Introduction
Colorectal cancer (CRC) accounted for ~1.4 million
of the new cancer cases diagnosed in 2012 worldwide and is
considered the third most common cancer (1). According to the International Agency
for Research on Cancer, by 2035, the estimated number of CRC cases
will reach 2.4 million cases diagnosed each year (1). Although a lot has been learned about
CRC, novel therapeutic strategies are needed in order to tackle
this cancer. Mammalian target of rapamycin (mTOR) is a catalytic
subunit of two large signaling complexes; mTORC1 and mTORC2, along
with key proteins, form these complexes. Regulatory-associated
protein of mTOR (RPTOR) is the unique component of mTORC1, whereas
rapamycin-insensitive companion of mTOR (RICTOR) is an exclusive
component of mTORC2. The two complexes play a central role in
tumorigenesis via phosphorylation of key proteins within the mTOR
pathway. mTORC1 regulates mRNA translation and elongation by
phosphorylating its downstream effectors, such as eukaryotic
initiation factor 4E-binding protein 1 and the p70 ribosomal S6
kinase 1 (2). mTORC2 phosphorylates
protein kinase B, promoting cell proliferation, apoptosis and
survival (3).
MicroRNAs (miRNAs/miRs) are single-stranded,
non-coding RNAs, ranging from 19–24 nucleotides in length. In order
to perform their regulatory functions, mature miRNAs bind most
often to the 3′untranslated region (UTR) of messenger RNAs, which
inhibit the translation of mRNA and result in the downregulation of
gene expression at the post-transcriptional level (4). Approximately 50% of human miRNA genes
are located in genomic regions that have fragile sites, and these
locations contain chromosomal abnormalities, such as deletions and
amplifications. These genomic regions are vulnerable to genetic
alteration in several types of human cancer (5,6).
Furthermore, a single miRNA can interact and regulate more than one
mRNA target (7).
In the last decade, a growing body of evidence has
revealed that miRNAs are involved in tumorigenesis and cancer
progression (8,9). Deregulation of miRNAs has been
demonstrated in several types of cancer, including CRC (10–16). In
cancer cells, alteration of miRNA expression levels can be
abnormally down- or upregulated, to function as either tumor
suppressors or oncogenes (17). In
multiple studies, miRNAs have been demonstrated to play an
important role in CRC. For example, miR-21, miR-31 and miR-223 are
upregulated in CRC, whereas miR-143, miR-145 and miR-126 are
downregulated (18–20).
The potential use of miRNA as diagnostic or
prognostic markers in the clinical setting has been highlighted
previously (21). Furthermore, miRNA
mimics can be used as therapeutic agents to restore miRNA function,
or miRNA inhibitors can be used to disrupt upregulated miRNA.
Therefore, identifying novel cancer-associated miRNAs is important
for the potential treatment of cancer.
Downregulated miRNAs are believed to act as tumor
suppressors, which may greatly contribute to colorectal
carcinogenesis (19,20,22,23). The
aim of the present study was to identify miRNA-mRNA associations
and evaluate miRNA expression in human CRC cells, by focusing on
the mTOR signaling pathway.
Materials and methods
Cell lines
The human CRC cell lines, HT29, HCT116, SW620, SW480
and the FHC normal fetal human colon epithelial cell line were
purchased from the American Type Culture Collection (ATCC). The
CSC480 cell line was purchased from BioMedicure (San Diego, CA,
USA) All cell lines were maintained in Dulbecco's modified Eagle's
medium (DMEM; 4.5 g/l D-Glucose, L-Glutamine, 110 mg/l sodium
pyruvate; Invitrogen; Thermo Fisher Scientific, Inc.) supplemented
with 10% fetal bovine serum (Invitrogen; Thermo Fisher Scientific,
Inc.) and cultured at 37°C in a humidified incubator with 5%
CO2. Cells were induced with insulin (200 nM) as
previously described (24). The FHC
cell line was cultured according to previously described culture
conditions (25).
Bioinformatics analysis
mRNA targets were predicted for 22 miRNAs of
interest using five miRNA target-prediction programs: DIANA-MICROT
(diana.imis.athena-innovation.gr) (26), miRWalk (umm.uni-heidelberg.de) (27), TargetScan (targetscan.org) (7),
PicTar (pictar.mdc-berlin.de) (28) and miRDB (mirdb.org)
(29). These databases use a
computational algorithm of miRNAs by searching for the presence of
conserved sites on the 3′UTR of mRNA that match the seed region of
each miRNA. The top target candidates according to the score in the
database for each miRNA provided by each program were chosen for
further molecular analysis.
RNA isolation and reverse
transcription polymerase chain reaction (RT-PCR)
Total RNA, including miRNA, was extracted from
1×106 cultured cells using an miRNeasy Mini kit (Qiagen,
Inc.), according to the manufacturer's protocol. Briefly, cells
were trypsinized, collected from a 10-cm cell-culture dish and
placed into a centrifuge tube, followed by the addition of 700 µl
TRIzol® reagent (Qiagen, Inc., Valencia, CA, USA) then
140 µl chloroform. The aqueous phase was separated by
centrifugation (5 min at room temperature) in a microfuge
(Eppendorf 5430; Eppendorf, Hamburg, Germany) followed by washing
sequentially with 100% ethanol. Samples were centrifuged in an
RNeasy Mini spin column membrane (Qiagen, Inc.) and RNA was then
eluted in RNase-free water. The concentration of RNA was determined
via absorbance measurements at 260 nm using a NanoDrop
spectrophotometer, and the RNA was checked by determining
absorbance ratios, 260/280 and 260/230 nm, for any contamination.
For mRNA expression, cDNAs were synthesized using a BluePrint 1st
Strand cDNA Synthesis kit (Takara Bio, Inc.), according to the
manufacturer's protocol. miRNAs were reverse-transcribed using an
miScript II RT kit (Qiagen, Inc.), also according to the
manufacturer's protocol. Briefly, single-stranded cDNA was
synthesized from RNA in a 20 µl reaction volume. The reactions were
incubated: first, at 37°C for 60 min, then the reverse
transcriptase mix was inactivated by incubation at 95°C for 5 min.
The cDNA generated was diluted 10-fold in RNase-free water.
RT-quantitative (q)PCR analysis of
mRNA
Quantitative, RT-qPCR was used to measure mRNA
expression levels for all three genes (mTOR, RPTOR and RICTOR).
qPCR was performed using a LightCycler 480 thermal cycling system
(Roche Diagnostics) with SYBR Premix Ex Taq II (Takara Bio, Inc.)
in a 20 µl reaction mixture. The sequences of primers used for qPCR
analysis are listed in Table I.
