be seen
from Fig. 3, the mean of the paired
data differences for the 60 patients was 0.3 cm2, and
the 95% agreement margin was 2.2 to −1.6 cm2, and 5%
(3/60) points were outside the 95% agreement limits, indicating a
strong agreement between the two methods.
Correlation analysis of CEUS and
enhanced CT in measuring maximum cross-sectional area after
treatment
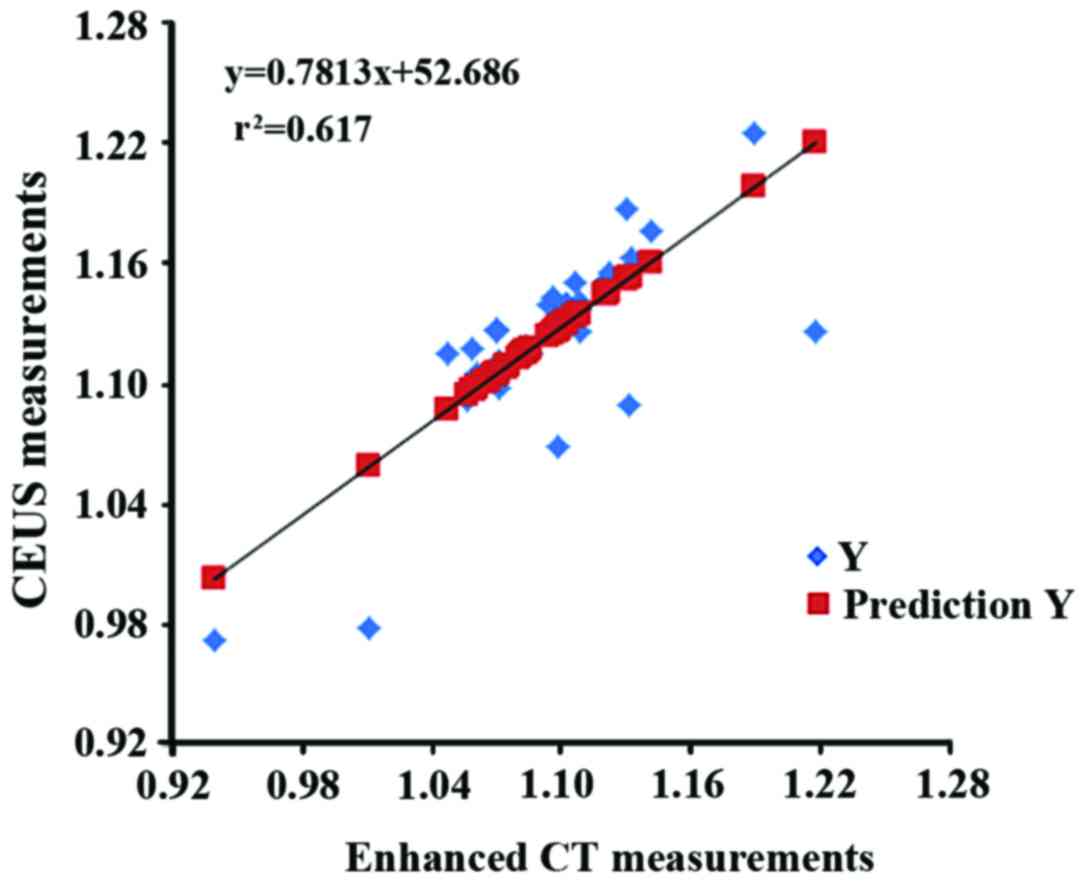
Pearson's correlation analysis equation was: Y =
0.7813X + 52.686 (Y, CEUS measurement; X, enhanced CT measurement).
Pearson's correlation analysis showed a strong correlation between
CEUS measurements and enhanced CT measurements
(r2=0.617, Fig. 4).
Discussion
Liver cancer often occurs in the intrahepatic bile
duct or liver cells, of which the former can account for >80% of
the cases, and its incidence and mortality ranks in the forefront
of all malignancies (11). Clinical
manifestations of liver cancer patients include loss of appetite,
liver pain, weight loss, jaundice and ascites, and even coma and
systemic failure (12). Liver cancer
can be caused by a variety of factors including viral infection,
cirrhosis, alcohol and tobacco consumption, chemical carcinogens,
drinking water pollution and genetic factors (13). Pathogenesis of liver cancer is
complex, and its occurrence is usually the result of long-term
cumulative changes in multiple factors. Interaction between
environmental factors and genetic polymorphisms plays an important
role in the occurrence and development of liver cancer (14). Attention should be paid to the regular
screening of liver cancer to achieve early detection, and early
diagnosis and early treatment are of great significance to improve
the prognosis and reduce the mortality of liver cancer.
