Introduction
Breast cancer had one of the highest incidence rates
and was a leading cause of cancer-associated mortality worldwide in
2016, and its incidence is continuously increasing (1). Breast cancer is a heterogeneous disease
comprising luminal type A, luminal type B, human epidermal growth
factor receptor 2 (HER2)-positive and basal-like breast cancer.
Clinical features, treatment and prognosis differ depending on the
subtype (2). The most common
metastatic sites of breast cancer are the bone, brain, liver and
lung, however, patterns of metastasis are affected by the breast
cancer subtype (3–5). By contrast, breast cancer rarely
metastasizes to the gastrointestinal tract, including the stomach,
and few studies and case reports concerning gastric metastasis from
breast cancer can be found (6–16).
Chemokines and their receptors, including CXCR4 and CCR7, are
critical for regulating the location of metastasis. Their
respective ligands CXCL12/SDF-1α and CCL21/6Ckine demonstrate peak
levels of expression in the primary organs of breast cancer
metastasis (17). Low incidence of
metastasis to the gastrointestinal tract may be due to low
expression of chemokine receptor ligands at this location. Due to
its rarity, breast cancer with gastric metastasis is occasionally
confused with a primary malignancy in the stomach. However,
discriminating characteristic features with clinical implications
may exist. Depending on whether it is primary or secondary, the
treatment and prognosis are different. The current study aimed to
identify the clinical features and prognosis of breast cancer with
gastric metastasis. A total of 13 patients with breast cancer and
gastric metastasis were analyzed. Invasive lobular carcinoma (ILC)
or luminal-type breast cancer tends to metastasize to the stomach
at a higher frequency compared with other types of breast cancer.
In addition, the stomach was one of the first metastatic lesion
locations in the patients in the present study. These patients had
a more favorable prognosis compared with patients with primary
gastric cancer.
Materials and methods
Study population
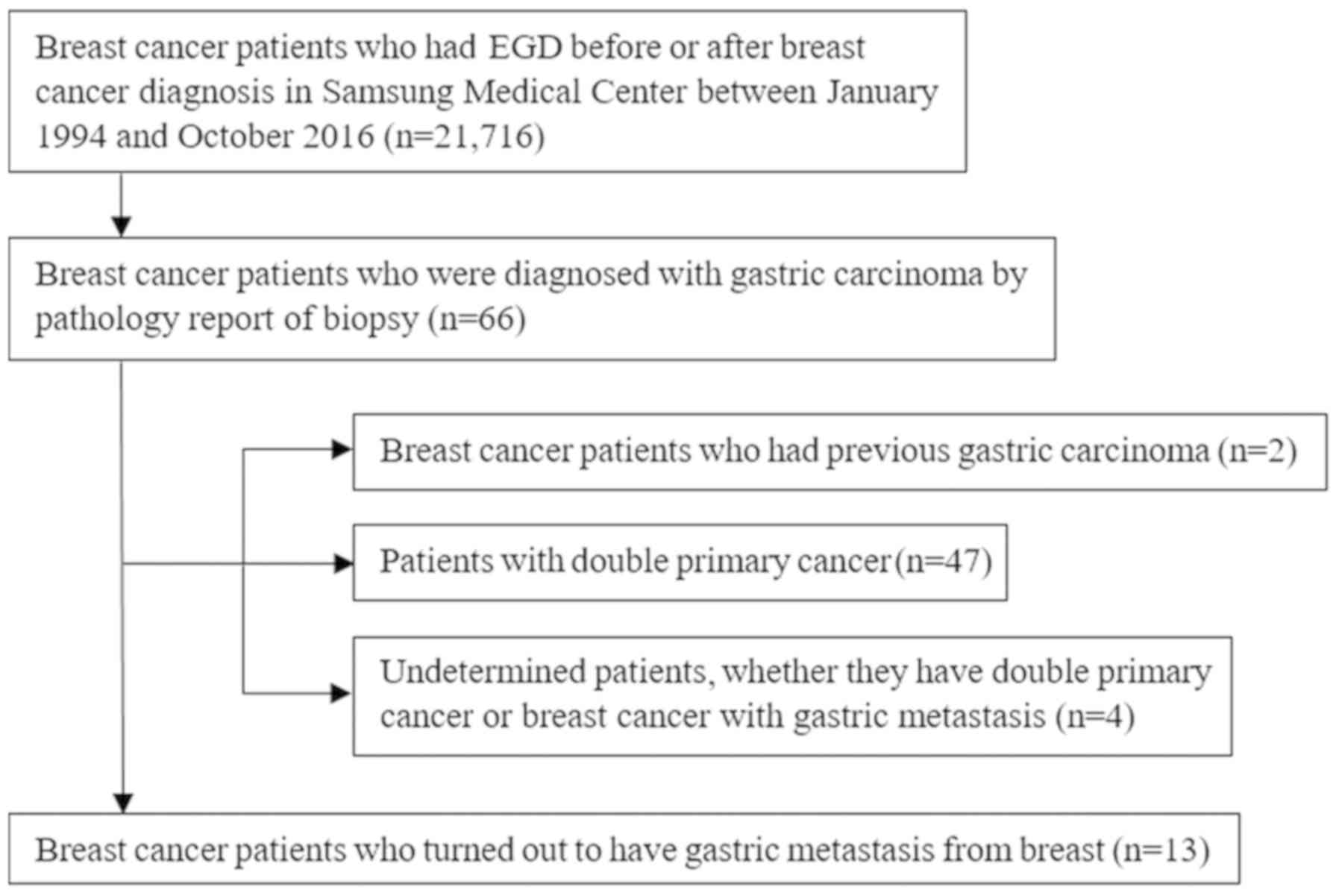
The present study was a retrospective, single-center
study. Between January 1994 and October 2016 in Samsung Medical
Center (Seoul, Korea), 21,716 patients with breast cancer underwent
esophagogastroduodenoscopy (EGD) prior to or following breast
cancer diagnosis. Among them, 66 patients with breast cancer had
gastric carcinoma according to the pathology reports from the
biopsies. A total of 13 patients (median age, 45 years; range,
38–65 years) with breast cancer presented with gastric metastasis
and 47 had double primary cancer. A total of 2 patients had a
previous gastric malignancy prior to breast cancer diagnosis. It
was not possible to determine whether 4 patients had double primary
cancer or breast cancer with gastric metastasis (Fig. 1). Pathology specialists in the
hospital determined whether a gastric lesion was primary or
secondary.