Thermal cycling conditions were stage 1: Initial denaturation (1
cycle) 95°C for 5 min; stage 2: PCR (40 cycles) 95°C for 10 sec
followed by 56°C for 10 sec and 72°C for 20 sec; and stage 3:
Melting curve analysis 95°C for 5 sec, 65°C for 60 sec and 97°C. A
non-template control (nuclease-free water) was included for each
primer set and each sample was analyzed in triplicate. Relative
expression was calculated using the 2−ΔΔCq method
(30).
 | Table I.mTOR, RPTOR and RICTOR primer
sequences. |
Table I.
mTOR, RPTOR and RICTOR primer
sequences.
| Primer | Sequence | Amplicon size |
|---|
| mTOR |
| 146 (bp) |
| F |
5′-AGCATCGGATGCTTAGGAGTGG-3′ |
|
| R |
5′-CAGCCAGTCATCTTTGGAGACC-3′ |
|
| RPTOR |
| 148 (bp) |
| F |
5′-GATCGTCAACAGCTATCACACGG-3′ |
|
| R |
5′-CGAGTCGAAGTTCTGCCAGATC-3′ |
|
| RICTOR |
| 125 (bp) |
| F |
5′-GCCAAACAGCTCACGGTTGTAG-3′ |
|
| R |
5′-CCAGATGAAGCATTGAGCCACTG-3′ |
|
| GAPDH |
| 131 (bp) |
| F |
GTCTCCTCTGACTTCAACAGCG |
|
| R |
ACCACCCTGTTGCTGTAGCCAA |
|
RT-qPCR analysis of miRNAs
RT-qPCR was performed on selected miRNAs using the
LightCycler 480 thermal cycling system (Roche Diagnostics).
Expression levels of mature miRNAs were quantified using an
miScript SYBR Green PCR kit (Qiagen), according to the
manufacturer's instructions. The small nucleolar RNA, C/D box 68
(SNORD68), and the U6 small nuclear 2 RNA (RNU6-2) (Qiagen, Inc.)
were used as endogenous controls, due to their relatively stable
expression. Forward primers (Table
II) were designed by first converting the miRNA sequences to
DNA and adjusting the melting temperature of the primer to 60°C,
either by adding a thymine to the 3′ of the primer or removing a
nucleotide from the 5′ end of the primer. Primers were purchased
from Integrated DNA Technologies, Inc., while the universal reverse
primer was included in the miScript SYBR Green kit. The
amplification reaction (10 µl) included 2X SYBR Green PCR master
mix (5 µl), universal primer (250 nM), miRNA-specific primer (250
nM), RNase-free water (1.75 µl) and cDNA (2 µl). Pipetting was
performed using the epMotion 5075 automated pipetting system
(Eppendorf). The reaction conditions were as follows: Initial
activation step (1 cycle), 95°C for 15 min followed by 3-step
cycling (40 cycles), (denaturation) 94°C for 15 sec, (annealing)
55°C for 30 sec and (extension) 70°C for 30 sec, with melting
curve: 95°C for 5 sec, 65°C for 60 sec and 97°C. All reactions were
performed in triplicate and the average values were used in
subsequent analysis. Expression was normalized to SNORD68 and
RNU6-2. The fold change was calculated using the 2−ΔΔCq
method (30) to acquire relative
expression levels.
 | Table II.Designed miRNA forward sequences. |
Table II.
Designed miRNA forward sequences.
| miRNA | Forward sequence
primer |
|---|
|
hsa-miR-1271-5p |
5′-TTGGCACCTAGCAAGCACTC-3′ |
| hsa-miR-496 |
5′-GCTGAGTATTACATGGCCAATCTC-3′ |
| hsa-miR-581 |
5′-GCGTCTTGTGTTCTCTAGATCAGTAAA-3′ |
|
hsa-miR-1185-3p |
5′-GCATATACAGGGGGAGACTCTTATAAA-3′ |
|
hsa-miR-767-3pa |
5′-CTGCTCATACCCCATGGTTTC-3′ |
| hsa-miR-96-5p |
5′-TTGGCACTAGCACATTTTTGC-3′ |
| hsa-miR-335-3p |
5′-GTTTTTCATTATTGCTCCTGACCA-3′ |
| hsa-miR-3182 |
5′-GCCGCTTCTGTAGTGTAGTCAAA-3′ |
| hsa-miR-1294 |
5′-TGTGAGGTTGGCATTGTTGTCT-3′ |
|
hsa-miR-2114-3p |
5′-GAGCCTCAAGCAAGGGACTT-3′ |
|
hsa-miR-3121-3p |
5′-GCTAAATAGAGTAGGCAAAGGACAAA-3′ |
| hsa-miR-3183 |
5′-CTCTCTCGGAGTCGCTCG-3′ |
| hsa-miR-340-3p |
5′-GCTCCGTCTCAGTTACTTTATAGCAA-3′ |
|
hsa-miR-4802-3p |
5′-TACATGGATGGAAACCTTCAAGC-3′ |
|
hsa-miR-548o-3p |
5′-CCAAAACTGCAGTTACTTTTGCA-3′ |
| hsa-miR-654-5p |
5′-GTGGGCCGCAGAACATG-3′ |
| hsa-miR-1323 |
5′-TCAAAACTGAGGGGCATTTTC-3′ |
| hsa-miR-142-3p |
5′-GCTGTAGTGTTTCCTACTTTATGGAAAA-3′ |
| hsa-miR-194-5p |
5′-TGTAACAGCAACTCCATGTGGA-3′ |
| hsa-miR-495 |
5′-GAAACAAACATGGTGCACTTCTTAA-3′ |
| hsa-miR-659-3p |
5′-TTGGTTCAGGGAGGGTCC-3′ |
| hsa-miR-98-5P |
5′-GCCTGAGGTAGTAAGTTGTATTGTTAAAA-3′ |
Statistical analysis
Data analysis was conducted using Prism software
(version 6.0; GraphPad Software, Inc.). One-way analysis of
variance (ANOVA) was used to determine statistical significance,
with the Dunnett's multiple comparisons test used as a post hoc
test. P<0.05 was considered to indicate a statistically
significant difference. In the ANOVA, each CRC line was compared to
the control FHC cell line, and the multiple group graphs represent
significance in regard to FHC. Data are presented as the mean of
three replicates with standard error.
Results
miRNA target prediction analysis of
mTOR pathway genes
In order to determine potential target genes and
signaling pathways implicated in mTOR, RPTOR and RICTOR, five
programs were used for miRNA target prediction analysis, namely
DIANA-MICROT, miRWalk, TargetScan, PicTar and miRDB. Using the
combination of five programs provided a more reliable model of
target prediction, due to a different computer-aided algorithm for
each target prediction program. From the miRNAs predicted for each
gene, the highest miRNAs predicted by at least 2 prediction
programs were chosen. A total of 22 miRNAs for all three genes were
selected for further analysis (Tables
III–V).
 | Table III.miRNA candidates targeting the mTOR
gene predicted by bioinformatics analysis. |
Table III.
miRNA candidates targeting the mTOR
gene predicted by bioinformatics analysis.
