Introduction
Pancreatic carcinoma (PC) was the fourth most common
cause of cancer-associated mortality in the United States in 2014
(1), with only a 5% 5-year survival
rate (2). Surgical treatment is the
only available therapeutic option with respect to PC (2). Despite certain breakthroughs in
treatment in recent years, the majority of patients with PC
eventually succumb due to recurrence (3). For this reason, in clinical practice,
the determination of an effective biomarker capable of predicting
tumour behaviour is of emergent importance.
The grainyhead-like transcription factor 2 (GRHL2)
gene is a member of the GRHL family of transcription factors that
contain a DNA-binding immunoglobulin fold homologous to the core
domain of key tumour suppressor p53 (4,5). Frisch
et al (6) indicated that GRHL2
suppresses the oncogenic epithelial-mesencyhmal transition, thereby
acting as a tumour suppressor. GRHL2 has also been implicated in
neural tube closure and in early embryonic development (7,8). A number
of studies have revealed that GRHL2 is associated with several
types of cancer, including those of the breast, prostate, renal
cells, cervix and liver (9–13).
In the present study, the GRHL2 expression in PC
tissues was investigated, along with its association with
clinicopathological factors and prognosis. GRHL2 may prove to be a
novel biomarker for PC.
Materials and methods
Clinical tissue samples
Overall, 92 PC samples and their corresponding
adjacent tissues were selected from specimens collected from
patients diagnosed at Anhui Provincial Hospital (Hefei, China)
between June 2008 and June 2012. Detailed pathological and clinical
data (including age, sex, tumour location, nerve invasion, degree
of differentiation, histological type, depth of invasion, lymph
node metastasis and TNM stage) were obtained from the medical
records of each patient. The samples were from 56 male and 36
female patients aged 38–77 years (median, 54 years). Samples were
included in the present study based on the 7th edition of the Union
for International Cancer Control TNM staging system (14). Patients who had received radiotherapy
or chemotherapy prior to surgery were not included. The specimens
were fixed in 4% formalin at 37°C for 2 h and embedded in paraffin
for pathological analysis and confirmation of the diagnosis. The
clinical follow-up data of the patients were obtained from the PC
database of Anhui Provincial Hospital. The present study was
approved by the Human Research Ethics Committee of the Anhui
Provincial Hospital and each patient provided informed consent.
Immunohistochemistry and scoring
Immunohistochemistry for GRHL2 was performed on each
tissue sample. The tissue samples were dissected into 4-µm sections
on silanised glass slides. Protein expression was detected with
2-step immunohistochemistry. In brief, deparaffinised and hydrated
sections were treated with 0.3% hydrogen peroxide in methanol for
15 min at room temperature in order to block endogenous peroxidase
activity, and washed in PBS (3 times for 3 min each), whereby
antigen retrieval was conducted in citrate buffer (cat. no. P0081;
Beyotime Institute of Biotechnology, Haimen, China; pH 6.0) for 10
min at 100°C. Following 3 more PBS washes (3 min each), the
sections were stained with a GRHL2 monoclonal antibody (cat. no.
ab86611, 1:200, Abcam, Cambridge, UK) for 2 h at 37°C, and washed
again with PBS (3 times for 3 min each). Subsequently, the sections
were incubated with horseradish peroxidase (HRP) universal IgG
antibody polymer (cat. no. sc69786, 1:1,000, Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) for 15 min at 37°C, followed
by 3 PBS washes (3 min each). Each section was treated with 50 µl
diaminobenzadine working solution (DAB Horseradish Peroxidase Color
Development kit; cat. no. P0202; Beyotime Institute of
Biotechnology) at room temperature for 3–10 min, followed by a wash
in PBS. All sections were counterstained with haematoxylin for 1–2
min at room temperature for the purpose of enabling the morphology
of the tissue to be observed using a light microscope.
