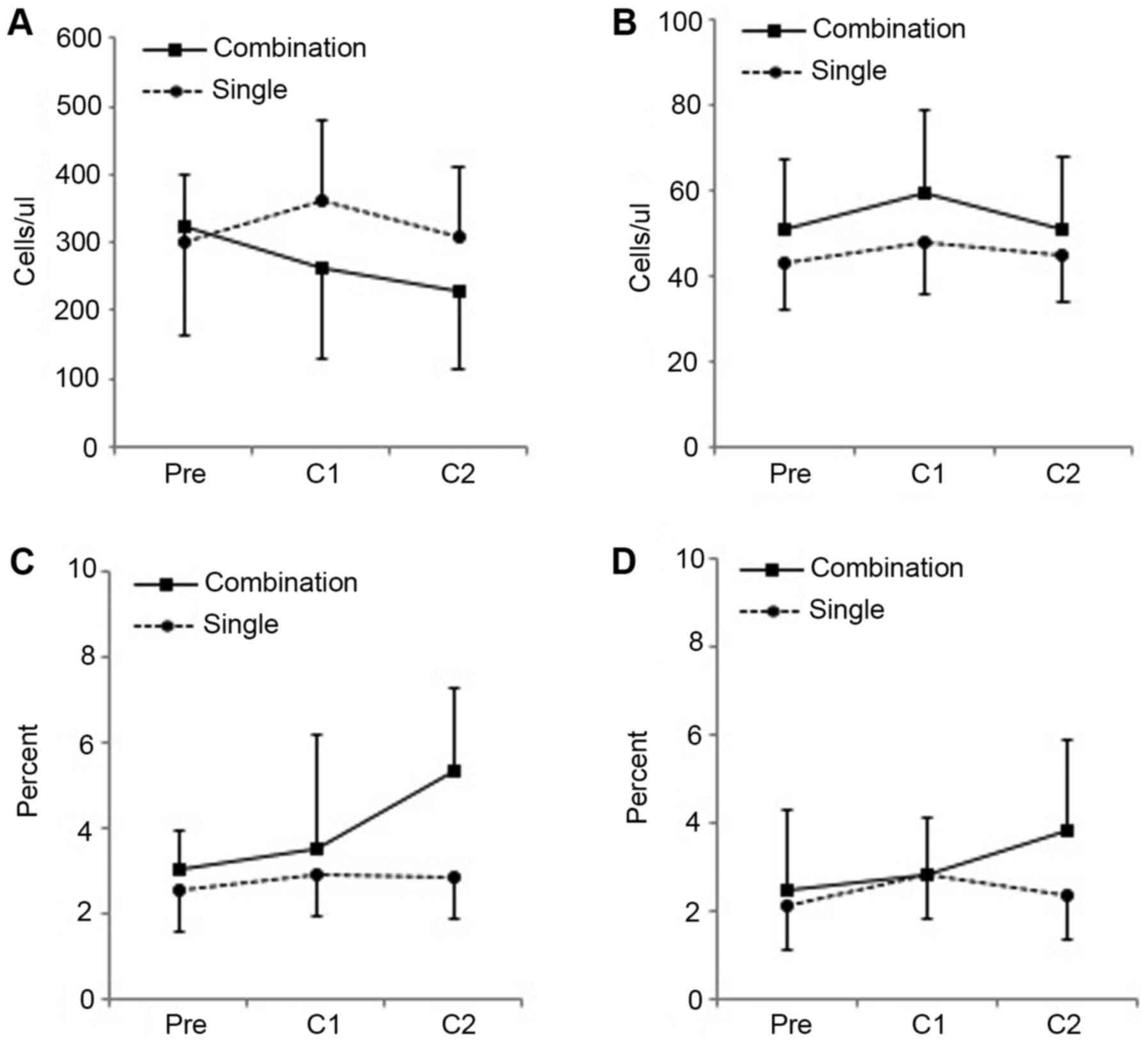
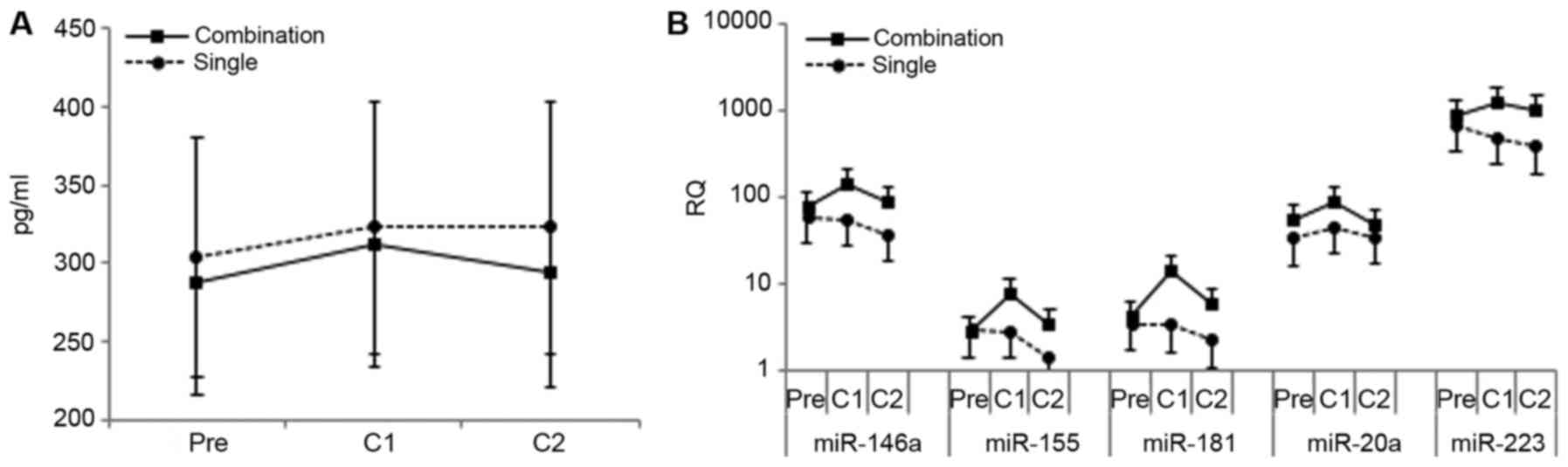
|
1
|
Azzoli CG, Temin S, Aliff T, Baker S Jr,
Brahmer J, Johnson DH, Laskin JL, Masters G, Milton D, Nordquist L,
et al: 2011 focused update of 2009 American society of clinical
oncology clinical practice guideline update on chemotherapy for
stage IV non-small-cell lung cancer. J Clin Oncol. 29:3825–3831.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Reck M, Rodríguez-Abreu D, Robinson AG,
Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe
S, et al: Pembrolizumab versus Chemotherapy for PD-L1-positive
non-small-cell lung cancer. N Engl J Med. 375:1823–1833. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brahmer J, Reckamp KL, Baas P, Crinò L,
Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE,
Holgado E, et al: Nivolumab versus docetaxel in advanced
squamous-cell non-small-cell lung cancer. N Engl J Med.
373:123–135. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rittmeyer A, Barlesi F, Waterkamp D, Park
K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols
MC, et al: Atezolizumab versus docetaxel in patients with
previously treated non-small-cell lung cancer (OAK): A phase 3,
open-label, multicentre randomised controlled trial. Lancet.
389:255–265. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rizvi NA, Hellmann MD, Brahmer JR,
Juergens RA, Borghaei H, Gettinger S, Chow LQ, Gerber DE, Laurie