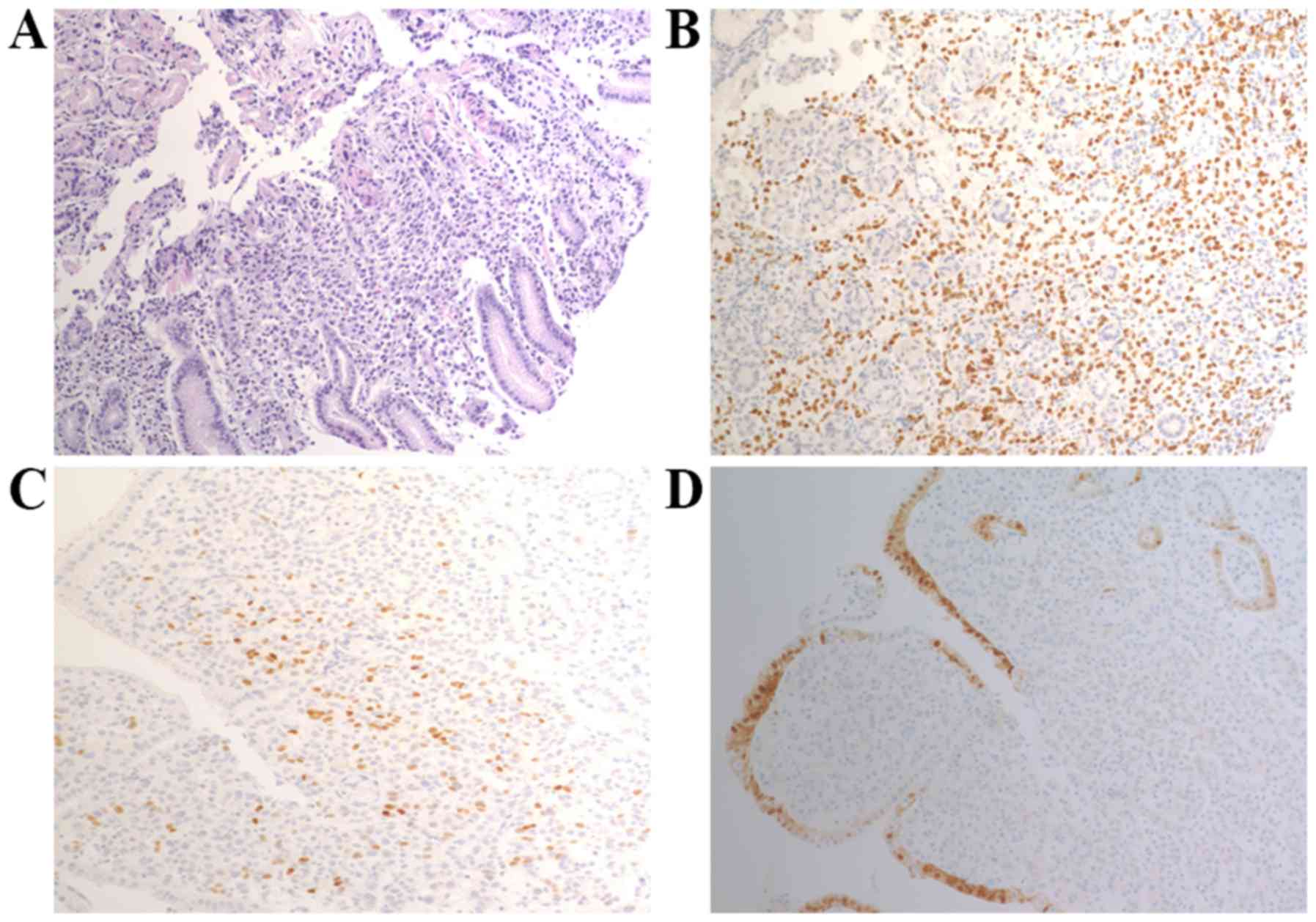
Out of the 13 patients, 11 were determined to have
breast cancer with gastric metastasis according to pathological
analysis using immunohistochemistry (IHC) staining with the
following biomarkers: Estrogen receptor (ER), progesterone receptor
(PR), HER2, gross cystic disease fluid protein 15 (GCDFP-15),
cytokeratin 7, GATA-binding protein 3 and E-cadherin. Only 2
patients underwent staining for GATA-3 and 1 patient underwent
staining for E-cadherin, therefore, these data are not shown. An
additional 2 patients with breast cancer, who had gastric carcinoma
at EGD, were also included into this group clinically, as their
gastric lesions responded to subsequent breast cancer chemotherapy.
The samples obtained from these 2 patients were assessed by EGD
without IHC staining.
Data collection
Clinicopathological data from the electronic medical
records were collected. The variables considered for the sample
collection of the present study were the following: Age, sex, stage
according to the 7th edition of the American Joint Committee on
Cancer (AJCC) staging system (18),
pathology, hormone receptor status of breast cancer, HER2 status,
treatment modalities, including surgery, chemotherapy, radiotherapy
and hormone therapy for breast cancer, time interval to other
metastasis and gastric metastasis, gastric symptoms, endoscopy
results according to Borrmann's classification (19), pathology and hormone receptor status
of metastasis, other IHC staining markers of gastric metastasis,
including GCDFP-15, treatment for gastric metastasis and follow-up
data.
IHC staining
Breast and gastric tissue samples were embedded in
paraffin and fixed in a 10% buffered formalin solution for 24–48 h
at room temperature. Each sample was cut into 5-mm thick sections.
In contrast with the samples of breast cancer, not all samples of
gastric metastasis underwent IHC staining with anti-ER (1:200,
catalog no. 6F11; Novocastra Laboratories Ltd., Newcastle upon
Tyne, UK), anti-PR (1:100; clone 16, Novocastra Laboratories Ltd.),
anti-HER2 (pre-diluted; clone 4B5; Ventana Medical Systems, Inc.,
Tucson, AZ, USA) or anti-GCDFP-15 (pre-diluted; clone EP1582Y;
Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) as not all samples
were originally considered to be metastasis. A total of 11 out of
13 samples underwent IHC staining for ER. A total of 10, 9 and 10
samples underwent staining for PR, HER2 and GCDFP-15, respectively.
Slides were incubated with anti-ER and anti-PR primary antibodies
at room temperature for 15 min or at 37°C for 32 min with anti-HER2
and anti-GCDFP-15 primary antibodies. Subsequently, Bond Polymer
Refine Detection kit (Leica Biosystems, Newcastle upon Tyne, UK)
was used to detect ER and PR, and ultraView Universal DAB Detection
kit (Ventana Medical Systems, Inc.) was used to detect HER2 and
GCDFP-15. Stained slides were evaluated using a light microscope
(magnification, ×100-200). A cut-off value of ≥1% stained tumor
nuclei was used to determine samples as ER-positive or PR-positive.
Samples were considered as HER2 positive if samples exhibited IHC
3+ positive staining or a positive silver in situ
hybridization (SISH) result. A diffuse intense circumferential
membrane ‘chicken-wire’ staining pattern in >10% of the tumor
was scored as IHC 3+. Tumors with circumferential membrane staining
demonstrating a thin pattern of staining and/or heterogeneity in
staining distribution in ≤10% of tumor cells were scored as IHC 2+.
