Introduction
Urothelial bladder cancer (BCa) is the ninth most
common cancer worldwide, with an estimated 430,000 new cases in
2012. More than 60% of all BCa cases and half of the total 165,000
BCa deaths occur in the less developed regions of the world. A
strong predominance of male patients is observed, with
three-quarters of all BCa cases occurring in men (1). Overall, it has a significant impact on
public health (2).
Approximately 75% of the newly diagnosed BCas are
non-muscle invasive bladder cancer (NMIBC) and have a high rate of
recurrence (60–70%) and progression to muscle-invasive disease
bladder cancer (MIBC, 10–20%), despite local therapy. The remaining
25% of the newly diagnosed BCas present with muscle invasion and
need either radical surgery or radiotherapy. However, these cases
often have poor outcomes despite systemic therapy (3,4). MIBC has
the worst prognosis, with a 5-year survival rate of less than 50%
of the cases (5).
BCa has a high degree of mutational heterogeneity
and a high frequency of somatic mutations compared with other solid
tumors. Mutational heterogeneity is mediated by multiple factors,
which include enzyme catalytic polypeptide family of enzymes and
smoking exposure among others. The mutational landscape of
urothelial carcinoma, including specific mutations in pathways and
driver genes, such as FGFR3, ERBB2, PIK3CA, TP53 (p53), and RB1,
affects tumor aggressiveness and response to therapy (6,7).
Mediator subunit 15 (MED15) is a subunit of the
multiprotein Mediator complex, which can be divided into four
modules: The head, middle, tail, and kinase, and is comprised of
≈33 subunits in humans. The Mediator complex functions as a bridge
between regulatory proteins and RNA polymerase II (Pol II), thereby
regulating the Pol II-dependent transcription (8–11). Several
studies reported altered expressions of distinct subunits in human
malignancy. Even in breast cancer, MED15 seems to be a crucial
cofactor for TGFβ signalling, and the MED15 deficiency
attenuates the TGFβ-targeted gene expression and hinders the
TGFβ-induced epithelial-mesenchymal transition (EMT) (12). Additionally, in head and neck squamous
cell carcinoma (HNSCC), MED15 has been found to be overexpressed,
especially in recurrent tumors possessing an aggressive phenotype
(13).
Till now, we have only a rudimentary knowledge of
the role of the Mediator complex in BCa. In comparison to benign
tissue, an overexpression of MED19 has been detected in BCa.
Additionally, the knockdown of MED19 leads to a reduced
growth of the carcinoma cell lines (14). Contrary to this, the expression of
MED1 significantly decreases during BCa progression from benign
urothelium to advanced BCa, and cancer-specific survival (CSS) was
significantly worse in the group that had low MED1 expression
(15).
Till now, nothing is known about MED15 in urothelial
BCa; so, this has led us to investigate its expression and
functional properties.
Materials and methods
Ethics statement
This investigation was conducted in accordance with
ethical standards, the Declaration of Helsinki, and national and
international guidelines. Ethical approval for using human material
in the present study was obtained from the Internal Review Board of
the University Hospital of Bonn (IRB nos. 036/08 and 093/12). The
participants of the study agreed in writing beforehand and were
anonymised before inclusion in the study cohort.
RNA expression analysis by
cBioPortal
The database cBioPortal for cancer genomics
(http://cbioportal.org; version 1.4.0) was
utilized in January 2017 to investigate the mRNA expression of
MED15 in BCa extended by the use of survival analysis.
cBioPortal provides a Web resource for exploring,
visualising, and analysing multidimensional cancer genomics data.
The portal reduces molecular profiling data from cancer tissues and
cell lines into readily understandable genetic, epigenetic, gene
expression and proteomic events (16,17). For
the analysis, RNA sequencing data from The Cancer Genome Atlas
(TCGA) (18) were used (RNA Seq V2
RSEM) with two SDs from the mean and a threshold of ±2.0. The mRNA
z-scores were precomputed from the expression values and were
compared to the expression distribution of each gene associated
with the tumors that are diploid for this gene (16). Two mRNA analyses for BCa, named
‘Bladder Urothelial Carcinoma (TCGA, Provisional)’ and ‘Bladder
Urothelial Carcinoma (TCGA, Nature 2014)’ are available on
cBioPortal with 408 and 129 samples, respectively.
Additionally, a mutation analysis was performed in
both data sets using cBioPortal [Copy number alterations, mRNA
expression (RNA Seq V RSEM)].
Immunohistochemistry (IHC)
Tissue microarray (TMA)
construction
A total of 182 specimens were used for this study.
The urothelial BCa cohort, provided by the Clinic for Urology of
the University Hospital Bonn, contained a total of 18 benign
samples, 126 BCa samples, and 38 metastases derived from BCa
(Table I).
 | Table I.Clinicopathological data of the
urothelial bladder cohort. |
Table I.
Clinicopathological data of the
urothelial bladder cohort.
| Characteristic | BCa Σ=126 (%) | Metastasis Σ=38
(%) | Benign Σ=18
(%) |
|---|
| Sex |
|
|
|
|
Male | 91 (72.2) | 11 (26.2) | 11 (61.1) |
|
Female | 35 (27.8) | 2 (4.8) | 7
(38.9) |
| Age, years |
|
|
|
|
Mean | 66.96 | 65.22 | 65.61 |
|
Median | 69.00 | 66.00 | 65.00 |
|
Range | 36–94 | 38–84 | 43–81 |
| TNM |
|
|
|
| T1 | 24 (19.0) | – | – |
| T2 | 39 (31.0) | – | – |
| T3 | 31 (24.6) | – | – |
| T4 | 32 (25.4) | – | – |
| N+ | 34 (27.0) | – | – |
| M+ | 4 (3.2) | – | – |
| Cancer
associated | 33 (26.2) | – | – |
|
mortality |
|
|
|
|
Relapse | 65 (51.6) | – | – |
| Grading |
|
|
|
| G1 | 2 (1.6) | – | – |
| G2 | 50 (39.7) | – | – |
| G3 | 74 (58.7) | – | – |
| NMIBC | 24 (19.0) | – | – |
| MIBC | 102 (81.0) | – | – |
TMAs were performed as described previously
(19,20). Briefly, formalin-fixed
paraffin-embedded (FFPE) tissues were cut into 4-µm-thick sections
and mounted on slides. After staining with haematoxylin and eosin
(H&E), relevant areas of benign tissue and primary tumor were
identified and circled by a pathologist. Each tumor and the
corresponding benign region were represented with up to three cores
measuring 0.6 mm in diameter on a TMA recipient block using a
semiautomatic tissue arrayer (Beecher Instruments, Sun Prairie, WI,
USA). H&E TMA sections were assessed again to confirm the
histology.