The expression of GRHL2 in the tumour samples was
semi-quantitatively assessed using ImageJ 1.8.0 software (National
Institutes of Health, Bethesda, MD, USA). The proportion of
positive cells was graded as follows: 0, <1%; 1, 1–30%; 2,
30–70%; and 3, >70%. Intensity of staining was stated as none
(score, 0), weak (score, 1), moderate (score, 2) or strong (score,
3). The immunoreactivity was calculated according to the intensity
of staining and the percentage of positive cells. The 2 scores were
multiplied with each other and the eventual immunostaining score
was determined: A final score of 0, 1, 2 or 3 was considered as low
expression, and a score of 4, 6 or 9 was classified as high
expression. All specimens were evaluated separately by 2
pathologists who were blinded to the clinical data, and
discrepancies were resolved by consensus.
Cell lines and culture
PANC-1, BxPC-3 and HPDE6-C7 were purchased from the
Cell Bank of Type Culture Collection of the Chinese Academy of
Sciences (Shanghai, China). Cell lines were grown in Dulbecco's
modified Eagle' medium (Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) at 37°C in a humidified atmosphere
containing 5% CO2.
Western blot analysis
The cells were lysed in RIPA cell lysis buffer (cat.
no. P0013B; Beyotime Institute of Biotechnology) with 1 mM
phenylmethylsulfonyl fluoride at 4°C for 30 min, with vortexing
every 10 min, followed by centrifugation at 13,800 × g for 10 min
at 4°C. BCA kit (cat. no. P0009; Beyotime Institute of
Biotechnology) was used to quantify the protein concentration. The
supernatant protein solutions were diluted to 4 mg/ml and kept at
−80°C until further use. A total of 50 µg of denatured protein for
each sample was separated on a 10% SDS-PAGE gel and transferred
onto a polyvinylidene fluoride membrane. The membranes were blocked
with 5% non-fat milk in TBS/Tween-20 for 2 h at room temperature
and blotted with primary antibodies of rabbit monoclonal anti-GRHL2
(cat. no. ab86611; 1:200; Abcam) and mouse monoclonal anti-β-actin
(cat. no. TA811000; 1:400; OriGene Technologies, Inc., Beijing,
China) overnight at 4°C. Following washing with TBS/Tween-20, the
membranes were incubated with anti-rabbit or anti-mouse secondary
antibodies, conjugated with HRP (catalog nos. A0208 and A0216;
1:1,000, Jackson ImmunoResearch Laboratories, Inc., West Grove, PA,
USA) for 1 h at room temperature. Following 3 washes, the membrane
was visualised with an enhanced chemiluminescence system (cat. no.
P0018AM; Beyotime Institute of Biotechnology). The housekeeping
control in this experiment was β-actin.
Statistical analysis
All statistical analyses were performed using the
statistical package SPSS 17.0 (SPSS Inc., Chicago, IL, USA). The
χ2 test was used to analyse the immunohistochemistry
results. Survival analysis was performed using Kaplan-Meier
survival curves with the log-rank test. A multivariate Cox
regression analysis was used to identify significant independent
prognostic factors. All P-values were 2-sided and P<0.05 was
considered to indicate statistically significant differences.