Tumors with absent or weak membrane staining were scored as 0 or
IHC 1+ (20). If samples exhibited
IHC 2+ staining, HER2 gene status was evaluated by HER2 SISH. HER2
SISH was considered as positive if the average HER2/CEP17 ratio was
≥2.0 or the average HER2 copy number was ≥6.0 signals/cell. For
GCDFP-15, any cytoplasmic staining was considered as positive.
Statistical analysis
Kaplan-Meier curves were used to measure overall
survival rate and time following gastric metastasis, and
significant differences were identified by a log-rank test.
P<0.05 was considered to indicate a statistically significant
difference. Statistical analysis was performed using SPSS software
(version 24; IBM Corp., Armonk, NY, USA). Data are presented as the
median ± standard deviation.
Ethical considerations
The present study was approved by the Institutional
Review Board of Samsung Medical Center (approval no.
2017-11-097-002). The requirement for informed consent was waived
due to the retrospective nature of the study.
Results
Patient characteristics
Characteristics of the 13 patients with breast
cancer are presented in Table I. The
median age of these patients at the time of breast cancer diagnosis
was 45 years. A total of 7 (53.8%) patients presented with ILC and
6 (46.2%) with invasive ductal carcinoma. Positivity in tissue
cancer samples was 92.3% for ER and 76.9% for PR. All samples were
negative for HER2. Out of the 13 tumors, 11 (84.6%) were stage
I–III and 2 patients (15.4%) had stage IV gastric metastasis at
initial breast cancer diagnosis based on the 7th edition of the
AJCC cancer staging system. All patients with de novo stage
I–III breast cancer underwent surgery, consisting of a mastectomy
in 9 (81.8%) patients and breast-conserving surgery in 2 (18.2%)
patients. 2 patients with stage IV breast cancer received
chemotherapy initially instead of surgery.
 | Table I.Characteristics of patients with
breast cancer. |
Table I.
Characteristics of patients with
breast cancer.
| Variable | Value |
|---|
| Median age (range),
years | 45 (38–65) |
| Sex, n (%) |
|
| Male | 0
(0.0) |
|
Female | 13 (100.0) |
| Pathology, n (%) |
|
| IDC | 6
(46.2) |
| ILC | 7
(53.8) |
| Breast cancer
receptor, n (%) |
|
| ER | 12 (92.3) |
| PR | 10 (76.9) |
| HER2 | 0
(0.0) |
| AJCC stage, n
(%) |
|
| Stage
I–III | 11 (84.6) |
| Stage
IV | 2
(15.4) |
| Surgery, n (%) |
|
|
Mastectomy | 9
(81.8) |
| BCS | 2
(18.2) |
Characteristics of the gastric
metastasis
Characteristics of the gastric metastasis from
breast cancer are presented in Table
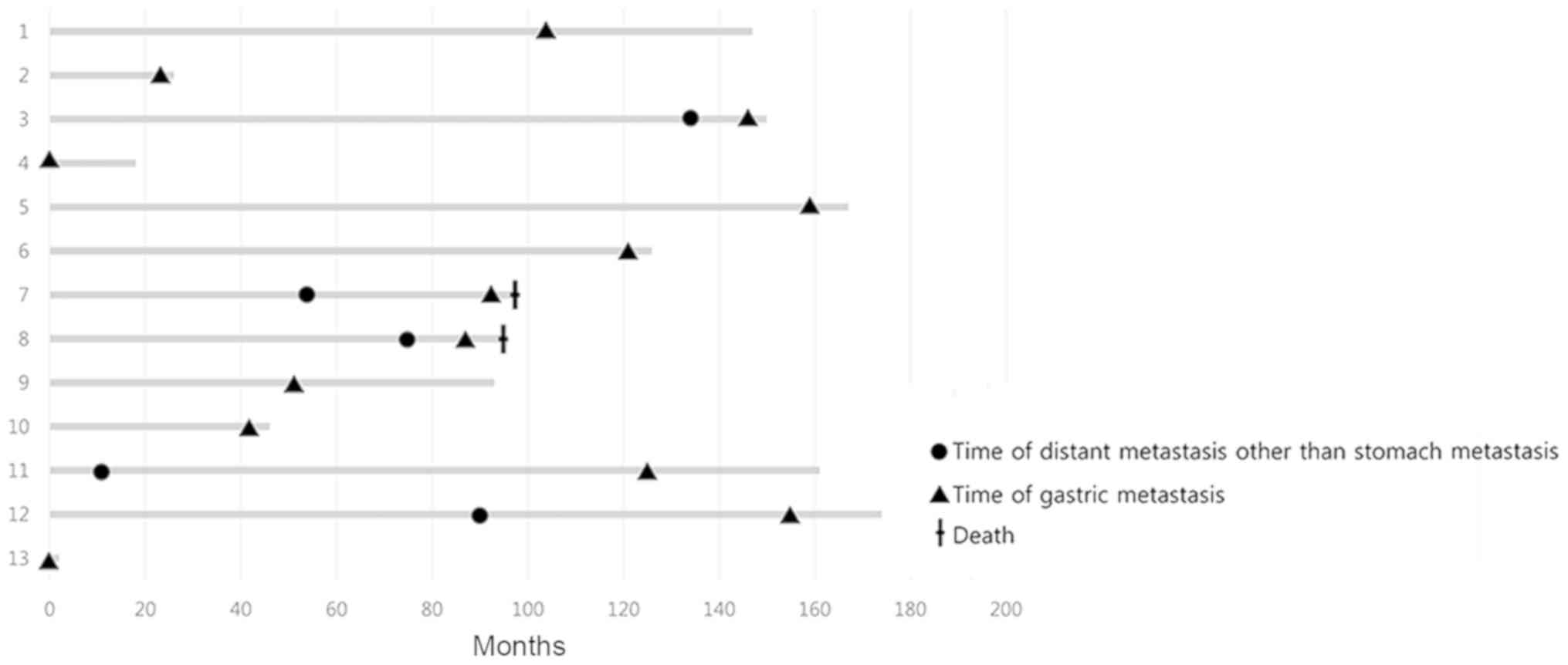
II. The stomach was the first metastatic lesion location in 6
(54.5%) out of the 11 patients with de novo stage I–III
cancer. The median time between breast cancer and gastric
metastasis was 104 months for these patients. For the 6 patients
where gastric metastasis was the initial metastatic lesion, the
median time to gastric metastasis was 77.5 months. For the other 5
patients, the median time between other initial distant metastasis
and gastric metastasis was 42 months, where initial metastatic
lesion location differed among patients. One of these 5 patients
had initial lung and bone metastasis following gastric metastasis,
while another patient also had bone metastasis initially, following
gastric metastasis. The third patient had liver metastasis
initially, followed by gastric metastasis and the fourth patient
had bone metastasis initially, followed by metastasis in each ovary
and then gastric metastasis. The last of these 5 patients had
pancreatic metastasis, followed by metastasis of the ureter and
finally gastric metastasis. A time graph for breast cancer and
gastric metastasis is presented in Fig.
