Introduction
Chemotherapy is the predominant option for advanced
breast cancer patients. Efficacious chemotherapeutic protocols will
prevent metastasis and recurrence, thereby to increase the
possibility of surgical resection and to extend survival time.
Single cytotoxic agents and combination chemotherapy regimens are
recommended for the treatment of patients with metastatic disease
(1,2).
Nowadays, the optional chemotherapy specimens included
anthracycline, paclitaxol and anti-metabolism medicines (3). Collective studies supported that
pemetrexed effectively prolongs survival estimation in a proportion
of advanced breast cancer patients, which was an optional specimen,
especially followed with anthracycline- and taxane-containing
regimens (4–6). However, the biomarkers which can
effectively screen out suitable patients to receive pemetexed
treatment is still unclear so far (5).
As a multi-targeted anti-metabolite, pemetrexed
inhibits multiple targets of folic acid metabolic pathway,
especially thymidylate synthase (TYMS) and other DNA synthase in
folic acid metabolism (7,8). Previous researches indicated that
pemetrexed efficacy was correlated with TYMS in lung adenocarcinoma
and gastric cancer (9,10). Therefore, these enzymes are promising
biomarkers for predicting the efficacy of pemetrexed chemotherapy.
Here in our study, we investigated the correlation between clinical
efficacy of pemetrexed chemotherapy and the expression of TYMS in
advanced breast cancer.
Materials and methods
Patients and pemetrexed
chemotherapy
Total 77 patients with advanced breast cancer at The
Second Hospital of Shandong University (Jinan, China) from 2013 to
2015 were collected in this retrospective study. Pemetrexed
chemotherapy was administrated in all the patients after
anthracycline- and taxane-containing regimens. The regimen plan was
pemetrexed (600 mg/m2) i.v. which was administered on
day 1 of each 21-day cycle until disease progression, unacceptable
toxicity or patient's refusal. Dexamethasone, folic acid and
vitamin B12 supplement were administered according to chemotherapy
protocol. Follow-up was performed to evaluate chemotherapy response
after every two cycles according to RECIST. The objective response
rate (ORR) was combined proportion of complete response (CR) and
partial response (PR). The disease control rate (DCR) was combined
proportion of CR, PR, and stable disease (SD).
Tumor specimens and tissue microarray
(TMA)
The resected specimens of primary breast cancer were
collected for TMAs, which were stored with formalin-fixed,
paraffin-embedded tissue blocks. The representative areas of tumors
were selected using hematoxylin and eosin (H&E) staining slides
of each tissue. TMA sections were prepared for immunohistochemical
(IHC) staining at 5 µm of thickness. The pathological
characteristics of each patient were determined by experienced
pathologists, including histologic grade, lymph node metastasis and
expression status of estrogen receptor (ER), progesterone receptor
(PR) and human epidermal growth factor receptor 2 (HER2). This
study was approved by the Institutional Review Board of The Second
Hospital of Shandong University.
IHC staining and evaluation of
staining
IHC staining was performed with TMAs according to
the following steps: Dewaxing with dimethylbenzene, hydration with
a gradient concentration of alcohol, antigen retrieval with citrate
buffer (pH 6.0), endogenous peroxidase blockage with 0.3%
H2O2 solution, TYMS antibody incubation (mAb;
clone TYMS106/4H4B1, 1:50 dilution; Zymed, San Francisco, CA, USA)
overnight at 4°C, staining with peroxidase-conjugated avidin and
3,3-diaminobenzidine tetrahydrochloride (DAB), hematoxylin blue
counterstain. Positive control was assigned IHC positive tissues,
and control IgG was used as negative control. TMAs were blindedly
evaluated by experienced pathologists. By using IHC staining, TYMS
is located in both cytoplasm and nucleus of cancer cells. Both
percentage and intensity of positive staining were evaluated for
TYMS scores with a semi-quantitative scale (11). The intensity was evaluated into three
groups: 0, no staining; 1, weak staining; and 2, strong staining.
Summed scores of each tissue are ranging from 0 to 2.