SA, Goldman JW, et al: Nivolumab in combination with platinum-based
doublet chemotherapy for first-line treatment of advanced
non-small-cell lung cancer. J Clin Oncol. 34:2969–2979. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Langer CJ, Gadgeel SM, Borghaei H,
Papadimitrakopoulou VA, Patnaik A, Powell SF, Gentzler RD, Martins
RG, Stevenson JP, Jalal SI, et al: Carboplatin and pemetrexed with
or without pembrolizumab for advanced, non-squamous non-small-cell
lung cancer: A randomised, phase 2 cohort of the open-label
KEYNOTE-021 study. Lancet Oncol. 17:1497–1508. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sculier JP, Chansky K, Crowley JJ, Van
Meerbeeck J and Goldstraw P: International Staging Committee and
Participating Institutions: The impact of additional prognostic
factors on survival and their relationship with the anatomical
extent of disease expressed by the 6th edition of the TNM
classification of malignant tumors and the proposals for the 7th
edition. J Thorac Oncol. 3:457–466. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bunn PA Jr: Chemotherapy for advanced
non-small-cell lung cancer: Who, what, when, why? J Clin Oncol. 20
18 Suppl:23S–33S. 2002.PubMed/NCBI
|
|
9
|
Tartarone A and Aieta M: Treatment of
performance status 2 patients with advanced non-small-cell lung
cancer: What we know and what we don't know. Future Oncol.