|
|
|
|
|
| Database
algorithm |
|---|
|
|
|
|
|
|
|
|---|
| miRNA ID | Proposed target
gene | Ensembl gene
ID | miRNA sequence |
| Diana | miRWalk | TargetScan | PicTar | miRDB |
|---|
| hsa-miR-581 | mTOR |
ENSG00000198793 |
UCUUGUGUUCUCUAGAUCAGU | Score | 0.95 | – | – | 3.42 | 63 |
|
|
|
|
| P-value | 0.04 | 0.014 | – | – | Rank 20 |
|
|
|
|
| Binding | 8mer | 8mer | – | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | – | UTR3 | UTR3 |
| hsa-miR-96-5p | mTOR |
ENSG00000198793 |
UUUGGCACUAGCACAUUUUUGCU | Score | 0.85 | – | 95 | – | 60 |
|
|
|
|
| P-value | 0.04 | – | 0.79 | – | Rank 24 |
|
|
|
|
| Binding | 7mer | – | 7mer-m8 | – | 7mer |
|
|
|
|
| Region | UTR3 | – | UTR3 | – | UTR3 |
|
hsa-miR-767-3pa | mTOR |
ENSG00000198793 |
UCUGCUCAUACCCCAUGGUUUCU | Score | 0.99 | – | – | – | 88 |
|
|
|
|
| P-value | 0.004 | 0.001 | – | – | Rank 4 |
|
|
|
|
| Binding | 6mer | 10mer | – | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | – | – | UTR3 |
|
hsa-miR-1185-3p | mTOR |
ENSG00000198793 |
AUAUACAGGGGGAGACUCUUAU | Score | 0.88 | – | – | – | 65 |
|
|
|
|
| P-value | 0.04 | – | – | – | Rank 15 |
|
|
|
|
| Binding | 8mer | – | – | – | 7mer |
|
|
|
|
| Region | UTR3 | – | – | – | UTR3 |
|
hsa-miR-1271-5p | mTOR |
ENSG00000198793 |
CUUGGCACCUAGCAAGCACUCA | Score | 0.84 | – | 95 | – | 61 |
|
|
|
|
| P-value | 0.03 | – | 0.79 | – | Rank 22 |
|
|
|
|
| Binding | 7mer | – | 7mer | – | 7mer |
|
|
|
|
| Region | UTR3 | – | UTR3 | – | UTR3 |
| hsa-miR-496 | mTOR |
ENSG00000198793 |
UGAGUAUUACAUGGCCAAUCUC | Score | 0.86 | – | – | – | 72 |
|
|
|
|
| P-value | 0.03 | 0.014 | – | – | Rank 9 |
|
|
|
|
| Binding | 8mer | 8mer | – | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | – | – | UTR3 |
| hsa-miR-335-3p | mTOR |
ENSG00000198793 |
UUUUUCAUUAUUGCUCCUGACC | Score | 0.85 | – | – | – | 67 |
|
|
|
|
| P-value | 0.03 | 0.014 | – | – | Rank 13 |
|
|
|
|
| Binding | 8mer | 8mer | – | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | – | – | UTR3 |
| hsa-miR-3182 | mTOR |
ENSG00000198793 |
GCUUCUGUAGUGUAGUC | Score | 0.85 | – | – | – | 79 |
|
|
|
|
| P-value | 0.028 | – | – | – | Rank 6 |
|
|
|
|
| Binding | 7mer | – | – | – | 7mer |
|
|
|
|
| Region | UTR3 | – | – | – | UTR3 |
 | Table V.miRNA candidates targeting the RICTOR
gene predicted by bioinformatics analysis. |
Table V.
miRNA candidates targeting the RICTOR
gene predicted by bioinformatics analysis.
|
|
|
|
|
| Database
algorithm |
|---|
| miRNA ID | Proposed target
gene | Ensembl gene
ID | miRNA sequence |
| Diana | miRWalk | TargetScan | PicTar | miRDB |
|---|
| hsa-miR-495 | RICTOR |
ENSG00000164327 |
AAACAAACAUGGUGCACUUCUU | Score | 0.871 | – | −0.17 | – | – |
|
|
|
|
| P-value | 0.002 | 0.0166 | N/A | – | – |
|
|
|
|
| Binding | 8mer | 9mer | 8mer | – | – |
|
|
|
|
| Region | UTR3 | UTR3 | UTR3 | – | – |
| hsa-miR-194-5p | RICTOR |
ENSG00000164327 |
UGUAACAGCAACUCCAUGUGGA | Score | 0.79 | – | – | – | 83 |
|
|
|
|
| P-value | 0.003 | – | – | – | Rank 28 |
|
|
|
|
| Binding | 6mer | – | – | – | 7mer |
|
|
|
|
| Region | UTR3 | – | – | – | UTR3 |
| hsa-miR-659-3p | RICTOR |
ENSG00000164327 |
CUUGUUCAGGGAGGGUCCCCA | Score | 0.986 | – | – | – | 75 |
|
|
|
|
| P-value | 0.008 | – | – | – | Rank 48 |
|
|
|
|
| Binding | 7mer | – | – | – | 7mer |
|
|
|
|
| Region | UTR3 | – | – | – | UTR3 |
| hsa-miR-142-3p | RICTOR |
ENSG00000164327 |
UGUAGUGUUUCCUACUUUAUGGA | Score | 0.999 | – | – | – | 100 |
|
|
|
|
| P-value | 0.03 | 0.0166 | 0.99 | – | Rank 61 |
|
|
|
|
| Binding | 8mer | 9mer | 8mer | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | UTR3 | – | UTR3 |
| hsa-miR-98-5P | RICTOR |
ENSG00000164327 |
UGAGGUAGUAAGUUGUAUU | Score | 0.999 | – | −0.14 | – |
|
|
|
|
|
| P-value | 0.007 | 0.0166 | 0.98 | – | Rank |
|
|
|
|
| Binding | 8mer | 9mer | 8mer | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | UTR3 | – | UTR3 |
| hsa-miR-1323 | RICTOR |
ENSG00000164327 |
UCAAAACUGAGGGGCAUUUUCU | Score | 0.75 | – | – | – | 80 |
|
|
|
|
| P-value | 0.006 | 0.004 | – | – | Rank 37 |
|
|
|
|
| Binding | 8mer | 10mer | – | – | 7mer |
|
|
|
|
| Region | UTR3 | UTR3 | – | – | UTR3 |
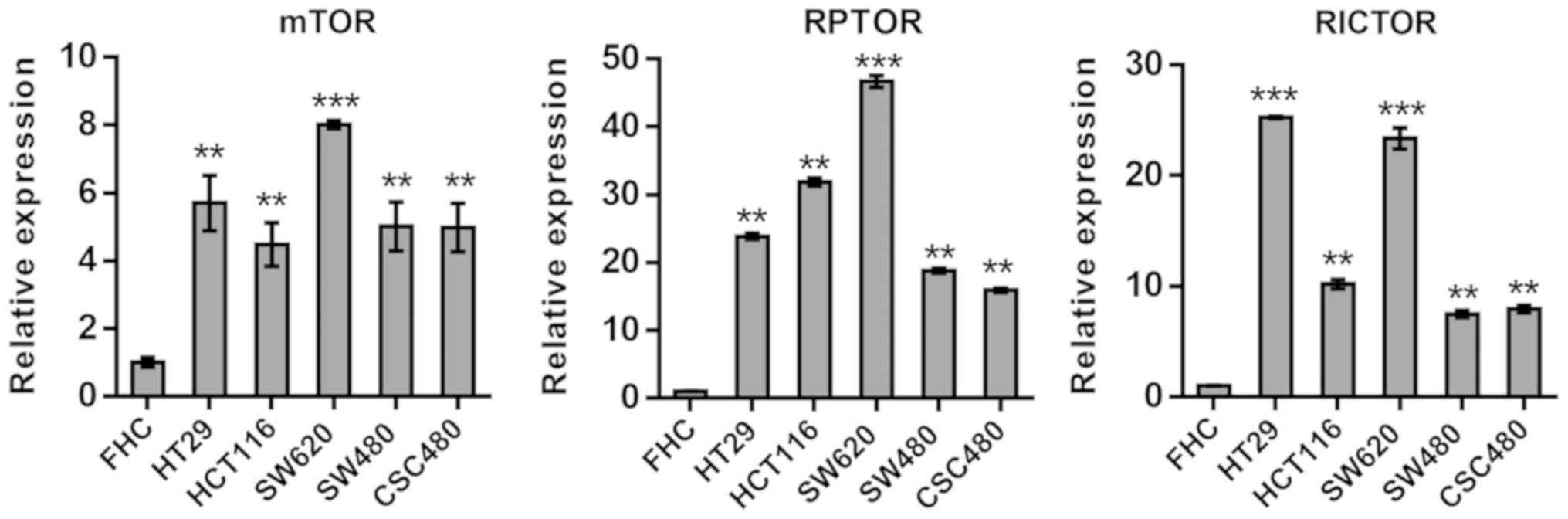
Gene expression of mTOR, RICTOR, RPTOR
and validation of specific miRNAs
To assess alterations of the mTOR pathway in CRC
cells, mTOR, RPTOR and RICTOR expression levels were investigated
in five human CRC cell lines, HT29, HCT116, SW620, SW480 and
CSC480, using RT-qPCR. The gene GAPDH was used as a reference. The
comparison of results from mTOR, RPTOR and RICTOR expression
revealed significantly (P<0.01) elevated mRNA expression levels
in all cell lines compared with normal FHC cells (Fig. 1).