Statistical analysis
Statistical analysis was performed with SPSS 19.0
(SPSS, Inc., Chicago, IL, USA). The cutoff values of TYMS IHC
scores were studied with receiver operating characteristic (ROC)
curves. The correlation between TYMS and other clinical
characteristics, therapeutic efficiency was studied with
χ2 test or Fisher's exact test. The effect of potential
predictive variable was analyzed with cox proportional hazards
regression model. The estimated relative risks were showed as
adjusted hazard ratios (HRs) and 95% confidence intervals (CIs).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Patient characteristics and pemetrexed
treatment response
All the 77 patients were administrated with
pemetrexed chemotherapy after anthracycline- and taxane-containing
regimens as followed chemotherapy for advanced disease (Table I). The median age of the patients at
diagnosis was 44 years (range 26-79 years). Approximately 54.55% of
the patients were HR-positive, 9.09% were HER2-positive but
HR-negative, and 36.36% were triple negative. Cancer metastasis was
observed in lymph nodes (37.66%), bone (31.17%), lung (25.97%),
liver (19.48%) and brain (6.49%). The median duration of pemetrexed
treatment was 3.66 months (range, 1.40-5.62 months), and two
patients only received three cycles of pemetrexed treatment because
of disease progression and serious side-effects. Efficacy
assessments showed that 3 cases had CR, 21 cases PR, 26 cases SD,
and 27 cases PD. The total ORR for the patients was 31.17%, and DCR
was 64.94%.
 | Table I.Patients' characteristics. |
Table I.
Patients' characteristics.
| Variable | No. of patients | Percentage, % |
|---|
| Median age
(years) | 44 (range,
26-79) |
|
| Median duration
(months) | 3.66
