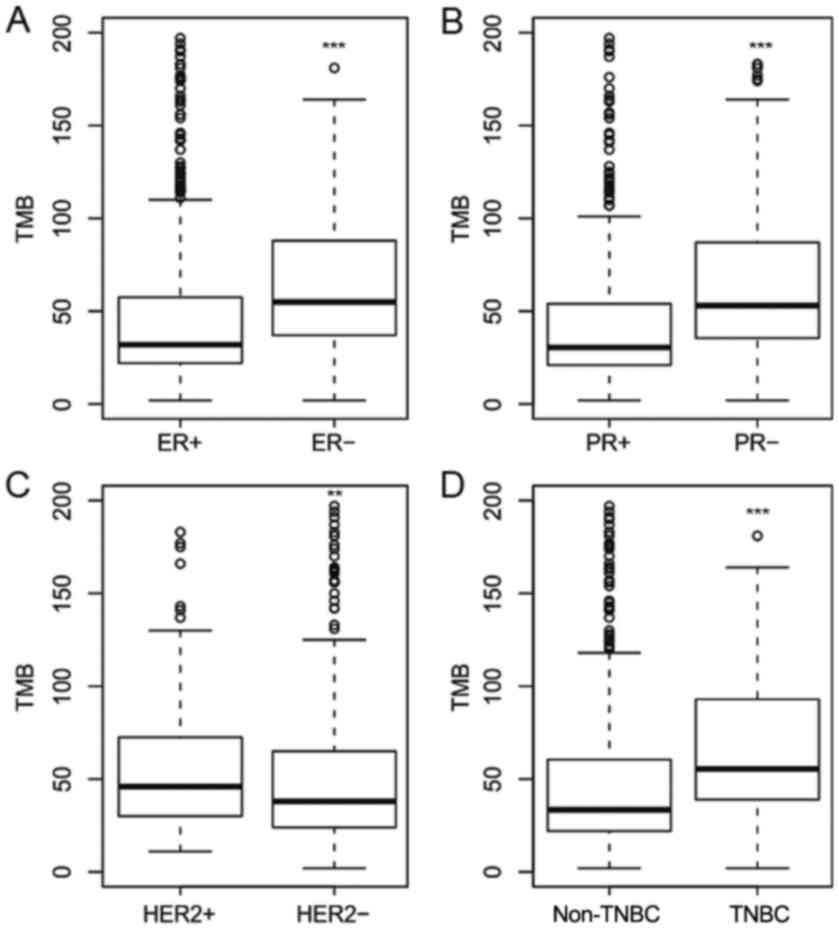
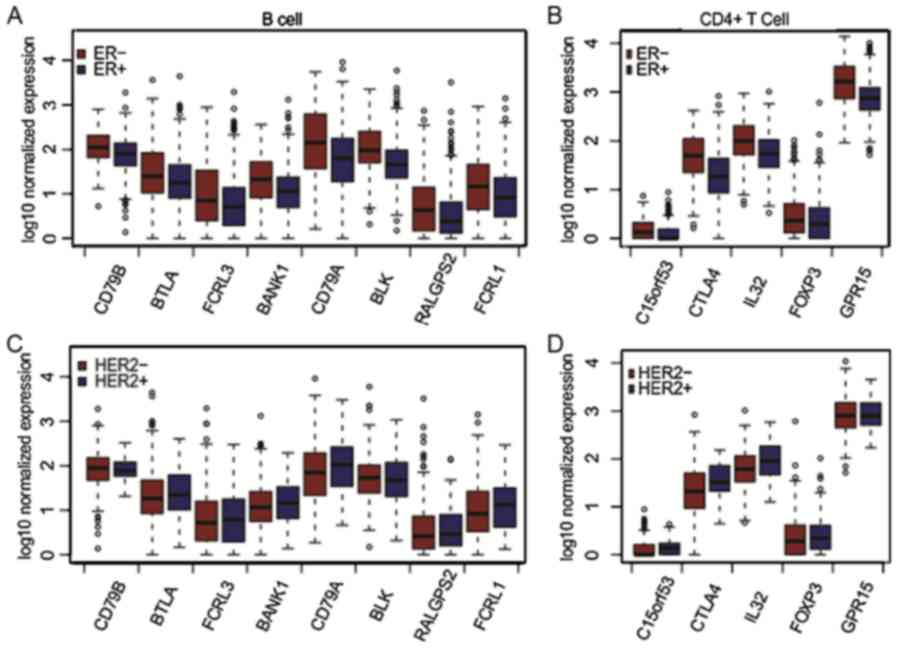
|
1
|
DeSantis CE, Ma J, Goding Sauer A, Newman
LA and Jemal A: Breast cancer statistics, 2017, racial disparity in
mortality by state. CA Cancer J Clin. 67:439–448. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Effi AB, Aman NA, Koui BS, Koffi KD,
Traore ZC and Kouyate M: Breast cancer molecular subtypes defined
by ER/PR and HER2 status: Association with clinicopathologic
parameters in ivorian patients. Asian Pac J Cancer Prev.