MED15 protein expression analysis by
IHC
Prior to performing IHC analyses in the selected
TMAs, the specificity of the antibody was confirmed according to
the manufacturer's instructions by using non-seminomatous germ cell
tumor (NSGCT) as a positive control. IHC was performed using the
Ventana Benchmark automated staining system (Ventana Medical
System, Tuscon, AZ, USA). In short, slides were incubated with the
primary antibody according to the manufacturer's instructions:
Anti-MED15 rabbit polyclonal (1:50, clone 11566-1-AP; ProteinTech
Group, Inc., Chicago, IL, USA) at room temperature; antibody
dilution was conducted using a Ventana diluent. For signal
detection, the ultraView Universal DAB detection kit (Ventana
Medical System) was used. Finally, slides were counterstained with
haematoxylin and bluing reagent, dehydrated, and mounted.
After IHC was performed, the staining was evaluated
independently by two pathologists (GK and TM). Only cases with at
least one assessable TMA core that had sufficient tumor tissue were
included in the analysis. Quantification of nuclear protein
expression was evaluated according to a score of 0 (=no expression)
to 3 (=high expression). The cut-off used to determine a nuclear
MED15 overexpression (≥2) was consistently set above the expression
of benign samples.
Functional investigations
Cell lines and culture conditions
Urothelial BCa cell lines T24 and TCCSUP were
purchased from the American Type Culture Collection (ATCC,
Manassas, VA, USA) and were grown in a 5% CO2 incubator
at 37°C and 85% humidity. Monolayer cultures were maintained in the
RPMI-1640 medium (Biochrom AG, Berlin, Germany) that was
supplemented with 10% heat-inactivated foetal calf serum (FCS;
Sigma-Aldrich; Merck KGaA, Darmstadt, Germany), 1%
streptomycin-penicillin antibiotics (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), and 1% glutamine (Thermo
Fisher Scientific, Inc., Darmstadt, Germany).
Small interfering (si)RNA mediated
MED15 knockdown
The cell transfections in the cell lines T24 and
TCCSUP were carried out with 100 nmol/l siRNA using Screenfect A
(Genaxxon Bioscience GmbH, Ulm, Germany) for 48 (for PCR) and 72 h
(for western blotting), respectively. MED15 siRNA (sc-75767; Santa
Cruz Biotechnology, Inc., Dallas, TX, USA) is a pool of three
target-specific siRNAs designed to knockdown gene expression. A
non-targeting scrambled siRNA was used as negative control (Exiqon
Life Sciences, Copenhagen, Denmark).
Western blotting
Post-transfected cells were washed with ice-cold
phosphate-buffered saline (PBS) and lysed in a protein extraction
buffer for 30 min. In addition, samples were sonicated for 2 min
and subsequently centrifuged at 16,000 × g for 30 min at 4°C.
Protein concentration was measured using bicinchoninic acid (BCA)
Protein Assay kit (Thermo Fisher Scientific, Inc., Waltham, MA,
USA), fractionated by SDS-PAGE and transferred to a nitrocellulose
membrane. After incubation with 5% non-fat milk in TBST (50 mM
Tris, 150 mM NaCl, 0.05% Tween-20, pH 7.5) for 60 min, the membrane
was incubated with anti-MED15 rabbit polyclonal (1:500, clone
11566-1-AP; ProteinTech Group, Inc.) or anti-GAPDH rabbit
monoclonal (1:2,000, clone 14C10; Cell Signaling Technology, Inc.,
Danvers, MA, USA) primary antibodies at 4°C overnight. Then, the
membrane is washed three times for 5 min with TBST and incubated
with horseradish peroxidase conjugated anti-rabbit antibody for 1 h
at room temperature. Staining was detected using SuperSignal West
Femto Maximum Sensitivity Substrate, an ultra-sensitive enhanced
chemiluminescence (ECL) substrate (Thermo Fisher Scientific,
Inc.).
Reverse transcription-quantitative PCR
(RT-qPCR)
RNA was isolated from cell line pellets using the
Total RNA Purification Mini Spin Column Kit (Genaxxon Bioscience
GmbH, Ulm, Germany). RNA quantity and quality were analysed using a
NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Inc.,
Wilmington, DE, USA). cDNA was synthesised using 200 ng total RNA
and the PrimeScript RT Reagent kit with gDNA Eraser (Takara Bio,
Saint-Germain-en-Laye, France). qPCR was performed using 5 ng/µl
cDNA, Takara Bio SYBR Premix Ex Taq II with ROX Plus, and 10
pmol/µl forward and reverse primer. The following primer sequences
were used: MED15, forward: 5′-TTGAGGATGATGAGCGGCAG-3′, reverse:
5′-GGAGGTCCTTGTCATCCAGC-3′; and β-actin, Forward:
5′-CCAACCGCGAGAAGATGA-3′, reverse: 5′-CCAGAGGCGTACAGGGATAG-3′. PCR
was performed on an ABIPrism 7900 HT Fast Real-Time PCR System
(Applied Biosystems; Thermo Fisher Scientific, Inc.). Data were
analysed using Qbase+ (Biogazelle, Ghent, Belgium) with β-actin as
reference gene, applying the 2−∆∆Cq algorithm.
EZ4U cell proliferation assay
The EZ4U cell proliferation assay kit was used
according to the manufacturer's protocol (EZ4U; Biomedica Group,
Vienna, Austria). The siRNA transfections for proliferation assays
were performed in 96-well plates. In each well of a flat-bottom
96-well plate, either 1.2×104 cells (T24) or
2.0×104 cells (TCCSUP) were seeded in 200 µl cell
culture medium. siRNA-mediated MED15 knockdown was then
performed, and cells were incubated to adhere and grow for 72 h.
After incubation, 20 µl of EZ4U substrate solutions were added
before incubating the resultant solution for 3 h (T24) and 4 h
(TCCSUP) until the color of the solution changed from yellow to
orange. The absorbance was measured using a microplate reader
(Tecan Spectra Rainbow microplate reader; Tecan Deutschland GmbH,
Crailsheim, Germany) at 450 nm wavelength. Each experiment was
repeated at least three times.
Migration assays
The siRNA transfections for migration assays were
performed in 6-well plates. 48 h post transfection, cells were
trypsinised and seeded into migration Boyden chambers.
3×104 (T24) or 5×104 cells (TCCSUP) were
plated in the upper chamber of migration inserts (VWR, Darmstadt,
Germany) containing 0% FCS medium. The lower chamber was filled
with medium containing 10% FCS for chemotactic attraction. After 24
h of incubation, cells on the upper surface were removed with the
help of a cotton swab. BCa cells invading the lower surface of
membrane were fixed with 4% paraformaldehyde (Merck KGaA,
Darmstadt, Germany), stained with haematoxylin (Waldeck, Münster,
Germany), and washed with water. Membranes were scanned and
manually evaluated in four randomly selected fields by counting.
