|
1
|
Caserta S, Kern F, Cohen J, Drage S,
Newbury SF and Llewelyn MJ: Circulating plasma microRNAs can
differentiate human sepsis and systemic inflammatory response
syndrome (SIRS). Sci Rep. 6:280062016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang H, Ward MF and Sama AE: Targeting
HMGB1 in the treatment of sepsis. Expert Opin Ther Targets.
18:257–268. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Huang W, Tang Y and Li L: HMGB1, a potent
proinflammatory cytokine in sepsis. Cytokine. 51:119–126. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
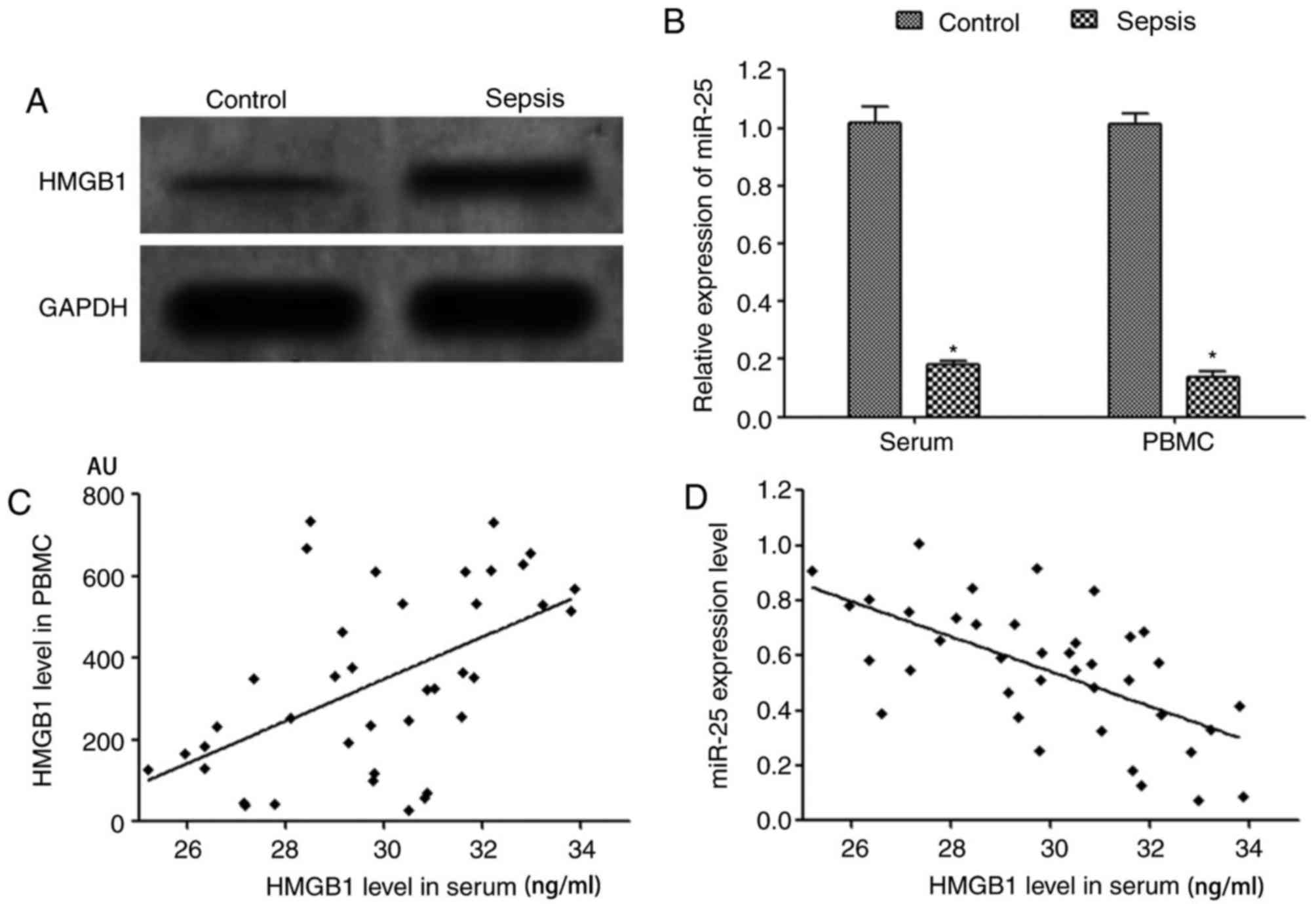