Results
Immunohistochemistry of GRHL2
expression in PC tissue samples
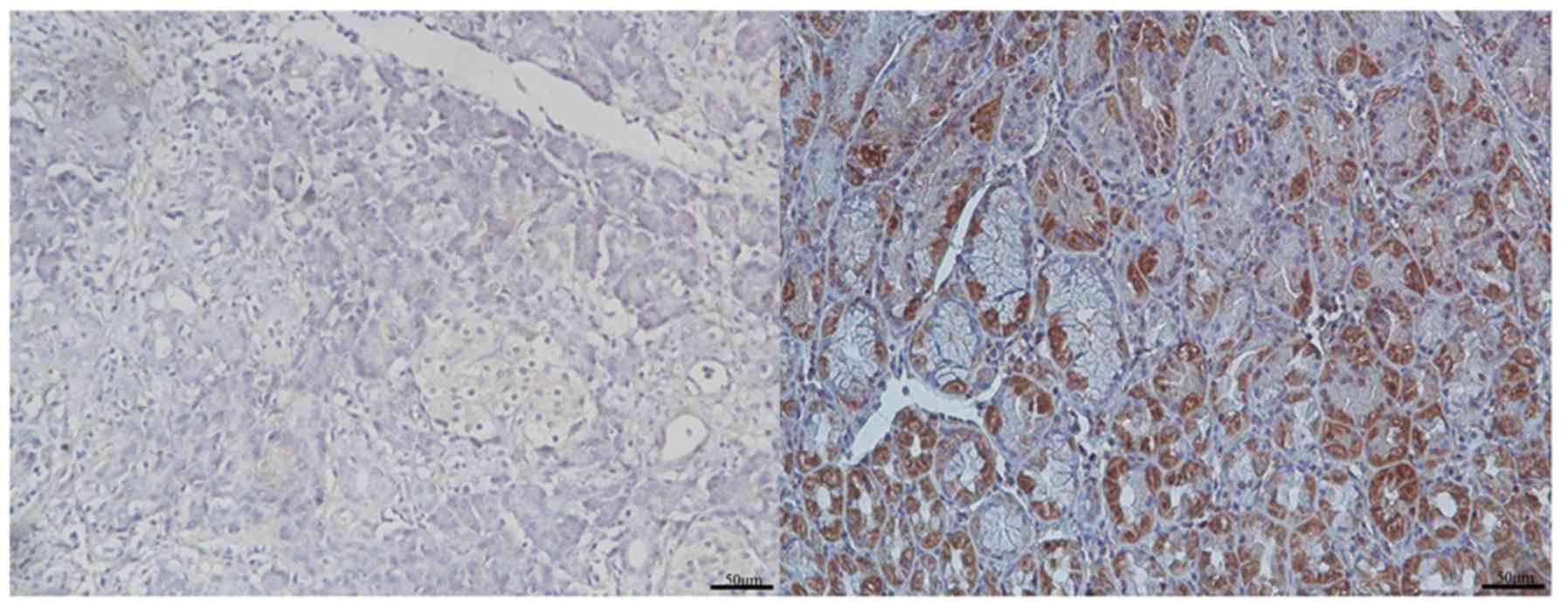
For the purpose of evaluating the levels of GRHL2
protein in PC tissue samples, immunohistochemistry was performed on
92 PC specimens and their matched adjacent healthy pancreatic
tissues. Representative images of high and low expression of GRHL2
in PC and adjacent normal tissue samples are presented in Fig. 1. The GRHL2 staining was primarily
observed in the cytoplasm of the tumour cells. High expression of
GRHL2 was observed in the majority of the PC tissues (50/92
samples) and in a number of the non-cancerous pancreatic tissues
(34/92 samples). The number of PC tissue samples with high GRHL2
expression was significantly higher than the number of
non-cancerous tissue samples of the same phenotype (P=0.018;
Table I).
 | Table I.Comparison between GRHL2 protein
expression (immunohistochemical staining) in PC and adjacent normal
pancreatic tissues. |
Table I.
Comparison between GRHL2 protein
expression (immunohistochemical staining) in PC and adjacent normal
pancreatic tissues.