Each experiment was repeated at least three times.
Invasion assays
The invasion analysis was carried out in a similar
as the migration except for the use of Matrigel invasion chambers
(VWR) and a cell number of 7.5×104 per well.
Clinical data and statistics
For IHC, associations with clinical-pathological
parameters were performed. Survival analysis was evaluated with the
help of the Kaplan-Meier estimator and log rank tests. Statistical
evaluation was performed in Microsoft Excel and SPSS. The IHC data
were not normal distributed, so we performed a non-parametric test.
When comparing more than two sets of numerical data, a multiple
group comparison test such as Kruskal-Wallis test was followed by a
post hoc test such as Dunn-Bonferroni test. When comparing two or
less sets of numerical data, the Mann-Whitney U test was applied.
The data were analysed anonymously.
For the statistical analysis of the functional
investigations in the cell lines, we compared the results of
scrambled RNA and target RNA on the one hand in TCCSUP and on the
other hand in the T24. The respective cell line had its own
scrambled control. The respective ‘scrambled’ control was set to 1.
Due to the fact that the data were distributed normally, the
parametric test was performed and because of two and less groups
the unpaired t-test was used.
The cell lines with each other have not been
compared statistically.
Results
Transcriptional expression of the
MED15 by cBioPortal
Using the cBioPortal database, we analysed the mRNA
expression of MED15 in BCa samples. In the two analyses, 408 and
129 samples of BCa were investigated. The results of the expression
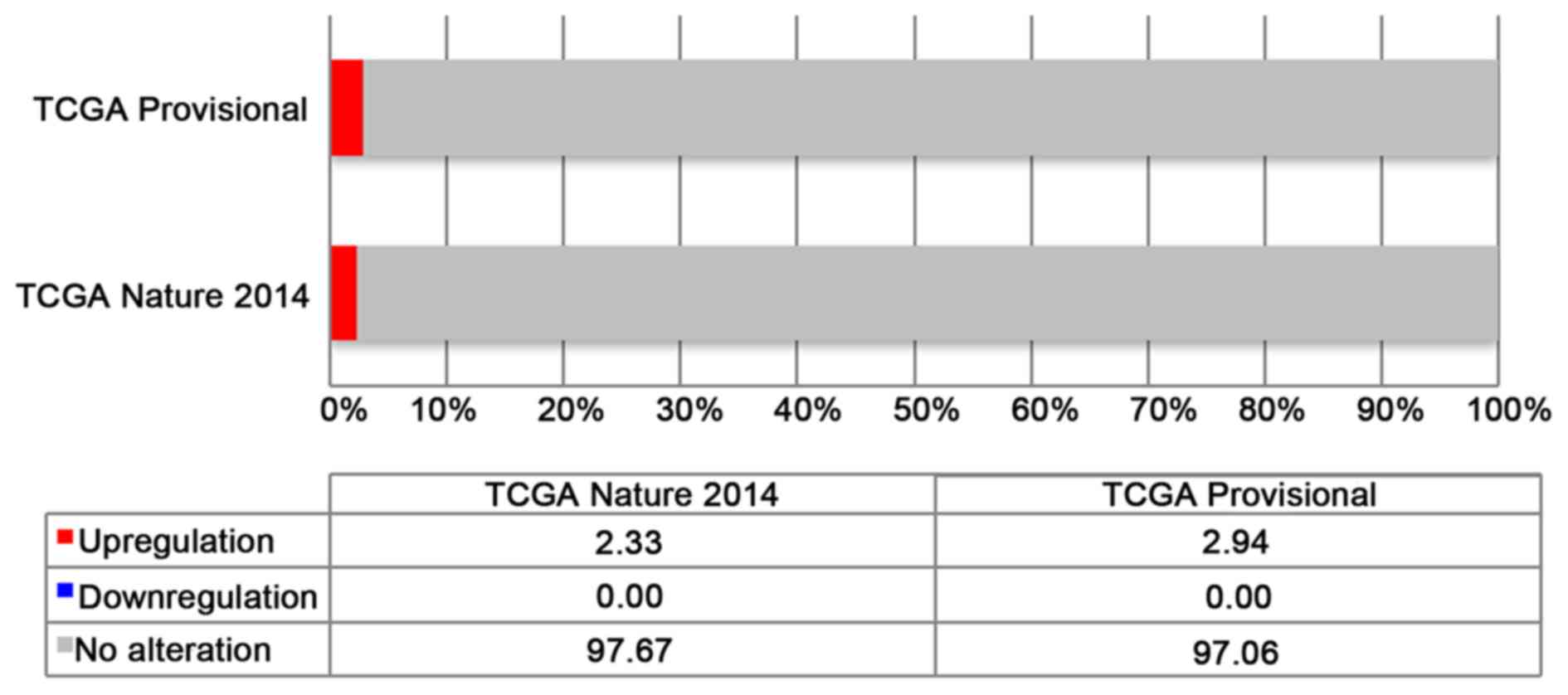
analyses are shown in Fig. 1.
Transcriptional expression regarding
the database cBioPortal
In both analyses, named ‘Bladder Urothelial
Carcinoma (TCGA, Provisional)’ and ‘Bladder Urothelial Carcinoma
(TCGA, Nature 2014)’, an alteration of MED15 is described in
2.9% (12/408) and 2.3% (3/129), respectively. All of the
alterations are upregulations of MED15 mRNA expression
(Fig. 1).
Survival analysis regarding
transcriptional expression
Due to the small number of alterations and events in
both the analyses, there are no significant differences in overall-
and progression-free-survival. Additionally, the analyses cannot
really determine a tendency (data not shown).