mTOR pathway-targets miRNAs
differentially regulated
Next, the most significant candidates of miRNAs were
selected, as predicted by the bioinformatics analysis, to target
the genes of interest for further confirmatory studies. A selection
of CRC cell lines, as well as the control (FHC), were used to
examine whether miRNAs were associated with transcriptional
expression of their relevant target gene and to identify miRNAs
involved in CRC cells. Based on the results from the bioinformatics
analysis, candidate miRNAs that were identified to be associated
with each gene were assessed using qPCR.
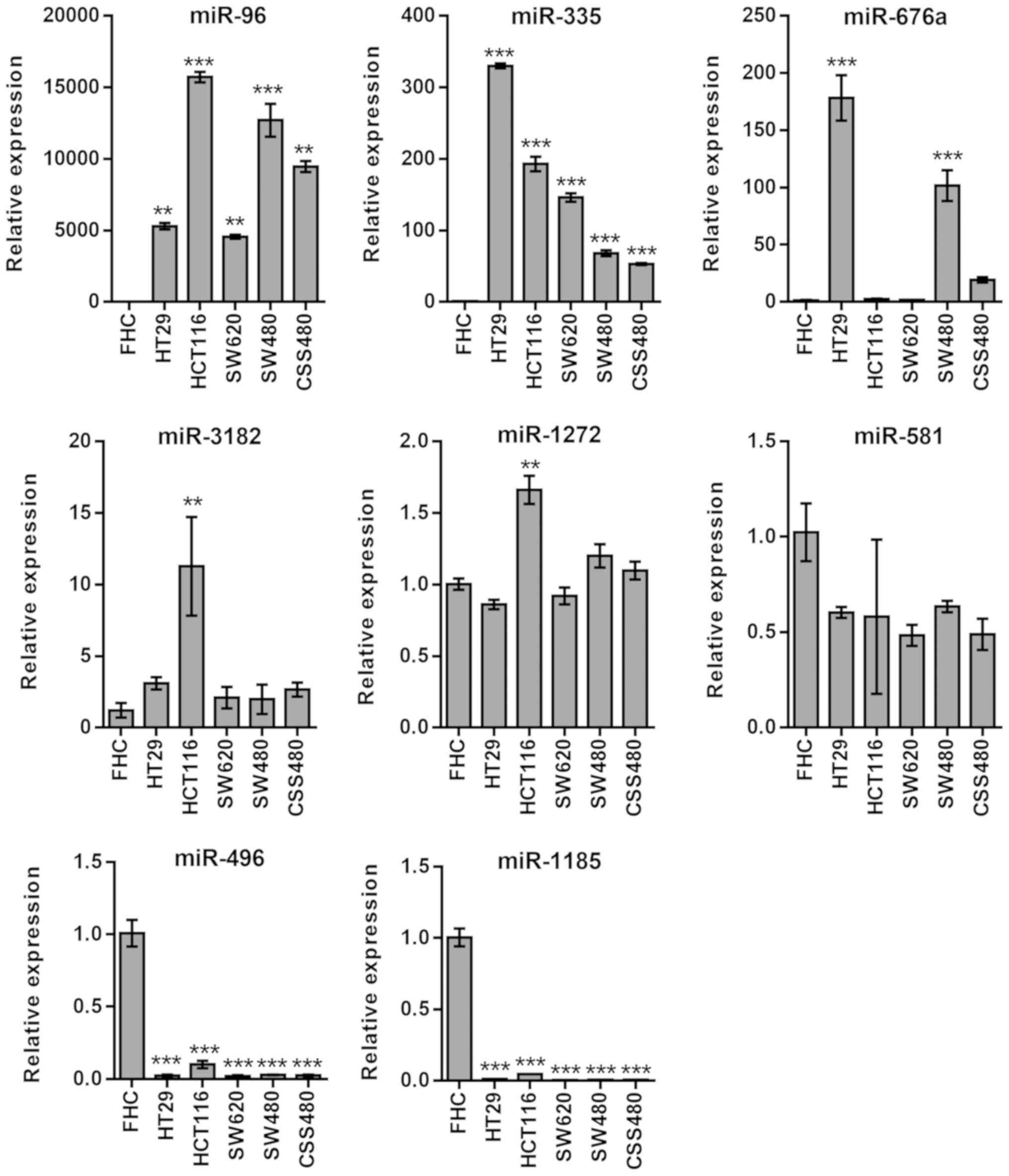
mTOR
A statistically significant upregulation in the
expression levels of miR-96 (P<0.01) and miR-335 (P<0.001),
which was associated with mTOR, was observed in all cell lines
(Fig. 2). miRNAs, such as miR-676a
(HT29 and SW480, P<0.001) and miR-3182 (HCT116 P<0.05), were
upregulated in certain cell lines, while miR-1272 and miR-581
expression remained unchanged. miR-496 and miR-1185 expression
levels were significantly downregulated (P<0.001) in all cancer
cell lines (Fig. 2).
