Introduction
An estimated 4,292,000 newly diagnosed cases of
cancer and 2,814,000 cancer-associated mortalities occurred in
China in 2015, and lung cancer accounted for nearly 20% of those
cases (1). With a low 5-year survival
rate of 17.8% in China (2), lung
cancer has become the most common type of cancer and the leading
cause of cancer-associated mortality over the past few decades
(3). It has been demonstrated that a
number of factors are associated with the progression of lung
cancer, including tobacco smoke, occupational exposure to asbestos
and radon, environmental pollution, chronic pulmonary inflammation
and a family history of lung cancer (2,4). In
Chinese women, where the prevalence of smoking is low, unpredicted
high incidences of lung cancer have been observed due to exposure
to indoor air pollution (5). A major
precaution in preventing lung cancer is smoking cessation, while
early diagnosis and treatment of lung cancer are of vital
importance in enhancing the prognosis of lung cancer (4).
Systemic inflammatory response (SIR) is associated
with survival in a variety of cancer types, including gastric,
esophageal and lung cancer (6–8). Previous
studies have examined the role of various SIR indicators in
predicting the outcomes of cancer patients (9–12). Albumin
(ALB) and globulin (GLB) are two major serum proteins. It is well
known that low ALB and high GLB levels are associated with
malnutrition and chronic inflammation (13). C-reactive protein (CRP) is an acute
phase protein, which is produced and secreted by hepatocytes via
several inflammatory stimuli, including interleukin (IL)-1 and IL-6
(14). Increased CRP levels have been
observed in numerous conditions, including inflammation, infection,
tissue infarction and malignancy (15). Lactate dehydrogenase (LDH), a
ubiquitous cellular enzyme, which catalyzes anaerobic glycolysis,
is increased in patients with malignant tumors (16). The neutrophil-lymphocyte ratio (NLR)
refers to the proportion of absolute neutrophils in the lymphocyte
count in the blood circulation. The platelet-to-lymphocyte ratio
(PLR), the proportion of absolute platelets to the lymphocyte count
in peripheral blood, has been proposed as a reliable prognostic
indicator for lung cancer (17,18). NLR
and PLR have been demonstrated to be associated with the prognosis
of a wide variety of tumors (19,20).
The present study evaluated whether these
SIR-associated indicators may provide beneficial prognostic
information for patients with resectable lung cancer.
Materials and methods
Subjects and inclusion criteria
The present study was conducted as a retrospective
investigation of patients with lung cancer that had been referred
to the First Affiliated Hospital of Soochow University (Jiangsu,
China) between January 2007 and May 2016. Approval for the study
was granted by the Medical Ethics Committees of the First
Affiliated Hospital of Soochow University. Clinical and
pathological records of all the patients participating in the study
were reviewed periodically.
A total of 101 patients with resectable lung cancer
were recruited in the present study. All cases were confirmed by
surgery and pathology. Among the 101 patients, 9 had small cell
lung cancer (SCLC). Of the 92 non-small cell lung cancer (NSCLC)
samples, 33 were cases of squamous and 59 were cases of
adenocarcinoma. The adenocarcinoma included 23 cases with an acinar
pattern, 19 cases with a papillary pattern, 11 cases with a
micropapillary pattern and 6 cases with a solid growth pattern. All
patients underwent pulmonary lobectomy and systematic lymph node
dissection. Patients with squamous carcinoma were treated with
cisplatin 75 mg/m2 day 1 and gemcitabine 1250
mg/m2 day 1,8. Patients with adenocarcinomas were
treated with cisplatin 75 mg/m2 day 1 and pemetrexed 500
mg/m2 day 1. Patients with resectable SCLC were treated
with cisplatin 80 mg/m2 day 1 and etoposide 100
mg/m2 day 1,2,3. Patient characteristics are detailed in
Table I. The median age of the 101
patients was 60 years (range, 27–80 years), and 63 patients were
male (age range, 27–78 years) and 38 were female (age range, 27–80
years). The performance status of the patients was evaluated using
the Eastern Cooperative Oncology Group (ECOG) performance status.
It was ensured that patients displayed a good performance status
(ECOG score ≤1). All the samples with coexisting diseases were
excluded to eliminate the differences in general performance
status. The staging of cancer was determined according to
Tumor-Node-Metastasis classification and was classified using the
American Joint Committee on Cancer (AJCC) recommendations (21). The prognostic analyses were performed
regarding overall survival (OS).
 | Table I.Clinicopathologic features. |
Table I.
Clinicopathologic features.
|
|
| CRP | ALB | GLB | LDH | NLR | PLR |
|---|
|
|
|
|
|
|
|
|
|
|---|
| Feature | n | Low, n | High, n | χ2 | P-value | Low, n | High, n | χ2 | P-value | Low, n | High, n | χ2 | P-value | Low, n | High, n | χ2 | P-value | Low, n | High, n | χ2 | P-value | Low, n | High, n | χ2 | P-value |
|---|
| Sex |
|
Male | 63 | 27 | 36 | 3.908 | 0.048a | 25 | 38 | 1.455 | 0.228 | 32 | 31 | 0.006 | 0.938 | 35 | 28 | 1.715 | 0.190 | 31 | 32 | 0.717 | 0.397 | 31 | 32 | 0.111 | 0.739 |
|
Female | 38 | 24 | 14 |
|
| 26 | 12 |
|
| 19 | 19 |
|
| 16 | 22 |
|
| 22 | 16 |
|
| 20 | 18 |
|
|
| Age, years |
|
≤60 | 56 | 32 | 24 | 2.222 | 0.136 | 26 | 30 | 0.831 | 0.362 | 26 | 30 | 0.831 | 0.362 | 26 | 30 | 0.831 | 0.362 | 32 | 24 | 1.098 | 0.295 | 27 | 29 | 0.262 | 0.609 |
|
>60 | 45 | 19 | 26 |
|
| 25 | 20 |
|
| 25 | 20 |
|
| 25 | 20 |
|
| 21 | 24 |
|
| 24 | 21 |
|
|
| Pathological
type |
|
NSCLC | 92 | 42 | 50 | 0.048 | 0.827 | 42 | 50 | 0.048 | 0.827 | 43 | 49 | 0.024 | 0.876 | 44 | 48 | 0.024 | 0.876 | 38 | 54 | 0.012 | 0.913 | 57 | 35 | 3.818 | 0.051 |
|
SCLC | 9 | 5 | 4 |
|
| 5 | 4 |
|
| 5 | 4 |
|
| 4 | 5 |
|
| 3 | 6 |
|
| 2 | 7 |
|
|
| Tumor size, cm |
| ≤5 | 73 | 45 | 28 | 13.093 | 0.000b | 41 | 32 | 3.386 | 0.066 | 39 | 34 | 0.904 | 0.342 | 35 | 38 | 4.929 | 0.026a | 37 | 36 | 0.338 | 0.561 | 39 | 34 | 0.904 | 0.342 |
|
>5 | 28 | 6 | 22 |
|
| 10 | 18 |
|
| 12 | 16 |
|
| 16 | 12 |
|
| 16 | 12 |
|
| 12 | 16 |
|
|
| T stage |
| T1,
T2 | 77 | 43 | 34 | 0.046 | 0.830 | 46 | 31 | 11.080 | 0.001b | 43 | 34 | 3.709 | 0.054 | 38 | 39 | 0.170 | 0.680 | 39 | 38 | 0.433 | 0.510 | 41 | 36 | 0.982 | 0.322 |
| T3,
T4 | 24 | 14 | 10 |
|
| 5 | 19 |
|
| 8 | 16 |
|
| 13 | 11 |
|
| 14 | 10 |
|
| 10 | 14 |
|
|
| N stage |
| N0,
N1 | 68 | 35 | 33 | 0.079 | 0.778 | 37 | 31 | 0.313 | 0.576 | 39 | 29 | 3.916 | 0.048a | 40 | 28 | 5.775 | 0.016a | 35 | 33 | 0.079 | 0.778 | 37 | 31 | 1.277 | 0.258 |
| N2 | 33 | 16 | 17 |
|
| 16 | 17 |
|
| 14 | 19 |
|
| 21 | 12 |
|
| 16 | 17 |
|
| 12 | 21 |
|
|
| AJCC stage |
| I,
II | 64 | 37 | 27 | 3.742 | 0.053 | 35 | 29 | 1.228 | 0.268 | 36 | 28 | 2.315 | 0.128 | 30 | 34 | 0.916 | 0.339 | 35 | 29 | 0.343 | 0.558 | 35 | 29 | 1.228 | 0.268 |
|
III | 37 | 14 | 23 |
|
| 16 | 21 |
|
| 15 | 22 |
|
| 21 | 16 |
|
| 18 | 19 |
|
| 16 | 21 |
|
|
Blood samples
Pre-surgery blood samples were collected within one
week prior to surgery. Post-surgery blood samples were regarded as
pre-chemotherapy samples and were collected three weeks after
surgery. Post-chemotherapy samples were collected following three
cycles of standard chemotherapy. Peripheral venous blood (5–7 ml)
samples were fasted and obtained between 6:30 and 7:30 am in order
to standardize the known impact of circulating hormones (circadian
rhythm) on the number and subtype distribution of the various white
blood cell indices. Blood samples were analyzed using a hematology
analyzer (Sysmex XE-2100; Sysmex Corporation, Kobe, Japan) or
biochemical analyzer (Olympus AU5421+ISE; Olympus Corporation,
Tokyo, Japan). ALB, GLB, LDH, NLR and PLR levels are presented in
Table I. The patients were divided
into two groups according to the median values. The
post/pre-treatment ratios were defined as the ratio of
pre-treatment SIR-related indicator values and the corresponding
ones obtained following therapy.