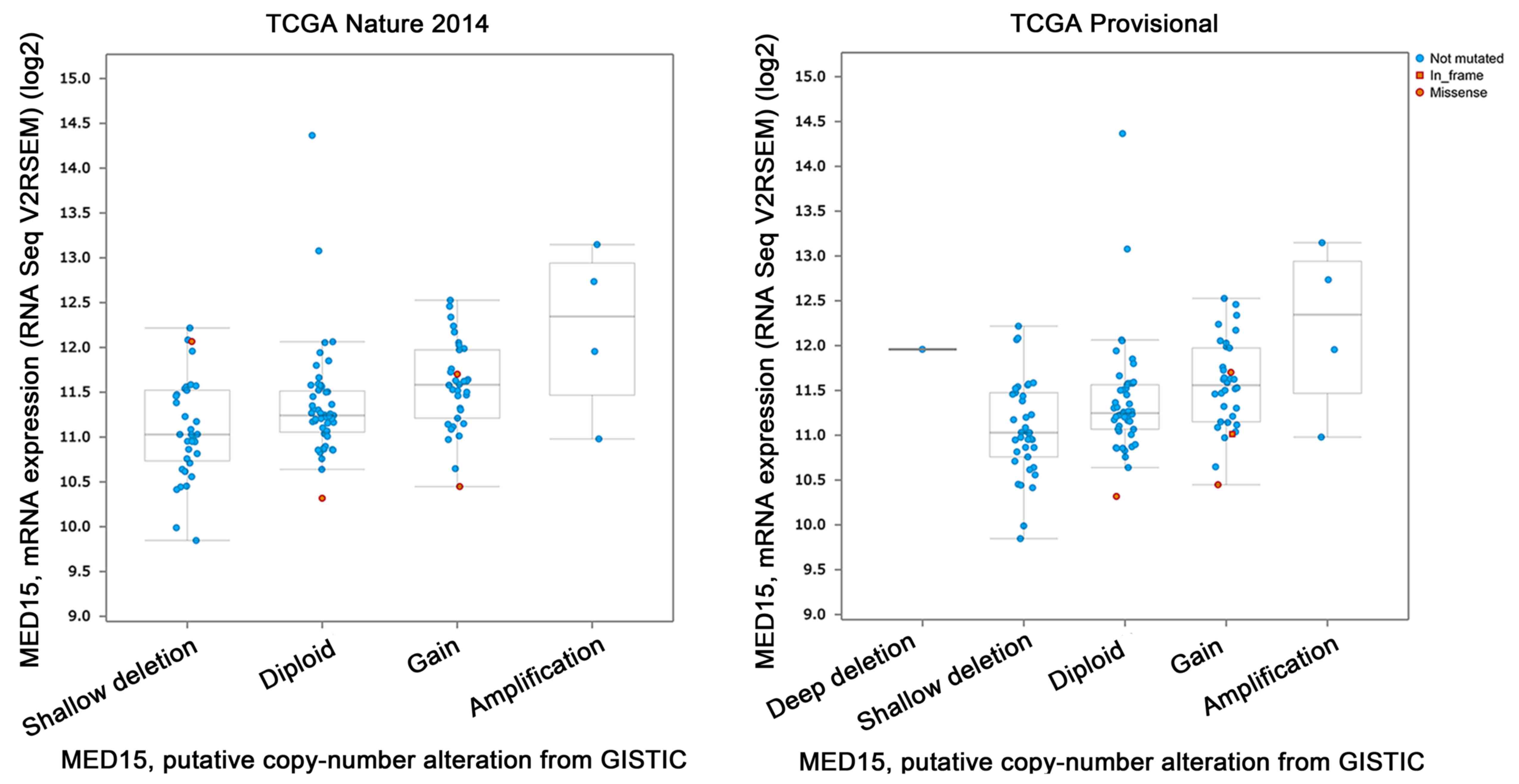
Mutation analysis of MED15 in BCa
As shown in Fig. 2, in
the TCGA 2014 analysis (n=130), the following mutation profiles
were found: Shallow deletions (n=38), diploid (n=51),
gain-of-function (n=34) and amplification (n=4). However, the
amplification logically led to the highest mRNA expression.
Missense mutations are found four times; no in-frame mutation.
The TCGA Provisional analysis (n=130) shows a
similar profile: Deep deletion (n=1), shallow deletion (n=39),
diploid (n=49), gain of function (n=34), amplification (n=4).
Again, the amplification leads to the highest mRNA expression. This
analysis revealed three missense and one in-frame mutations.
IHC and functional investigations
In order to further illuminate the expression and
role of MED15 in BCa, we performed IHC staining on a TMA cohort
with the available clinical information and functional analysis in
cell lines of essential tumor parameters.
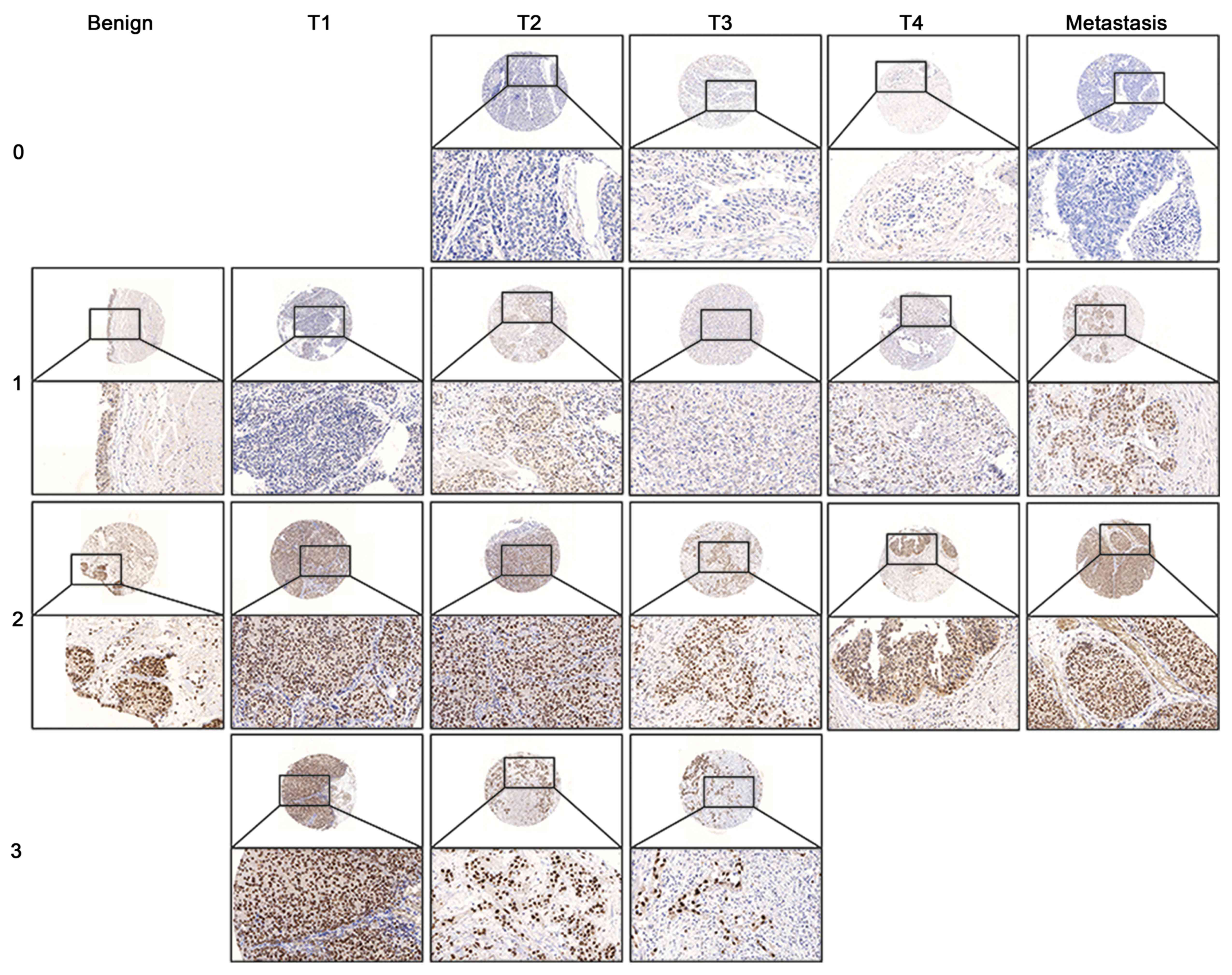
IHC staining shows MED15 nuclear expression in
benign, tumor and metastasis tissue (Fig.