(range, 1.40-5.62) |
|
| Subtype |
| HR+ | 42 | 54.55 |
|
HER2+ | 7 | 9.09 |
| TN | 28 | 36.36 |
| Metastases |
|
Liver | 15 | 19.48 |
| Lung | 20 | 25.97 |
| Bone | 24 | 31.17 |
|
Brain | 5 | 6.49 |
| Lymph
nodes | 29 | 37.66 |
| Response |
| CR | 3 | 3.90 |
| PR | 21 | 27.27 |
| SD | 26 | 33.77 |
| PD | 27 | 35.06 |
| ORR | 24 | 31.17 |
| DCR | 50 | 64.94 |
Relationship between TYMS and
clinicopathological parameters
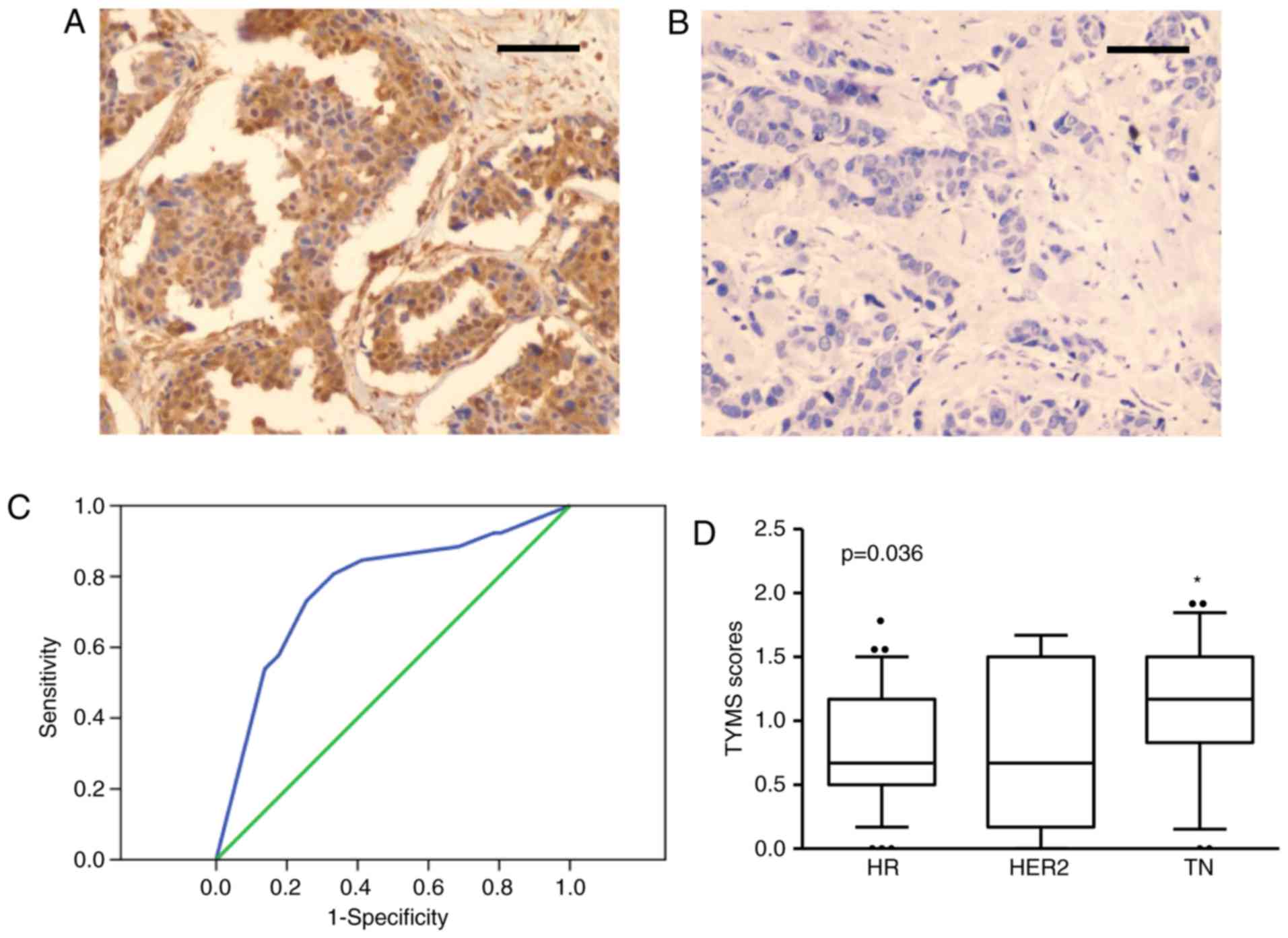
IHC staining shows that TYMS was mainly localized in
cytoplasm and nucleus of the breast cancer cells (Fig. 1A). As described previously, TYMS
expression was also observed in a large proportion of normal
tissues, which is consistent with previous report (12). To further evaluate the correlation
between TYMS and clinicopathological parameters, the patients were
analyzed into two groups according TYMS scores: TYMS-high and
TYMS-low. The cutoff value of TYMS score was 1.09 according to ROC
curves analysis (AUC=0.768, P<0.001, 95% CI: 0.651-0.886,
Fig. 1B). Totally 32 patients are in
TYMS-high group, and 45 patients in TYMS-low group. No significant
correlation was observed between TYMS expression and age, tumor
size, expression of ER, PR and HER-2 (P>0.05 for each; Table II). However, high TYMS expression
correlated with high histopathological grade and lymph node
metastasis (P<0.05 for each; Table
II). Notably, high TYMS expression was more common in patients
with the triple-negative (TN) subtype than in those with other
subtypes (P=0.036; Fig. 1C and
D).
 | Table II.Association between clinical
characteristics and TYMS expression. |
Table II.
Association between clinical
characteristics and TYMS expression.
|
|
| TYMS
expression |
|
|---|
|
|
|
|
|
|---|