2. Of the 11 patients for whom details regarding any related
symptoms were retrieved by electronic medical records, 7 (63.6%)
had symptoms, including melena, hematemesis, vomiting, poor oral
intake, dyspepsia and abdominal pain, while 4 (36.4%) patients
exhibited no symptoms. A total of 7 out of 13 patients underwent
EGD due to the aforementioned symptoms, 3 underwent EGD for routine
check-ups without symptoms and 1 underwent EGD as gastric
malignancy was suspected at follow-up abdominal computed tomography
(CT) scan for disease evaluation of chemotherapy. The remaining 2
patients underwent EGD at other hospitals for unknown reasons;
therefore information regarding their symptoms could not be
collected. A total of 9 (81.8%) patients with gastric cancer had
tumors that were ER-positive, and 5 out of 10 (50%) had tumors that
were PR-positive. Among the nine gastric tumor samples that
underwent IHC staining for HER2, all tumors were HER2-negative.
Among the 10 patients who underwent GCDFP-15 staining, 6 (60%)
exhibited positive results.
 | Table II.Characteristics of patients with
gastric metastasis from breast cancer. |
Table II.
Characteristics of patients with
gastric metastasis from breast cancer.
| Variable | Value |
|---|
| Median (range) time
to gastric metastasis from breast cancer (n=11)a, months | 104 (24–159) |
| Median (range) time
to gastric metastasis as initial metastasis from breast cancer
(n=6) | 77.5 (24–159) |
| Median (range) time
to gastric metastasis from another initial distant metastasis
(n=5) | 42 (12–114) |
| Symptoms, n (%) |
|
| Yes | 7 (63.6) |
| No | 4 (36.4) |
| Stomach cancer
receptor, n (%) |
|
| ER | 9 (81.8) |
| PR | 5 (50.0) |
| HER2 | 0 (0.0) |
| GCDFP-15 | 6 (60.0) |
Treatment modalities of the breast
cancer
The treatment of the breast cancer, including
surgery, adjuvant chemotherapy, adjuvant radiotherapy, hormone
therapy, palliative chemotherapy and intrathecal methotrexate is
presented in Table III. In
addition, Table III also presents
the treatment of the gastric metastasis, including palliative
chemotherapy, intrathecal methotrexate, radiotherapy, hormone
therapy and whole-brain radiotherapy.
 | Table III.Clinical information for 13 female
patients with breast cancer with gastric metastasis. |
Table III.
Clinical information for 13 female
patients with breast cancer with gastric metastasis.
| Patient no. | Age, years | Pathology | Breast hormone
receptor (ER/PR/HER2) | Surgery | Subsequent
treatment | Status | Gastric symptoms | Time from breast
cancer to distant metastasis, months | Time from distant
metastasis to gastric metastasis | Gastric hormone
receptor (ER/PR/HER2) | Subsequent
treatment | Follow-up
status |
|---|
| 1 | 38 | IDC | +/−/− | Mastectomy | Adj. CTx, RT,
HT | NETR | Melena | 104 | 0 | +/+/− | CTx., IT MTX | AWD |
| 2 | 58 | IDC | −/−/− | BCS | Adj. CTx, RT | NETR | Vomiting, poor oral
intake | 24 | 0 | Not tested | Not done | Lost to
follow-up |
| 3 | 40 | IDC | +/+/− | Mastectomy | Adj. CTx | NETR | Not described | 134 | 12 | +/−/− | RT, CTx. | Lost to
follow-up |
| 4 | 65 | ILC | +/−/−/ | Not performed | Pall. CTx, IT
MTX | Pall. CTx | None | 0 | 0 | +/not tested/− | As breast cancer
treatment | AWD |
| 5 | 38 | ILC | +/+/− | Mastectomy | Adj. CTx, RT,
HT | NETR | Soreness, fullness,
hematemesis | 159 | 0 | +/−/− | CTx., HT | AWD |
| 6 | 40 | IDC | +/+/− | Mastectomy | Adj. CTx, HT | NETR | Soreness | 121 | 0 | +/+/− | CTx. | Lost to
follow-up |
| 7 | 45 | ILC | +/+/− | BCS | Adj. CTx, HT | While adj. HT, bone
meta occurred, treated with letrozole | Melena, abdominal
discomfort | 54 | 42 | −/−/not tested | CTx. | STD |
| 8 | 41 | ILC | +/+/− | Mastectomy | Adj. CTx, RT,
HT | After NETR period,
liver meta occurred, treated with letrozole | None | 75 | 12 | +/+/not tested | WBRT, CTx. | STD |
| 9 | 46 | ILC | +/+/− | Mastectomy | Adj. CTx, RT,
HT | While adj. HT,
gastric meta occurred | None | 51 | 0 | +/+/− | HT, CTx. | AWD |
| 10 | 41 | ILC | +/+/− | Mastectomy | Adj. CTx, RT,
HT | While adj. HT,
gastric meta occurred | Dyspepsia,
vomiting, weight loss | 42 | 0 | +/−/− | CTx. | AWD |
| 11 | 45 | ILC | +/+/− | Mastectomy | Adj. CTx, RT,
HT | While adj. HT, bone
meta occurred, treated with letrozole, Pall. CTx done, both ovarian
meta treated by BSO, spine meta treated by Pall. RT, brain meta
treated by GKS | Not described | 11 | 114 | −/−/− | As breast cancer
treatment | AWD |
| 12 | 55 | IDC | +/+/− | Mastectomy | Neoadj. CTx, adj.
CTx, RT, HT | After NETR period,
pancreas meta occurred, treated by a Whipple procedure, Pall.