3). A MED15 cytoplasmic expression seemed not to be specific,
since almost every sample shows a weak cytoplasmic signal. There
are no differences here. The expression of MED15 in BCa was just
found in epithelial cells. In the stroma, only a few fibroblasts
displayed a weak non-specific expression, comparable to the
cytoplasm. For IHC, we found MED15 to have a higher
expression in NMIBC (staining score=2.0) compared to benign
(staining score=1.5, P=0.028) and MIBC (staining score=1.0,
P≤0.001; Fig. 4A and B). The
overexpression of MED15 in BCa did not associate with
positive lymphnode- or distant-metastasis stage [N: P=0.252 (data
not shown), M: P=0.434; Fig. 4C] or
the grading status of the tumor (Fig.
4D). Unfortunately, no significant differences between samples
with or without overexpression (staining score >2) of
MED15 have been found for CSS (P=0.770) and progression-free
survival (PFS, P=0.503) (data not shown) for survival analysis of
the IHC. An evaluation of the pure MED15 expression (division into
positive and negative, without definition of an intensity or
overexpression) on the basis of the clinicopathological data can be
found in Table II.
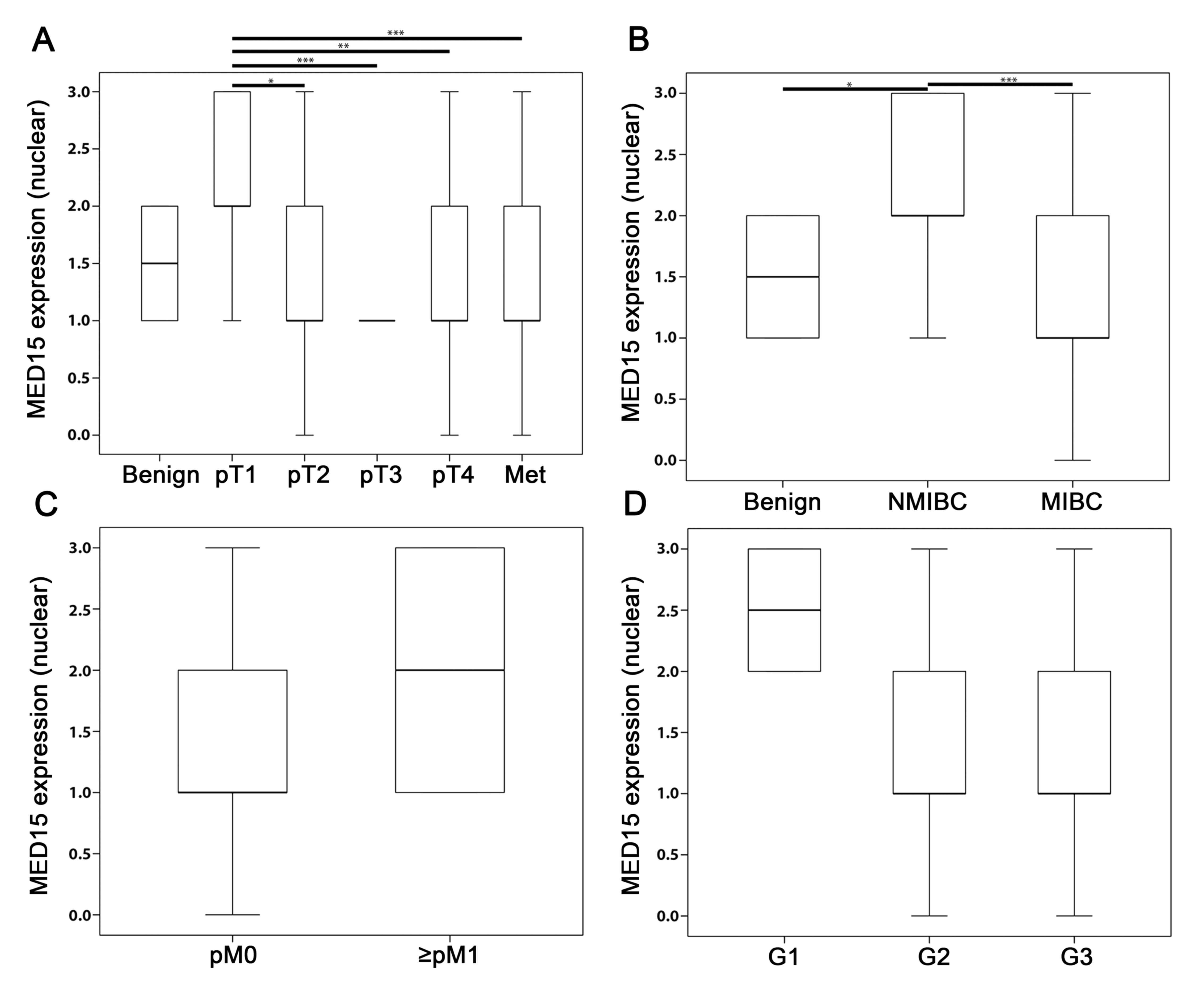 | Figure 4.IHC analysis of MED15 in BCa. (A)
Mean IRS score for MED15 protein expression profile for all tumor
stages of the total bladder cohort including benign and metastases
tissue. Significances of the individual tumor stages (pT2, pT3, pT4
and Met) were tested against pT1. (B) NMIBC showed the highest
MED15 expression in comparison to benign and MIBC (significances
were tested: benign vs. NMIBC and NMIBC vs. MIBC). The presence of
(C) distant metastases and, (D) on the other hand, low grading
states are associated with higher levels of MED15 expression,
although the results show no significance. Bars and error bars
indicate mean IRS ± SD. Due to either low case numbers of
individual analyzes (M1 and G1) or the same result values of the
samples (benign, pT3), there are no error bars in individual box
plots. Sample number of the individual groups: benign n=18, pT1
n=24, pT2 n=39, pT3 n=31, pT4 n=32, Met n=38; NMIB n=24, MIBC
n=102; pM0 n=122, ≥pM1 n=4; G1 n=2, G2 n=50, G3 n=74.