| Characteristic | Number (%) | Positive (%) | Negative (%) | P-value |
|---|
| Total | 77 | (32, 41.6) | (45, 58.4) |
|
| Age, years |
|
|
|
|
|
<50 | 43 (55.8) | 17 (22.1) | 26 (33.8) | 0.685 |
|
≥50 | 34 (44.2) | 15 (19.5) | 19 (24.7) |
|
| Tumor size, cm |
|
|
|
|
| ≤2 | 17 (25.0) | 6 (7.8) | 11 (14.3) | 0.267 |
|
2~5 | 45 (58.4) | 17 (22.1) | 28 (36.4) |
|
| ≥5 | 15 (19.5) | 9 (11.7) | 6 (7.8) |
|
| Histological
status |
|
|
|
|
| I | 19 (24.7) | 3 (3.9) | 16 (20.8) | 0.012 |
| II | 48 (62.3) | 22 (28.6) | 26 (33.8) |
|
|
III | 10 (13.0) | 7 (9.1) | 3 (3.9) |
|
| Lymph node
status |
|
|
|
|
| 0 | 22 (28.6) | 4 (5.2) | 18 (23.4) | 0.031 |
|
1-3 | 40 (51.9) | 20 (26.0) | 20 (26.0) |
|
| ≥4 | 15 (19.5) | 8 (10.4) | 7 (9.1) |
|
| ER |
|
|
|
|
|
Positive | 38 (49.4) | 14 (18.2) | 22 (31.2) | 0.407 |
|
Negative | 39 (50.6) | 18 (23.4) | 23 (27.3) |
|
| PR |
|
|
|
|
|
Positive | 42 (54.5) | 14 (18.2) | 28 (36.4) | 0.109 |
|
Negative | 35 (45.5) | 18 (23.4) | 17 (22.1) |
|
| HER2 |
|
|
|
|
|
Positive | 14 (18.2) | 3 (3.9) | 11 (14.3) | 0.091 |
|
Negative | 63 (81.8) | 29 (37.7) | 34 (44.2) |
|
Clinical response and TYMS
expression
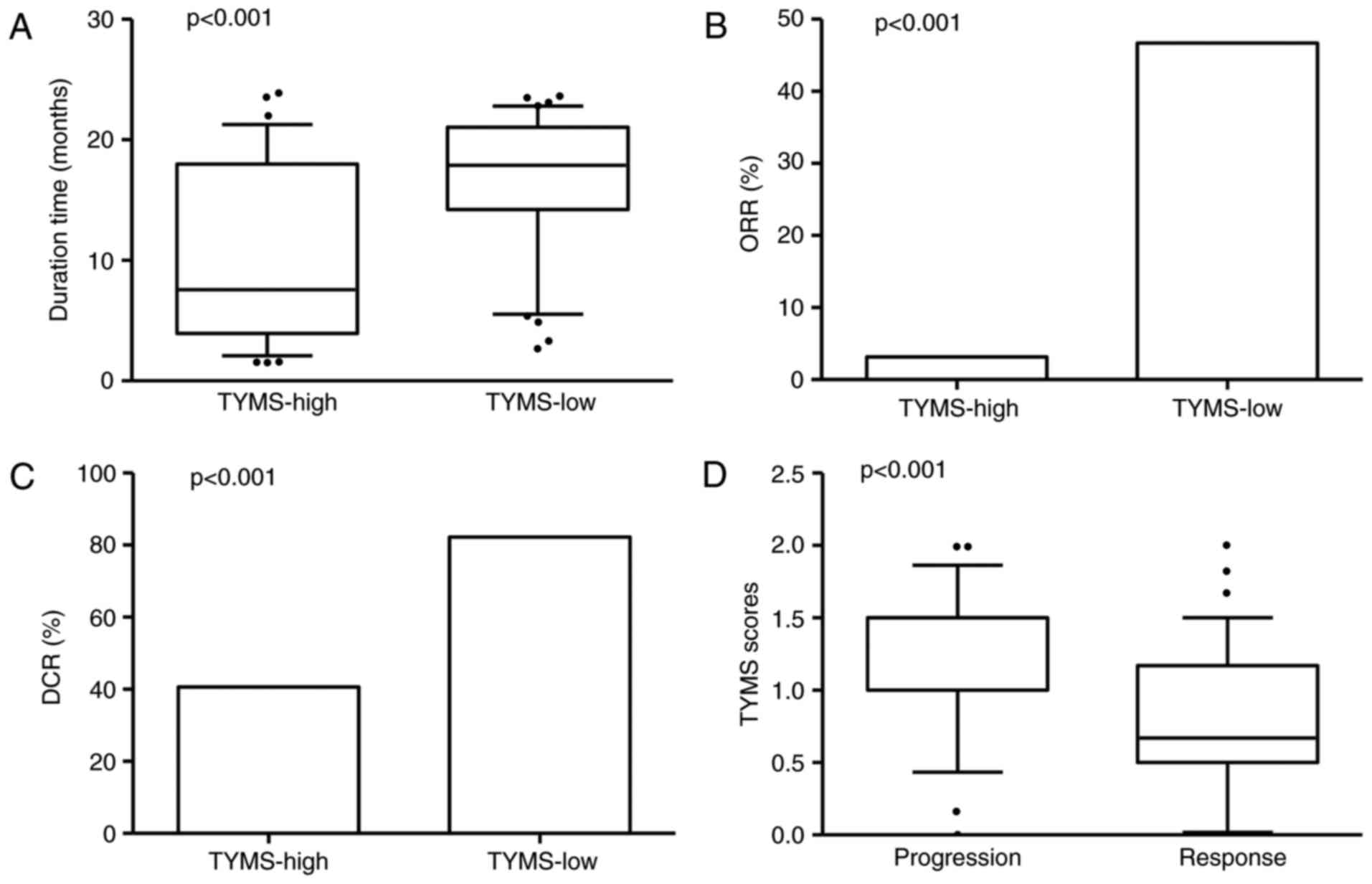
The duration of response to pemetrexed chemotherapy
in TYMS-high group was shorter than TYMS-low group (10.60 vs. 16.95
months, P<0.001; Fig. 2A).
TYMS-high patients showed significant lower overall response rate
(ORR) compared with TYMS-low ones (3.13 vs. 46.67%, P<0.001;
Fig. 2B). TYMS-high patients also
showed significant lower DCR than TYMS-low ones (40.63 vs. 82.22%,
P<0.001; Fig. 2C). Notably,
significantly higher TYMS scores were observed in the
disease-progression patients in comparison with those responses to
chemotherapy (P=0.0002; Fig. 2D).