|
| GRHL2 expression |
|
|
|---|
|
|
|
|
|
|---|
| Tissue type | High, n | Low, n | χ2 | P-value |
|---|
| PC | 50 | 42 | 5.608 | 0.018 |
| Adjacent | 34 | 58 |
|
|
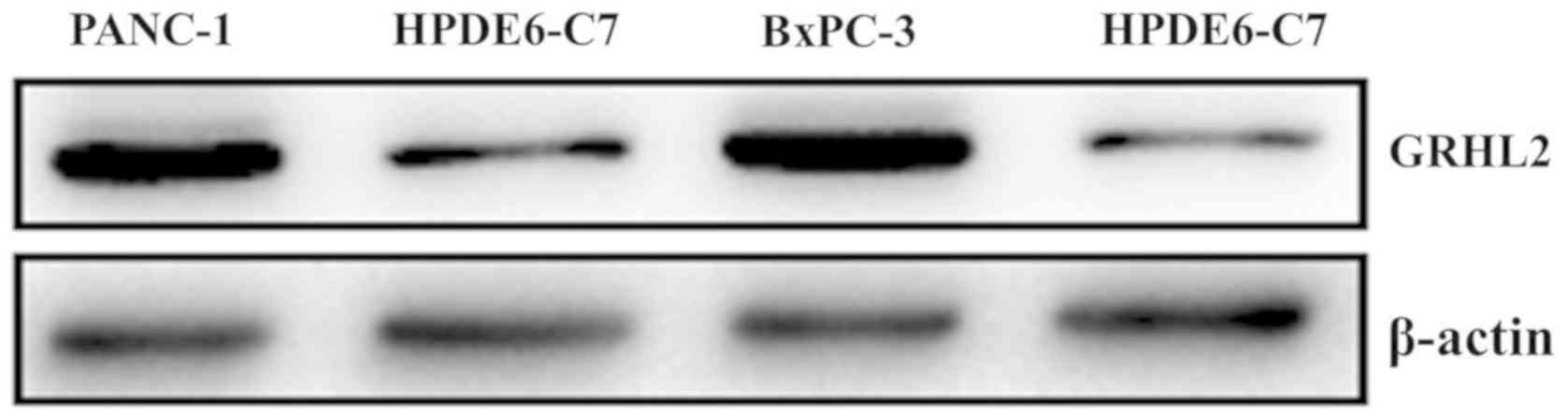
Western blot analysis of the GRHL2
protein level in the PC cell lines
The results of the western blotting were in
agreement with those of the immunohistochemistry. As demonstrated
in Fig. 2, the amount of GRHL2
protein in the PC cell lines (PANC-1 and BxPC-3) was higher
compared with that from the normal pancreatic cell line
(HPDE6-C7).
Association of GRHL2 expression with
patient clinicopathological parameters
In order to evaluate the biological significance of
GRHL2 in PC, the associations between PC tissue GRHL2 levels and
clinicopathological parameters, including age, sex, tumour size,
tumour location, nerve invasion, lymphatic metastasis, pathological
grade, depth of invasion and TNM stage, were analyzed. As
demonstrated in Table II, the
increased GRHL2 expression was associated with higher tumour
differentiation (P=0.018) and N classification (P=0.024). By
contrast, no significant association was identified between the
expression of GRHL2 and any of the other parameters tested. These
findings imply that GRHL2 may serve an important role in the
progression of PC.
 | Table II.Associations between GRHL2 protein
expression (immunohistochemical staining) in pancreatic carcinoma
and various clinicopathological variables. |
Table II.
Associations between GRHL2 protein
expression (immunohistochemical staining) in pancreatic carcinoma
and various clinicopathological variables.