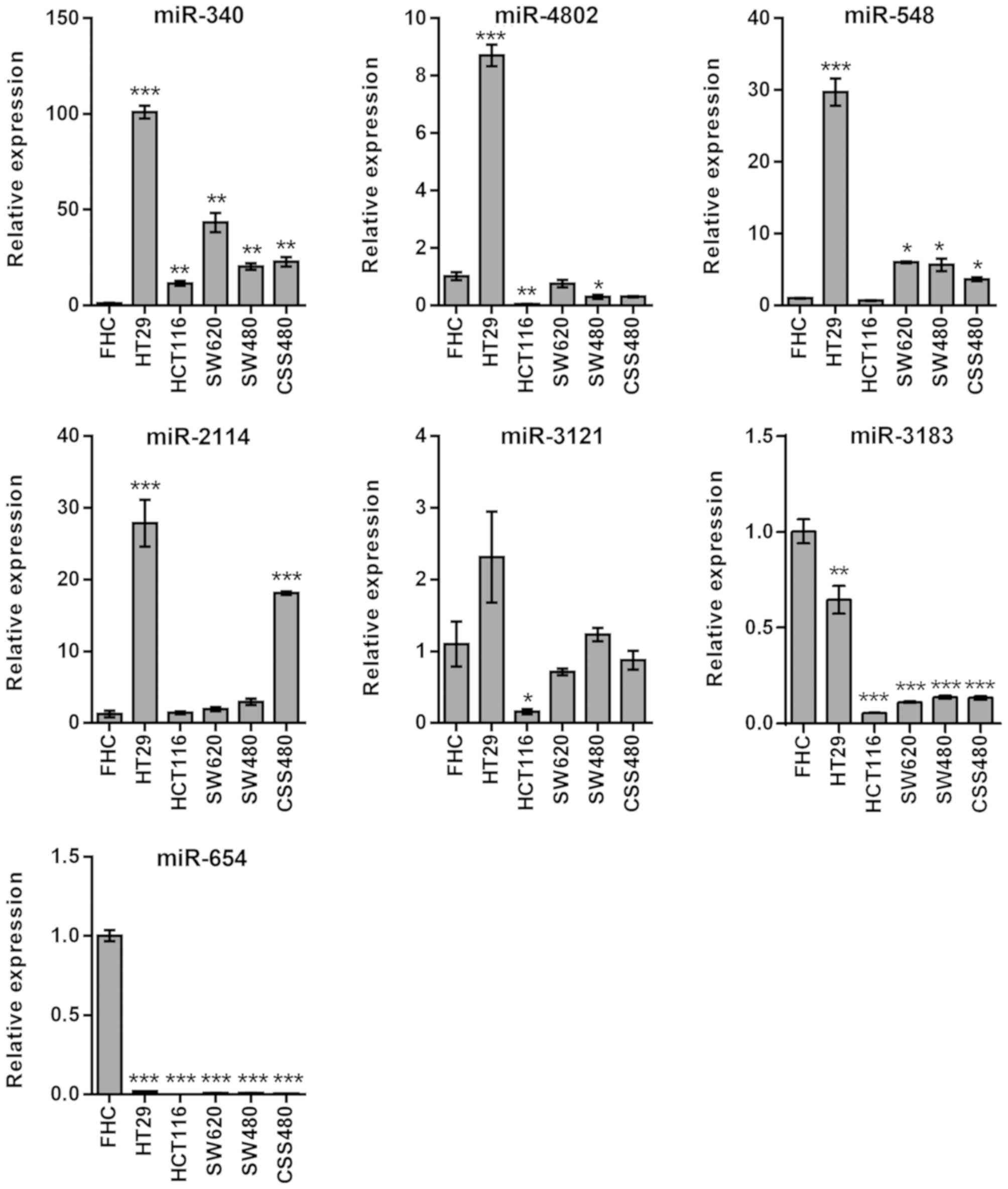
RPTOR
In miRNAs associated with RPTOR, miR-340 exhibited
high expression levels in all cell lines (P<0.01), whereas
miR-4802 was upregulated (P<0.001) in HT29 cells only (Fig. 3). Furthermore, miR-548 was
upregulated in all cell lines (P<0.05) except HCT116. HT29 and
CSC480 were the only cell lines that exhibited upregulated miR-2114
(P<0.001). There was no statistically significant difference in
the expression levels of miR-3121, except for the downregulation in
HCT116 cells (P<0.05). However, miR-654 (P<0.01) and miR-3183
(P<0.001) expression levels were downregulated in all cell lines
(Fig. 3).
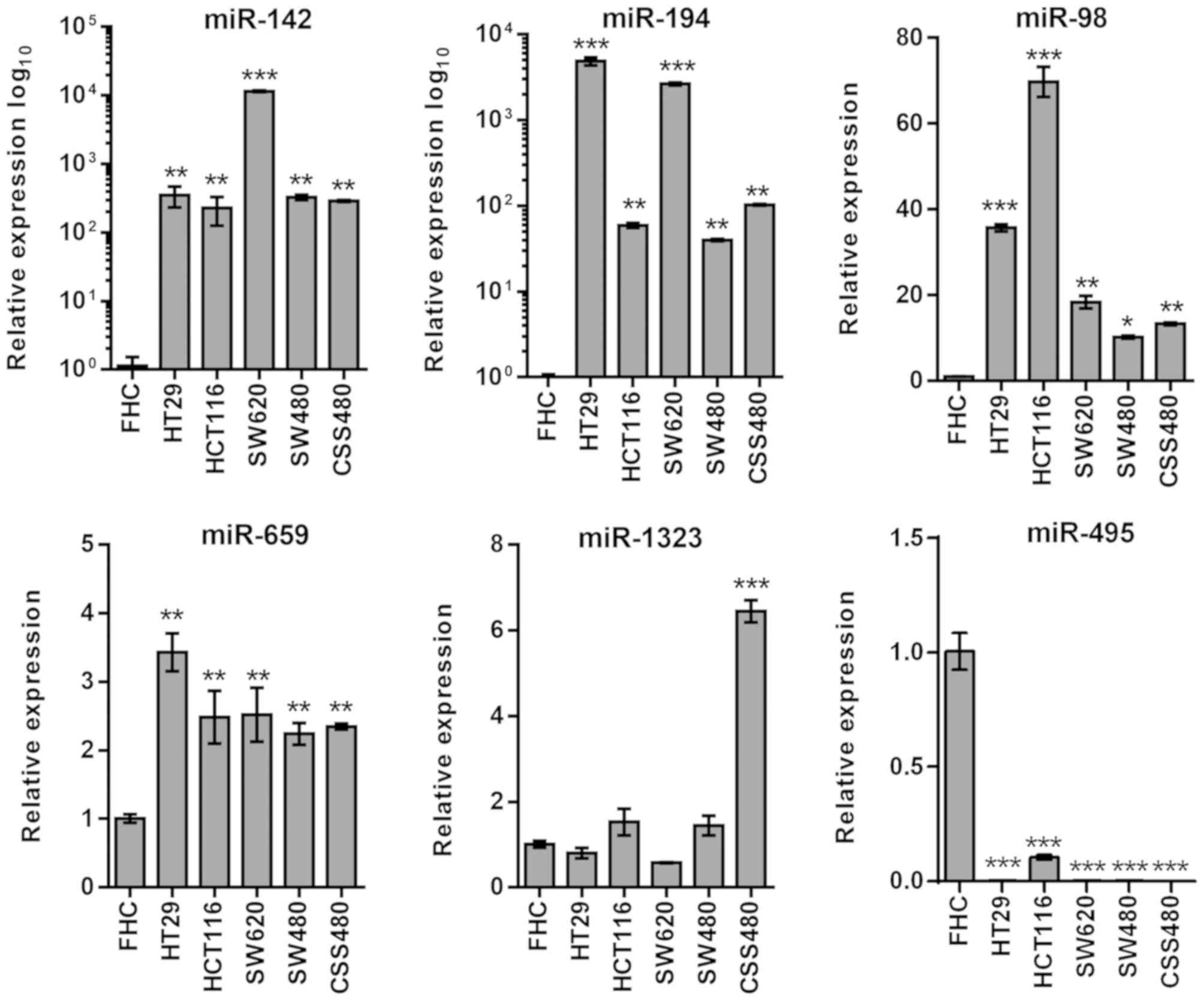
RICTOR
In miRNAs associated with RICTOR, miR-142, miR-194,
miR-98 and miR-659 demonstrated high expression levels in all cells
(P<0.01), while miR-1323 was upregulated (P<0.001) in CSC480
cells only (Fig. 4). The expression
levels of miR-495 were downregulated in all cell lines
(P<0.001).