17:1973–1978. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dunnwald LK, Rossing MA and Li CI: Hormone
receptor status, tumor characteristics, and prognosis: A
prospective cohort of breast cancer patients. Breast Cancer Res.
9:R62007. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Davis SL, Eckhardt SG, Tentler JJ and
Diamond JR: Triple-negative breast cancer: Bridging the gap from
cancer genomics to predictive biomarkers. Ther Adv Med Oncol.
6:88–100. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nanda R, Chow LQ, Dees EC, Berger R, Gupta
S, Geva R, Pusztai L, Pathiraja K, Aktan G, Cheng JD, et al:
Pembrolizumab in patients with advanced triple-negative breast
cancer: Phase Ib KEYNOTE-012 study. J Clin Oncol. 34:2460–2467.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hartman ZC, Poage GM, den Hollander P,
Tsimelzon A, Hill J, Panupinthu N, Zhang Y, Mazumdar A, Hilsenbeck
SG, Mills GB and Brown PH: Growth of triple-negative breast cancer
cells relies upon coordinate autocrine expression of the
proinflammatory cytokines IL-6 and IL-8. Cancer Res. 73:3470–3480.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Adams S, Schmid P, Rugo HS, Winer EP,
Loirat D, Awada A, Cescon DW, Iwata H, Campone M, Nanda R, et al:
Pembrolizumab monotherapy for previously treated metastatic
triple-negative breast cancer: Cohort A of the phase II KEYNOTE-086
study. Ann Oncol. 30:397–404. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Adams S, Loi S, Toppmeyer D, Cescon DW, De
Laurentiis M, Nanda R, Winer EP, Mukai H, Tamura K, Armstrong A, et
al: Pembrolizumab monotherapy for previously untreated,
PD-L1-positive, metastatic triple-negative breast cancer: Cohort B
of the phase II KEYNOTE-086 study. Ann Oncol. 30:405–411. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schmid P, Adams S, Rugo HS, Schneeweiss A,
Barrios CH, Iwata H, Diéras V, Hegg R, Im SA, Shaw Wright G, et al:
Atezolizumab and nab-paclitaxel in advanced triple-negative breast
cancer. N Engl J Med. 379:2108–2121. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Criscitiello C and Curigliano G:
Immunotherapy of breast cancer. Prog Tumor Res. 42:30–43. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rummel SK, Lovejoy L, Shriver CD and
Ellsworth RE: Contribution of germline mutations in cancer
predisposition genes to tumor etiology in young women diagnosed
with invasive breast cancer. Breast Cancer Res Treat. 164:593–601.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chalmers ZR, Connelly CF, Fabrizio D, Gay
L, Ali SM, Ennis R, Schrock A, Campbell B, Shlien A, Chmielecki J,
et al: Analysis of 100,000 human cancer genomes reveals the
landscape of tumor mutational burden. Genome Med. 9:342017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tomczak K, Czerwińska P and Wiznerowicz M:
The cancer genome atlas (TCGA): An immeasurable source of
knowledge. Contemp Oncol (Pozn). 19:A68–A77. 2015.PubMed/NCBI
|
|
14
|
Boland CR and Goel A: Microsatellite
instability in colorectal cancer. Gastroenterology. 138:2073–2087,
e2073. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rooney MS, Shukla SA, Wu CJ, Getz G and
Hacohen N: Molecular and genetic properties of tumors associated
with local immune cytolytic activity. Cell. 160:48–61. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Danaher P, Warren S, Dennis L, D'Amico L,
White A, Disis ML, Geller MA, Odunsi K, Beechem J and Fling SP:
Gene expression markers of Tumor Infiltrating Leukocytes. J
Immunother Cancer. 5:182017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Low SK, Zembutsu H and Nakamura Y: Breast
cancer: The translation of big genomic data to cancer precision
medicine. Cancer Sci. 109:497–506. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kraya AA, Maxwell KN, Wubbenhorst B, Wenz
BM, Pluta J, Rech AJ, Dorfman LM, Lunceford N, Barrett A, Mitra N,
et al: Genomic signatures predict the immunogenicity of
BRCA-deficient breast cancer. Clin Cancer Res.