Independent-samples t-test, *P≤0.05, **P≤0.01, ***P≤0.001. NMIBC,
non-muscle invasive bladder cancer; MIBC, muscle invasive bladder
cancer; Y-axis, nuclear MED15 expression. MED15, mediator subunit
15; IHC, Immunohistochemistry. |
 | Table II.Overview of MED15 expression in the
BCa cohort divided into positive and negative expression based on
the clinicopathological parameters. |
Table II.
Overview of MED15 expression in the
BCa cohort divided into positive and negative expression based on
the clinicopathological parameters.
|
| BCa Σ=126 (%) | Metastasis Σ=38
(%) | Benign Σ=18
(%) |
|---|
|
|
|
|
|
|---|
|
|
| Positive | Positive | Negative | Positive | Negative |
|---|
| Characteristic | Total | Negative | 30 (79.0) | 8 (21.0) | 18 (100.0) | 0 (0.0) |
|---|
| TNM |
|
|
|
|
|
|
| T1 | 24 (19.0) | 24 (19.0) 0
(0.0) | – | – |
| T2 | 39 (31.0) | 33 (26.2) 6
(4.8) | – | – |
| T3 | 31 (24.6) | 25 (19.8) 6
(4.8) | – | – |
| T4 | 32 (25.4) | 28 (22.2) 4
(3.2) | – | – |
| N+ | 34 (27.0) | 30 (23.8) 4
(3.2) | – | – |
| M+ | 4 (3.2) | 4 (3.2) 0
(0.0) | – | – |
| Cancer
associated death | 33 (26.2) | 30 (23.8) 30
(2.4) | – | – |
|
Relapse | 65 (51.6) | 58 (46.0) 7
(5.6) | – | – |
| Grading |
|
|
|
|
|
|
| G1 | 2 (1.6) | 2 (1.6) 0
(0.0) | – | – |
| G2 | 50 (39.7) | 40 (31.7) 10
(8.0) | – | – |
| G3 | 74 (58.7) | 68 (54.0) 6
(4.7) | – | – |
| NMIBC | 24 (19.0) | 24 (19.0) 0
(0.0) | – | – |
| MIBC | 102 (81.0) | 86 (68.3) 16
(12.7) | – | – |
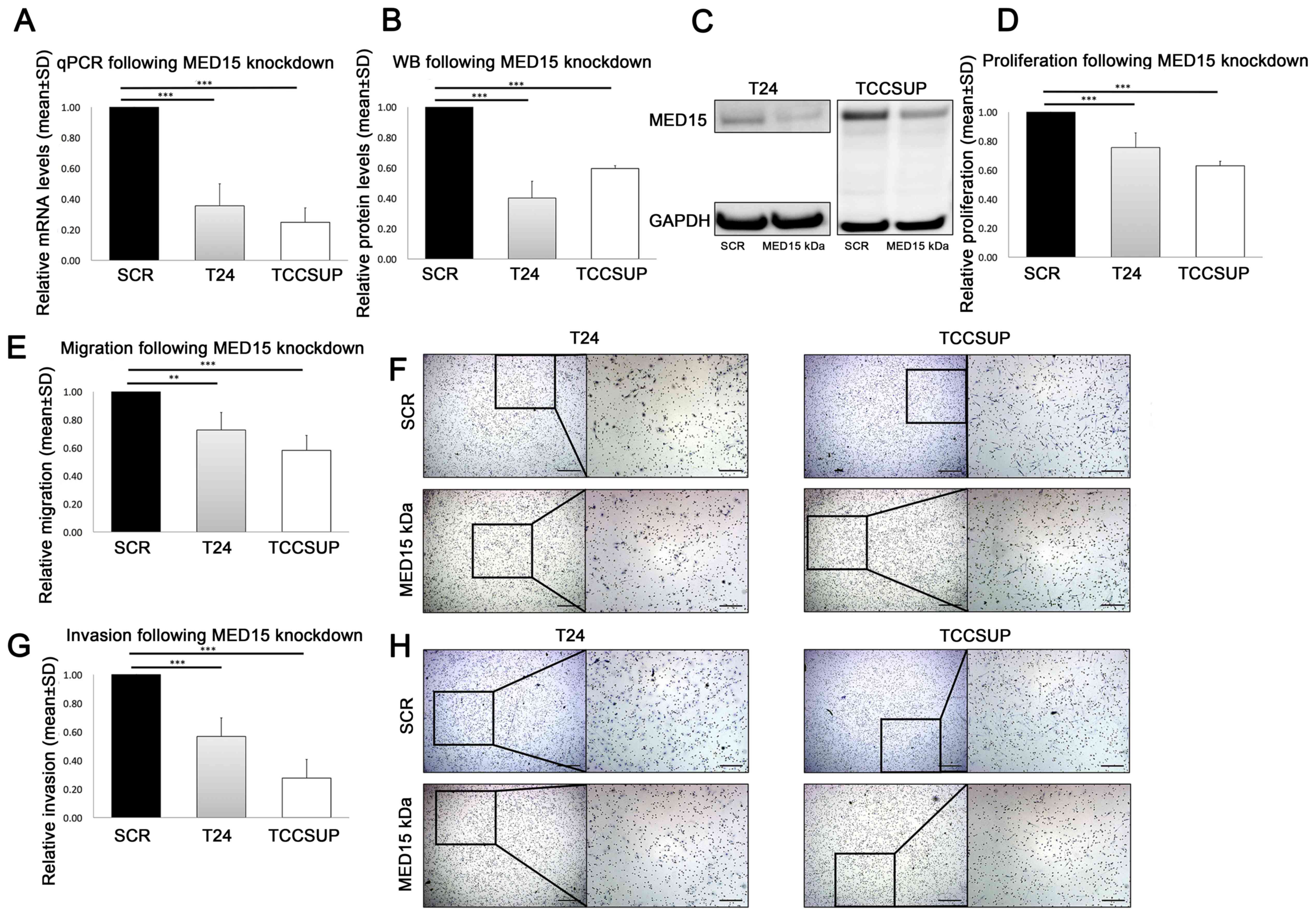
To investigate the functional role of the subunit
MED15 in malignant progression and metastasis, we performed in
vitro siRNA-mediated knockdown of MED15 in the BCa cell
lines T24 (P≤0.001) and TCCSUP (P≤0.001; Fig. 5A-C). Thereafter, we compared the
measured proliferative activity of scrambled control and
MED15 knockdown cells and observed a significant decrease in
T24 (P=≤0.001) and TCCSUP (P≤0.001; Fig.