Evaluation
Computed tomography (CT) scans were performed for
the assessment of response every 2 months and evaluated according
to the Response Evaluation Criteria in Solid Tumors 1.1 (22).
Follow-up
All the patients were followed-up post-operatively
for between 16 and 90 months, with a median follow-up period of 36
months. Survival time was measured from the date of diagnosis until
mortality or last clinical evaluation. The prognostic analyses were
performed regarding overall survival (OS). OS was defined as the
time between the diagnosis date and mortality from any cause.
Statistical analysis
All statistical analyses were performed using SPSS
19.0 software (IBM Corp., Armonk, NY, USA). The associations
between blood parameter status and clinicopathological features
were determined using χ2 tests. For analysis of survival
data, Kaplan-Meier curves were constructed, and statistical
analyses was performed using the log-rank test. Receiver operating
characteristic (ROC) curve analysis was performed to evaluate the
predictive values of SIR-related indicators for resectable lung
cancer and to determine the best cut-off value of SIR-related
indicators. The associations between changes in the status of
SIR-related indicators and surgery or chemotherapy were assessed by
Student's t-tests. The multivariate logistic regression model was
employed to identify the independent risk factors associated with
resectable lung cancer. Numerical data are presented as the mean ±
standard error. P<0.05 was considered to indicate a
statistically significant difference.
Results
Pre-treatment ALB level is associated
with outcomes in patients with resectable lung cancer
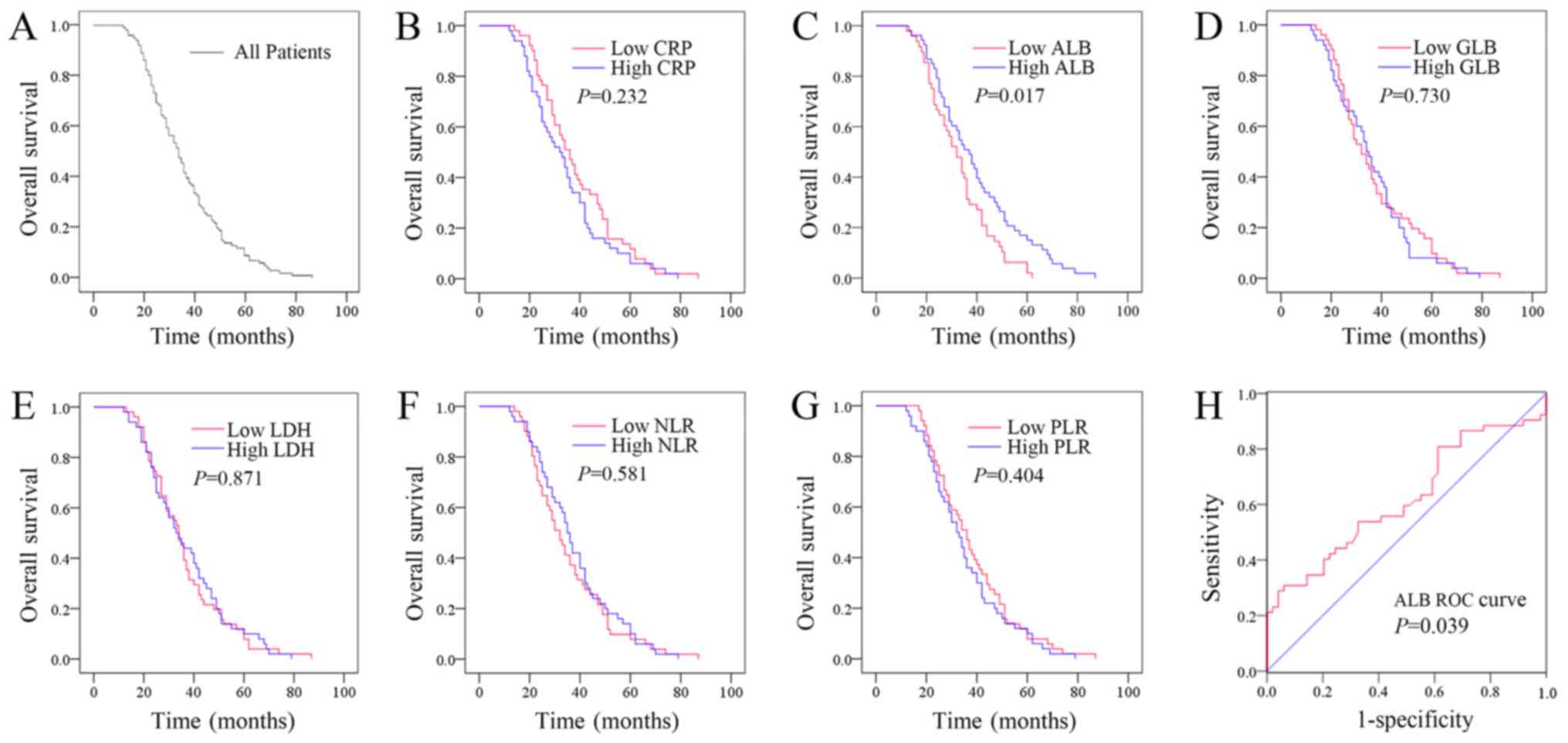
The median OS time for all the patients with
resectable lung cancer was 36 (33.329–39.542) months (Fig. 1A). Kaplan-Meier plots were used to
determine the effect of pre-treatment CRP, ALB, GLB, LDH, NLR and
PLR status on OS (Fig. 1B-G). The
patients were divided into two groups according to the median
values of CRP (low CRP, ≤1.426 mg/l or high CRP, >1.426 mg/l),
ALB (low ALB, ≤42.600 g/l or high ALB, >42.600 g/l), GLB (low
GLB, ≤28.227 g/l or high GLB, >28.227 g/l), LDH (low LDH,
≤178.965 U/l or high LDH, >178.965 U/l), NLR (low NLR, ≤2.049 or
high NLR, >2.049), or PLR (low PLR, ≤113.534 or high PLR,
>113.534). The median OS time of the high CRP group was 32
(25.070–38.930) months, while that of the low CRP group was 36
(30.760–41.240) months (P=0.232). The median OS time of the high
ALB group was 37 (30.758–43.242) months, while that of the low ALB
group was 32 (27.154–36.846) months (P=0.017). The median OS time
of the high GLB group was 34 (30.040–37.960) months, while that of
the low GLB group was 32 (26.558–37.442) months (P=0.730). The
median OS time of the high LDH group was 33 (26.070–39.930) months,
while that of the low LDH group was 34 (29.342–38.658) months
(P=0.871). The median OS time of the high NLR group was 35
(31.535–38.465) months, while that of the low NLR group was 32
(27.342–36.658) months (P=0.581). The median OS time of the high
PLR group was 32 (27.050–36.950) months, while that of the low PLR
group was 36 (31.010–40.990) months (P=0.404). Therefore, the
patients whose pre-treatment ALB levels were lower exhibited a
poorer prognosis. However, pre-treatment levels of CRP, GLB, LDH,
NLR or PLR had no effect on OS.
ROC curve analysis was subsequently performed to
evaluate the predictive value of pre-treatment ALB for resectable
lung cancer and determine the optimum cut-off value. As
demonstrated in Fig. 1H, the area
under the curve of pre-treatment ALB was 0.619 (95% CI 0.509–0.727;
P=0.039), and the optimum cut-off point of pre-treatment ALB was
47.850 g/l with a sensitivity of 28.8% and a specificity of
95.9%.