5:837–841. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lilenbaum RC, Cashy J, Hensing TA, Young S
and Cella D: Prevalence of poor performance status in lung cancer
patients: Implications for research. J Thorac Oncol. 3:125–129.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Correale P, Cerretani D, Remondo C,
Martellucci I, Marsili S, La Placa M, Sciandivasci A, Paolelli L,
Pascucci A, Rossi M, et al: A novel metronomic chemotherapy regimen
of weekly platinum and daily oral etoposide in high-risk non-small
cell lung cancer patients. Oncol Rep. 16:133–140. 2006.PubMed/NCBI
|
|
12
|
Bilir C, Durak S, Kızılkaya B,
Hacıbekiroglu I, Nayır E and Engin H: Efficacy of metronomic
vinorelbine in elderly patients with advanced non-small-cell lung
cancer and poor performance status. Curr Oncol. 24:e199–e204. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zwitter M, Kovac V, Rajer M, Vrankar M and
Smrdel U: Two schedules of chemotherapy for patients with non-small
cell lung cancer in poor performance status: A phase II randomized
trial. Anticancer Drugs. 21:662–668. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee KW, Lim JH, Kim JH, Lee CT and Lee JS:
Weekly low-dose docetaxel for salvage chemotherapy in pretreated
elderly or poor performance status patients with non-small cell
lung cancer. J Korean Med Sci. 23:992–998. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Juan O, Albert A, Ordoño F, Casany R,
Carañana V, Campos JM and Alberola V: Low-dose weekly paclitaxel as
second-line treatment for advanced non-small cell lung cancer: A
phase II study. Jpn J Clin Oncol. 32:449–454. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hainsworth JD, Burris HA III and Greco FA:
Weekly docetaxel as a single agent and in combination with
gemcitabine in elderly and poor performance status patients with
advanced non-small cell lung cancer. Semin Oncol. 28 3 Suppl
9:S21–S25. 2001. View Article : Google Scholar
|
|
17
|
Santegoets SJ, Welters MJ and van der Burg
SH: Monitoring of the immune dysfunction in cancer patients.
Vaccines (Basel). 4:pii: E29. 2016.PubMed/NCBI
|
|
18
|
de Biasi AR, Villena-Vargas J and
Adusumilli PS: Cisplatin-induced antitumor immunomodulation: A
review of preclinical and clinical evidence. Clin Cancer Res.
20:5384–5391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Javeed A, Ashraf M, Riaz A, Ghafoor A,
Afzal S and Mukhtar MM: Paclitaxel and immune system. Eur J Pharm
Sci. 38:283–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang CL, Hsu YT, Wu CC, Lai YZ, Wang C,
Yang YC, Wu TC and Hung CF: Dose-dense chemotherapy improves
mechanisms of antitumor immune response. Cancer Res. 73:119–127.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–47.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang A, Zhang B, Wang B, ZAhang F, Fan KX
and Guo YJ: Increased CD14(+)HLA-DR (−/low) myeloid-derived
suppressor cells correlate with extrathoracic metastasis and poor
response to chemotherapy in non-small cell lung cancer patients.
Cancer Immunol Immunother. 62:1439–1451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kamphorst AO, Pillai RN, Yang S, Nasti TH,
Akondy RS, Wieland A, Sica GL, Yu K, Koenig L, Patel NT, et al:
Proliferation of PD-1+ CD8 T cells in peripheral blood after
PD-1-targeted therapy in lung cancer patients. Proc Natl Acad Sci
USA. 114:4993–4998. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou J, Mahoney KM, Giobbie-Hurder A, Zhao
F, Lee S, Liao X, Rodig S, Li J, Wu X, Butterfield LH, et al:
Soluble PD-L1 as a biomarker in malignant melanoma treated with
checkpoint blockade. Cancer Immunol Res. 5:480–492. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Okuma Y, Hosomi Y, Nakahara Y, Watanabe K,
Sagawa Y and Homma S: High plasma levels of soluble programmed cell
death ligand 1 are prognostic for reduced survival in advanced lung
cancer. Lung Cancer. 104:1–6. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang J, Gao J, Li Y, Nie J, Dai L, Hu W,
Chen X, Han J, Ma X, Tian G, et al: Circulating PD-L1 in NSCLC
patients and the correlation between the level of PD-L1 expression
and the clinical characteristics. Thorac Cancer. 6:534–538. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dronca RS, Mansfield AS, Liu X, Harrington
S, Enninga EA, Kottschade LA, Koo CW, McWilliams RR, Block MS,
Nevala WK, et al: Bim and soluble PD-L1 (sPD-L1) as predictive
biomarkers of response to anti-PD-1 therapy in patients with
melanoma and lung carcinoma. J Clin Oncol. 35 15 Suppl:S115342017.