5D). Further, migration and invasion were significantly reduced
in T24 (migration P=0.003, invasion P≤0.001) and TCCSUP (migration
P≤0.001, invasion P≤0.001) cells following the transient
MED15 knockdown, compared to scrambled control cells
(Fig. 5E-H). In conclusion,
MED15 knockdown led to reduced malignant behaviour in the
BCa cell lines T24 and TCCSUP.
Discussion
Considering its high incidence, urothelial BCa is a
high-priority concern in urology. In the non-muscle invasive stage,
the BCa requires intensive treatment due to the frequent
recurrences. Additionally, the muscle invasive stage demands
extensive therapy and care (2–4). Due to
this, comprehensive studies of BCa have to be continued and
expanded.
Over the last ten years, the Mediator complex has
been well investigated and illuminated in humans, yeast, and plants
(21,22). The Mediator complex is a multi-protein
complex that links enhancer bound transcription factors (TF) to
specific promoter regions and, thereby, activates transcription. In
mammals, this complex consists of ≈33 subunits, and its composition
can change to alter its biological function. Furthermore, the
specificity of the Mediator complex is mediated by various domains
that interact with different TFs (23). The precise mechanisms through which
the Mediator complex regulates Pol II activity remain poorly
understood; However it has been observed that they involve
extensive protein-protein interactions between the Mediator
complex, pol II, and other general and gene-specific transcription
regulatory factors (21).
The roles of individual subunits have been described
in various diseases, such as intellectual disability (24), cardiovascular disease (25), and carcinogenesis (26). However, much remains unknown.
Little information is available pertaining to the
BCa especially. Till now, only the overexpression of MED19
has been observed to be associated with higher tumor stages
(14), while a MED1
underexpression accompanies a worse survival rate (15).
In our investigation, we found out that the
knockdown of MED15 in BCa cell lines leads to a
downregulation of the tumor parameters of proliferation, migration,
and invasion in the functional analysis. These parameters are
decisive for the recurrence and progression of urothelial BCa.
Unfortunately, the survival data of mRNA and IHC analyses did not
show any significant differences in the overexpression of
MED15. Some results from the IHC analysis also contrast the
results obtained from the cell line examinations (higher expression
of MED15 in NMIBC or in low grading status). The cause of these
deviant results may be the rather small number of samples in the
IHC analysis. In addition, the cohort shows significant differences
in the individual group sizes (NMIBC n=24, MIBC n=102, benign
n=18). Expecting, a larger and balanced number of samples would
lead to possibly more clear results.
Controversial expression profiles can also be found
for the same subunit in other studies, MED1 for example. In
osteosarcoma, the knockdown of MED1 reduced the
proliferation (27), whereas in
non-small lung cancer the loss of MED1 led to an increase of
invasion and metastases (28).
Possible explanations for aberrant expression
profiles can be found in the connection to different pathways. For
MED1, the interaction with receptor tyrosine kinases (RTKs)
resulted in Tamoxifen resistance (29). The involvement of the Apaf-1-dependent
pathway/Apaf1-dependent apoptotic pathway was found in the
downregulation of MED19 in human laryngocarcinoma HEp2 cells
(30). For MED15, a connection to the
TGFβ- (13) and PI3K/mTOR- (31) signalling pathways was already
described in head and neck cancer and prostate cancer. Whether this
is also true for BCa is to be determined in subsequent
investigations. Certainly, for BCa, the involvement of these and
more signalling pathways have been described: The PI3K/Akt-, as a
cellular signalling pathway, rapidly activates downstream pathways
of inflammation and neoplasia in bladder urothelium (32). Additionally, the p53/pRb signal
pathway regulates the BCa cell cycle (33). Furthermore, the tyrosine kinase
pathway with the epidermal growth factor receptor-(EGFR) family was
reported to be overexpressed in BCa, and tyrosine kinase inhibitors
(TKIs) have been suggested as treatment by the strong inhibition of
the cell proliferation and induction of cell cycle G1 arrest and
apoptosis in UM-UC-5 cells (34).
Thus, further analyses are necessary, especially
that will include larger sample sizes. Additionally, whether the
mentioned and already described signalling pathways for MED15 or
BCa are also included in the regulation of MED15 in BCa, needs to
be investigated in further studies. However, we concluded that
MED15 is involved in the important tumor parameters proliferation,
migration and invasion.
We concluded that the knockdown of the Mediator
complex subunit MED15 restrains urothelial BCa cells' malignancy.
Our work is to be considered as the first overview-study on the
role of MED15 in BCa, and further investigations pertaining to the
underlying signal pathways are necessary.
Acknowledgements
Not applicable.
Funding
The study was supported by the Ferdinand Eisenberger
Fellowship of the German Society of Urology (DGU; SYI1/FE-13) and
the Maria von Linden-Program of the University of Bonn for IS.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
IS, RW and JE designed the study. IS, RW, DS, SS
performed the experiments. IS, RW, TM, GK, SCM and JE analyzed and
interpreted the results. IS wrote the manuscript with input from
all authors. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
This investigation was conducted in accordance with
ethical standards, the Declaraion of Helsinki, and national and
international guidelines. Ethical approval for using human material
in the present study was obtained from the Internal Review Board of
the University Hospital of Bonn (IRB nos. 036/08 and 093/12). The
participants of the study agreed in writing beforehand and were
anonymised before inclusion in the study cohort.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Antoni S, Ferlay J, Soerjomataram I, Znaor
A, Jemal A and Bray F: Bladder cancer incidence and mortality: A
global overview and recent trends. Eur Urol. 71:96–108. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sievert KD, Amend B, Nagele U, Schilling
D, Bedke J, Horstmann M, Hennenlotter J, Kruck S and Stenzl A:
Economic aspects of bladder cancer: What are the benefits and
costs? World J Urol. 27:295–300. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Babjuk M, Oosterlinck W, Sylvester R,
Kaasinen E, Böhle A, Palou-Redorta J and Rouprêt M: European
association of urology (EAU): EAU guidelines on non-muscle-invasive
urothelial carcinoma of the bladder, the 2011 update. Eur Urol.
59:997–1008. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stenzl A, Cowan NC, De Santis M, Kuczyk
MA, Merseburger AS, Ribal MJ, Sherif A and Witjes JA: European
association of urology (EAU): Treatment of muscle-invasive and
metastatic bladder cancer: Update of the EAU guidelines. Eur Urol.
59:1009–1018. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wei L, Chintala S, Ciamporcero E,
Ramakrishnan S, Elbanna M, Wang J, Hu Q, Glenn ST, Murakami M, Liu
L, et al: Genomic profiling is predictive of response to cisplatin
treatment but not to PI3K inhibition in bladder cancer
patient-derived xenografts. Oncotarget. 7:76374–76389. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Glaser AP, Fantini D, Shilatifard A,
Schaeffer EM and Meeks JJ: The evolving genomic landscape of
urothelial carcinoma. Nat Rev Urol. 14:215–229. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Choi W, Ochoa A, McConkey DJ, Aine M,
Höglund M, Kim WY, Real FX, Kiltie AE, Milsom I, Dyrskjøt L and
Lerner SP: Genetic alterations in the molecular subtypes of bladder
cancer: Illustration in the cancer genome atlas dataset. Eur Urol.