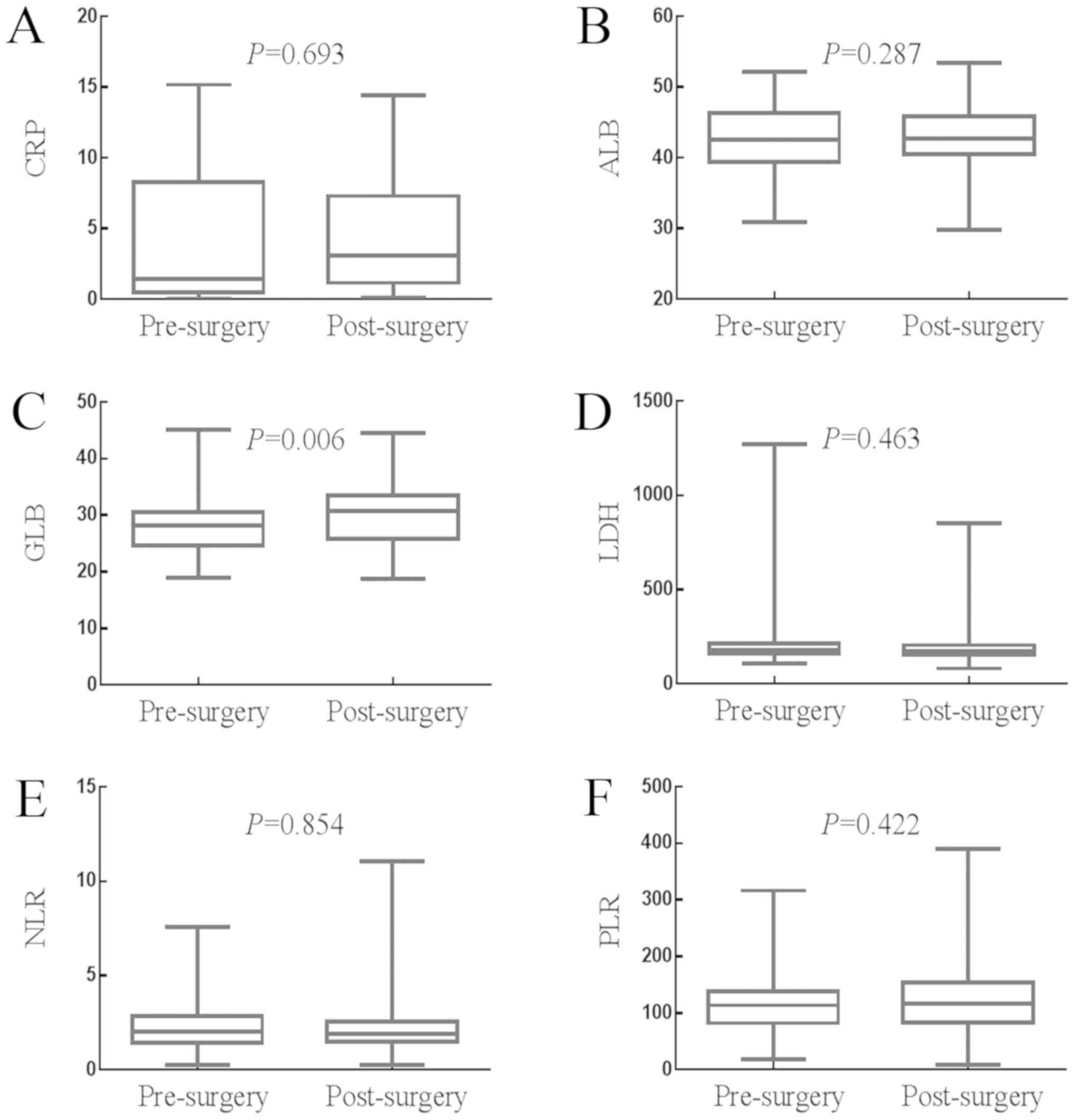
Effects of surgery on the values of
SIR-related indicators
The effects of surgery on the levels of SIR-related
indicators are presented in Fig.
2A-F. The median value of CRP was 1.430 mg/l (0.980–2.900 mg/l)
prior to surgery and 3.070 mg/l (2.140–4.360 mg/l) following
surgery (P=0.693). The median value of ALB was 42.600 g/l
(41.700–43.880 g/l) prior to surgery and 42.640 g/l (41.900–43.800
g/l) following surgery (P=0.287). The median value of GLB was
28.230 g/l (27.400–29.100 g/l) prior to surgery and 30.800 g/l
(29.250–31.430 g/l) following surgery (P=0.006). The median value
of LDH was 178.970 U/l (170.040–192.610 U/l) prior to surgery and
173.000 U/l (165.000–181.390 U/l) following surgery (P=0.463). The
median value of NLR was 2.050 (1.790–2.289) prior to surgery and
1.920 (1.740–2.080) following surgery (P=0.854). The median value
of PLR was 113.530 (100.960–125.930) prior to surgery and 116.790
(100.751–135.360) following surgery (P=0.423). Therefore, surgery
significantly increased the value of GLB, but had no significant
impact on the values of CRP, ALB, LDH, NLR or PLR.
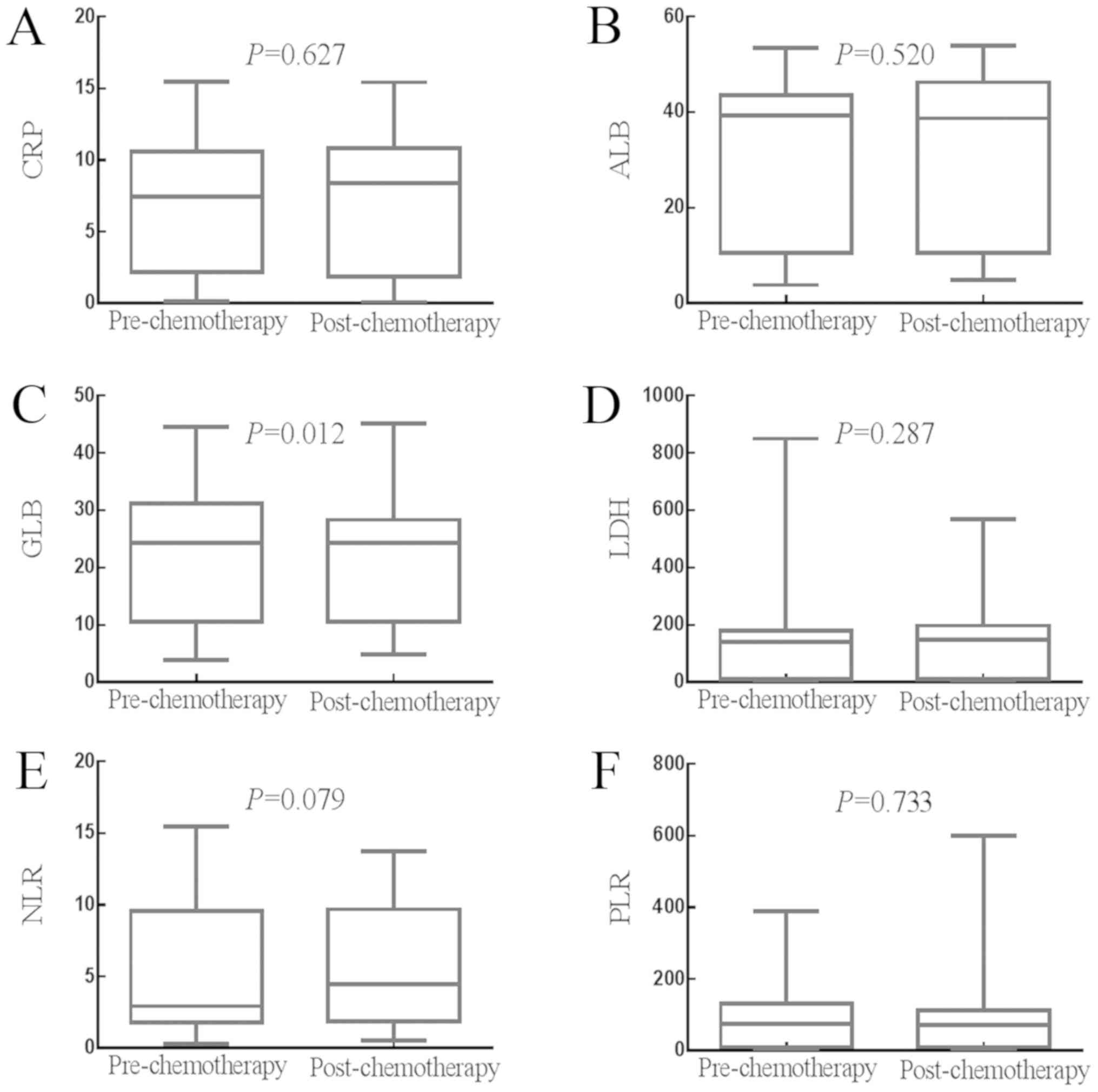
Effects of adjuvant chemotherapy on
the values of SIR-related indicators
The effects of adjuvant chemotherapy on the status
of SIR-related indicators are shown in Fig. 3A-F. The median value of CRP was 3.070
mg/l (2.140–4.360 mg/l) prior to chemotherapy and 2.100 mg/l
(1.780–3.870 mg/l) following chemotherapy (P=0.627). The median
value of ALB was 42.640 g/l (41.900–43.800 g/l) prior to
chemotherapy and 45.590 g/l (43.880–46.600 g/l) following
chemotherapy (P=0.520). The median value of GLB was 30.800 g/l
(29.250–31.430 g/l) prior to chemotherapy and 27.500 g/l
(26.700–28.600 g/l) following chemotherapy (P=0.012). The median
value of LDH was 173.000 U/l (165.000–181.390 U/l) prior to
chemotherapy and 189.150 U/l (178.000–198.850 U/l) following
chemotherapy (P=0.287). The median value of NLR was 1.920
(1.740–2.080) prior to chemotherapy and 1.980 (1.830–2.260)
following chemotherapy (P=0.079). The median value of PLR was
116.790 (100.751–135.360) prior to chemotherapy and 105.170
(95.442–114.040) following chemotherapy (P=0.733). Therefore,
adjuvant chemotherapy significantly decreased the value of GLB, but
had no significant impact on the values of CRP, ALB, LDH, NLR or
PLR.