With the rapid development of medical imaging
technology, it plays an important role in detecting,
characterizing, locating and staging liver cancer. Its diagnostic
value far exceeds that of serology and has been widely used
clinically. CT scanning has the characteristics of fast scanning,
clear image and minimal influence from the surrounding organs. With
the emergence of multi-slice CT, scanning time becomes even
shorter, motion artifact interference is even less, Z-axis
resolution and the diagnostic accuracy are even higher (15). Enhanced CT scans through iohexol can
not only effectively detect liver cancer lesions, but also improve
the accuracy of its qualitative judgment, which in turn increase
liver cancer detection rate (16).
Results of this study showed that, compared with the results of
pathological diagnosis, the accuracy of enhanced CT in the
diagnosis of liver cancer was 91.67%, area under the ROC curve was
0.907, with a sensitivity of 91.8% and a specificity of 89.7%.
Diagnostic accuracy of CEUS was 91.67%. Area under ROC curve was
0.896, the sensitivity was 90.2% and specificity was 88.7%. No
significant differences in diagnostic accuracy was found between
the two methods (p>0.05), and diagnostic accuracy of both
methods was high. This is because enhanced scan in three phases
performed according to the three kinds of blood supply
characteristics of liver cancer (hepatic artery blood supply,
double hepatic artery and portal vein blood supply to portal
vein-based hepatic artery less blood supply) can obtain data of
arterial, stable and delayed phases of liver tissue and get
information on the internal tumor hemodynamic changes, which can
accurately reflect the characteristics of the blood supply, so as
to improve the accuracy of diagnosis of liver cancer (17). With the application of contrast agent,
diagnostic level of ultrasound is improved. CEUS can objectively
display the shape, size and number of tumors, and at the same time,
it can reflect the characteristics of blood flow inside the tumor
(18), which is similar to the
function of enhanced CT. CEUS can be an alternative to enhanced CT
in the diagnosis of liver cancer, and can provide a reliable basis
for treatment of liver cancer.
Treatment of liver cancer with RFA is easy and
repeatable without radioactive injury. The principle is: Ηigh
frequency AC electromagnetic waves generate biological energy to
heat the lesions to reach 43–60°C, so as to cause tumor cell
protein denaturation and tissue coagulation necrosis. At the same
time, it can also cause coagulation reaction around the tumor,
which in turn inhibits tumor metastasis without inducing
significant damage to surrounding healthy tissues (19). After treatment, the curative effect
needs to be evaluated to determine whether it is necessary to
repeat the treatment. Clinical evaluation of RFA is usually
performed by imaging examination, which can include ultrasound, CT
and MRI (20). The results of this
study showed that there was no difference in evaluating efficacy of
RFA between CEUS and enhanced CT, and results of Bland-Altman test
showed that there was a strong consistency between CEUS and CT.
Linear regression analysis showed a strong linear correlation
between the two methods (r2=0.617). The data suggest
that CEUS can completely reflect the degree of tumor necrosis after
RFA treatment, so as to avoid residual tumor recurrence after RFA
due to inaccurate assessment, and its evaluation efficiency is
comparable to that of enhanced CT. Clinical application of CT has a
certain degree of radioactive damage and imaging section is fixed,
while CEUS has the characteristics of convenient and rapid
operation, non-radiation, non-space and time constraints, and high
repeatability. So its clinical value is higher than that of
CEUS.
In summary, CEUS and enhanced CT have high accuracy
and consistency in diagnosis if liver cancer and evaluation of RFA
efficacy, they can both provide a reliable basis for the diagnosis
and treatment of liver cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
ShuhongK and XY conceived and designed the study.
ShengK and YR were responsible for the collection, analysis and
interpretation of the data. ShuhongK drafted the manuscript. YR
revised the manuscript critically for important intellectual
content. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Dongying People's Hospital (Dongying, China). Signed informed
consents were obtained from the patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Younossi ZM, Otgonsuren M, Henry L,
Venkatesan C, Mishra A, Erario M and Hunt S: Association of
nonalcoholic fatty liver disease (NAFLD) with hepatocellular
carcinoma (HCC) in the United States from 2004 to 2009. Hepatology.
62:1723–1730. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lee JM, Park JW and Choi BI: 2014
KLCSG-NCC Korea Practice Guidelines for the management of
hepatocellular carcinoma: HCC diagnostic algorithm. Dig Dis.