Discussion
Deregulation of the mTOR pathway occurs frequently
in human cancer types (3,24,31,32).
Regulation of mTOR by miRNAs has been investigated in a number of
different types of cancer, including CRC (33,34). In
the present study, it was revealed that 6 miRNAs (miR-96, miR-335,
miR-340, miR-142, miR-194, miR-659 and miR-98) were upregulated in
the 5 investigated human CRC cell lines when comparing with FHC.
Furthermore, among 20 miRNAs, 5 (miR-496, miR-1185, miR-3183,
miR-654 and miR-495) revealed significant downregulation in
association with the 3 key mTOR signaling pathway genes, mTOR,
RPTOR and RICTOR.
Several software programs are available to aid in
identifying miRNA target prediction. The majority of these
approaches use the seed region, which is approximately 6–8
nucleotides in length within the miRNA, as methods to target mRNA
and bind at the 3′UTR of the target gene when searching for
complementary strands (35). The
present study used five algorithmic tools, Diana-MicroT, miRWalk,
TargetScan, PicTar and miRDB, for miRNA target prediction analysis,
which also considered seed-based methods, and miRDB analyses
identified those genes affected by miRNAs, using a support vector
machine trained on multiple microarray datasets (29). Despite the recent advances in miRNA
target prediction databases (26–29), the
results of miRNA-mRNA association analyses were varied depending on
the database. A possible explanation is that each program has its
methodological variances with respect to detecting miRNA-binding
regions. To minimize the differences in miRNA target predictions,
miRNAs that were identified by at least three prediction programs
were selected for analysis.
Consistent with the results from the previous study,
miR-335 has been demonstrated to be overexpressed in gastric cancer
and involved in the oncogenic mTOR signaling pathway (36). In addition, miR-335 has been revealed
as upregulated in a number of different types of cancer, including
CRC (37–39). In numerous studies, the miRNAs miR-96
(40–45), miR-340 (46), miR-142 (47–49),
miR-194 (50–52) and miR-98 (53–55),
have demonstrated overexpression in a number of different types of
cancer, including CRC.
Numerous studies have demonstrated downregulation of
miRNAs in different types of cancer and their capacity to play a
role in the mTOR signaling pathway (56–59). In
the present study, the expression analysis was focused on the most
highly downregulated miRNAs that were associated with upregulated
mTOR, RPTOR and RICTOR gene expression in CRC cell lines. Among all
five downregulated miRNAs investigated in the present study,
miR-495, miR-1185, miR-654 and miR-496 have previously been
reported in various types of cancer (56–59).
When regarding the role of miR-495 in cancer,
several studies have revealed decreased miR-495 expression levels
in a number of different types of cancer, such as CRC, prostate
cancer, glioma, leukemia and lung cancer (58,60–63); in
addition, miR-495 has been identified as a tumor suppressor in
these cancer types. However, in a study conducted on breast cancer
stem cells, miR-495 overexpression promoted oncogenesis (64).
Xu et al (65)
demonstrated downregulation of miR-1185 in stage IV colorectal
carcinoma. Tan et al (66)
revealed that miR-654 acts as a tumor suppressor in breast cancer,
by modulation of its target EPSTI1. Furthermore, another study
indicated that miR-654 has tumor suppressor properties in papillary
thyroid cancer (67). miR-495 was
revealed to be downregulated in malignant cells and tissues of the
breast (68), while its
overexpression acts as a critical tumor suppressor in CRC cells,
through targeting FAM83D (69).
Previous in vitro findings have identified an
inverse correlation between miR-496 and miR-1185 expression and
mTOR (1), miR-654 and miR-3183
expression and RPTOR (2), miR-495
expression and RICTOR (3) in human
CRC cells. These downregulated miRNAs highlight the importance of
miRNAs for use as potential tumor suppressors via targeting the
mTOR signaling pathway. Further studies using 3′ luciferase
reporters are needed to confirm the targets of these miRNAs in the
mTOR pathway. Increasing miRNA expression levels using mimics to
examine mTOR signaling and cancer progression is an important
approach in CRC research. Furthermore, the role of these miRNAs
requires confirmation by applying more functional in vitro
and in vivo studies, such as miRNA inhibitor studies.
Acknowledgements
Not applicable.
Funding
This present study was supported by a PhD
scholarship awarded to Naif Alqurashi by the Imam Abdulrahman Bin
Faisal University. Funding at Griffith University was provided to
Ming Wei through the higher degree research office.
Availability of data and materials
The data and materials including within the present
study are available from the corresponding author upon reasonable
request.
Authors contributions
SMH conceptualized the design of the present study.
NA curated the data, while SMH and AF analyzed the data. FA and MQW
acquired funding for the present study. NA, SMH and FA performed
the experiments. MQW and SI supervised and provided administrative
support for the study. NA wrote the manuscript, and SMH, FA, SI, AF
and MQW critically reviewed the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflicts of
interest.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zoncu R, Efeyan A and Sabatini DM: mTOR:
From growth signal integration to cancer, diabetes and ageing. Nat
Rev Mol Cell Biol. 12:21–35. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sarbassov DD, Guertin DA, Ali SM and
Sabatini DM: Phosphorylation and regulation of Akt/PKB by the
rictor-mTOR complex. Science. 307:1098–1101. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bagga S, Bracht J, Hunter S, Massirer K,
Holtz J, Eachus R and Pasquinelli AE: Regulation by let-7 and lin-4
miRNAs results in target mRNA degradation. Cell. 122:553–563. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Calin GA, Sevignani C, Dumitru CD, Hyslop
T, Noch E, Yendamuri S, Shimizu M, Rattan S, Bullrich F, Negrini M
and Croce CM: Human microRNA genes are frequently located at
fragile sites and genomic regions involved in cancers. Proc Natl
Acad Sci USA. 101:2999–3004. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kitade Y and Akao Y: MicroRNAs and their
therapeutic potential for human diseases: microRNAs, miR-143 and
−145, function as anti-oncomirs and the application of chemically
modified miR-143 as an anti-cancer drug. J Pharmacol Sci.
114:276–280. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Krek A, Grün D, Poy MN, Wolf R, Rosenberg
L, Epstein EJ, MacMenamin P, da Piedade I, Gunsalus KC, Stoffel M
and Rajewsky N: Combinatorial microRNA target predictions. Nat
Genet. 37:495–500. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lu J, Getz G, Miska EA, Alvarez-Saavedra
E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA,
et al: MicroRNA expression profiles classify human cancers. Nature.