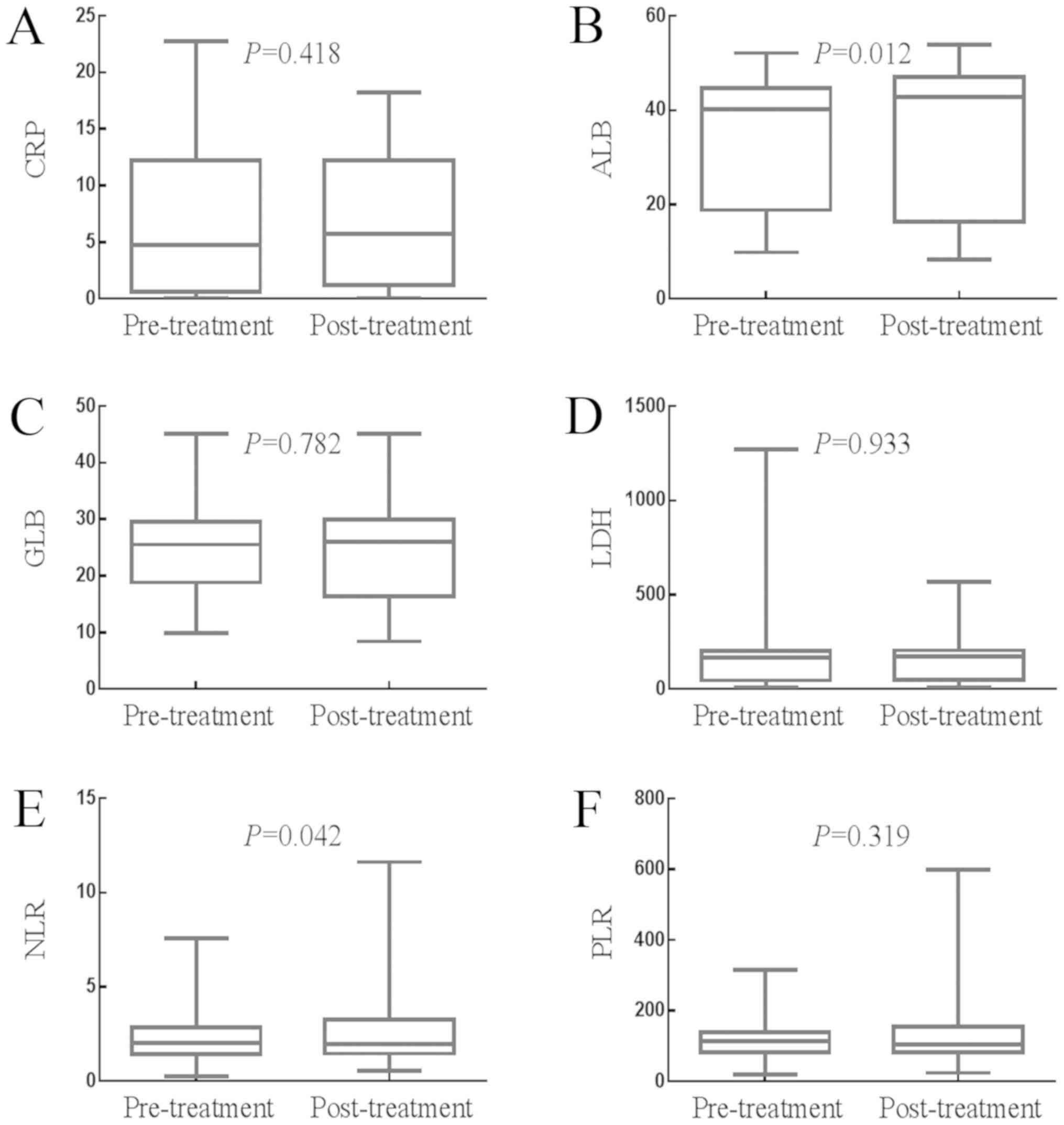
Effects of whole course of treatment
on the values of SIR-related indicators
The impact of whole course of treatment (surgery and
adjuvant chemotherapy) on the values of the SIR-related indicators
is presented in Fig. 4A-F. The median
value of CRP was 1.430 mg/l (0.980–2.900 mg/l) prior to treatment
and 2.100 mg/l (1.780–3.870 mg/l) following treatment (P=0.418).
The median value of ALB was 42.600 g/l (41.700–43.880 g/l) prior to
treatment and 45.590 g/l (43.880–46.600 g/l) following treatment
(P=0.012). The median value of GLB was 28.230 g/l (27.400–29.100
g/l) prior to treatment and 27.500 g/l (26.700–28.600 g/l)
following treatment (P=0.782). The median value of LDH was 178.970
U/l (170.040–192.610 U/l) prior to treatment and 189.150 U/l
(178.000–198.850 U/l) following treatment (P=0.933). The median
value of NLR was 2.050 (1.790–2.289) prior to treatment and 1.980
(1.830–2.260) following treatment (P=0.042). The median value of
PLR was 113.530 (100.960–125.930) prior to treatment and 105.170
(95.442–114.040) following treatment (P=0.319). Therefore, whole
course of treatment significantly increased the value of ALB, but
significantly decreased the value of NLR, but had no significant
effect on the values of CRP, GLB, LDH or PLR.
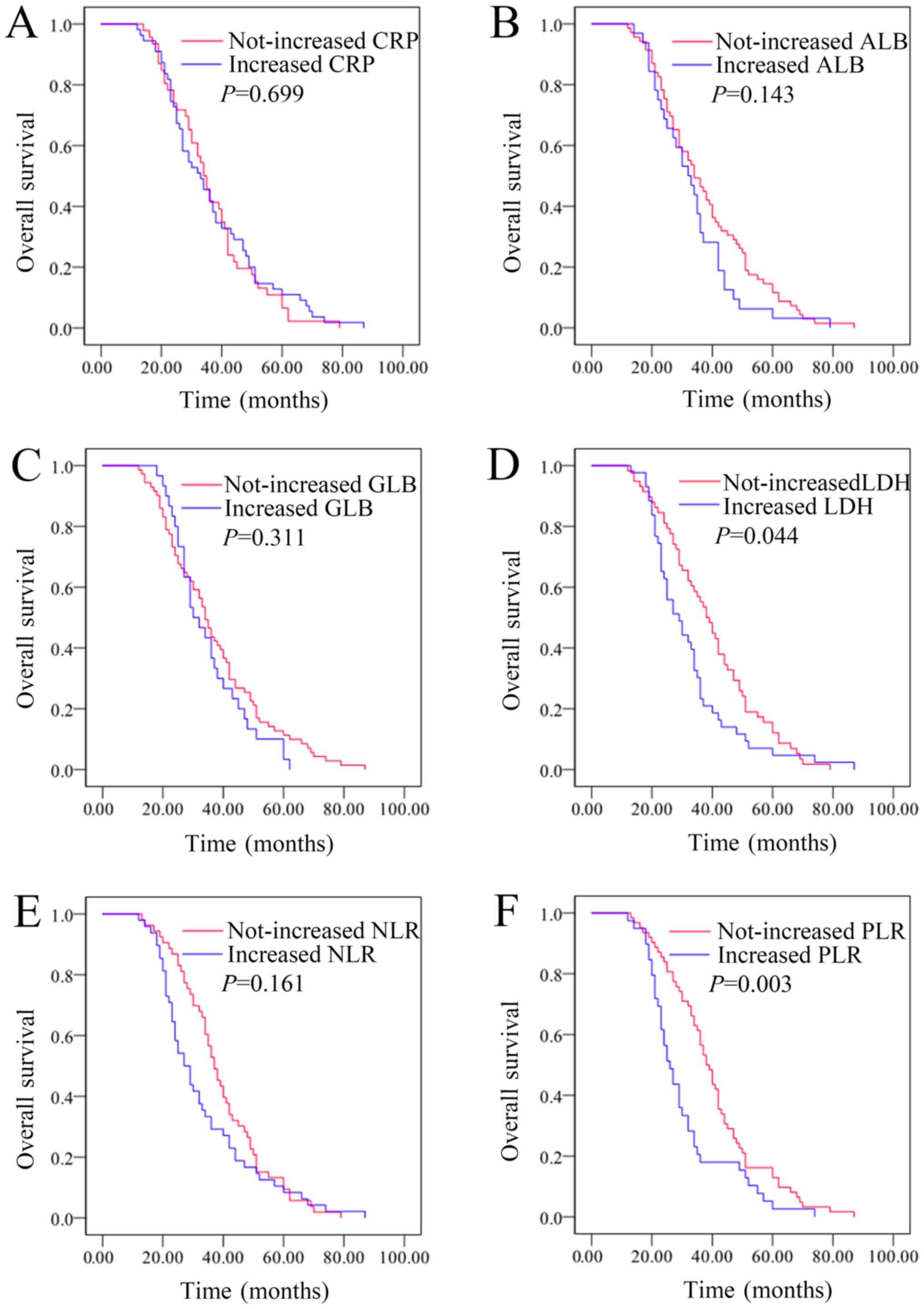
Changes in LDH and PLR levels
following whole course of treatment were associated with the
outcomes in patients with resectable lung cancer, while CRP, ALB,
GLB, and NLR levels were not associated with outcomes
Kaplan-Meier plots were used to determine the effect
of individual changes in CRP, ALB, GLB, LDH, NLR and PLR status on
OS (Fig. 5A-F). The median OS time of
patients whose CRP levels increased following whole course of
treatment was 33 (25.733–40.267) months, while that of the
not-increased CRP group was 34 (30.202–37.798) months (P=0.699).