View Article : Google Scholar
|
|
29
|
Paladini L, Fabris L, Bottai G, Raschioni
C, Calin GA and Santarpia L: Targeting microRNAs as key modulators
of tumor immune response. J Exp Clin Cancer Res. 35:1032016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Triozzi PL, Achberger S, Aldrich W, Elson
P, Garcia J and Dreicer R: Differential immunologic and microRNA
effects of 2 dosing regimens of recombinant human
granulocyte/macrophage colony stimulating factor. J Immunother.
35:587–594. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Achberger S, Aldrich W, Tubbs R, Crabb JW,
Singh AD and Triozzi PL: Circulating immune cell and microRNA in
patients with uveal melanoma developing metastatic disease. Mol
Immunol. 58:182–186. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen J, Yuan L, Fan Q, Su F, Chen Y and Hu
S: Adjuvant effect of docetaxel on the immune responses to
influenza A H1N1 vaccine in mice. BMC Immunol. 13:362012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yuan L, Wu L, Chen J, Wu Q and Hu S:
Paclitaxel acts as an adjuvant to promote both Th1 and Th2 immune
responses induced by ovalbumin in mice. Vaccine. 28:4402–4410.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Volk V, Cathomas R, Mark M, von Moos R,
Klingbiel D, Brossart P and Mey U: Weekly carboplatin in
combination with weekly paclitaxel in the treatment of metastatic
non-small cell lung cancer: A single center 10-year experience.
Support Care Cancer. 24:2119–2128. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Schuette W, Blankenburg T, Guschall W,
Dittrich I, Schroeder M, Schweisfurth H, Chemaissani A, Schumann C,
Dickgreber N, Appel T and Ukena D: Multicenter randomized trial for
stage IIIB/IV non-small-cell lung cancer using every-3-week versus
weekly paclitaxel/carboplatin. Clin Lung Cancer. 7:338–343. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Caiado J and Castells M: Presentation and
diagnosis of hypersensitivity to platinum drugs. Curr Allergy
Asthma Rep. 15:152015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Carthon BC, Wolchok JD, Yuan J, Kamat A,
Tang Ng DS, Sun J, Ku G, Troncoso P, Logothetis CJ, Allison JP and
Sharma P: Preoperative CTLA-4 blockade: Tolerability and immune
monitoring in the setting of a presurgical clinical trial. Clin
Cancer Res. 16:2861–2871. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ribas A, Shin DS, Zaretsky J, Frederiksen
J, Cornish A, Avramis E, Seja E, Kivork C, Siebert J, Kaplan-Lefko
P, et al: PD-1 blockade expands intratumoral memory T cells. Cancer
Immunol Res. 4:194–203. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Weber JS, Kudchadkar RR, Yu B, Gallenstein
D, Horak CE, Inzunza HD, Zhao X, Martinez AJ, Wang W, Gibney G, et
al: Safety, efficacy, and biomarkers of nivolumab with vaccine in
ipilimumab-refractory or -naive melanoma. J Clin Oncol.
31:4311–4318. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Heegaard NH, Schetter AJ, Welsh JA, Yoneda
M, Bowman ED and Harris CC: Circulating micro-RNA expression
profiles in early stage nonsmall cell lung cancer. Int J Cancer.
130:1378–2886. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shen J, Todd NW, Zhang H, Yu L, Lingxiao
X, Mei Y, Guarnera M, Liao J, Chou A, Lu CL, et al: Plasma
microRNAs as potential biomarkers for non-small-cell lung cancer.
Lab Invest. 91:579–587. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zheng D, Haddadin S, Wang Y, Gu LQ, Perry
MC, Freter CE and Wang MX: Plasma microRNAs as novel biomarkers for
early detection of lung cancer. Int J Clin Exp Pathol. 4:575–586.