pii:clincanres.0468.2018. 2019.
|
|
19
|
Stovgaard ES, Nielsen D, Hogdall E and
Balslev E: Triple negative breast cancer-prognostic role of
immune-related factors: A systematic review. Acta Oncol. 57:74–82.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang J, Xu J, E Y and Sun T: Predictive
and prognostic value of circulating blood lymphocyte subsets in
metastatic breast cancer. Cancer Med. 8:492–500. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu J, Jiang L, Cao H, Jia Y, Wu S, Jiang C
and Sun T: Predictive value of CD4+/CD8+
ratio in patients with breast cancer receiving recombinant human
thrombopoietin. J Interferon Cytokine Res. 38:213–220. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
McGrail DJ, Federico L, Li Y, Dai H, Lu Y,
Mills GB, Yi S, Lin SY and Sahni N: Multi-omics analysis reveals
neoantigen-independent immune cell infiltration in copy-number
driven cancers. Nat Commun. 9:13172018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shaw JA, Guttery DS, Hills A,
Fernandez-Garcia D, Page K, Rosales BM, Goddard KS, Hastings RK,
Luo J, Ogle O, et al: Mutation analysis of cell-free DNA and single
circulating tumor cells in metastatic breast cancer patients with
high circulating tumor cell counts. Clin Cancer Res. 23:88–96.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bidard FC, Hajage D, Bachelot T, Delaloge
S, Brain E, Campone M, Cottu P, Beuzeboc P, Rolland E, Mathiot C
and Pierga JY: Assessment of circulating tumor cells and serum
markers for progression-free survival prediction in metastatic
breast cancer: A prospective observational study. Breast Cancer
Res. 14:R292012. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xu J, Sun T, Guo X, Wang Y and Jing M:
Estrogen receptor-α promoter methylation is a biomarker for outcome
prediction of cisplatin resistance in triple-negative breast
cancer. Oncol Lett. 15:2855–2862. 2018.PubMed/NCBI
|
|
26
|
Birkbak NJ, Kochupurakkal B, Izarzugaza
JM, Eklund AC, Li Y, Liu J, Szallasi Z, Matulonis UA, Richardson
AL, Iglehart JD and Wang ZC: Tumor mutation burden forecasts
outcome in ovarian cancer with BRCA1 or BRCA2 mutations. PLoS One.
8:e800232013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xu J, Guo X, Jing M and Sun T: Prediction
of tumor mutation burden in breast cancer based on the expression
of ER, PR, HER-2, and Ki-67. Onco Targets Ther. 11:2269–2275. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Win AK, Lindor NM and Jenkins MA: Risk of
breast cancer in Lynch syndrome: A systematic review. Breast Cancer
Res. 15:R272013. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kappil M, Terry MB, Delgado-Cruzata L,
Liao Y and Santella RM: Mismatch repair polymorphisms as markers of
breast cancer prevalence in the breast cancer family registry.
Anticancer Res. 36:4437–4441. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Loi S, Giobbie-Hurder A, Gombos A,
Bachelot T, Hui R, Curigliano G, Campone M, Biganzoli L, Bonnefoi
H, Jerusalem G, et al: Pembrolizumab plus trastuzumab in
trastuzumab-resistant, advanced, HER2-positive breast cancer
(PANACEA): A single-arm, multicentre, phase 1b-2 trial. Lancet
Oncol. 20:371–382. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rugo HS, Delord JP, Im SA, Ott PA,
Piha-Paul SA, Bedard PL, Sachdev J, Tourneau CL, van Brummelen EMJ,
Varga A, et al: Safety and antitumor activity of pembrolizumab in
patients with estrogen receptor-positive/human epidermal growth
factor receptor 2-negative advanced breast cancer. Clin Cancer Res.
24:2804–2811. 2018. View Article : Google Scholar : PubMed/NCBI
|