The median OS time of patients whose ALB levels increased following
whole course of treatment was 32 (25.348–38.652) months, while that
of the not-increased ALB group was 34 (28.186–39.814) months
(P=0.143). The median OS time of patients whose GLB levels
increased following whole course of treatment was 30
(23.738–36.262) months, while that of the not-increased GLB group
was 34 (30.330–37.670) months (P=0.311). The median OS time of
patients whose LDH levels increased following whole course of
treatment was 29 (22.575–35.425) months, while that of the
not-increased LDH group was 38 (33.024–42.976) months (P=0.044).
The median OS time of patients whose NLR levels increased following
whole course of treatment was 27 (22.150–31.850) months, while that
of the not-increased NLR group was 37 (32.924–41.076) months
(P=0.161). The median OS time of patients whose PLR levels
increased following whole course of treatment was 26
(22.329–29.671) months, while that of the not-increased PLR group
was 38 (34.142–41.858) months (P=0.003). Therefore, changes in LDH
and PLR levels following whole course of treatment were associated
with outcomes in patients with resectable lung cancer, while CRP,
ALB, GLB and NLR levels were not associated with the outcomes
Prognostic factors for resectable lung
cancer
Sex, age, pathological type, tumor size, T stage, N
stage, AJCC stage, as well as baseline CRP, ALB, GLB, LDH, NLR and
PLR, post-/pre-treatment ratios of CRP, ALB, GLB, LDH, NLR and PLR
were evaluated by univariate analyses. Risk factors (P<0.1) were
evaluated by multivariate analysis. Univariate analyses
demonstrated that AJCC stage III [hazard ratio (HR), 1.538; 95%
confidence interval (CI), 1.010–2.340; P=0.045], low pre-treatment
ALB (HR, 1.625; 95% CI, 1.077–2.452; P=0.021), post-/pre-treatment
PLR ratio (HR, 1.810; 95% CI,1.201–2.729; P=0.005) were significant
risk factors for a poor prognosis (Table
II). In multivariate analysis, low pre-treatment ALB (HR,1.738;
95% CI,1.143–2.643; P=0.010) and increased post-/pre-treatment PLR
(HR, 1.890; 95% CI,1.238–2.887; P=0.003) were revealed to be
independently associated with poor survival.
 | Table II.Univariate and multivariate logistic
regression analysis of resectable lung cancer risk factors. |
Table II.
Univariate and multivariate logistic
regression analysis of resectable lung cancer risk factors.
| A, Univariate
analysis. |
|---|
|
|---|
|
| Overall survival
(OS) |
|---|
|
|
|
|---|
| Risk factors | OR (95% CI) | P-value |
|---|
| Sex |
| (Female
or Male) | 0.782
(0.517–1.183) | 0.245 |
| Age |
| (>60
years or ≤60 years) | 1.059
(0.713–1.572) | 0.776 |
| Pathologic
type |
| (NSCLC
or SCLC) | 0.890
(0.446–1.774) | 0.740 |
| Tumor size
(cm) |
| (>5
or ≤5) | 0.851
(0546–1.326) | 0.476 |
| Depth of
invasion |
| (T3-4
or T1-2) | 1.126
(0.708–1.789) | 0.617 |
| Lymphonodus
metastasis |
| (N2 or
N0-1) | 1.474
(0.959–2.264) | 0.077 |
| AJCC stage |
| (III or
I–II) | 1.538
(1.010–2.340) | 0.045a |
| Pre-treatment
CRP |
|
(>1.430 mg/l or ≤1.430
mg/l) | 1.265
(0.853–1.875) | 0.243 |
| Pre-treatment
ALB |
|
(≤42.600 g/l or >42.600
g/l) | 1.625
(1.077–2.452) | 0.021a |
| Pre-treatment
GLB |
|
(>28.227 g/l or ≤28.227
g/l) | 1.071
(0.721–1.589) | 0.735 |
| Pre-treatment
LDH |
|
(>178.965 U/l or ≤178.965
U/l) | 0.969
(0.654–1.436) | 0.874 |
| Pre-treatment
NLR |
|
(>2.049 or ≤2.049) | 0.897
(0.606–1.330) | 0.590 |
| Pre-treatment
PLR |
|
(>113.534 or ≤113.534) | 1.178
(0.795–1.746) | 0.414 |
| Post-/pre-treatment
CRP ratio |
|
(>1.1 or ≤1.1) | 0.926
(0.623–1.377) | 0.705 |
| Post-/pre-treatment
ALB ratio |
|
(>1.1 or ≤1.1) | 1.366
(0.891–2.094) | 0.152 |
| Post-/pre-treatment
GLB ratio |
|
(>1.1 or ≤1.1) | 1.246
(0.806–1.928) | 0.323 |
| Post-/pre-treatment
LDH ratio |
|
(>1.1 or ≤1.1) | 1.498
(1.000–2.244) | 0.050 |
| Post-/pre-treatment
NLR ratio |
|
(>1.1 or ≤1.1) | 1.318
(0.888–1.957) | 0.171 |
| Post-/pre-treatment
PLR ratio |
|
(>1.1 or ≤1.1) | 1.810
(1.201–2.729) | 0.005b |
|
| B, Multivariate
analysis. |
|
|
| Overall survival
(OS) |
|
|
|
| Risk
factors | OR (95%
CI) | P-value |
|
| Lymphonodus
metastasis |
| (N2 or
N0-1) | 1.423
(0.714–2.838) | 0.317 |
| AJCC stage |
| (III or
I–II) | 1.163
(0.596–2.267) | 0.658 |
| Pre-treatment
ALB |
|
(≤42.600 g/l or >42.600
g/l) | 1.738
(1.143–2.643) | 0.010a |
| Post-/pre-treatment
LDH ratio |
|
(>1.1 or ≤1.1) | 1.515
(0.997–2.304) | 0.052 |
| Post-/pre-treatment
PLR ratio |
|
(>1.1 or ≤1.1) | 1.890
(1.238–2.887) | 0.003b |
Discussion
Cancer-related SIR is associated with the genetic
instability of cancer cells, serving a crucial role in tumor
development, including proliferation of malignant cells,
angiogenesis, metastasis, immune escape and resistance to
chemotherapeutic agents (23–25). Chronic obstructive pulmonary disease
(COPD) and lung cancer share a common etiological factor, cigarette
smoking (26). Furthermore, chronic
pulmonary inflammatory diseases, particularly COPD, are risk
factors for developing lung cancer, irrespective of smoking history
(27). Therefore, a previous study
confirmed an association among smoking, COPD and lung cancer
(28). Possible mechanisms involving
cigarette smoking and chronic inflammation are listed as follows:
Firstly, tobacco smoke compromises the integrity of the respiratory
epithelium, impairs mucociliary clearance and attenuates the
defense against harmful environmental agents (29). Secondly, long-term cigarette smoking
activates alveolar macrophages, leading to increased secretion of
pro-inflammatory cytokines and reactive oxygen species (ROS), which
leads to chronic inflammatory infiltration and tissue damage
(30). Reiterative injury of
epithelia has been proven to be involved in tumor initiation
(31). Thirdly, cigarette smoking and
host systemic inflammation may provoke the excessive production of
ROS, which causes direct damage to DNA and leads to further somatic
mutations, thereby increasing the predisposition to malignant
tumors (32). As systemic
inflammation is associated with cancer development, the prognostic
significance of indicators that assess the state of SIR requires
further investigation. For example, NLR, PLR and the CRP/ALB ratio
were associated with the prognosis of several types of cancer,
including breast, lung and gastric cancer (33–35). The
present study investigated the predictive values of CRP, ALB, GLB,
LDH, NLR and PLR in patients with resectable lung cancer.
Previous studies have suggested that a high level of
CRP is correlated with a poorer prognosis inpatients with
resectable lung cancer (36,37). For instance, Hara et al
(36) demonstrated that
disease-specific survival and OS rates in the high-CRP group (CRP
≥5 mg/l) were significantly lower than in the low CRP group (CRP
<5 mg/l) in patients with resectable lung cancer. In a study
undertaken by Lee et al (37),
a high pre-operative serum CRP level was considered an independent
prognostic indicator in patients with resectable lung cancer. A
higher CRP level was correlated with a larger tumor size, increased
lymph node metastasis and vascular invasion in patients with NSCLC
(37). Tumor-derived inflammatory
cytokines (including IL-6), which can block p53-induced apoptosis
and maintain a suitable tumor microenvironment for the survival of
malignant cells, have been demonstrated to be a primary inducer of
CRP production (38). Therefore, a
higher CRP level could be an indicator of a poor prognosis in lung
cancer. The present study demonstrated that a high pre-treatment
CRP level was associated with a lager tumor size. However, surgery
combined with adjuvant chemotherapy had no significant effects on
the level of CRP. In addition, the pre-treatment level and
post-/pre-treatment ratio of CRP had no significant impact on
OS.