435:834–838. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Farazi TA, Spitzer JI, Morozov P and
Tuschl T: miRNAs in human cancer. J Pathol. 223:102–115. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ciafrè SA, Galardi S, Mangiola A, Ferracin
M, Liu CG, Sabatino G, Negrini M, Maira G, Croce CM and Farace MG:
Extensive modulation of a set of microRNAs in primary glioblastoma.
Biochem Biophys Res Commun. 334:1351–1358. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim YK, Yu J, Han TS, Park SY, Namkoong B,
Kim DH, Hur K, Yoo MW, Lee HJ, Yang HK and Kim VN: Functional links
between clustered microRNAs: Suppression of cell-cycle inhibitors
by microRNA clusters in gastric cancer. Nucleic Acids Res.
37:1672–1681. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Iorio MV, Ferracin M, Liu CG, Veronese A,
Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M,
et al: MicroRNA gene expression deregulation in human breast
cancer. Cancer Res. 65:7065–7070. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wu WK, Law PT, Lee CW, Cho CH, Fan D, Wu
K, Yu J and Sung JJ: MicroRNA in colorectal cancer: From benchtop
to bedside. Carcinogenesis. 32:247–253. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Michael MZ, SM OC, van Holst Pellekaan NG,
Young GP and James RJ: Reduced accumulation of specific microRNAs
in colorectal neoplasia. Mol Cancer Res. 1:882–891. 2003.PubMed/NCBI
|
|
16
|
Takamizawa J, Konishi H, Yanagisawa K,
Tomida S, Osada H, Endoh H, Harano T, Yatabe Y, Nagino M, Nimura Y,
et al: Reduced expression of the let-7 microRNAs in human lung
cancers in association with shortened postoperative survival.
Cancer Res. 64:3753–3756. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pradhan AK, Emdad L, Das SK, Sarkar D and
Fisher PB: The enigma of miRNA regulation in cancer. Adv Cancer
Res. 135:25–52. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Slaby O, Svoboda M, Fabian P, Smerdova T,
Knoflickova D, Bednarikova M, Nenutil R and Vyzula R: Altered
expression of miR-21, miR-31, miR-143 and miR-145 is related to
clinicopathologic features of colorectal cancer. Oncology.
72:397–402. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guo C, Sah JF, Beard L, Willson JK,
Markowitz SD and Guda K: The noncoding RNA, miR-126, suppresses the
growth of neoplastic cells by targeting phosphatidylinositol
3-kinase signaling and is frequently lost in colon cancers. Genes
Chromosomes Cancer. 47:939–946. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gaedcke J, Grade M, Camps J, Søkilde R,
Kaczkowski B, Schetter AJ, Difilippantonio MJ, Harris CC, Ghadimi
BM, Møller S, et al: The rectal cancer microRNAome-microRNA
expression in rectal cancer and matched normal mucosa. Clin Cancer
Res. 18:4919–4930. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shah MY, Ferrajoli A, Sood AK,
Lopez-Berestein G and Calin GA: microRNA therapeutics in cancer-an
emerging concept. EBioMedicine. 12:34–42. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Grady WM, Parkin RK, Mitchell PS, Lee JH,
Kim YH, Tsuchiya KD, Washington MK, Paraskeva C, Willson JK, Kaz
AM, et al: Epigenetic silencing of the intronic microRNA
hsa-miR-342 and its host gene EVL in colorectal cancer. Oncogene.
27:3880–3888. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tazawa H, Tsuchiya N, Izumiya M and
Nakagama H: Tumor-suppressive miR-34a induces senescence-like
growth arrest through modulation of the E2F pathway in human colon
cancer cells. Proc Natl Acad Sci USA. 104:15472–15477. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Alqurashi N, Hashimi SM, Alowaidi F,
Ivanovski S and Wei MQ: Dual mTOR/PI3K inhibitor NVP-BEZ235 arrests
colorectal cancer cell growth and displays differential inhibition
of 4E-BP1. Oncol Rep. 40:1083–1092. 2018.PubMed/NCBI
|
|
25
|
Alowaidi F, Hashimi SM, Alqurashi N,
Alhulais R, Ivanovski S, Bellette B, Meedenyia A, Lam A and Wood S:
Assessing stemness and proliferation properties of the newly
established colon cancer ‘stem’ cell line, CSC480 and novel
approaches to identify dormant cancer cells. Oncol Rep.
39:2881–2891. 2018.PubMed/NCBI
|
|
26
|
Kiriakidou M, Nelson PT, Kouranov A,
Fitziev P, Bouyioukos C, Mourelatos Z and Hatzigeorgiou A: A
combined computational-experimental approach predicts human
microRNA targets. Genes Dev. 18:1165–1178. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dweep H, Sticht C, Pandey P and Gretz N:
miRWalk-database: Prediction of possible miRNA binding sites by
‘walking’ the genes of three genomes. J Biomed Inform. 44:839–847.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Friedman RC, Farh KK, Burge CB and Bartel
DP: Most mammalian mRNAs are conserved targets of microRNAs. Genome
Res. 19:92–105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang X and El Naqa IM: Prediction of both
conserved and nonconserved microRNA targets in animals.
Bioinformatics. 24:325–332. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dowling RJ, Topisirovic I, Alain T,
Bidinosti M, Fonseca BD, Petroulakis E, Wang X, Larsson O, Selvaraj
A, Liu Y, et al: mTORC1-mediated cell proliferation, but not cell
growth, controlled by the 4E-BPs. Science. 328:1172–1176. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Laplante M and Sabatini DM: mTOR signaling
in growth control and disease. Cell. 149:274–293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Melo SA and Esteller M: Dysregulation of
microRNAs in cancer: Playing with fire. FEBS Lett. 585:2087–2099.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
AlQurashi N, Hashimi S and Wei M: Chemical
inhibitors and microRNAs (miRNA) targeting the mammalian target of
rapamycin (mTOR) pathway: Potential for novel anticancer
therapeutics. Int J Mol Sci. 14:3874–3900. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Oğul H: Computational prediction of
microRNA function and activity. Methods Mol Biol. 1107:243–256.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yan Z, Xiong Y, Xu W, Gao J, Cheng Y, Wang
Z, Chen F and Zheng G: Identification of hsa-miR-335 as a
prognostic signature in gastric cancer. PLoS One. 7:e400372012.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Peng HH, Zhang YD, Gong LS, Liu WD and
Zhang Y: Increased expression of microRNA-335 predicts a favorable
prognosis in primary gallbladder carcinoma. Onco Targets Ther.