|
|
| GRHL2 expression |
|
|
|---|
|
|
|
|
|
|
|---|
| Variables | Total, n | Low (n=42) | High (n=50) | χ2 | P-value |
|---|
| Sex |
|
|
| 0.035 | 0.852 |
| Male | 56 | 26 | 30 |
|
|
|
Female | 36 | 16 | 20 |
|
|
| Age, years |
|
|
| 0.207 | 0.649 |
| ≤60 | 44 | 19 | 25 |
|
|
|
>60 | 48 | 23 | 25 |
|
|
| Tumour diameter,
cm |
|
|
| 0.372 | 0.542 |
| ≤4 | 47 | 20 | 27 |
|
|
|
>4 | 45 | 22 | 23 |
|
|
| Tumour location |
|
|
| 0.589 | 0.443 |
| Head and
neck | 50 | 21 | 29 |
|
|
| Body and
tail | 42 | 21 | 21 |
|
|
| Nerve invasion |
|
|
| 2.170 | 0.141 |
|
Negative | 56 | 29 | 27 |
|
|
|
Positive | 36 | 13 | 23 |
|
|
| Differentiation |
|
|
| 5.571 | 0.018a |
|
Well/moderate | 63 | 34 | 29 |
|
|
|
None/poor | 29 | 8 | 21 |
|
|
| T classification |
|
|
| 0.058 | 0.809 |
|
T1+T2 | 69 | 31 | 38 |
|
|
|
T3+T4 | 23 | 11 | 12 |
|
|
| M classification |
|
|
| 0.032 | 0.858 |
| M0 | 88 | 40 | 48 |
|
|
| M1 | 4 | 2 | 2 |
|
|
| N classification |
|
|
| 5.124 | 0.024a |
| N0 | 66 | 35 | 31 |
|
|
| N1 | 26 | 7 | 19 |
|
|
| TNM stage |
|
|
| 0.049 | 0.825 |
|
I–II | 86 | 39 | 47 |
|
|
|
III–IV | 6 | 3 | 3 |
|
|
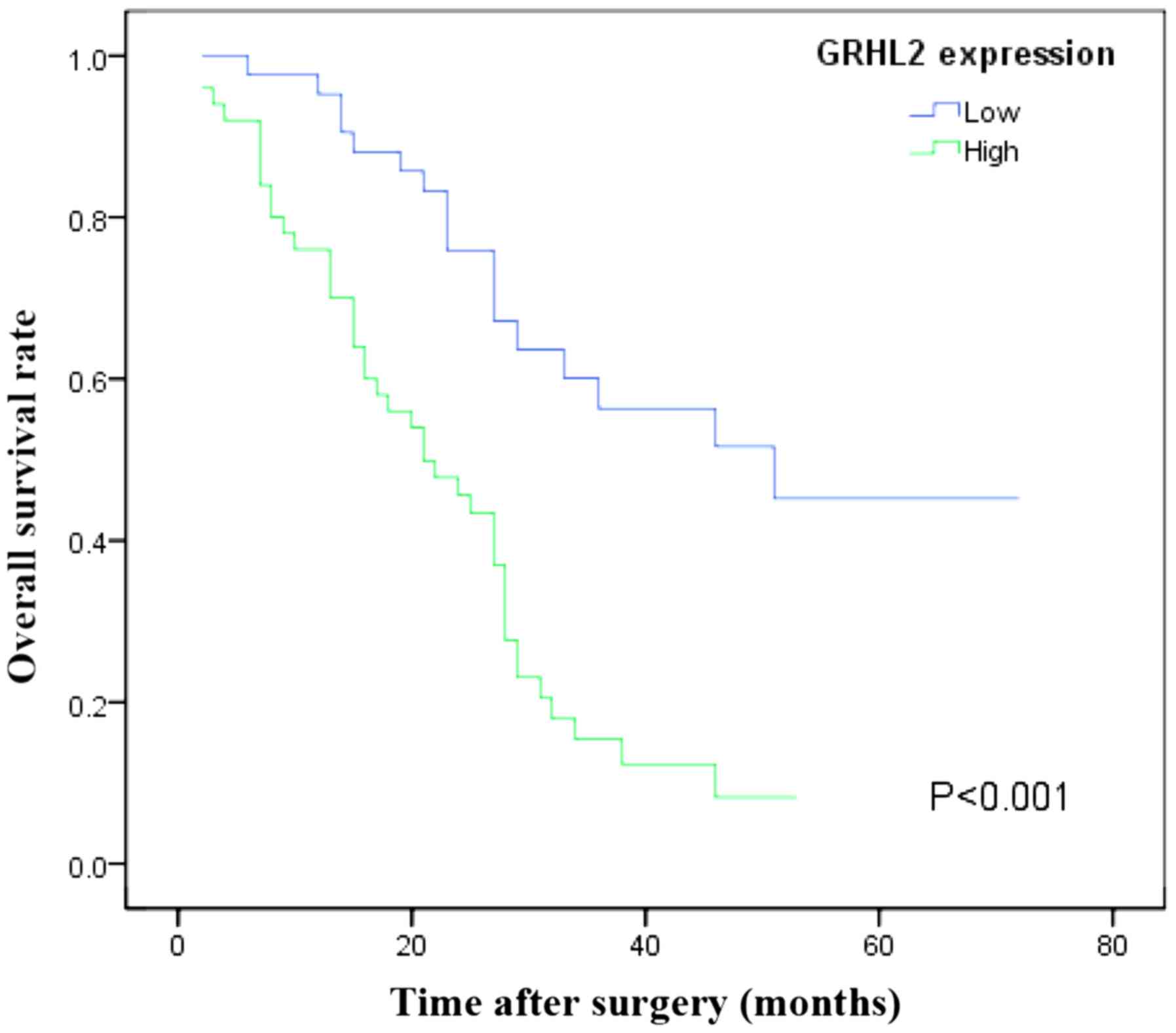
Association of GRHL2 expression with
PC prognosis
A Kaplan-Meier survival curve was plotted in order
to compare the OS time and the GRHL2 expression data (Fig. 3). Patients whose samples revealed high
expression of GRHL2 presented with a worse prognosis than those
with low GRHL2 expression (P<0.001; Fig. 3). Multivariate survival analysis
further revealed the high GRHL2 levels in the tumour tissue to be
an independent prognostic marker for poor OS time (P=0.001;