ALB, a major type of human plasma protein
synthesized by the liver, is commonly used as a marker for
assessment of individual nutritional status (13). On account of the fact that
malnutrition and SIR are induced by malignant cells, the synthesis
of ALB was suppressed and the level of serum ALB decreased sharply
in patients with advanced cancer (39). A variety of mechanisms are involved in
the association between a low ALB level and a poor prognosis.
Firstly, patients with malignant tumors suffer from weight loss,
nutrition depletion and even cachexia, typically with decreased
serum ALB levels. Secondly, persistent inflammation in convalescent
patients following resection are characterized with insufficient
ALB recovery, leading to the proliferation of persistent
post-operative tumor cells, which leads to a poorer prognosis and
early recurrence (40,41). Furthermore, cancer-induced
malnutrition leads to numerous clinical consequences, including
decreased treatment response, increased treatment-related toxicity
and decreased quality of life (11).
Numerous studies have evaluated the association
between the serum level of ALB and the survival of patients with
cancer. It was identified that a lower serum ALB level is an
independent indicator of a poorer survival in various types of
cancer (42). For instance, Tolia
et al (43) indicated that the
serum ALB level was significantly associated with OS in univariate
analysis. Additionally, Jin et al (44) analyzed 101 samples from patients with
stage I NSCLC and concluded that patients with low pre-operative
ALB levels (<35 g/l) had a significantly poorer survival rate
than patients with normal pre-operative serum ALB levels (≥35 g/l).
Furthermore, patients with low post-operative ALB levels had a
poorer survival rate when compared with patients with normal
post-operative serum ALB levels (44). As demonstrated in Table I, the pre-treatment ALB level was
associated with T stage. However, the pre-treatment ALB level was
not associated with tumor size, N stage or AJCC stage. The present
study concluded that a whole course of treatment significantly
increased the value of ALB, which was accompanied by improvements
in individual nutritional status and reductions in tumor burden.
Patients with low pre-treatment ALB levels had poorer outcomes.
Multivariate analyses demonstrated that a low pre-treatment ALB
level was an independent risk factor for prognosis. The ROC curve
analysis demonstrated that a pre-treatment ALB value of 47.850 g/l
was considered to be the optimal cut-off value for prognosis, and
the sensitivity was 28.8% and specificity was 95.9%. The
post-/pre-treatment ratio of ALB had no significant effect on OS.
In summary, a high pre-treatment ALB level could be a favorable
prognostic indicator in resectable lung cancer.
As an indicator of SIR status, GLB is synthesized by
the human monocyte-phagocyte system, serving a crucial role in the
antitumor immune response (45). Qu
et al (46) indicated that a
higher percentage of α1-GLB in the serum was significantly
associated with a higher pathological stage and poorer tumor status
(46). A major possible mechanism to
explain the findings of Qu et al (46) is that α1-antitrypsin (AAT), a major
component of GLB, was increased in several types of tumor,
including lung cancer. AAT may regulate host immunodefence
mechanisms and may promote tumor progression by inhibiting T
cell-mediated cytotoxicity, antibody-dependent cell-mediated
cytotoxicity and activity of natural killer cells (47). In the present study, although surgery
upregulated the level of GLB and adjuvant chemotherapy
downregulated the level of GLB, a whole course of treatment (a
combination of surgery and adjuvant chemotherapy) had no
significant effect on the level of GLB. Neither the pre-treatment
GLB level nor post-/pre-treatment ratio of GLB had a significant
impact on OS.
LDH, which is established as a universal enzyme,
catalyzes anaerobic glycolysis, and it is a ubiquitously increased
indicator in patients with malignant tumors (16,11,48,49).
The present study included components of the mechanisms that are
involved in a poor prognosis, which are associated with LDH.
Firstly, hypoxia, high rates of glucose uptake and lactate
production are characteristics of malignant tumors, which
facilitate anaerobic glycolysis and promote the proliferation of
cancer cells (50,51). Therefore, a high level of LDH reflects
a highly metabolic and more aggressive tumor status. Secondly,
previous studies have focused on the association between LDH levels
and tumor angiogenesis, and it was demonstrated that high LDH-5
levels are associated with the overexpression of vascular
endothelial growth factor-A (VEGF-A) and vascular endothelial
growth factor receptor-1 (VEGFR-1), which facilitate hematogenous
metastasis and result in a poorer prognosis (52,53).
Thirdly, hypoxia mediated the overexpression of hypoxia-inducible
factor-α1 (HIF-α1), which may upregulate LDH-5 activity and, in
turn, facilitate the secretion of VEGF and angiogenesis (54–56).
However, few studies have focused on the prognostic value of serum
LDH in NSCLC. For instance, in a recent study, Koh et al
(57) revealed that a higher level of
LDHB, a subunit of LDH, was significantly associated with the level
of serum LDH and improved clinical outcomes in NSCLC. In the
present study, the pre-treatment LDH level was not correlated with
the outcomes of patients with resectable lung cancer. Neither
surgery nor adjuvant chemotherapy had significant effects on the
LDH level. Furthermore, patients with increased post-/pre-treatment
LDH ratios had better outcomes than those with not-increased LDH
ratios.
NLR is accepted as a useful and independent
predictor of gastric cancer and hepatocellular carcinoma, in
addition to early and advanced stage NSCLC (20,58–61). The
mechanisms of poor outcomes that are associated with a high NLR
value remain under investigation. Firstly, a high NLR reflects a
relative increase outcomes that count and/or lymphopenia. An
increased neutrophil response facilitates tumor growth and
metastasis by inhibiting the function of the cytotoxic lymphocytes
and remodeling the tumor extracellular matrix (62). Secondly, granulocyte colony
stimulating factor (GCSF) derived from malignant cells could
increase the level of circulating leukocytes, which may further
inhibit the activation of cytotoxic lymphocytes, weaken
immune-surveillance, remodel the tumor extracellular matrix and
promote tumor progression (62–65).
Furthermore, lymphocytes are responsible for the adaptive immune
response and serve a crucial antitumor role in immunological
surveillance and immunoediting (66,67).
Therefore, a relative decrease in lymphocytes may also lead to an
increased NLR and promote neutrophil-associated inhibition of
antitumor cytotoxic lymphocytes. These mechanisms contribute toward
increased neutrophils and decreased lymphocytes, which eventually
leads to a higher NLR level and poorer survival, suggesting that
NLR could be a prognostic indicator. Dirican et al (68) concluded that a high level of NLR was
associated with poorer outcomes in patients with NSCLC (68). The present study revealed that neither
surgery nor adjuvant chemotherapy had significant effects on serum
NLR levels, while a whole course of treatment significantly
decreased the level of NLR. However, neither the pre-treatment
level nor the post-/pre-treatment ratio of NLR had an impact on
OS.
As an indicator of systemic inflammation, PLR has
been demonstrated to be a prognostic indicator in resectable lung
cancer. Platelets facilitate tumor growth by promoting tumor
angiogenesis via the secretion of several types of cytokines and
chemokines, including vascular endothelial growth factors.
Furthermore, an adhesion molecule from platelets directly binds to
malignant cells and facilitates tumor metastasis (69–71).
Additionally, platelets are proposed to protect tumor cells from
immunological elimination and serve a negative role in the host
immune attack against tumor cells, as well as in restraining the
cytolytic activity of natural killer cells (72,73). For
example, Yuan et al (17)
concluded that high PLR levels (>204.00) indicated a poorer
prognosis in patients with NSCLC (17). Similarly, Toda et al (18) indicated that increased post-operative
PLR predicted a poorer prognosis in patients with NSCLC,
particularly in those who received adjuvant chemotherapy (14). In the present study, the baseline PLR
value was not associated with OS, while an increased
post-/pre-treatment ratio of PLR was associated with poorer
outcomes in patients with resectable lung cancer. Multivariate
analysis revealed that an increased post-/pre-treatment PLR ratio
was an independent risk factor affecting OS. In addition, treatment
had no significant impact on PLR levels.
The present study investigated the predictive values
of CRP, ALB, GLB, LDH, NLR and PLR in patients with resectable lung
cancer and concluded that patients with low pre-treatment ALB
levels and increased post-/pre-treatment PLR ratios following whole
course treatment had poorer outcomes, and a low pre-treatment ALB
level and increased post-/pre-treatment ratio of PLR were
independent risk factors for OS. Since ALB and PLR are inexpensive
and easily accessible indicators, they can be easily incorporated
into routine use as prognostic factors, combined with tumor markers
and imaging examination. However, the present study has a number of
limitations. For example, insufficient sample size was attributed
to limited manpower and material resources. In order to eliminate
the difference in general performance status of patients, those
with coexisting diseases, including chronic infection, rheumatic
diseases and other chronic inflammatory diseases, were excluded,
and only 101 samples were included. Furthermore, the data were
obtained from a single center, and the duration of follow-up was
relatively short.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant nos. 81472199, 81472296,
81602091, 81402176, 81402093, 81272542 and 81200369), the Six Major
Talent Peak Project of Jiangsu Province (grant no. 2015-WSN-022),
the Project of Invigorating Health Care through Science, Technology
and Education, Jiangsu Provincial Medical Youth Talent (grant no.