Relationship between therapeutic
outcomes and TYMS expression
Univariate and multivariate analysis were performed
to evaluate therapeutic outcomes of pemetrexed treated patients. As
shown in Table III, elevated TYMS
expression significantly correlated with poor PFS (HR, 4.775; 95%
CI, 2.004-11.379, P<0.001) and OS (HR, 3.786; 95% CI,
1.734-8.265; P=0.001). Multivariate analysis also showed that high
TYMS expression was a detrimental factor in DFS (HR, 4.321; 95% CI,
1.442-12.943; P=0.009), and also for OS (HR, 4.569; 95% CI,
1.657-12.595; P=0.003). Moreover, ER expression was also correlated
to a better prognosis for pemetrexed treated advanced breast cancer
patients in multivariate analysis (HR, 0.139; 95% CI, 0.027-0.706;
P=0.017). However, HER2 expression was a detrimental factor for
both DFS and OS (HR, 4.281; 95% CI, 1.222-14.996; P=0.023; HR,
5.035; 95% CI, 1.686-15.040; P=0.004).
 | Table III.Cox regression analyses of
disease-free survival and overall survival for TYMS expression. |
Table III.
Cox regression analyses of
disease-free survival and overall survival for TYMS expression.
|
| DFS | OS |
|---|
|
|
|
|
|---|
| Variable
analysis | HR | 95.0% CI | P | HR | 95.0% CI | P |
|---|
| Univariate |
|
|
|
|
|
|
|
TYMS | 4.775 | 2.004-11.379 | <0.001 | 3.786 | 1.734-8.265 | 0.001 |
| Multivariate |
|
|
|
|
|
|
|
Age | 1.235 | 0.449-3.401 | 0.683 | 1.541 | 0.610-3.893 | 0.361 |
|
Size | 1.132 | 0.409-3.133 | 0.812 | 1.172 | 0.481-2.857 | 0.727 |
|
LNM | 3.950 | 0.833-18.739 | 0.084 | 3.726 | 0.824-16.845 | 0.088 |
|
Grade | 0.761 | 0.194-2.982 | 0.695 | 1.179 | 0.308-4.514 | 0.810 |
| ER | 0.139 | 0.027-0.706 | 0.017 | 0.385 | 0.100-1.481 | 0.165 |
| PR | 1.188 | 0.322-4.376 | 0.796 | 0.847 | 0.229-3.132 | 0.804 |
|
HER2 | 4.281 | 1.222-14.996 | 0.023 | 5.035 | 1.686-15.040 | 0.004 |
|
TYMS | 4.321 | 1.442-12.943 | 0.009 | 4.569 | 1.657-12.595 | 0.003 |
Discussion
Pemetrexed chemotherapy was a choice for advanced
breast cancer patients (13).
However, only a part of patients benefit from pemetrexed
chemotherapy. Our study indicated that TYMS expression correlated
with high histopathological grade and lymph node metastasis. More
importantly, high TYMS expression predicts therapeutic sensitivity
of pemetrexed chemotherapy in advanced breast cancer, suggesting
that it may be a useful biomarker to choose chemotherapy
regimens.
Anthracycline- and taxane-based chemotherapy
regimens is a common treatment for advanced breast cancer. As a
third-line chemotherapy specimen, pemetrexed is a multitarget
anti-metabolite chemotherapy drug that inhibits folate metabolism
and DNA synthesis enzymes. It has been widely used in non-small
cell lung cancer, gastrointestinal cancer during recent years
(14–16). However, variable treatment response of
pemetrexed chemotherapy was observed in patients with different
pathological type of tumors (17).
Here in this study, 77 patients with advanced breast cancer who
received pemetrexed chemotherapy were evaluated for treatment
efficiency. The ORR of these patients was 31.17% and DCR was
64.94%, which were similar to the efficacy of pemetrexed combined
with cyclophosphamide in the treatment of advanced breast cancer
(6). A large proportion of patients
suffered disease progression during pemetrexed treatment (5,17). Thus,
appropriate chemotherapy options are valuable for advanced breast
cancer patients.
Previous studies have shown that gene expression
differences are responsible for chemotherapeutic response
variability between individuals (18). Selected patients according to
biomarker will improve the chemotherapy efficacy (19). TYMS participates in deoxythymidine
monophosphate synthesis, which is critical for DNA synthesis and
repair. Breast cancer specimens have showed increased mRNA and
protein expression level of TYMS. The breast cancer with TYMS
expression showed a significant aggressive phenotype and poor
prognosis (20,21). Among the 77 patients in this study,
the TYMS scores are variable from 0 to 2 by IHC staining,
suggesting the diversity of TYMS expression in different breast
cancer patients. Elevated TYMS expression is correlated with high
histological grade and lymph node metastasis, rather than ER, PR
and HER2 expression, which is consistent with previous report
(22). Our study indicated that TYMS
was involved in disease progression and treatment resistance of
advanced breast cancer (23).