Table III).
 | Table III.Univariate and multivariate analyses
of the clinicopathological parameters and overall survival time of
patients with pancreatic carcinoma. |
Table III.
Univariate and multivariate analyses
of the clinicopathological parameters and overall survival time of
patients with pancreatic carcinoma.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Sex |
|
|
|
|
|
|
|
Male | 1.270 | 0.759–2.123 | 0.363 |
|
|
|
|
Female |
|
|
|
|
|
|
| Age, years |
|
|
|
|
|
|
|
≤60 | 1.319 | 0.794–2.191 | 0.285 |
|
|
|
|
>60 |
|
|
|
|
|
|
| Tumour size
(diameter), cm |
|
|
|
|
|
|
| ≤4 | 1.115 | 0.672–1.851 | 0.674 |
|
|
|
|
>4 |
|
|
|
|
|
|
|
Differentiation |
|
|
|
|
|
|
|
Well/moderate | 1.721 | 1.022–2.901 | 0.041a | 1.216 | 0.646–2.290 | 0.545 |
|
None/poor |
|
|
|
|
|
|
| Tumour
location |
|
|
|
|
|
|
| Head
and neck | 0.977 | 0.587–1.627 | 0.930 |
|
|
|
| Body
and tail |
|
|
|
|
|
|
| Nerve invasion |
|
|
|
|
|
|
|
Positive | 1.531 | 0.921–2.544 | 0.100 |
|
|
|
|
Negative |
|
|
|
|
|
|
| T
classification |
|
|
|
|
|
|
|
T1+T2 | 1.019 | 0.558–1.861 | 0.950 |
|
|
|
|
T3+T4 |
|
|
|
|
|
|
| M
classification |
|
|
|
|
|
|
| M0 | 1.173 | 0.365–3.768 | 0.789 |
|
|
|
| M1 |
|
|
|
|
|
|
| N
classification |
|
|
|
|
|
|
| N0 | 2.301 | 1.371–3.861 | 0.002a | 1.780 | 0.952–3.326 | 0.071 |
| N1 |
|
|
|
|
|
|
| TNM stage |
|
|
|
|
|
|
|
I–II | 1.425 | 0.569–3.566 | 0.450 |
|
|
|
|
III–IV |
|
|
|
|
|
|
| GRHL2 level |
|
|
|
|
|
|
|
Low | 3.247 | 1.847–5.708 |
<0.01a | 2.901 | 1.547–5.439 | 0.001a |
|
High |
|
|
|
|
|
|
Discussion
GRHL was first identified in the fruit fly
Drosophila melanogaster (15).