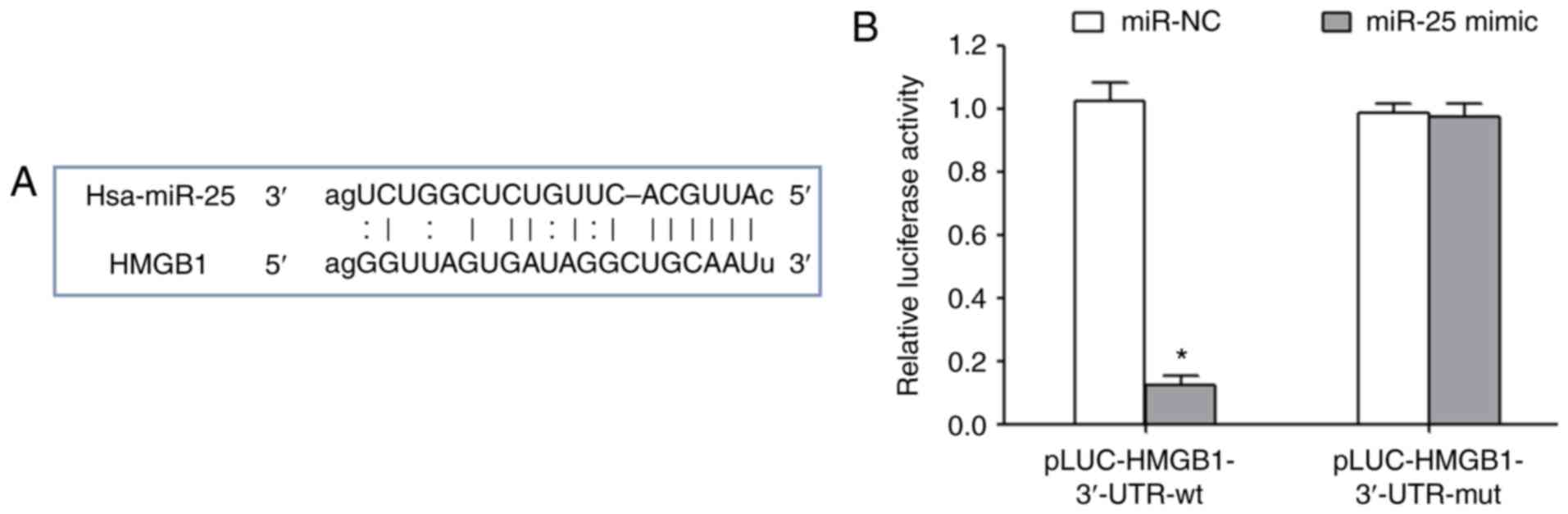
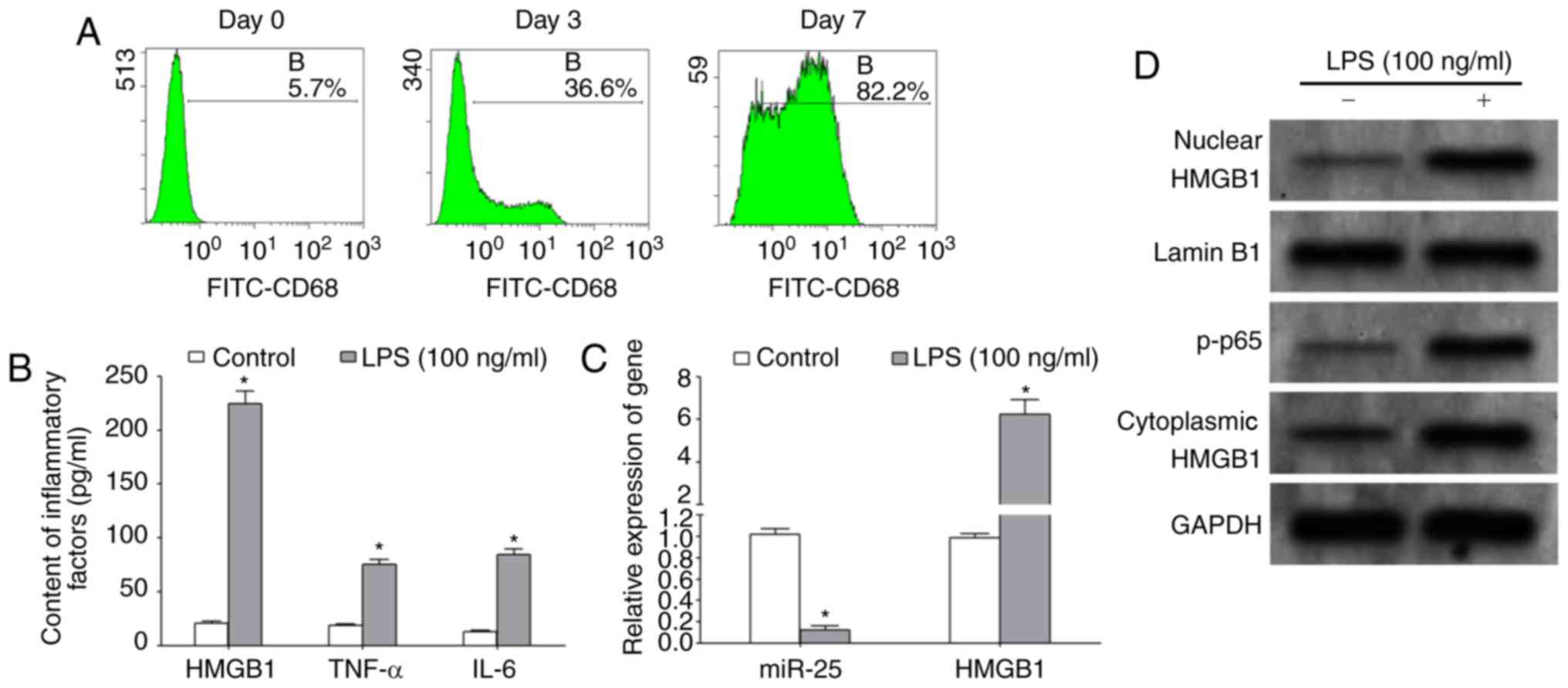
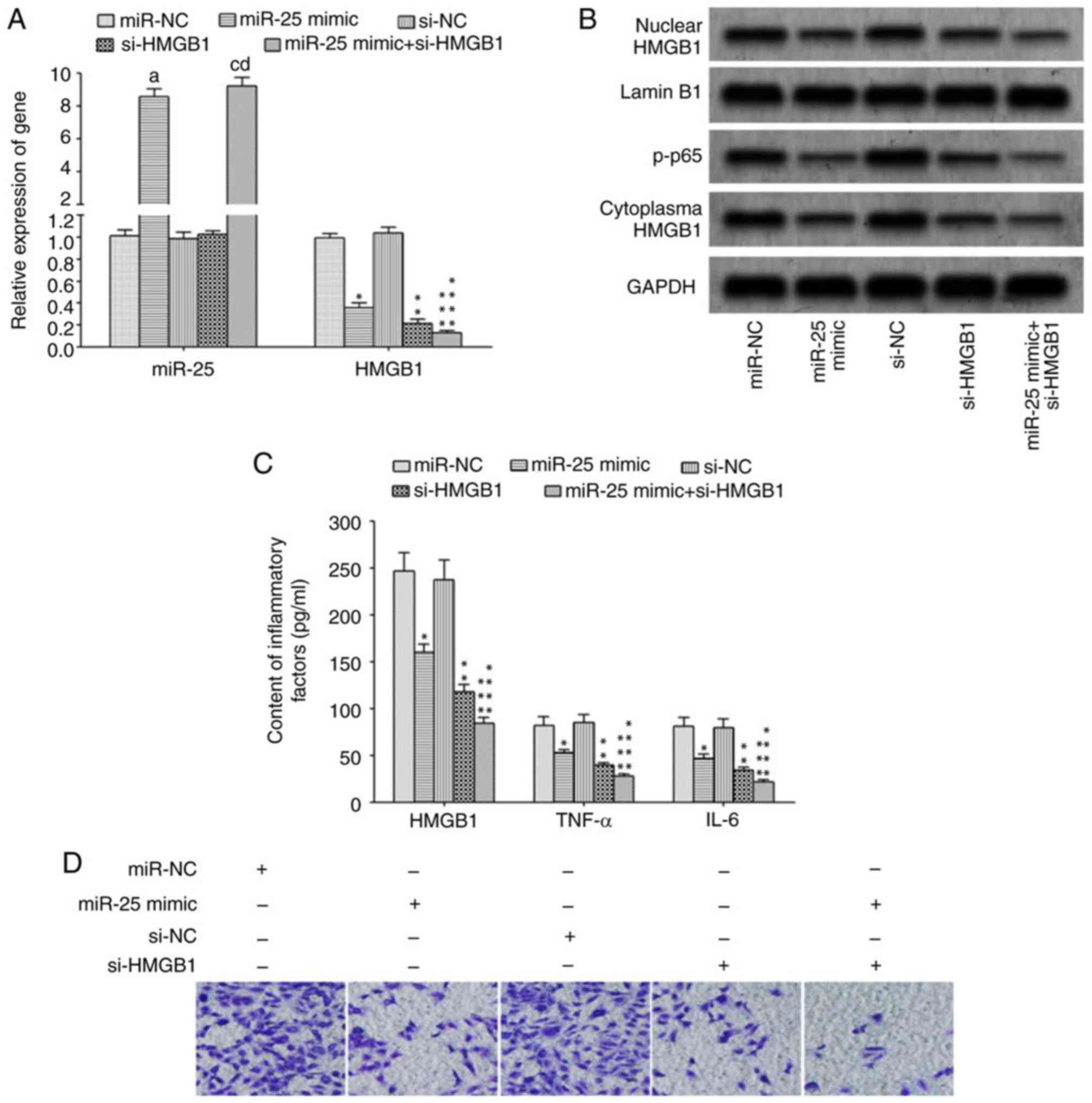
Huston JM, Wang H, Ochani M, Ochani K,
Rosas-Ballina M, Gallowitsch-Puerta M, Ashok M, Yang L, Tracey KJ
and Yang H: Splenectomy protects against sepsis lethality and
reduces serum HMGB1 levels. J Immunol. 181:3535–3539. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang SY, Li ZJ, Wang X, Li WF and Lin ZF:
Effect of ulinastatin on HMGB1 expression in rats with acute lung
injury induced by sepsis. Genet Mol Res. 14:4344–4353. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bulun SE and Nezhat C: Aromatase,
microRNA, and inflammation: A complex relationship. Fertil Steril.
106:552–553. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang X, Guo Y, Wang C, Yu X and Yu H:
MicroRNA-142-3p inhibits chondrocyte apoptosis and inflammation in
osteoarthritis by targeting HMGB1. Inflammation. 39:1718–1728.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhou W, Wang J, Li Z, Li J and Sang M:
MicroRNA-2055b inhibits HMGB1 expression in LPS-induced sepsis. Int
J Mol Med. 38:312–318. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Benz F, Roy S, Trautwein C, Roderburg C
and Luedde T: Circulating microRNAs as biomarkers for sepsis. Int J
Mol Sci. 17:782016. View Article : Google Scholar
|
|
10
|
Weiss M, Huber-Lang M, Taenzer M, Traeger
K, Altherr J, Kron M, Hay B and Schneider M: Different patient case
mix by applying the 2003 SCCM/ESICM/ACCP/ATS/SIS sepsis definitions
instead of the 1992 ACCP/SCCM sepsis definitions in surgical
patients: A retrospective observational study. BMC Med Inform Decis
Mak. 9:252009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ritchie W, Rasko JE and Flamant S:
MicroRNA target prediction and validation. Adv Exp Med Biol.