As a multitargeted antifolate, pemetrexed inhibits
several de novo synthesis enzymes for purine and pyrimidine,
including TYMS. Previous clinical and in vitro studies
showed that cancer cell lines with TYMS expression showed poor
sensitivity to cisplatin- and taxane-based chemotherapy regimens
(24,25). Pemetrexed treatment in non-small cell
lung cancer patients indicated that low TYMS mRNA expression was
associated with increased ORR (26),
and TYMS was an appropriate biomarker for pemetrexed chemotherapy
response in non-small cell lung cancer (9,10,27). Furthermore, breast cancer with
elevated TYMS expression showed poor prognosis in a long-term
follow-up study (10,28). Our study indicated that the expression
of TYMS was correlated with treatment resistance to pemetrexed in
advanced breast cancer. More importantly, significantly elevated
TYMS expression was observed in the patients with resistance than
those with objective response. Moreover, TYMS low expression group
showed significantly higher ORR than those with high expression
group. Consistent with above conclusions, our study further
confirmed that TYMS is an important marker for pemetrexed
chemotherapy efficacy. Previous studies suggested pemetrexed
resistance correlated with membrane transport deficiency and acidic
microenvironment (7,29). Further studies will be performed to
verify their correlation to TYMS expression.
As a candidate option, pemetrexed chemotherapy
efficacy provided a promising choice for advanced breast cancer
patients (6,30). However, our study only analyzed
short-term clinical efficacy of pemetrexed treatment of advanced
breast cancer due to limited cases and short observation time.
Moreover, it was reported that breast cancer patients with TYMS
polymorphism of a 6-bp deletion within TYMS 3′-UTR would benefit
from 5-FU and capecitabine chemotherapy (28,31–33).
Further studies will be employed to analyze the long-term efficacy
and gene sequencing in the future, which will provide a firm
evidence for best chemotherapy options by detecting TYMS expression
levels.
In conclusion, TYMS expression levels predicts
therapeutic sensitivity of pemetrexed chemotherapy in advanced
breast cancer. The breast cancer cells with high TYMS expression
are more likely resistant to pemetrexed chemotherapy. These
patients should be excluded from pemetrexed chemotherapy
candidate.
Acknowledgements
The authors would like to thank the Department of
Pathology of The Second Hospital of Shandong University for their
helpful assistance.
Funding
This study was supported by granst from Shinan
District of Science and Technology plan item 2016-3-020-YY and
National Natural Science Foundation of China (grant no.
81502283).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
Preparation of the tissue microarrays, IHC staining
and clinical data collection were performed by FS and YLS. The
slides were analyzed by YLL and QW. Statistical analysis was
performed by FS and QW. The manuscript was written by FS and
QW.
Ethics approval and consent to
participate
The study protocol was approved by the Ethical and
Protocol Review Committee of The Second Hospital of Shandong
University. All procedures performed in studies involving human
participants were in accordance with the ethical standards of the
Second Hospital of Shandong University and with the 1964 Helsinki
declaration and its later amendments or comparable ethical
standards. Written informed consent was obtained from all
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Van Poznak C, Somerfield MR, Bast RC,
Cristofanilli M, Goetz MP, Gonzalez-Angulo AM, Hicks DG, Hill EG,
Liu MC, Lucas W, et al: Use of biomarkers to guide decisions on
systemic therapy for women with metastatic breast cancer: American
society of clinical oncology clinical practice guideline. J Clin
Oncol. 33:2695–2704. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Khatcheressian JL, Hurley P, Bantug E,
Esserman LJ, Grunfeld E, Halberg F, Hantel A, Henry NL, Muss HB,
Smith TJ, et al: Breast cancer follow-up and management after
primary treatment: American Society of Clinical Oncology clinical
practice guideline update. J Clin Oncol. 31:961–965. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Deng QQ, Huang XE, Ye LH, Lu YY, Liang Y
and Xiang J: Phase II trial of Loubo® (Lobaplatin) and
pemetrexed for patients with metastatic breast cancer not
responding to anthracycline or taxanes. Asian Pac J Cancer Prev.
14:413–417. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Garin A, Manikhas A, Biakhov M, Chezhin M,
Ivanchenko T, Krejcy K, Karaseva V and Tjulandin S: A phase II
study of pemetrexed and carboplatin in patients with locally
advanced or metastatic breast cancer. Breast Cancer Res Treat.