32:764–777. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kew MC: Hepatocellular carcinoma:
Epidemiology and risk factors. J Hepatocell Carcinoma. 1:115–125.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hann HW, Coben R, Brown D, Needleman L,
Rosato E, Min A, Hann RS, Park KB, Dunn S and DiMarino AJ: A
long-term study of the effects of antiviral therapy on survival of
patients with HBV-associated hepatocellular carcinoma (HCC)
following local tumor ablation. Cancer Med. 3:390–396. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vardal J, Salo RA, Larsson C, Dale AM,
Holland D, Groote IR and Bjørnerud A: Correction of B0-distortions
in echo-planar-imaging-based perfusion-weighted MRI. J Magn Reson
Imaging. 39:722–728. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yu MH, Kim JH, Yoon JH, Kim HC, Chung JW,
Han JK and Choi BI: Small (≤1-cm) hepatocellular carcinoma:
Diagnostic performance and imaging features at gadoxetic
acid-enhanced MR imaging. Radiology. 271:748–760. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jiang N, Xie B, Zhang X, He M, Li K, Bai
J, Wang Z, He J and Zhang L: Enhancing ablation effects of a
microbubble-enhancing contrast agent (‘SonoVue’) in the treatment
of uterine fibroids with high-intensity focused ultrasound: A
randomized controlled trial. Cardiovasc Intervent Radiol.
37:1321–1328. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bends R, Brössner A, Felberbaum R and
Römer T: Myoma in statu nascendi after transcervical radiofrequency
ablation of a transmural uterine leiomyoma. Gynakologische
Endokrinologie. 14:291–294. 2016.(In German). View Article : Google Scholar
|
|
9
|
Wang W, Liu JY, Yang Z, Wang YF, Shen SL,
Yi FL, Huang Y, Xu EJ, Xie XY, Lu MD, et al: Hepatocellular
adenoma: Comparison between real-time contrast-enhanced ultrasound
and dynamic computed tomography. Springerplus. 5:9512016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sung JY, Baek JH, Jung SL, Kim JH, Kim KS,
Lee D, Kim WB and Na DG: Radiofrequency ablation for autonomously
functioning thyroid nodules: A multicenter study. Thyroid.
25:112–117. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikai I, Arii S, Okazaki M, Okita K, Omata
M, Kojiro M, Takayasu K, Nakanuma Y, Makuuchi M, Matsuyama Y, et
al: Report of the 17th nationwide follow-up survey of primary liver
cancer in japan. Hepatol Res. 37:676–691. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hung HH, Chao Y, Chiou YY, Li CP, Lee RC,
Huo TI, Huang YH, Chau GY, Su CW, Yeh YC, et al: A comparison of
clinical manifestations and prognoses between patients with
hepatocellular carcinoma and Child-Pugh scores of 5 or 6. Medicine
(Baltimore). 93:e3482014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu F, Wang J, Chang H, Lu J and Li H:
Relevance between HLA-DP gene rs2281388 polymorphism and
hepatocellular carcinoma risk. Int J Clin Exp Pathol. 8:7431–7435.
2015.PubMed/NCBI
|
|
14
|
Hong YM, Yoon KT, Cho M, Heo J, Woo HY and
Lim W: A case of small hepatocellular carcinoma with an extensive
lymph node metastasis at diagnosis. Clin Mol Hepatol. 20:310–312.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chou CT, Chen RC, Lin WC, Ko CJ, Chen CB
and Chen YL: Prediction of microvascular invasion of hepatocellular
carcinoma: Preoperative CT and histopathologic correlation. AJR Am
J Roentgenol. 203:W253–W259. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
George RT, Mehra VC, Chen MY, Kitagawa K,
Arbab-Zadeh A, Miller JM, Matheson MB, Vavere AL, Kofoed KF,
Rochitte CE, et al: Myocardial CT perfusion imaging and SPECT for
the diagnosis of coronary artery disease: A head-to-head comparison
from the CORE320 multicenter diagnostic performance study.
Radiology. 272:407–416. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu J-J, Li H-X, Chen Z-B, Yang W-P, Zhao
S-F, Chen J, Bai T, Li H and Li L-Q: Consistency analysis of
contrast-enhanced ultrasound and contrast-enhanced CT in diagnosis
of small hepatocellular carcinoma. Int J Clin Exp Med.
8:21466–21471. 2015.PubMed/NCBI
|
|
18
|
Trenker C, Neesse A and Görg C:
Sonographic patterns of renal lymphoma in B-mode imaging and in
contrast-enhanced ultrasound (CEUS) - a retrospective evaluation.
Eur J Radiol. 84:807–810. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang ZG, Zhang WG, Wu YH, Liang HF, Zhang
BX and Chen XP: Incomplete RFA-generated heat shock response
provokes colorectal cancer liver metastases (CRLMs) recurrence by
inducing cancer cell stemness and invasion. Mol Cancer Res. 13(10
Suppl): Abst B472015. View Article : Google Scholar
|
|
20
|
Bo XW, Xu HX, Sun LP, Zheng SG, Guo LH, Lu
F, Wu J and Xu XH: Bipolar radiofrequency ablation for liver
tumors: Comparison of contrast-enhanced ultrasound with
contrast-enhanced MRI/CT in the posttreatment imaging evaluation.
Int J Clin Exp Pathol. 7:6108–6116. 2014.PubMed/NCBI
|