CTx | None | 90 | 65 | Not tested/not
tested/not tested | As breast cancer
treatment | AWD |
| 13 | 45 | IDC | +/+/− | Not performed | Pall. RT, CTx | Pall. CTx | Abdominal pain | 0 | 0 | +/+/− | As breast cancer
treatment | AWD |
Prognosis of gastric metastasis
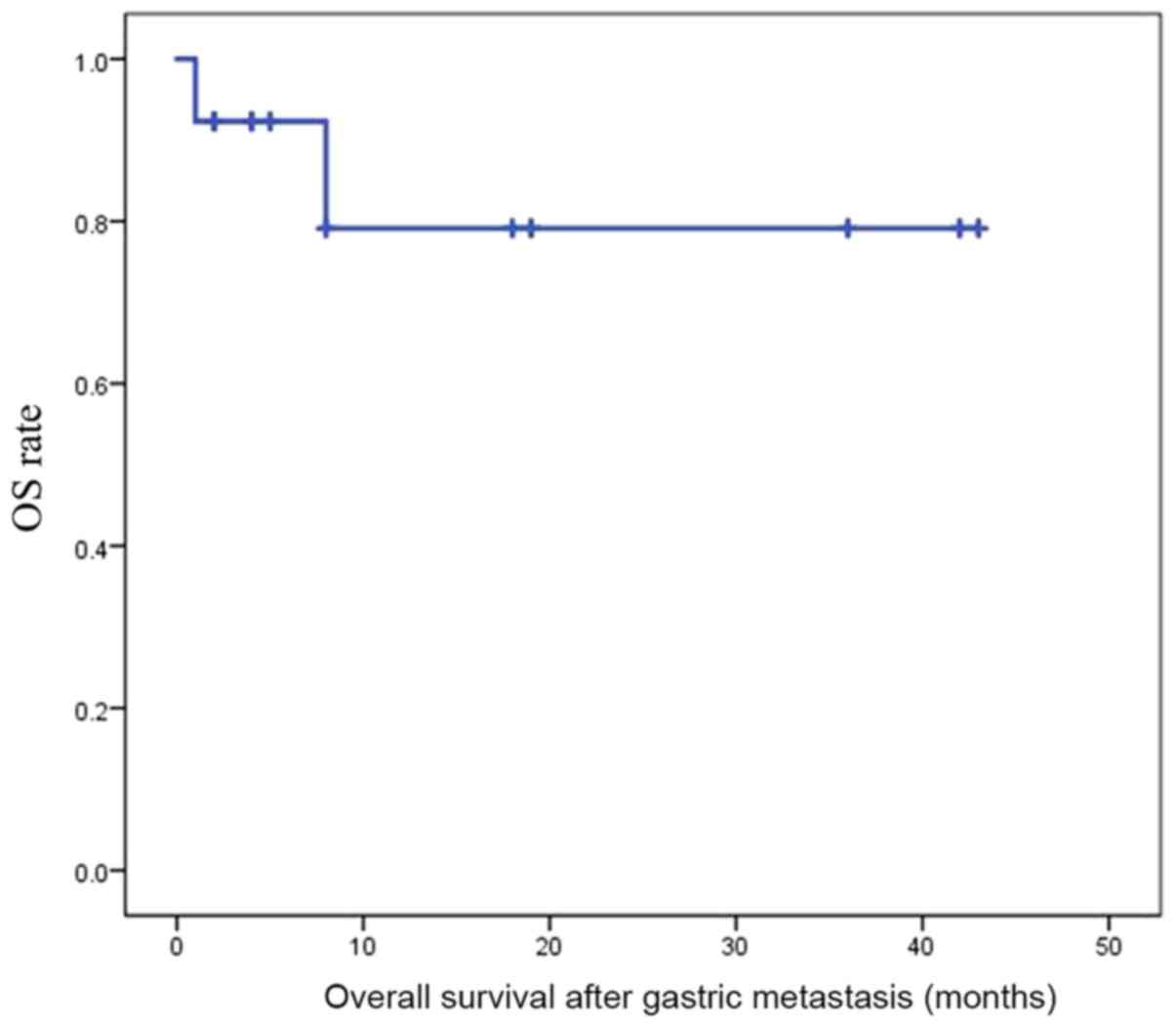
Kaplan-Meier curves are presented in Fig. 3. The 3-year survival rate was 79.1%,
and the estimated mean survival time was 35.1 months. A total of 2
patients succumbed to their disease, and 8 patients remained alive
with the disease at follow-up. When comparing 6 patients who had
initial gastric metastasis with 5 patients who had subsequent
gastric metastasis following initial other distant metastasis, the
3-year survival rate of the former was 100%, while that of the
latter was 53.3%. However, there was no significant difference
(P=0.138) between the two, due to the small number of cases.
Further extensive follow-up is required to confirm the 5-year
survival rate and events.
A patient with gastric metastasis as
the initial metastasis
Among the 13 patients with breast cancer and gastric
metastasis, only 1 had sole gastric metastasis as the initial
metastasis. Even though the stomach lesion was a sole cancer lesion
in this patient, it was determined not to be primary, but
metastatic, following thorough review and discussion with a
pathology specialist. Therefore, rather than surgery or endoscopic
treatment, this patient was treated with 2,000 mg capecitabine
(Roche Diagnostics, Basel, Switzerland) every 3 weeks for 4 cycles
and a reduced dose of 1,500 mg due to diarrhea every 3 weeks for 3
cycles. The last noted response was partial. Another patient had
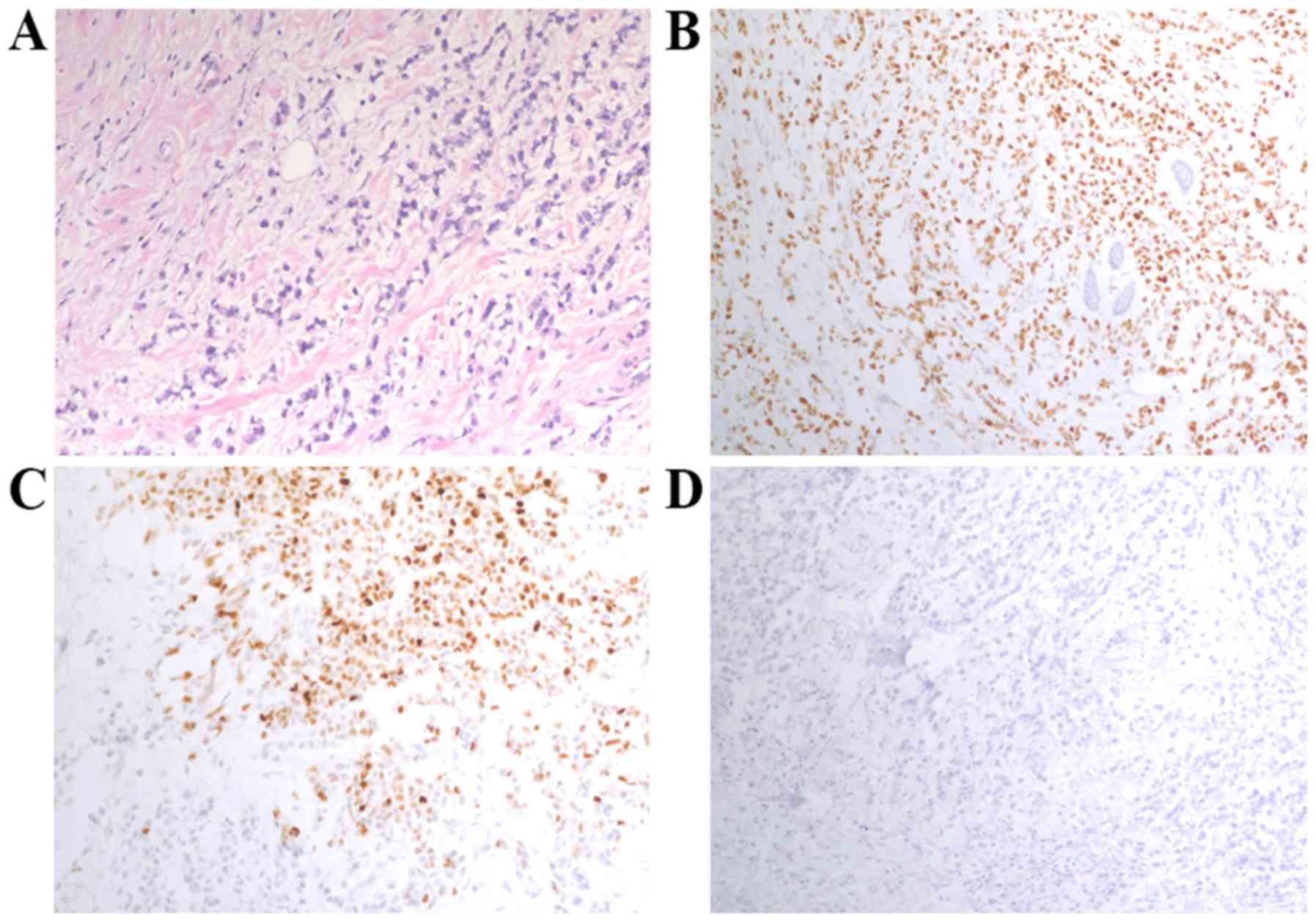
gastric metastasis with regional lymph node metastasis at the
initial metastasis (Figs. 4 and
5). Treatment consisted of letrozole
(Novartis International AG, Basel, Switzerland), exemestane (Pfizer
Inc., New York, NY, USA) plus everolimus (Novartis International),
weekly paclitaxel (Hospira; Pfizer, Inc.), doxorubicin (Boryung,
Seoul, Korea) plus cyclophosphamide (Baxter Inc., Deerfield, IL,
USA), and capecitabine (Roche Diagnostics) until the last
follow-up. The remaining patients had other distant metastasis in
addition to that of the stomach. These patients did not undergo
stomach surgery, but instead received chemotherapy, radiotherapy,
hormone therapy or no treatment (Table
III).