QNRC2016709), the Project of Jiangsu Provincial Commission of
Health and Family Planning (grant no. H201518), the Science and
Education for Health Foundation of Suzhou for Youth (grant nos.
kjxw2015002 and kjxw2015003), the Science and Technology Foundation
of Suzhou Xiangcheng (grant nos. XJ201642 and XJ201702) and the
Science and Technology Project Foundation of Suzhou (grant nos.
SYS201464 and SYS201504).
Availability of data and materials
The datasets used or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WL and MYW made substantial contributions to the
conception and design of the work; WJW, RR, MDX and KC revised the
manuscript critically for important intellectual content and
acquired the data; JZ, LL, WD, FRG, MT, and QZ analyzed and
interpreted the data for the present study. All authors have read
and approved the final version of the manuscript.
Ethics statement and consent to
participate
The present study was approved by the Medical Ethics
Committees of The First Affiliated Hospital of Soochow University
(Suzhou, China). Written informed consent was obtained from all
patients.
Patient consent for publication
All patients provided consent for the publication of
their data and associated images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hu J, Qian GS and Bai CX: Lung Cancer
Study Group of Chinese Thoracic Society and Chinese Alliance
Against Lung Cancer Expert Group: Chinese consensus on early
diagnosis of primary lung cancer (2014 version). Cancer. 17 Suppl
121:3157–3164. 2015. View Article : Google Scholar
|
|
3
|
Chen W, Zheng R, Zhang S, Zeng H, Xia C,
Zuo T, Yang Z, Zou X and He J: Cancer incidence and mortality in
China, 2013. Cancer Lett. 401:63–71. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Huang JY, Jian ZH, Nfor ON, Ku WY, Ko PC,
Lung CC, Ho CC, Pan HH, Huang CY, Liang YC and Liaw YP: The effects
of pulmonary diseases on histologic types of lung cancer in both
sexes: A population-based study in Taiwan. BMC Cancer. 15:8342015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Torre LA, Siegel RL, Ward EM and Jemal A:
Global cancer incidence and mortality rates and trends--an update.
Cancer Epidemiol Biomarkers Prev. 25:16–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li S, Lan X, Gao H, Li Z, Chen L, Wang W,
Song S, Wang Y, Li C, Zhang H and Xue Y: Systemic Inflammation
Response Index (SIRI), cancer stem cells and survival of localised
gastric adenocarcinoma after curative resection. J Cancer Res Clin
Oncol. 143:2455–2468. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tong YS, Tan J, Zhou XL, Song YQ and Song
YJ: Systemic immune-inflammation index predicting chemoradiation
resistance and poor outcome in patients with stage III non-small
cell lung cancer. J Transl Med. 15:2212017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Feng JF, Chen S and Yang X: Systemic
immune-inflammation index (SII) is a useful prognostic indicator
for patients with squamous cell carcinoma of the esophagus.
Medicine (Baltimore). 96:e58862017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhong JH, Huang DH and Chen ZY: Prognostic
role of systemic immune-inflammation index in solid tumors: A
systematic review and meta-analysis. Oncotarget. 8:75381–75388.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Han LH, Jia YB, Song QX, Wang JB, Wang NN
and Cheng YF: Prognostic significance of preoperative
lymphocyte-monocyte ratio in patients with resectable esophageal
squamous cell carcinoma. Asian Pac J Cancer Prev. 16:2245–2250.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu X, Meng QH, Ye Y, Hildebrandt MA, Gu J
and Wu X: Prognostic significance of pretreatment serum levels of
albumin, LDH and total bilirubin in patients with non-metastatic
breast cancer. Carcinogenesis. 36:243–248. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tas F, Aydiner A, Demir C and Topuz E:
Serum lactate dehydrogenase levels at presentation predict outcome
of patients with limited-stage small-cell lung cancer. Am J Clin
Oncol. 24:376–378. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gabay C and Kushner I: Acute-phase
proteins and other systemic responses to inflammation. N Engl J
Med. 340:448–454. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Groblewska M, Mroczko B,
Wereszczynska-Siemiatkowska U, Kedra B, Lukaszewicz M, Baniukiewicz
A and Szmitkowski M: Serum interleukin 6 (IL-6) and C-reactive
protein (CRP) levels in colorectal adenoma and cancer patients.
Clin Chem Lab Med. 46:1423–1428. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gozlan Y, Ben-Ari Z, Moscona R, Shirazi R,
Rakovsky A, Kabat A, Veizman E, Berdichevski T, Weiss P, Cohen-Ezra
O, et al: HCV genotype-1 subtypes and resistance-associated
substitutions in drug-naive and in direct-acting antiviral
treatment failure patients. Antivir Ther. 22:431–441. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jurisic V, Radenkovic S and Konjevic G:
The actual role of LDH as tumor marker, biochemical and clinical
aspects. Adv Exp Med Biol. 867:115–124. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yuan C, Li N, Mao X, Liu Z, Ou W and Wang
SY: Elevated pretreatment neutrophil/white blood cell ratio and
monocyte/lymphocyte ratio predict poor survival in patients with
curatively resected non-small cell lung cancer: Results from a
large cohort. Thorac Cancer. 8:350–358. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Toda M, Tsukioka T, Izumi N, Komatsu H,
Okada S, Hara K, Miyamoto H, Ito R, Shibata T and Nishiyama N:
Platelet-to-lymphocyte ratio predicts the prognosis of patients
with non-small cell lung cancer treated with surgery and
postoperative adjuvant chemotherapy. Thorac Cancer. 9:112–119.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lorente D, Mateo J, Templeton AJ,
Zafeiriou Z, Bianchini D, Ferraldeschi R, Bahl A, Shen L, Su Z,
Sartor O and de Bono JS: Baseline neutrophil-lymphocyte ratio (NLR)
is associated with survival and response to treatment with
second-line chemotherapy for advanced prostate cancer independent
of baseline steroid use. Ann Oncol. 26:750–755. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shen L, Zhang H, Liang L, Li G, Fan M, Wu
Y, Zhu J and Zhang Z: Baseline neutrophil-lymphocyte ratio
(>/=2.8) as a prognostic factor for patients with locally
advanced rectal cancer undergoing neoadjuvant chemoradiation.
Radiat Oncol. 9:2952014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Edge SB and Compton CC: The american joint
committee on cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Diakos CI, Charles KA, McMillan DC and
Clarke SJ: Cancer-related inflammation and treatment effectiveness.
Lancet Oncol. 15:e493–503. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Proctor MJ, Morrison DS, Talwar D, Balmer
SM, O'Reilly DS, Foulis AK, Horgan PG and McMillan DC: An
inflammation-based prognostic score (mGPS) predicts cancer survival
independent of tumour site: A glasgow inflammation outcome study.
Br J Cancer. 104:726–734. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wilson DO, Weissfeld JL, Balkan A,
Schragin JG, Fuhrman CR, Fisher SN, Wilson J, Leader JK, Siegfried
JM, Shapiro SD and Sciurba FC: Association of radiographic
emphysema and airflow obstruction with lung cancer. Am J Respir
Crit Care Med. 178:738–744. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wasswa-Kintu S, Gan WQ, Man SF, Pare PD
and Sin DD: Relationship between reduced forced expiratory volume
in one second and the risk of lung cancer: A systematic review and
meta-analysis. Thorax. 60:570–575. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ng Kee Kwong F, Nicholson AG, Harrison CL,
Hansbro PM, Adcock IM and Chung KF: Is mitochondrial dysfunction a
driving mechanism linking COPD to nonsmall cell lung carcinoma? Eur
Respir Rev. 26:1700402017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dye JA and Adler KB: Effects of cigarette
smoke on epithelial cells of the respiratory tract. Thorax.
49:825–834. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sopori M: Effects of cigarette smoke on
the immune system. Nat Rev Immunol. 2:372–377. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Houghton AM: Mechanistic links between
COPD and lung cancer. Nat Rev Cancer. 13:233–245. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Marnett LJ: Oxyradicals and DNA damage.
Carcinogenesis. 21:361–370. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gu X, Sun S, Gao XS, Xiong W, Qin S, Qi X,
Ma M, Li X, Zhou D, Wang W and Yu H: Prognostic value of platelet
to lymphocyte ratio in non-small cell lung cancer: Evidence from
3,430 patients. Sci Rep. 6:238932016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Koh CH, Bhoo-Pathy N, Ng KL, Jabir RS, Tan
GH, See MH, Jamaris S and Taib NA: Utility of pre-treatment
neutrophil-lymphocyte ratio and platelet-lymphocyte ratio as
prognostic factors in breast cancer. Br J Cancer. 113:150–158.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li Y, Wang C, Xu M, Kong C, Qu A, Zhang M,
Zheng Z and Zhang G: Preoperative NLR for predicting survival rate
after radical resection combined with adjuvant immunotherapy with
CIK and postoperative chemotherapy in gastric cancer. J Cancer Res
Clin Oncol. 143:861–871. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hara M, Matsuzaki Y, Shimuzu T, Tomita M,
Ayabe T, Enomoto Y and Onitsuka T: Preoperative serum C-reactive
protein level in non-small cell lung cancer. Anticancer Res.
