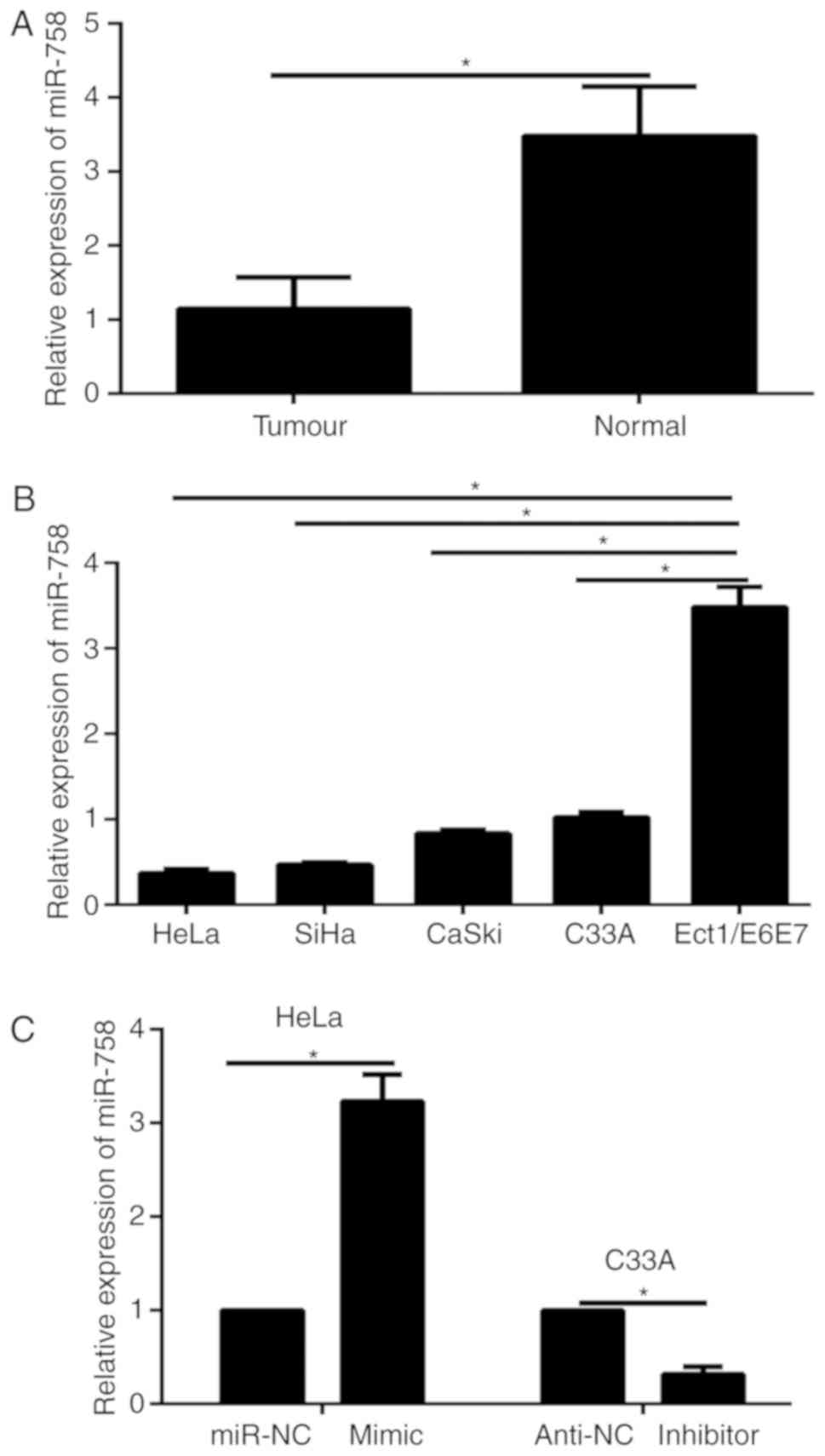
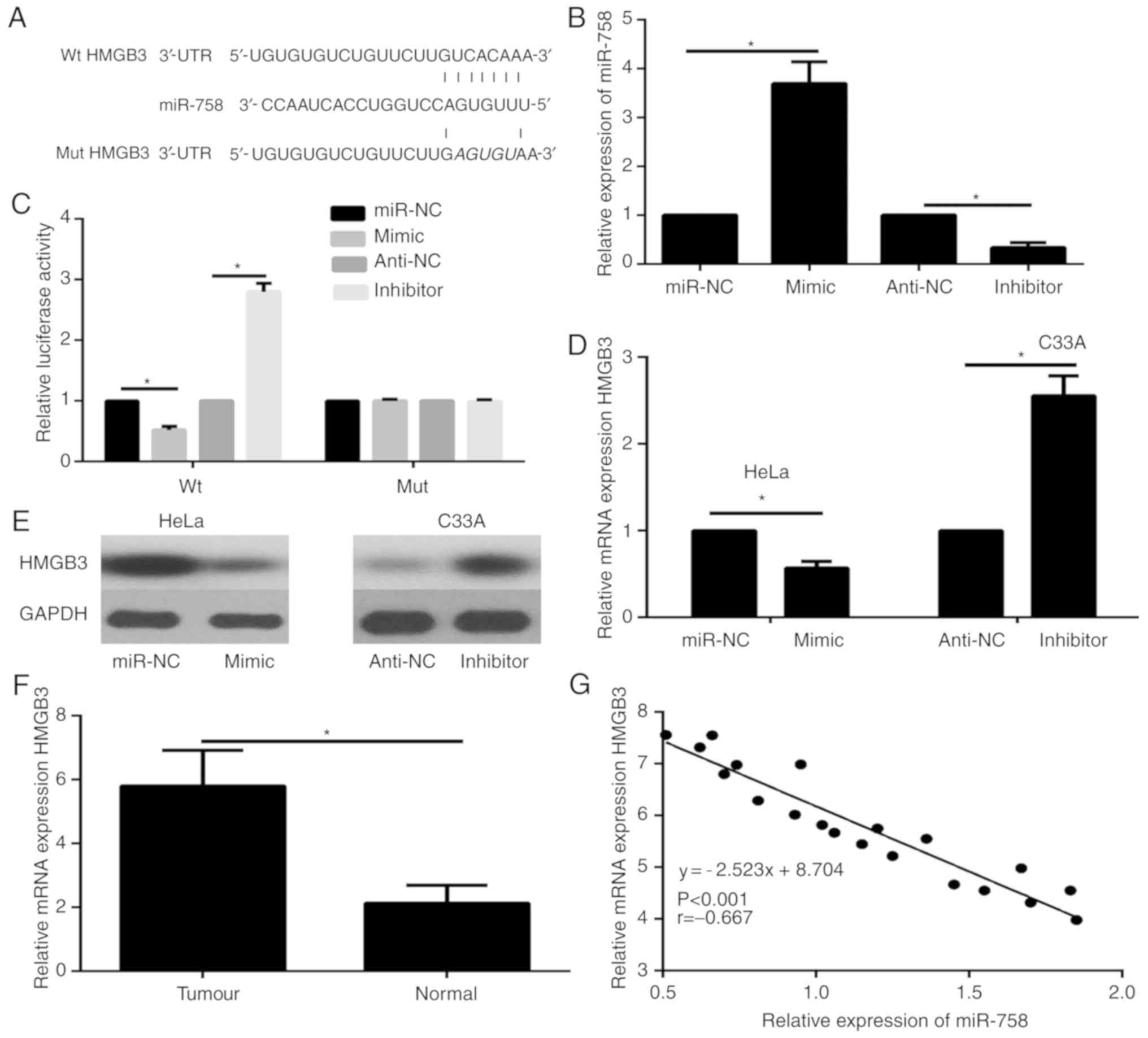
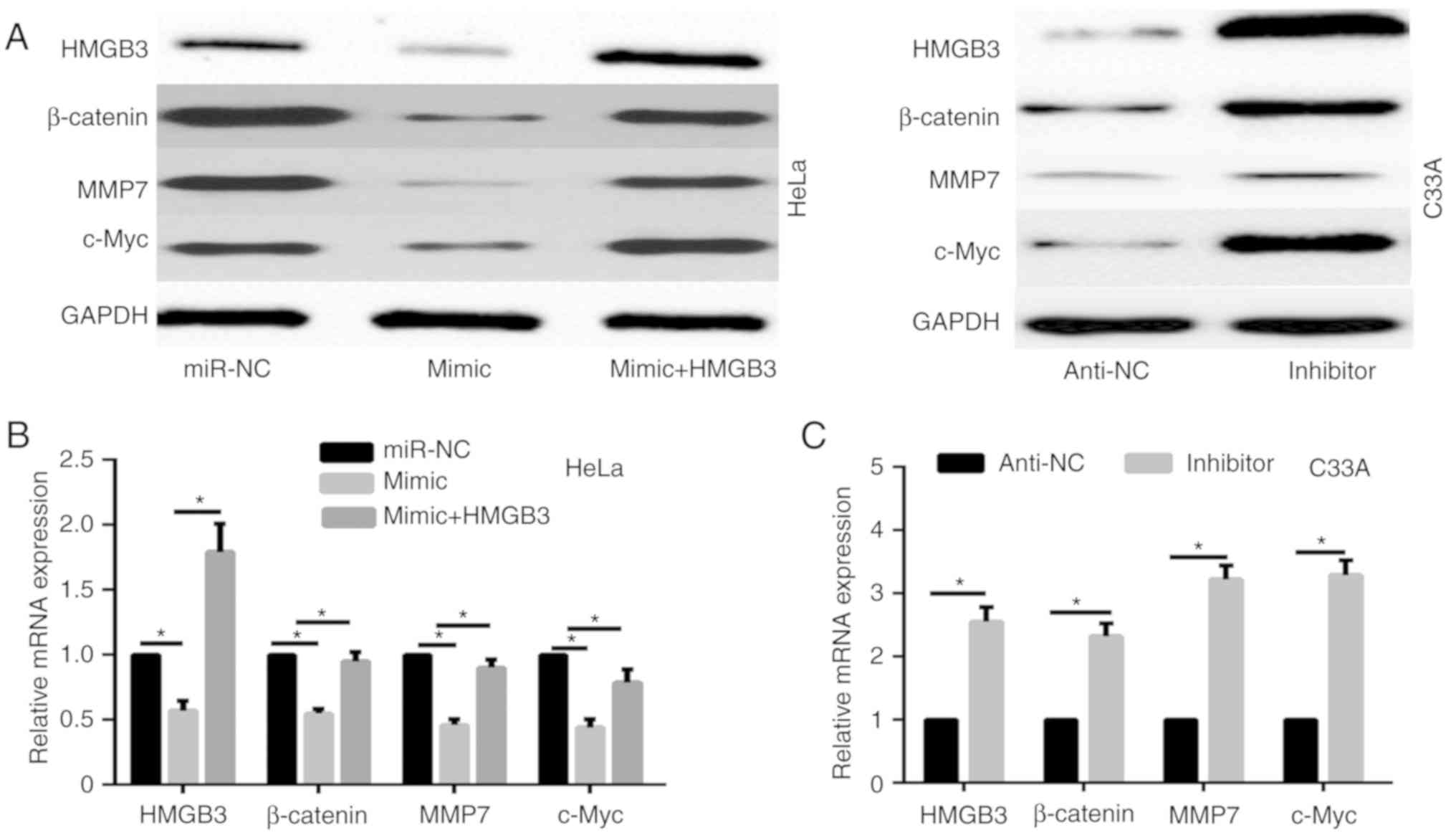
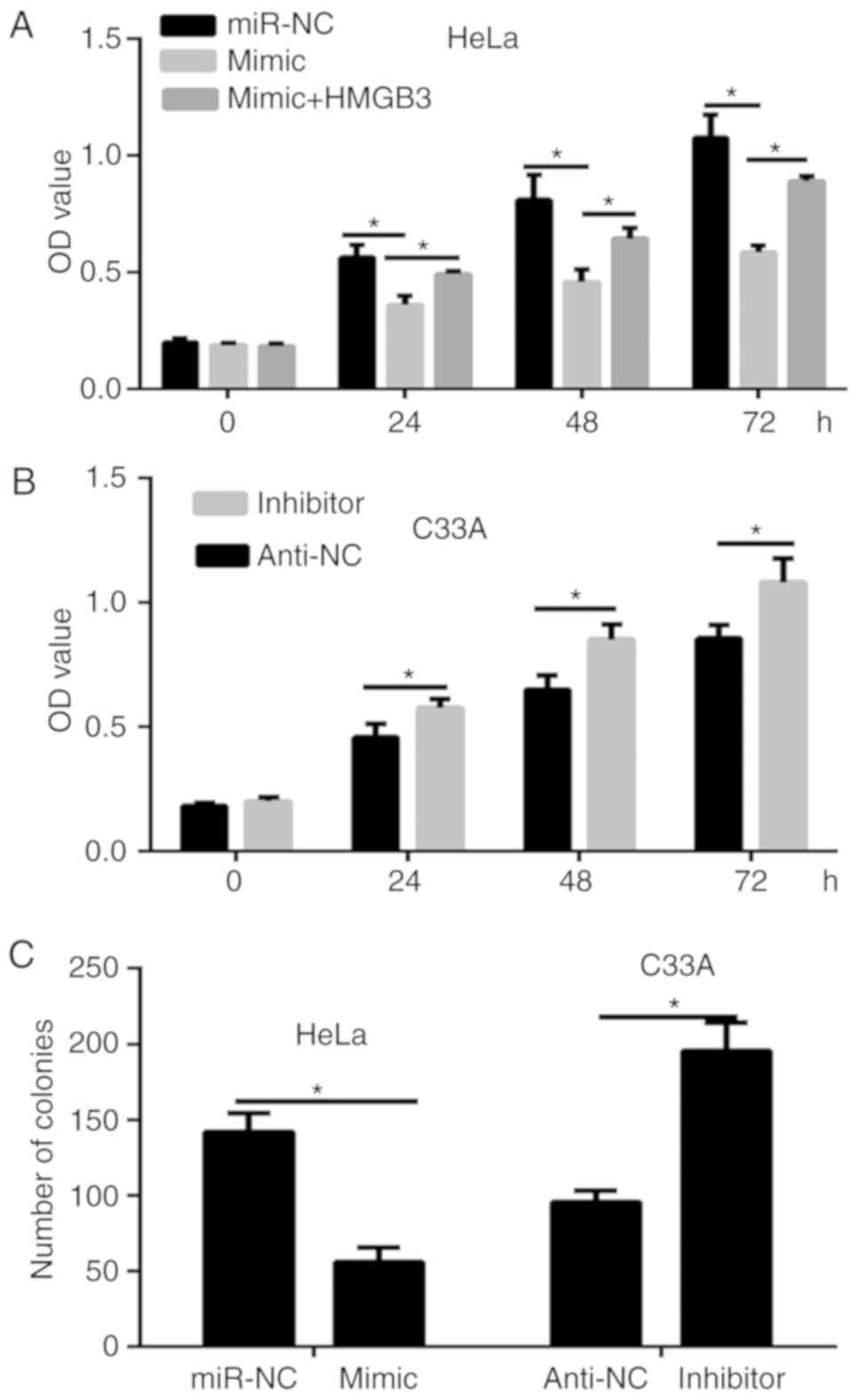
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Smith RA, Brooks D, Cokkinides V, Saslow D
and Brawley OW: Cancer screening in the United States, 2013: A
review of current American cancer society guidelines, current
issues in cancer screening, and new guidance on cervical cancer
screening and lung cancer screening. CA Cancer J Clin. 63:88–105.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dai S, Lu Y, Long Y, Lai Y, Du P, Ding N
and Yao D: Prognostic value of microRNAs in cervical carcinoma: A
systematic review and meta-analysis. Oncotarget. 7:35369–35378.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kanekura K, Nishi H, Isaka K and Kuroda M:
MicroRNA and gynecologic cancers. J Obstet Gynaecol Res.