EGD results of gastric metastases
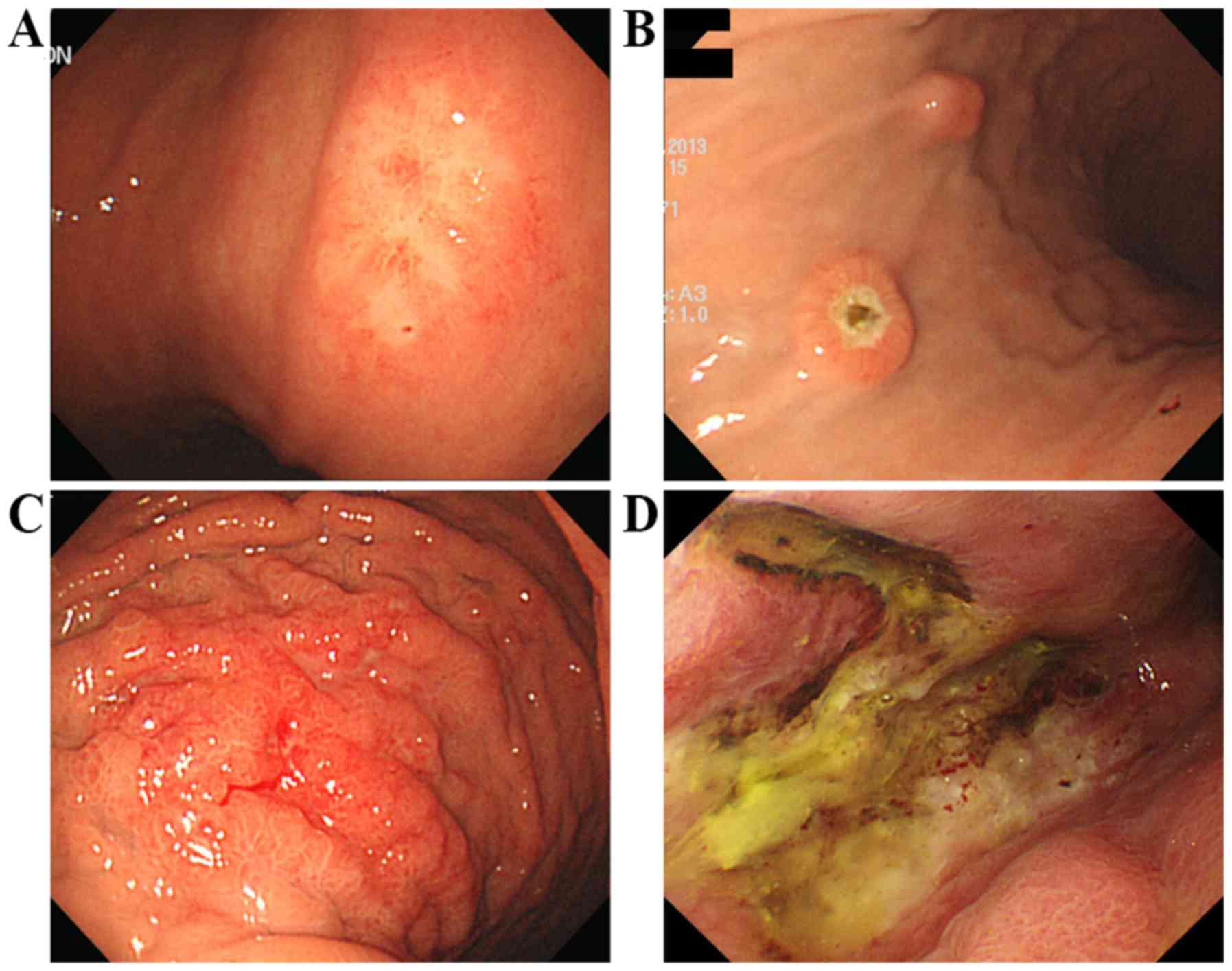
EGD results of patients with gastric metastasis were
diverse in terms of location, size, number, shape, and presence or
absence of bleeding, erosion or ulceration (Fig. 6). Fig.
6A presents white discolored mucosal lesion with speculated
edge, Fig. 6B presents round polypoid
lesions with or without ulceration, Fig.