110:309–315. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Llombart-Cussac A, Theodoulou M, Rowland
K, Clark RS, Nakamura T, Carrasco E and Cruciani G: Pemetrexed in
patients with locally advanced or metastatic breast cancer who had
received previous anthracycline and taxane treatment: Phase II
study. Clin Breast Cancer. 7:380–385. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dittrich C, Solska E, Manikhas A, Eniu A,
Tjulandin S, Anghel R, Musib L, Frimodt-Moller B, Liu Y, Krejcy K
and Láng I: A phase II multicenter study of two different dosages
of pemetrexed given in combination with cyclophosphamide as
first-line treatment in patients with locally advanced or
metastatic breast cancer. Cancer Invest. 30:309–316. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vandana M and Sahoo SK: Reduced folate
carrier independent internalization of PEGylated pemetrexed: A
potential nanomedicinal approach for breast cancer therapy. Mol
Pharm. 9:2828–2843. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pandyra AA, Berg R, Vincent M and
Koropatnick J: Combination silencer RNA (siRNA) targeting Bcl-2
antagonizes siRNA against thymidylate synthase in human tumor cell
lines. J Pharmacol Exp Ther. 322:123–132. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang M, Fan WF, Pu XL, Meng LJ and Wang J:
The role of thymidylate synthase in non-small cell lung cancer
treated with pemetrexed continuation maintenance therapy. J
Chemother. 29:106–112. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen CY, Chang YL, Shih JY, Lin JW, Chen
KY, Yang CH, Yu CJ and Yang PC: Thymidylate synthase and
dihydrofolate reductase expression in non-small cell lung
carcinoma: The association with treatment efficacy of pemetrexed.
Lung Cancer. 74:132–138. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Akbar S, Jordan LB, Purdie CA, Thompson AM
and McKenna SJ: Comparing computer-generated and
pathologist-generated tumour segmentations for immunohistochemical
scoring of breast tissue microarrays. Br J Cancer. 113:1075–1080.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fukushima M, Morita M, Ikeda K and
Nagayama S: Population study of expression of thymidylate synthase
and dihydropyrimidine dehydrogenase in patients with solid tumors.
Int J Mol Med. 12:839–844. 2003.PubMed/NCBI
|
|
13
|
Llombart-Cussac A, Martin M, Harbeck N,
Anghel RM, Eniu AE, Verrill MW, Neven P, De Grève J, Melemed AS,
Clark R, et al: A randomized, double-blind, phase II study of two
doses of pemetrexed as first-line chemotherapy for advanced breast
cancer. Clin Cancer Res. 13:3652–3659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Richards DA, Loesch D, Vukelja SJ, Wu H,
Hyman WJ, Nieves J, Wang Y, Hu S, Shonukan OO and Tai DF: Phase I
study of pemetrexed and pegylated liposomal doxorubicin in patients
with refractory breast, ovarian, primary peritoneal, or fallopian
tube cancer. Invest New Drugs. 29:963–970. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jensen SA, Vainer B, Witton CJ, Jørgensen
JT and Sørensen JB: Prognostic significance of numeric aberrations
of genes for thymidylate synthase, thymidine phosphorylase and
dihydrofolate reductase in colorectal cancer. Acta Oncol.
47:1054–1061. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Le François BG, Maroun JA and Birnboim HC:
Expression of thymidylate synthase in human cells is an early G(1)
event regulated by CDK4 and p16INK4A but not E2F. Br J Cancer.
97:1242–1250. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Robert NJ, Conkling PR, O'Rourke MA,
Kuefler PR, McIntyre KJ, Zhan F, Asmar L, Wang Y, Shonukan OO and
O'Shaughnessy JA: Results of a phase II study of pemetrexed as
first-line chemotherapy in patients with advanced or metastatic
breast cancer. Breast Cancer Res Treat. 126:101–108. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Denduluri N, Somerfield MR, Eisen A,
Holloway JN, Hurria A, King TA, Lyman GH, Partridge AH, Telli ML,
Trudeau ME and Wolff AC: Selection of optimal adjuvant chemotherapy
regimens for human epidermal growth factor receptor 2
(HER2)-negative and adjuvant targeted therapy for HER2-positive
breast cancers: An American Society of Clinical Oncology Guideline
adaptation of the cancer care ontario clinical practice guideline.