2011.PubMed/NCBI
|
|
43
|
Zhu W, He J, Chen D, Zhang B, Xu L, Ma H,
Liu X, Zhang Y and Le H: Expression of miR-29c, miR-93, and miR-429
as potential biomarkers for detection of early stage non-small lung
cancer. PLoS One. 9:e877802014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang H, Mao F, Shen T, Luo Q, Ding Z,
Qian L and Huang J: Plasma miR-145, miR-20a, miR-21 and miR-223 as
novel biomarkers for screening early-stage non-small cell lung
cancer. Oncol Lett. 13:669–676. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wu C, Cao Y, He Z, He J, Hu C, Duan H and
Jiang J: Serum levels of miR-19b and miR-146a as prognostic
biomarkers for non-small cell lung cancer. Tohoku J Exp Med.
232:85–95. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhou M, Liu Z, Zhao Y, Ding Y, Liu H, Xi
Y, Xiong W, Li G, Lu J, Fodstad O, Riker AI and Tan M:
MicroRNA-125b confers the resistance of breast cancer cells to
paclitaxel through suppression of pro-apoptotic Bcl-2 antagonist
killer 1 (Bak1) expression. J Biol Chem. 285:21496–21507. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kong F, Sun C, Wang Z, Han L, Weng D, Lu Y
and Chen G: miR-125b confers resistance of ovarian cancer cells to
cisplatin by targeting pro-apoptotic Bcl-2 antagonist killer 1. J
Huazhong Univ Sci Technolog Med Sci. 31:5432011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Liu M, Wang J, Huang H, Hou J, Zhang B and
Wang A: miR-181a-Twist1 pathway in the chemoresistance of tongue
squamous cell carcinoma. Biochem Biophys Res Commun. 441:364–370.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pogribny IP, Filkowski JN, Tryndyak VP,
Golubov A, Shpyleva SI and Kovalchuk O: Alterations of microRNAs
and their targets are associated with acquired resistance of MCF-7
breast cancer cells to cisplatin. Int J Cancer. 127:1785–1794.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Boldin MP, Taganov KD, Rao DS, Yang L,
Zhao JL, Kalwani M, Garcia-Flores Y, Luong M, Devrekanli A, Xu J,
et al: miR-146a is a significant brake on autoimmunity,
myeloproliferation, and cancer in mice. J Exp Med. 208:1189–1201.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li L, Zhang J, Diao W, Wang D, Wei Y,
Zhang CY and Zen K: MicroRNA-155 and MicroRNA-21 promote the
expansion of functional myeloid-derived suppressor cells. J
Immunol. 192:1034–1043. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang M, Liu Q, Mi S, Liang X, Zhang Z, Su
X, Liu J, Chen Y, Wang M, Zhang Y, et al: Both miR-17-5p and
miR-20a alleviate suppressive potential of myeloid-derived
suppressor cellsby modulating STAT3 expression. J Immunol.
186:4716–4724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Liu Q, Zhang M, Jiang X, Zhang Z, Dai L,
Min S, Wu X, He Q, Liu J, Zhang Y, et al: miR-223 suppresses
differentiation of tumor-induced CD11b+ Gr1+
myeloid-derived suppressor cells from bone marrow cells. Int J
Cancer. 129:2662–2673. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Dooley J, Linterman MA and Liston A:
MicroRNA regulation of T-cell development. Immunol Rev. 253:53–64.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Galluzzi L, Morselli E, Vitale I, Kepp O,
Senovilla L, Criollo A, Servant N, Paccard C, Hupé P, Robert T, et
al: miR-181a and miR-630 regulate cisplatin-induced cancer cell
death. Cancer Res. 70:1793–1803. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li H, Zhang P, Sun X, Sun Y, Shi C, Liu H
and Liu X: MicroRNA-181a regulates epithelial-mesenchymal
transition by targeting PTEN in drug-resistant lung adenocarcinoma
cells. Int J Oncol. 47:1379–1392. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhu M, Zhou X, Du Y, Huang Z, Zhu J, Xu J,
Cheng G, Shu Y, Liu P, Zhu W and Wang T: miR-20a induces cisplatin
resistance of a human gastric cancer cell line via targeting CYLD.
Mol Med Rep. 14:1742–1750. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lin HM, Castillo L, Mahon KL, Chiam K, Lee
BY, Nguyen Q, Boyer MJ, Stockler MR, Pavlakis N, Marx G, et al:
Circulating microRNAs are associated with docetaxel chemotherapy
outcome in castration-resistant prostate cancer. Br J Cancer.
110:2462–2471. 2014. View Article : Google Scholar : PubMed/NCBI
|