To date, a total of 3 members of the GRHL family, termed GRHL1-3,
have been detected in mammals (16,17).
GRHL2, an important member of the GRHL family, is quintessential in
embryonic neural tube development (7). A number of studies have revealed that
altered expression of GRHL2 is associated with development,
progression, tumourigenesis and poor prognosis in a variety of
different cancer types, including colorectal cancer, oral squamous
cell carcinoma and liver cancer (4,11,18,19). Quan
et al (18) investigated the
GRHL2 expression in 171 colorectal cancer and paired normal colon
mucosa samples, and revealed that GRHL2 is an independent
prognostic factor for OS, as well as recurrence-free survival
times. Butz et al (11)
studied the GRHL2 expression in 593 clear cell renal cell carcinoma
and 389 normal kidney specimens, and demonstrated that GRHL2
expression was associated with higher chances of disease recurrence
and that the disease-free survival times of GRHL2-positive patients
were lower.
In the present study, the GRHL2 protein expression
levels were examined in samples of patients with PC who had
undergone surgery, along with the potential association between
GRHL2 expression levels and clinicopathological characteristics of
the tumour. Furthermore, GRHL2 overexpression was determined in 2
human PC cell lines. An association between increased GRHL2
expression and lymphatic metastasis and tumour differentiation was
identified, while a χ2 test revealed that there were
significantly more PC tissue samples with high GRHL2 protein levels
compared with the corresponding adjacent non-cancerous tissue
samples. Additionally, the OS time in patients with low levels of
GRHL2 was increased compared with that of patients with elevated
GRHL2 levels. The Kaplan-Meier and multivariate analyses suggested
that the expression level of GRHL2 was a potential independent
prognostic predictor of poor OS time. It was revealed that the
increased expression level of GRHL2 was associated with the
prognosis of PC.
In summary, the present study suggests that elevated
GRHL2 protein levels may be a malignant phenotypic characteristic
of PC. Furthermore, this is the first report of GRHL2 protein
expression being a novel independent prognostic marker for PC.
Further research, including cell function and pathway experiments,
is therefore required for the purpose of exploring the molecular
mechanism of GRHL2, along with investigating its potential as a
therapeutic target in PC. A limitation of the present study is the
small sample size from a single centre. Future studies with larger
sample sizes are required to further validate the association
between GRHL2 expression and prognosis.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CW conceived and designed the study. JP and LZ
analyzed and interpreted the patient data. GW, JP and LZ
contributed to the acquisition of data, data analysis and data
interpretation. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Human Research
Ethics Committee of the Anhui Provincial Hospital and each patient
provided informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wolfgang CL, Herman JM, Laheru DA, Klein
AP, Erdek MA, Fishman EK and Hruban RH: Recent progress in
pancreatic cancer. CA Cancer J Clin. 63:318–348. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Groot VP, van Santvoort HC, Rombouts SJ,
Hagendoorn J, Rinkes Borel IH, van Vulpen M, Herman JM, Wolfgang
CL, Besselink MG and Molenaar IQ: Systematic review on the
treatment of isolated local recurrence of pancreatic cancer after
surgery; re-resection, chemoradiotherapy and SBRT. HPB (Oxford).
19:83–92. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mlacki M, Kikulska A, Krzywinska E, Pawlak
M and Wilanowski T: Recent discoveries concerning the involvement
of transcription factors from the Grainyhead-like family in cancer.
Exp Biol Med (Maywood). 240:1396–1401. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kokoszynska K, Ostrowski J, Rychlewski L
and Wyrwicz LS: The fold recognition of CP2 transcription factors
gives new insights into the function and evolution of tumor
suppressor protein p53. Cell Cycle. 7:2907–2915. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Frisch SM, Farris JC and Pifer PM: Roles
of Grainyhead-like transcription factors in cancer. Oncogene.