6C presents diffuse infiltrative lesion with spontaneous
bleeding and fold thickening mimicking Bormann type IV, and
Fig. 6D presents deep ulcerative
lesion with yellowish exudate and spontaneous bleeding. Gastric
metastasis can be located anywhere from the fundus to the antrum,
even in the lesser curvature and posterior wall side of the remnant
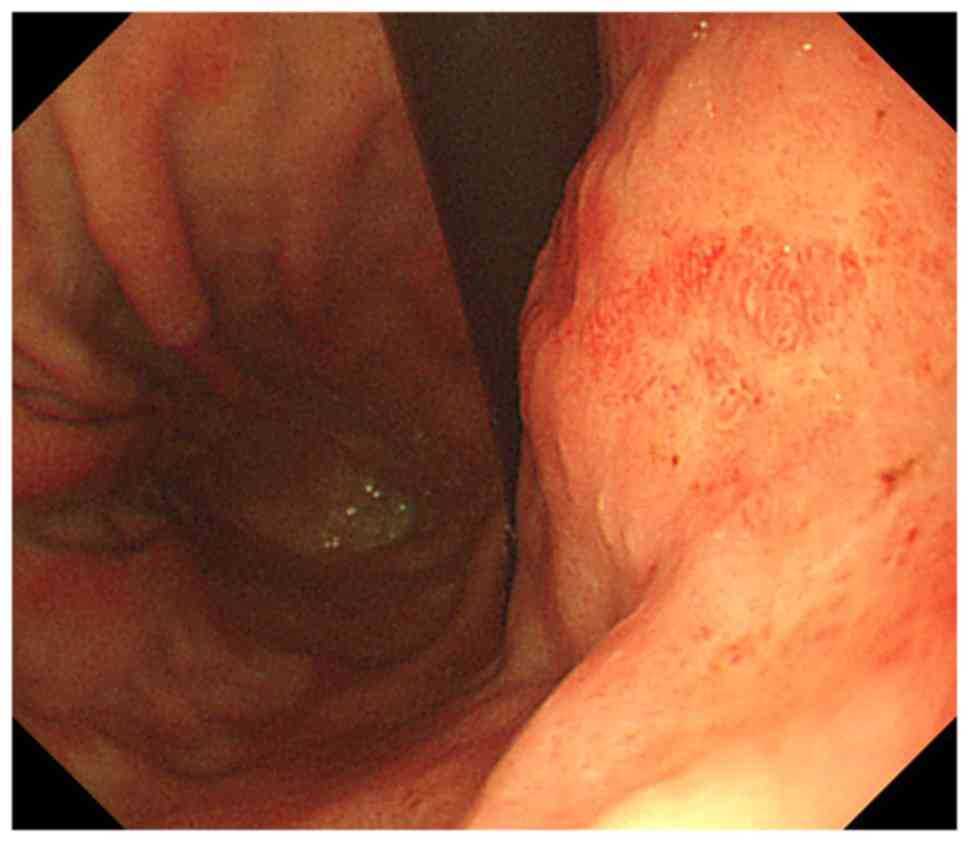
stomach. One of the patients underwent a Whipple procedure for
pancreatic metastasis prior to gastric metastasis. A gastric lesion
was suspected subsequent to a follow-up abdominal CT scan for
disease evaluation of chemotherapy. EGD was performed and a gastric
lesion in the remnant stomach was found (Fig. 7). Sizes ranged between 0.5 and 10 cm.
While 9 patients (69.2%) presented with solitary lesions, the other
patients (30.8%) presented with 2 or multiple lesions that
increased the suspicion of metastasis. Shapes were diverse,
including elevated mucosal lesions or polyps, with or without
erosion or ulceration, and infiltrative or ulcerative lesions,
which can mimic early gastric cancer to advanced gastric cancer. In
terms of bleeding, 1 patient had a mucosal elevated lesion with
oozing bleeding, and another patient had an ill-defined nodular
lesion with spontaneous bleeding. In addition, 1 patient who
presented with melena received hypertonic saline mixed with
epinephrine and argon plasma coagulation for spontaneous bleeding.
There were no specific EGD results to diagnose gastric metastasis.
It is difficult to consider gastric metastasis, particularly in the
setting of a solitary gastric lesion. Taking the aforementioned
into consideration, clinical suspicion and obtaining a full history
of patients in addition to an adequate biopsy of the gastric lesion
are crucial.
Undetermined patients
A total of 4 patients had undetermined status, as
data were not available. Terminal-stage breast cancer was indicated
in 3 patients who were diagnosed with gastric carcinoma as they had
gastric symptoms. Since the patients were diagnosed with
terminal-stage breast cancer, further evaluation and management was
deferred, and they succumbed in the following months.
Discussion
Breast cancer is a heterogeneous disease. In the
present study, it was not possible to precisely discern which type
of breast cancer metastasized most often to the stomach. However,
luminal type A or B breast cancer tended to commonly metastasize to
the stomach. Furthermore, ILC metastasis to the stomach was more
frequent when considering pathological type.
There are several case reports including only a few
cases due to low incidence of breast cancer with gastric metastasis
(6–16). Taal et al (21) reported 51 cases of breast cancer with
gastric metastasis, and 36 of the 51 patients had ILC. In contrast
with a favorable outcome in the current study, median survival was
10 months and the 2-year survival rate was 23% in Taal et al
study. This may be due to advancements in breast cancer treatment
as enrollment of patients was between 1977 and 1997, and cancer
treatment was administered prior to 2000. Xu et al (22) also reported 78 cases of metastatic
gastric cancer from breast carcinoma from a literature search,
which included Taal et al study. Of the 78 cases, 51 had ILC
and positive IHC staining for ER, PR and HER-2 was 94.0, 68.3 and
5.9%, respectively. This result is very similar to the current
study, in which positive IHC staining for ER, PR and HER-2 was
81.8, 50, and 0%, respectively. However, in contrast with Xu et
al's study, which comprised a broad spectrum of patients
regarding study time and medical centers, the current study only
collected data from a single center and in a shorter time
period.
Among the 66 patients diagnosed with gastric
carcinoma by biopsy pathology, 47 had double primary cancer based
on clinical situation or pathology reports. Out of the 47 patients
with double primary cancer, 38 had no evidence of breast or gastric
cancer recurrence according to their follow-up. Among these 38
patients, 34 underwent gastrectomy and 4 underwent endoscopic
resection. Certain patients who had double primary cancer may have
had breast cancer with gastric metastasis, which may have not been
identified due to lack of attention to patient history regarding
breast cancer at the time of diagnosis. Therefore, only 2 out of 47
patients with double primary cancer had stomach samples with ER/PR
IHC staining, and 3 had stomach samples with GCDFP-15 IHC staining.
These results were in contrast with those of the 13 patients with
breast cancer and gastric metastasis, 11 of whom had IHC staining
results for ER, 10 for PR, 9 for HER2 and 10 for GCDFP-15. HER2 IHC
staining was performed for samples from 37 patients, as HER2 is a
routine IHC examination for gastric cancer. Nevertheless, 38 out of
47 patients had no evidence of recurrence. Even if recurrence
following gastrectomy occurred, pathological stage was higher than
stage III. In summary, a number of indications may be considered at
the time of IHC staining, particularly the results of ER/PR and
GCDFP-15 IHC staining. The stomach was the first metastatic lesion
location in 6 (54.5%) out of 11 patients with breast cancer de
novo stage I–III cancer, and the median time interval from
breast cancer diagnosis to gastric metastasis was 77.5 months. This
interval of >5 years may be long enough to consider a gastric
malignancy as a primary cancer. The results of the present study
suggests that clinical suspicion and thorough pathological review
are required.
The 3-year survival rate following gastric
metastasis was 79.1% and the estimated mean survival time was 35.1
months. The 13 patients with breast cancer with gastric metastases
had a more favorable outcome compared with patients with gastric
cancer stage IV (23). A total of 2
patients succumbed, emphasizing that classifying gastric malignancy
as primary or secondary is important for deciding adequate
treatment without unnecessary surgery in addition to estimating
prognosis.