J Clin Oncol. 34:2416–2427. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Krop I, Ismaila N, Andre F, Bast RC,
Barlow W, Collyar DE, Hammond ME, Kuderer NM, Liu MC, Mennel RG, et
al: Use of biomarkers to guide decisions on adjuvant systemic
therapy for women with early-stage invasive breast cancer: American
Society of Clinical Oncology Clinical Practice Guideline Focused
Update. J Clin Oncol. 35:2838–2847. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yu Z, Sun J, Zhen J, Zhang Q and Yang Q:
Thymidylate synthase predicts for clinical outcome in invasive
breast cancer. Histol Histopathol. 20:871–878. 2005.PubMed/NCBI
|
|
21
|
Kaira K, Okumura T, Ohde Y, Takahashi T,
Murakami H, Kondo H, Nakajima T and Yamamoto N: Prognostic
significance of thymidylate synthase expression in the adjuvant
chemotherapy after resection for pulmonary metastases from
colorectal cancer. Anticancer Res. 31:2763–2771. 2011.PubMed/NCBI
|
|
22
|
Takagi K, Miki Y, Nakamura Y, Hirakawa H,
Kakugawa Y, Amano G, Watanabe M, Ishida T, Sasano H and Suzuki T:
Immunolocalization of thymidylate synthase as a favorable
prognostic marker in estrogen receptor-positive breast carcinoma.
Histol Histopathol. 30:1223–1232. 2015.PubMed/NCBI
|
|
23
|
Marverti G, Ligabue A, Paglietti G, Corona
P, Piras S, Vitale G, Guerrieri D, Luciani R, Costi MP, Frassineti
C and Moruzzi MS: Collateral sensitivity to novel thymidylate
synthase inhibitors correlates with folate cycle enzymes impairment
in cisplatin-resistant human ovarian cancer cells. Eur J Pharmacol.
615:17–26. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Calascibetta A, Martorana A, Cabibi D,
Aragona F and Sanguedolce R: Relationship between thymidylate
synthase and p53 and response to FEC versus taxane adjuvant
chemotherapy for breast carcinoma. J Chemother. 23:354–357. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Quintero-Ramos A, Gutiérrez-Rubio SA, Del
Toro-Arreola A, Franco-Topete RA, Oceguera-Villanueva A,
Jiménez-Pérez LM, Castro-Cervantes JM, Barragán-Ruiz A,
Vázquez-Camacho JG and Daneri-Navarro A: Association between
polymorphisms in the thymidylate synthase gene and risk of breast
cancer in a Mexican population. Genet Mol Res. 13:8749–8756. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shimizu T, Nakanishi Y, Nakagawa Y,
Tsujino I, Takahashi N, Nemoto N and Hashimoto S: Association
between expression of thymidylate synthase, dihydrofolate
reductase, and glycinamide ribonucleotide formyltransferase and
efficacy of pemetrexed in advanced non-small cell lung cancer.
Anticancer Res. 32:4589–4596. 2012.PubMed/NCBI
|
|
27
|
Ceppi P, Papotti M and Scagliotti G: New
strategies for targeting the therapy of NSCLC: The role of ERCC1
and TS. Adv Med Sci. 55:22–25. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee SJ, Choi YL, Park YH, Kim ST, Cho EY,
Ahn JS and Im YH: Thymidylate synthase and thymidine phosphorylase
as predictive markers of capecitabine monotherapy in patients with
anthracycline- and taxane-pretreated metastatic breast cancer.
Cancer Chemother Pharmacol. 68:743–751. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li L, Sham YY, Bikadi Z and Elmquist WF:
pH-dependent transport of pemetrexed by breast cancer resistance
protein. Drug Metab Dispos. 39:1478–1485. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wan F, Chen X, Dong LF, Cheng YH and Long
JP: A systemic analysis on pemetrexed in treating patients with
breast cancer. Asian Pac J Cancer Prev. 15:4567–4570. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fujishima M, Inui H, Hashimoto Y, Azumi T,
Yamamoto N, Kato H, Hojo T, Yamato M, Matsunami N, Shiozaki H and
Watatani M: Relationship between thymidylate synthase (TYMS) gene
polymorphism and TYMS protein levels in patients with high-risk
breast cancer. Anticancer Res. 30:4373–4739. 2010.PubMed/NCBI
|
|
32
|
da Silva Nogueira J Jr, de Lima Marson FA
and Silvia Bertuzzo C: Thymidylate synthase gene (TYMS)
polymorphisms in sporadic and hereditary breast cancer. BMC Res
Notes. 5:6762012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang B, Walsh SJ and Saif MW: Pancytopenia
and severe gastrointestinal toxicities associated with
5-fluorouracil in a patient with Thymidylate Synthase (TYMS)
Polymorphism. Cureus. 8:e7982016.PubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBIPubMed/NCBI
|