36:6067–6073. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ray HJ and Niswander LA: Grainyhead-like 2
downstream targets act to suppress epithelial-to-mesenchymal
transition during neural tube closure. Development. 143:1192–1204.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rifat Y, Parekh V, Wilanowski T, Hislop
NR, Auden A, Ting SB, Cunningham JM and Jane SM: Regional neural
tube closure defined by the Grainy head-like transcription factors.
Dev Biol. 345:237–245. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang X, Vasudevan P, Parekh V, Penev A and
Cunningham JM: Bridging cancer biology with the clinic: Relative
expression of a GRHL2-mediated gene-set pair predicts breast cancer
metastasis. PLoS One. 8:e561952013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Danila DC, Anand A, Schultz N, Heller G,
Wan M, Sung CC, Dai C, Khanin R, Fleisher M, Lilja H and Scher HI:
Analytic and clinical validation of a prostate cancer-enhanced
messenger RNA detection assay in wholeblood as a prognostic
biomarker for survival. Eur Urol. 65:1191–1197. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Butz H, Szabo PM, Nofech-Mozes R, Rotondo
F, Kovacs K, Mirham L, Girgis H, Boles D, Patocs A and Yousef GM:
Integrative bioinformatics analysis reveals new prognostic
biomarkers of clear cell renal cell carcinoma. Clin Chem.
60:1314–1326. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Torres-Reyes LA, Alvarado-Ruiz L,
Pina-Sanchez P, Martínez-Silva MG, Ramos-Solano M, Olimón-Andalón
V, Ortiz-Lazareno PC, Hernández-Flores G, Bravo-Cuellar A,
Aguilar-Lemarroy A and Jave-Suarez LF: Expression of transcription
factor grainyhead-like 2 is diminished incervical cancer. Int J
Clin Exp Pathol. 7:7409–7418. 2014.PubMed/NCBI
|
|
13
|
Tanaka Y, Kanai F, Tada M, Tateishi R,
Sanada M, Nannya Y, Ohta M, Asaoka Y, Seto M, Shiina S, et al: Gain
of GRHL2 is associated with early recurrence of hepatocellular
carcinoma. J Hepatol. 49:746–757. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sobin LH, Gospodarowicz MK and Wittekind
C: PancreasInternational Union against Cancer (UICC) TNM
Classification of Malignant Tumours. 7th edition. Wiley-Blackwell;
New York, NY: pp. 132–135. 2010
|
|
15
|
Dynlacht BD, Attardi LD, Admon A, Freeman
M and Tjian R: Functional analysis of NTF-1, a developmentally
regulated Drosophila transcription factor that binds neuronal cis
elements. Genes Dev. 3:1677–1688. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ting SB, Wilanowski T, Cerruti L, Zhao LL,
Cunningham JM and Jane SM: The identification and characterization
of human sister-of-mammalian grainyhead (SOM) expands the
grainyhead-like family of developmental transcription factors.
Biochem J. 370:953–962. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wilanowski T, Tuckfield A, Cerruti L,
O'Connell S, Saint R, Parekh V, Tao J, Cunningham JM and Jane SM: A
highly conserved novel family of mammalian developmental
transcription factors related to drosophila grainyhead. Mech Dev.
114:37–50. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Quan Y, Xu M, Cui P, Ye M, Zhuang B and
Min Z: Grainyhead-like 2 promotes tumor growth and is associated
with poor prognosis in colorectal cancer. J Cancer. 6:342–350.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhao Q, Caballero OL, Davis ID, Jonasch E,
Tamboli P, Yung WK and Weinstein JN: Kenna Shaw for TCGA research
network, Strausberg RL and Yao J: Tumor-specific isoform switch of
the fibroblast growth factor receptor 2 underlies the mesenchymal
and malignant phenotypes of clear cell renal cell carcinomas. Clin
Cancer Res. 19:2460–2472. 2013. View Article : Google Scholar : PubMed/NCBI
|