Limitations of the present study included that it
was small, retrospective and based in a single center. Due to these
limitations, it was not possible to demonstrate the associations
among markers associated with breast cancer, including receptor
status, tumor grade and survival. Additional cases are required to
strengthen the present study. Secondly, information was incomplete.
For example, certain IHC staining results were missing. With
cautious suspicion, the number of cases of breast cancer with
gastric metastasis may increase, and additional information may be
gathered. The number of patients with double primary cancer may
have been underestimated in the present study. As gastric cancer
was the variable searched for in pathology reports, data on gastric
lymphoma, gastrointestinal stromal tumors, gastric neuroendocrine
tumors and other types of carcinoma may have been missed. However,
as the proportion of the aforementioned types of cancer was low and
the aim of the present study was to analyze breast cancer with
gastric metastasis, patients may not have been missed from the
analysis process after all. Further translational research for
gastric metastases from breast cancer to elucidate the underlying
biological mechanism is required.
The present study concluded that breast cancer with
gastric metastasis is rare. Luminal-type breast cancer or ILC
tended to metastasize to the stomach more than other types of
breast cancer. The stomach was one of the first metastatic lesion
locations among patients with breast cancer and gastric metastasis,
and these patients had a more favorable prognosis compared with
patients with primary gastric cancer. Since gastric metastasis from
breast cancer is rare, a thorough pathology review and greater
clinical suspicion are required.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JH gathered, analyzed and interpreted the patient
data and wrote the article. YK, JC, SWL, SEP, HKK and HL gathered
and analyzed the patient data. SYC analyzed and interpreted the
pathology of the patients. JK, JSA, YHI and YHP analyzed and
interpreted the patient data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
This study was approved by the Institutional Review
Board of Samsung Medical Center (approval no. 2017-11-097-002), and
the requirement for informed consent was waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Foulkes WD, Smith IE and Reis-Filho JS:
Triple-negative breast cancer. N Engl J Med. 363:1938–1948. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Harris M, Howell A, Chrissohou M, Swindell
RI, Hudson M and Sellwood RA: A comparison of the metastatic
pattern of infiltrating lobular carcinoma and infiltrating duct
carcinoma of the breast. Br J Cancer. 50:231984. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kennecke H, Yerushalmi R, Woods R, Cheang
MCU, Voduc D, Speers CH, Nielsen TO and Gelmon K: Metastatic
behavior of breast cancer subtypes. J Clin Oncol. 28:3271–3277.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Patanaphan V, Salazar OM and Risco R:
Breast cancer: Metastatic patterns and their prognosis. South Med
J. 81:1109–1112. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Abe T, Yoshidome K, Iijima H, Oyama T,
Akamatsu H, Kawai N, Yasumaru M, Tsujimoto M, Nishida T and Tsujii
M: Metastatic breast cancer mimicking primary depressed gastric
cancer. Gastroint Endosc. 70:1241–1242. 2009. View Article : Google Scholar
|
|
7
|
Arrangoiz R, Papavasiliou P, Dushkin H and
Farma JM: Case report and literature review: Metastatic lobular
carcinoma of the breast an unusual presentation. Int J Case Rep.
2:301–305. 2011. View Article : Google Scholar
|
|
8
|
Aurello P, D'Angelo F, Cosenza G, Petrocca
S, Stoppacciaro A, Ramacciato G and Ziparo V: Gastric metastasis 14
years after mastectomy for breast lobular carcinoma: Case Report
and Literature Review. Am Surg. 72:456–460. 2006.PubMed/NCBI
|
|
9
|
Brandi G, Campadelli E, Nobili E and Leone
O: Breast carcinoma presenting as linitis plastica. Dig Liver Dis.
42:3062010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dumoulin FL and Gupta RS: Breast cancer
metastasis to the stomach resembling small benign gastric polyps.
Gastrointest Endosc. 69:174–175. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ellis MC, Mason T, Barnett J, Kiesow LL
and Vetto JT: Gastric malignancies in breast cancer survivors:
Pathology and outcomes. Am J Surg. 197:633–636. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kudo T, Matsumoto T, Nakamura S, Nakamura
S, Esaki M, Yada S, Hirahashi M, Yao T and Iida M: Solitary minute
metastasis from breast cancer mimicking primary intramucosal
gastric signet-cell cancer. Gastrointest Endosc. 62:139–140. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pectasides D, Psyrri A, Pliarchopoulou K,
Floros T, Papaxoinis G, Skondra M, Papatsibas G, Macheras A,
Athanasas G, Arapantoni-Datioti P and Economopoulos T: Gastric
metastases originating from breast cancer: Report of 8 cases and
review of the literature. Anticancer Res. 29:4759–4763.
2009.PubMed/NCBI
|
|
14
|
Rusticeanu M, Schuster M, Moga SL,
Solomayer E-F, Bohle RM, Lammert F and Zimmer V: Metastatic lobular
breast cancer presenting as gastric linitis plastica. Am J Med.
124:e5–e6. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schwarz RE, Klimstra DS and Turnbull ADM:
Metastatic breast cancer masquerading as gastrointestinal primary.
Am J Gastroenterol. 93:111–114. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Whitty LA, Crawford DL, Woodland JH, Patel
JC, Nattier B and Thomas CR: Metastatic breast cancer presenting as
linitis plastica of the stomach. Gastric Cancer. 8:193–197. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Müller A, Homey B, Soto H, Ge N, Catron D,
Buchanan ME, McClanahan T, Murphy E, Yuan W, Wagner SN, et al:
Involvement of chemokine receptors in breast cancer metastasis.
Nature. 410:50–60. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Edge SB and Compton CC: The American Joint
Committee on Cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Laurén P: The two histological main types
of gastric carcinoma: Diffuse and so-called intestinal-type
carcinoma. Acta Pathol Microbiol Scand. 64:31–49. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hicks DG and Schiffhauer L: Standardized
assessment of the HER2 status in breast cancer by
immunohistochemistry. Lab Med. 42:459–467. 2011. View Article : Google Scholar
|
|
21
|
Taal BG, Peterse H and Boot H: Clinical
presentation, endoscopic features, and treatment of gastric
metastases from breast carcinoma. Cancer. 89:2214–2221. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu L, Liang S, Yan N, Zhang L, Gu H, Fei
X, Xu Y and Zhang F: Metastatic gastric cancer from breast
carcinoma: A report of 78 cases. Oncol Lett. 14:4069–4077. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Orditura M, Galizia G, Sforza V,
Gambardella V, Fabozzi A, Laterza MM, Andreozzi F, Ventriglia J,
Savastano B, Mabilia A, Lieto E, et al: Treatment of gastric
cancer. World J Gastroenterol. 20:1635–1649. 2014. View Article : Google Scholar : PubMed/NCBI
|