72:354–365. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Malik S and Roeder RG: The metazoan
mediator co-activator complex as an integrative hub for
transcriptional regulation. Nat Rev Genet. 11:761–772. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cai G, Imasaki T, Takagi Y and Asturias
FJ: Mediator structural conservation and implications for the
regulation mechanism. Structure. 17:559–567. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ansari SA and Morse RH: Mechanisms of
mediator complex action in transcriptional activation. Cell Mol
Life Sci. 70:2743–2756. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Boube M, Joulia L, Cribbs DL and Bourbon
HM: Evidence for a mediator of RNA polymerase II transcriptional
regulation conserved from yeast to man. Cell. 110:143–151. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao M, Yang X, Fu Y, Wang H, Ning Y, Yan
J, Chen YG and Wang G: Mediator MED15 modulates transforming growth
factor beta (TGFβ)/Smad signaling and breast cancer cell
metastasis. J Mol Cell Biol. 5:57–60. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Offermann A, Shaikhibrahim Z and Perner S:
MED15: A potential biomarker for head and neck squamous cell
carcinoma? Biomark Med. 9:939–941. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang H, Jiang H, Wang W, Gong J, Zhang L,
Chen Z and Ding Q: Expression of Med19 in bladder cancer tissues
and its role on bladder cancer cell growth. Urol Oncol. 30:920–927.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Klümper N, Syring I, Vogel W, Schmidt D,
Müller SC, Ellinger J, Shaikhibrahim Z, Brägelmann J and Perner S:
Mediator complex subunit MED1 protein expression Is decreased
during bladder cancer progression. Front Med (Lausanne).
4:302017.PubMed/NCBI
|
|
16
|
Gao J, Aksoy BA, Dogrusoz U, Dresdner G,
Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, et al:
Integrative analysis of complex cancer genomics and clinical
profiles using the cBioPortal. Sci Signal. 6:pl12013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cerami E, Gao J, Dogrusoz U, Gross BE,
Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, et
al: The cBio cancer genomics portal: An open platform for exploring
multidimensional cancer genomics data. Cancer Discov. 2:401–404.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tomczak K, Czerwińska P and Wiznerowicz M:
The cancer genome atlas (TCGA): An immeasurable source of
knowledge. Contemp Oncol (Pozn). 19:A68–A77. 2015.PubMed/NCBI
|
|
19
|
Scheble VJ, Braun M, Wilbertz T, Stiedl
AC, Petersen K, Schilling D, Reischl M, Seitz G, Fend F,
Kristiansen G and Perner S: ERG rearrangement in small cell
prostatic and lung cancer. Histopathology. 56:937–943. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Scheble VJ, Braun M, Beroukhim R, Mermel
CH, Ruiz C, Wilbertz T, Stiedl AC, Petersen K, Reischl M, Kuefer R,
et al: ERG rearrangement is specific to prostate cancer and does
not occur in any other common tumor. Mod Pathol. 23:1061–1067.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Allen BL and Taatjes DJ: The mediator
complex: A central integrator of transcription. Nat Rev Mol Cell
Biol. 16:155–166. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dolan WL and Chapple C: Conservation and
divergence of Mediator structure and function: Insights from
plants. Plant Cell Physiol. 58:4–21. 2017.PubMed/NCBI
|
|
23
|
Kosan C and Godmann M: Bringing light into
gene regulation in hematopoietic stem cells by the Mediator
complex. Stem Cell Investig. 4:102017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Caro-Llopis A, Rosello M, Orellana C,
Oltra S, Monfort S, Mayo S and Martinez F: De novo mutations in
genes of Mediator complex causing syndromic intellectual
disability: Mediatorpathy or transcriptomopathy? Pediatr Res.
80:809–815. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schiano C, Casamassimi A, Vietri MT,
Rienzo M and Napoli C: The roles of Mediator complex in
cardiovascular diseases. Biochim Biophys Acta. 1839:444–451. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schiano C, Casamassimi A, Rienzo M, de
Nigris F, Sommese L and Napoli C: Involvement of Mediator complex
in malignancy. Biochim Biophys Acta. 1845:66–83. 2014.PubMed/NCBI
|
|
27
|
Jiang C, Chen H, Shao L and Wang Q:
MicroRNA-1 functions as a potential tumor suppressor in
osteosarcoma by targeting Med1 and Med31. Oncol Rep. 32:1249–1256.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim HJ, Roh MS, Son CH, Kim AJ, Jee HJ,
Song N, Kim M, Seo SY, Yoo YH and Yun J: Loss of Med1/TRAP220
promotes the invasion and metastasis of human non-small-cell lung
cancer cells by modulating the expression of metastasis-related
genes. Cancer Lett. 321:195–202. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mansouri S, Naghavi-Al-Hosseini F,
Farahmand L and Majidzadeh-A K: MED1 may explain the interaction
between receptor tyrosine kinases and ERα66 in the complicated
network of Tamoxifen resistance. Eur J Pharmacol. 804:78–81. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhao Y, Meng Q, Gao X, Zhang L and An L:
Down-regulation of Mediator complex subunit 19 (Med19) induces
apoptosis in human laryngocarcinoma HEp2 cells in an
Apaf-1-dependent pathway. Am J Transl Res. 9:755–761.
2017.PubMed/NCBI
|
|
31
|
Offermann A, Vlasic I, Syring I, Vogel W,
Ruiz C, Zellweger T, Rentsch CA, Hagedorn S, Behrends J, Nowak M,
et al: MED15 overexpression in prostate cancer arises during
androgen deprivation therapy via PI3K/mTOR signaling. Oncotarget.
8:7964–7976. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tamarkin FJ, Kang WS, Cohen JJ, Wheeler MA
and Weiss RM: A role for Akt in the rapid regulation of
inflammatory and apoptotic pathways in mouse bladder. Naunyn
Schmiedebergs Arch Pharmacol. 373:349–359. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kiselyov A, Bunimovich-Mendrazitsky S and
Startsev V: Key signaling pathways in the muscle-invasive bladder
carcinoma: Clinical markers for disease modeling and optimized
treatment. Int J Cancer. 138:2562–2569. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li J, Lv B, Li X, He Z and Zhou K:
Apoptosis-related molecular differences for response to tyrosin
kinase inhibitors in drug-sensitive and drug-resistant human
bladder cancer cells. J Cancer Res Ther. 9:668–671. 2013.
View Article : Google Scholar : PubMed/NCBI
|