27:3001–3004. 2007.PubMed/NCBI
|
|
37
|
Lee JG, Cho BC, Bae MK, Lee CY, Park IK,
Kim DJ, Ahn SV and Chung KY: Preoperative C-reactive protein levels
are associated with tumor size and lymphovascular invasion in
resected non-small cell lung cancer. Lung Cancer. 63:106–110. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Alifano M, Falcoz PE, Seegers V, Roche N,
Schussler O, Younes M, Antonacci F, Forgez P, Dechartres A, Massard
G, et al: Preresection serum C-reactive protein measurement and
survival among patients with resectable non-small cell lung cancer.
J Thorac Cardiovasc Surg. 142:1161–1167. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yeun JY and Kaysen GA: Factors influencing
serum albumin in dialysis patients. Am J kidney Dis. 32 Suppl
4:S118–S125. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Okumura H, Uchikado Y, Setoyama T,
Matsumoto M, Owaki T, Ishigami S and Natsugoe S: Biomarkers for
predicting the response of esophageal squamous cell carcinoma to
neoadjuvant chemoradiation therapy. Surg Today. 44:421–428. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lu CY, Uen YH, Tsai HL, Chuang SC, Hou MF,
Wu DC, Juo SH, Lin SR and Wang JY: Molecular detection of
persistent postoperative circulating tumour cells in stages II and
III colon cancer patients via multiple blood sampling: Prognostic
significance of detection for early relapse. Br J Cancer.
104:1178–1184. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gupta D and Lis CG: Pretreatment serum
albumin as a predictor of cancer survival: A systematic review of
the epidemiological literature. Nutr J. 9:692010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Tolia M, Tsoukalas N, Kyrgias G, Mosa E,
Maras A, Kokakis I, Liakouli Z, Kouvaris JR, Liaskonis K,
Charalampakis N, et al: Prognostic significance of serum
inflammatory response markers in newly diagnosed non-small cell
lung cancer before chemoirradiation. BioMed Res Int.
2015:4857322015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jin Y, Zhao L and Peng F: Prognostic
impact of serum albumin levels on the recurrence of stage I
non-small cell lung cancer. Clinics (Sao Paulo). 68:686–693. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen J, Zhou Y, Xu Y, Zhu HY and Shi YQ:
Low pretreatment serum globulin may predict favorable prognosis for
gastric cancer patients. Tumour Biol. 37:3905–3911. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Qu X, Pang Z, Yi W, Wang Y, Wang K, Liu Q
and Du J: High percentage of α1-globulin in serum protein is
associated with unfavorable prognosis in non-small cell lung
cancer. Med Oncol. 31:2382014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Higashiyama M, Doi O, Kodama K, Yokouchi H
and Tateishi R: An evaluation of the prognostic significance of
alpha-1-antitrypsin expression in adenocarcinomas of the lung: An
immunohistochemical analysis. Br J Cancer. 65:300–302. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tas F, Karabulut S, Ciftci R, Sen F, Sakar
B, Disci R and Duranyildiz D: Serum levels of LDH, CEA, and CA19-9
have prognostic roles on survival in patients with metastatic
pancreatic cancer receiving gemcitabine-based chemotherapy. Cancer
Chemother Pharmacol. 73:1163–1171. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yin C, Jiang C, Liao F, Rong Y, Cai X, Guo
G, Qiu H, Chen X, Zhang B, He W and Xia L: Initial LDH level can
predict the survival benefit from bevacizumab in the first-line
setting in chinese patients with metastatic colorectal cancer.
OncoTargets Ther. 7:1415–1422. 2014. View Article : Google Scholar
|
|
50
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the warburg effect: The metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sun X, Sun Z, Zhu Z, Guan H, Zhang J,
Zhang Y, Xu H and Sun M: Clinicopathological significance and
prognostic value of lactate dehydrogenase A expression in gastric
cancer patients. PLoS One. 9:e910682014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Harris AL: Hypoxia-a key regulatory factor
in tumour growth. Nat Rev Cancer. 2:38–47. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Azuma M, Shi M, Danenberg KD, Gardner H,
Barrett C, Jacques CJ, Sherod A, Iqbal S, El-Khoueiry A, Yang D, et
al: Serum lactate dehydrogenase levels and glycolysis significantly
correlate with tumor VEGFA and VEGFR expression in metastatic CRC
patients. Pharmacogenomics. 8:1705–1713. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Koukourakis MI, Giatromanolaki A,
Simopoulos C, Polychronidis A and Sivridis E: Lactate dehydrogenase
5 (LDH5) relates to up-regulated hypoxia inducible factor pathway
and metastasis in colorectal cancer. Clin Exp Metastasis. 22:25–30.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Toffoli S and Michiels C: Intermittent
hypoxia is a key regulator of cancer cell and endothelial cell
interplay in tumours. FEBS J. 275:2991–3002. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lu H, Forbes RA and Verma A:
Hypoxia-inducible factor 1 activation by aerobic glycolysis
implicates the Warburg effect in carcinogenesis. J Biol Chem.
277:23111–23115. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Koh YW, Lee SJ and Park SY: Prognostic
significance of lactate dehydrogenase B according to histologic
type of non-small-cell lung cancer and its association with serum
lactate dehydrogenase. Pathol Res Pract. 213:1134–1138. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Harimoto N, Shirabe K, Nakagawara H,
Toshima T, Yamashita Y, Ikegami T, Yoshizumi T, Soejima Y, Ikeda T
and Maehara Y: Prognostic factors affecting survival at recurrence
of hepatocellular carcinoma after living-donor liver
transplantation: with special reference to neutrophil/lymphocyte
ratio. Transplantation. 96:1008–1012. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Tanaka N, Kikuchi E, Kanao K, Matsumoto K,
Shirotake S, Miyazaki Y, Kobayashi H, Kaneko G, Hagiwara M, Ide H,
et al: A multi-institutional validation of the prognostic value of
the neutrophil-to-lymphocyte ratio for upper tract urothelial
carcinoma treated with radical nephroureterectomy. Ann surg Oncol.
21:4041–4048. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Jin H, Zhang G, Liu X, Liu X, Chen C, Yu
H, Huang X, Zhang Q and Yu J: Blood neutrophil-lymphocyte ratio
predicts survival for stages III–IV gastric cancer treated with
neoadjuvant chemotherapy. World J Surg Oncol. 11:1122013.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Choi JE, Villarreal J, Lasala J,
Gottumukkala V, Mehran RJ, Rice D, Yu J, Feng L and Cata JP:
Perioperative neutrophil:Lymphocyte ratio and postoperative NSAID
use as predictors of survival after lung cancer surgery: A
retrospective study. Cancer Med. 4:825–833. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Goubran HA, Burnouf T, Radosevic M and
El-Ekiaby M: The platelet-cancer loop. Eur J Intern Med.
24:393–400. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Jeong E, Hyun SH, Moon SH, Cho YS, Kim BT
and Lee KH: Relation between tumor FDG uptake and hematologic
prognostic indicators in stage I lung cancer patients following
curative resection. Medicine (Baltimore). 96:e59352017. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Sanchez-Salcedo P, de-Torres JP,
Martinez-Urbistondo D, Gonzalez-Gutierrez J, Berto J, Campo A,
Alcaide AB and Zulueta JJ: The neutrophil to lymphocyte and
platelet to lymphocyte ratios as biomarkers for lung cancer
development. Lung Cancer. 97:28–34. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Nikolic I, Kukulj S, Samaržija M, Jeleč V,
Žarak M, Orehovec B, Taradi I, Romić D, Kolak T and Patrlj L:
Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio help
identify patients with lung cancer, but do not differentiate
between lung cancer subtypes. Croat Med J. 57:287–292. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Dunn GP, Old LJ and Schreiber RD: The
immunobiology of cancer immunosurveillance and immunoediting.
Immunity. 21:137–148. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ohtani H: Focus on TILs: Prognostic
significance of tumor infiltrating lymphocytes in human colorectal
cancer. Cancer Immun. 7:42007.PubMed/NCBI
|
|
68
|
Dirican N, Dirican A, Anar C, Atalay S,
Ozturk O, Bircan A, Akkaya A and Cakir M: A new inflammatory
prognostic index, based on C-reactive protein, the neutrophil to
lymphocyte ratio and serum albumin is useful for predicting
prognosis in non-small cell lung cancer cases. Asian Pac J Cancer
Prev. 17:5101–5106. 2016.PubMed/NCBI
|
|
69
|
Suzuki K, Aiura K, Ueda M and Kitajima M:
The influence of platelets on the promotion of invasion by tumor
cells and inhibition by antiplatelet agents. Pancreas. 29:132–140.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Sabrkhany S, Griffioen AW and Oude Egbrink
MG: The role of blood platelets in tumor angiogenesis. Biochim
Biophys Acta. 1815:189–196. 2011.PubMed/NCBI
|
|
71
|
Kim YJ, Borsig L, Varki NM and Varki A:
P-selectin deficiency attenuates tumor growth and metastasis. Proc
Natl Acad Sci U S A. 95:9325–9330. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Nieswandt B, Hafner M, Echtenacher B and
Mannel DN: Lysis of tumor cells by natural killer cells in mice is
impeded by platelets. Cancer Res. 59:1295–1300. 1999.PubMed/NCBI
|
|
73
|
Maini MK and Schurich A: Platelets harness
the immune response to drive liver cancer. Proc Natl Acad Sci U S
A. 109:12840–12841. 2012. View Article : Google Scholar : PubMed/NCBI
|