42:612–617. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kwan JY, Psarianos P, Bruce JP, Yip KW and
Liu FF: The complexity of microRNAs in human cancer. J Radiat Res.
57 (Suppl 1):i106–i111. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu J, Jiang J, Hui X, Wang W, Fang D and
Ding L: Mir-758-5p suppresses glioblastoma proliferation, migration
and invasion by targeting ZBTB20. Cell Physiol Biochem.
48:2074–2083. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang S and Jiang M: The long non-coding
RNA-DANCR exerts oncogenic functions in non-small cell lung cancer
via miR-758-3p. Biomed Pharmacother. 103:94–100. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jiang D, Cho W, Li Z, Xu X, Qu Y, Jiang Z,
Guo L and Xu G: MiR-758-3p suppresses proliferation, migration and
invasion of hepatocellular carcinoma cells via targeting MDM2 and
mTOR. Biomed Pharmacother. 96:535–544. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Meng X, Zhao Y, Wang J, Gao Z, Geng Q and
Liu X: Regulatory roles of miRNA-758 and matrix extracellular
phosphoglycoprotein in cervical cancer. Exp Ther Med. 14:2789–2794.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang Z, Chang Y, Zhang J, Lu Y, Zheng L,
Hu Y, Zhang F and Li X, Zhang W and Li X: HMGB3 promotes growth and
migration in colorectal cancer by regulating WNT/β-catenin pathway.
PLoS One. 12:e01797412017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vu M, Yu J, Awolude OA and Chuang L:
Cervical cancer worldwide. Curr Probl Cancer. 42:457–465. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Nemeth MJ, Curtis DJ, Kirby MR,
Garrett-Beal LJ, Seidel NE, Cline AP and Bodine DM: Hmgb3: An
HMG-box family member expressed in primitive hematopoietic cells
that inhibits myeloid and B-cell differentiation. Blood.
102:1298–1306. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen X and Zeng L: Ginkgo biloba extract
761 enhances 5-fluorouracil chemosensitivity in colorectal cancer
cells through regulation of high mobility group-box 3 expression.
Am J Transl Res. 10:1773–1783. 2018.PubMed/NCBI
|
|
15
|
Yamada Y, Nishikawa R, Kato M, Okato A,
Arai T, Kojima S, Yamazaki K, Naya Y, Ichikawa T and Seki N:
Regulation of HMGB3 by antitumor miR-205-5p inhibits cancer cell
aggressiveness and is involved in prostate cancer pathogenesis. J
Hum Genet. 63:195–205. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao J, Zou Z, Gao J, Zhang H, Lin Z, Zhang
Y, Luo X, Liu C, Xie J and Cai C: Increased expression of HMGB3: A
novel independent prognostic marker of worse outcome in patients
with esophageal squamous cell carcinoma. Int J Clin Exp Pathol.
8:345–352. 2015.PubMed/NCBI
|
|
17
|
Song N, Liu B, Wu JL, Zhang RF, Duan L, He
WS and Zhang CM: Prognostic value of HMGB3 expression in patients
with non-small cell lung cancer. Tumour Biol. 34:2599–2603. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bahrami A, Hasanzadeh M, ShahidSales S,
Yousefi Z, Kadkhodayan S, Farazestanian M, Joudi Mashhad M, Gharib
M, Mahdi Hassanian S and Avan A: Clinical significance and
prognosis value of Wnt signaling pathway in cervical cancer. J Cell
Biochem. 118:3028–3033. 2017. View Article : Google Scholar : PubMed/NCBI
|