774:39–53. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Boyd JH, Russell JA and Fjell CD: The
meta-genome of sepsis: Host genetics, pathogens and the acute
immune response. J Innate Immun. 6:272–283. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Czura CJ, Yang H, Amella CA and Tracey KJ:
HMGB1 in the immunology of sepsis (not septic shock) and arthritis.
Adv Immunol. 84:181–200. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lin SM, Chung FT, Kuo CH, Chou PC, Wang
TY, Chang PJ, Lo YL, Huang CD, Lin HC, Wang CH and Kuo HP:
Circulating angiopopietin-1 correlates with the clinical course of
multiple organ dysfunction syndrome and mortality in patients with
severe sepsis. Medicine (Baltimore). 94:e8782015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guirgis FW, Khadpe JD, Kuntz GM, Wears RL,
Kalynych CJ and Jones AE: Persistent organ dysfunction after severe
sepsis: A systematic review. J Crit Care. 29:320–326. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chavan SS, Huerta PT, Robbiati S,
Valdes-Ferrer SI, Ochani M, Dancho M, Frankfurt M, Volpe BT, Tracey
KJ and Diamond B: HMGB1 mediates cognitive impairment in sepsis
survivors. Mol Med. 18:930–937. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gentile LF and Moldawer LL: HMGB1 as a
therapeutic target for sepsis: It's all in the timing! Expert Opin
Ther Targets. 18:1–245. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shao J, Zhao M, Tong M, Wei J, Wise MR,
Stone P, Chamley L and Chen Q: Increased levels of HMGB1 in
trophoblastic debris may contribute to preeclampsia. Reproduction.
152:775–784. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang KC, Ko YS, Kim HJ, Nam DY and Lee
DU: 13-Methylberberine reduces HMGB1 release in LPS-activated
RAW264.7 cells and increases the survival of septic mice through
AMPK/P38 MAPK activation. Int Immunopharmacol. 40:269–276. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhao H, Liu Z, Liu W, Han X and Zhao M:
Betulin attenuates lung and liver injuries in sepsis. Int
Immunopharmacol. 30:50–56. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gil M, Kim YK, Hong SB and Lee KJ:
Naringin decreases TNF-α and HMGB1 release from LPS-stimulated
macrophages and improves survival in a CLP-induced sepsis mice.
PLoS One. 11:e01641862016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kumar V: Targeting macrophage
immunometabolism: Dawn in the darkness of sepsis. Int
Immunopharmacol. 58:173–185. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang Z, Zhang L, Zhou C and Wu H:
Ketamine inhibits LPS-induced HGMB1 release in vitro and in vivo.
Int Immunopharmacol. 23:14–26. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yao L, Liu Z, Zhu J, Li B, Chai C and Tian
Y: Clinical evaluation of circulating microRNA-25 level change in
sepsis and its potential relationship with oxidative stress. Int J
Clin Exp Pathol. 8:7675–7684. 2015.PubMed/NCBI
|
|
26
|
Karlsson S, Pettila V, Tenhunen J,
Laru-Sompa R, Hynninen M and Ruokonen E: HMGB1 as a predictor of
organ dysfunction and outcome in patients with severe sepsis.
Intensive Care Med. 34:1046–1053. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen XL, Sun L, Guo F, Wang F, Liu S,
Liang X, Wang RS, Wang YJ and Sun YX: High-mobility group box-1
induces proinflammatory cytokines production of Kupffer cells
through TLRs-dependent signaling pathway after burn injury. PLoS
One. 7:e506682012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mudaliar H, Pollock C, Komala MG, Chadban
S, Wu H and Panchapakesan U: The role of toll-like receptor
proteins (TLR) 2 and 4 in mediating inflammation in proximal
tubules. Am J Physiol Renal Physiol. 305:F143–F154. 2013.
View Article : Google Scholar : PubMed/NCBI
|