6:1625–1630. 2013.PubMed/NCBI
|
|
38
|
Wang YX, Zhang XY, Zhang BF, Yang CQ, Chen
XM and Gao HJ: Initial study of microRNA expression profiles of
colonic cancer without lymph node metastasis. J Dig Dis. 11:50–54.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Vickers MM, Bar J, Gorn-Hondermann I,
Yarom N, Daneshmand M, Hanson JE, Addison CL, Asmis TR, Jonker DJ,
Maroun J, et al: Stage-dependent differential expression of
microRNAs in colorectal cancer: Potential role as markers of
metastatic disease. Clin Exp Metastasis. 29:123–132. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sarver AL, French AJ, Borralho PM,
Thayanithy V, Oberg AL, Silverstein KA, Morlan BW, Riska SM,
Boardman LA, Cunningham JM, et al: Human colon cancer profiles show
differential microRNA expression depending on mismatch repair
status and are characteristic of undifferentiated proliferative
states. BMC Cancer. 9:4012009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bandrés E, Cubedo E, Agirre X, Malumbres
R, Zárate R, Ramirez N, Abajo A, Navarro A, Moreno I, Monzó M and
García-Foncillas J: Identification by Real-time PCR of 13 mature
microRNAs differentially expressed in colorectal cancer and
non-tumoral tissues. Mol Cancer. 5:292006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang J, Kong X, Li J, Luo Q, Li X, Shen
L, Chen L and Fang L: miR-96 promotes tumor proliferation and
invasion by targeting RECK in breast cancer. Oncol Rep.
31:1357–1363. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pineau P, Volinia S, McJunkin K, Marchio
A, Battiston C, Terris B, Mazzaferro V, Lowe SW, Croce CM and
Dejean A: miR-221 overexpression contributes to liver
tumorigenesis. Proc Natl Acad Sci USA. 107:264–269. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Navon R, Wang H, Steinfeld I, Tsalenko A,
Ben-Dor A and Yakhini Z: Novel rank-based statistical methods
reveal microRNAs with differential expression in multiple cancer
types. PLoS One. 4:e80032009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang L, Zhu MJ, Ren AM, Wu HF, Han WM, Tan
RY and Tu RQ: A ten-microRNA signature identified from a
genome-wide microRNA expression profiling in human epithelial
ovarian cancer. PLoS One. 9:e964722014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yao Y, Suo AL, Li ZF, Liu LY, Tian T, Ni
L, Zhang WG, Nan KJ, Song TS and Huang C: MicroRNA profiling of
human gastric cancer. Mol Med Rep. 2:963–970. 2009.PubMed/NCBI
|
|
47
|
Wang XY, Wu MH, Liu F, Li Y, Li N, Li GY
and Shen SR: Differential miRNA expression and their target genes
between NGX6-positive and negative colon cancer cells. Mol Cell
Biochem. 345:283–290. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chen WC, Lin MS, Ye YL, Gao HJ, Song ZY
and Shen XY: microRNA expression pattern and its alteration
following celecoxib intervention in human colorectal cancer. Exp
Ther Med. 3:1039–1048. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang B, Li J, Sun M, Sun L and Zhang X:
miRNA expression in breast cancer varies with lymph node metastasis
and other clinicopathologic features. IUBMB Life. 66:371–377. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gu J, Wang Y and Wu X: MicroRNA in the
pathogenesis and prognosis of esophageal cancer. Curr Pharm Des.
19:1292–1300. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sundaram P, Hultine S, Smith LM, Dews M,
Fox JL, Biyashev D, Schelter JM, Huang Q, Cleary MA, Volpert OV and
Thomas-Tikhonenko A: p53-responsive miR-194 inhibits
thrombospondin-1 and promotes angiogenesis in colon cancers. Cancer
Res. 71:7490–7501. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang J, Zhao CY, Zhang SH, Yu DH, Chen Y,
Liu QH, Shi M, Ni CR and Zhu MH: Upregulation of miR-194
contributes to tumor growth and progression in pancreatic ductal
adenocarcinoma. Oncol Rep. 31:1157–1164. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sampson VB, Rong NH, Han J, Yang Q, Aris
V, Soteropoulos P, Petrelli NJ, Dunn SP and Krueger LJ: MicroRNA
let-7a down-regulates MYC and reverts MYC-induced growth in Burkitt
lymphoma cells. Cancer Res. 67:9762–9770. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Du L, Schageman JJ, Subauste MC, Saber B,
Hammond SM, Prudkin L, Wistuba II, Ji L, Roth JA, Minna JD and
Pertsemlidis A: miR-93, miR-98, and miR-197 regulate expression of
tumor suppressor gene FUS1. Mol Cancer Res. 7:1234–1243. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Deng ZQ, Yin JY, Tang Q, Liu FQ, Qian J,
Lin J, Shao R, Zhang M and He L: Over-expression of miR-98 in FFPE
tissues might serve as a valuable source for biomarker discovery in
breast cancer patients. Int J Clin Exp Pathol. 7:1166–1171.
2014.PubMed/NCBI
|
|
56
|
Nagaraja AK, Creighton CJ, Yu Z, Zhu H,
Gunaratne PH, Reid JG, Olokpa E, Itamochi H, Ueno NT, Hawkins SM,
et al: A link between mir-100 and FRAP1/mTOR in clear cell ovarian
cancer. Mol Endocrinol. 24:447–463. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Duan Z, Choy E, Harmon D, Liu X, Susa M,
Mankin H and Hornicek F: MicroRNA-199a-3p is downregulated in human
osteosarcoma and regulates cell proliferation and migration. Mol
Cancer Ther. 10:1337–1345. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Chen SM, Chen HC, Chen SJ, Huang CY, Chen
PY, Wu TW, Feng LY, Tsai HC, Lui TN, Hsueh C and Wei KC:
MicroRNA-495 inhibits proliferation of glioblastoma multiforme
cells by downregulating cyclin-dependent kinase 6. World J Surg
Oncol. 11:872013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Fornari F, Milazzo M, Chieco P, Negrini M,
Calin GA, Grazi GL, Pollutri D, Croce CM, Bolondi L and Gramantieri
L: miR-199a-3p regulates mTOR and c-Met to influence the
doxorubicin sensitivity of human hepatocarcinoma cells. Cancer Res.
70:5184–5193. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Formosa A, Markert EK, Lena AM, Italiano
D, Finazzi-Agro' E, Levine AJ, Bernardini S, Garabadgiu AV, Melino
G and Candi E: MicroRNAs, miR-154, miR-299-5p, miR-376a, miR-376c,
miR-377, miR-381, miR-487b, miR-485-3p, miR-495 and miR-654-3p,
mapped to the 14q32.31 locus, regulate proliferation, apoptosis,
migration and invasion in metastatic prostate cancer cells.
Oncogene. 33:5173–5182. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Jiang X, Huang H, Li Z, He C, Li Y, Chen
P, Gurbuxani S, Arnovitz S, Hong GM, Price C, et al: miR-495 is a
tumor-suppressor microRNA down-regulated in MLL-rearranged
leukemia. Proc Natl Acad Sci USA. 109:19397–19402. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Chu H, Chen X, Wang H, Du Y, Wang Y, Zang
W, Li P, Li J, Chang J, Zhao G and Zhang G: miR-495 regulates
proliferation and migration in NSCLC by targeting MTA3. Tumour
Biol. 35:3487–3494. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Chuang AY, Chuang JC, Zhai Z, Wu F and
Kwon JH: NOD2 expression is regulated by microRNAs in colonic
epithelial HCT116 cells. Inflamm Bowel Dis. 20:126–135. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Hwang-Verslues WW, Chang PH, Wei PC, Yang
CY, Huang CK, Kuo WH, Shew JY, Chang KJ, Lee EY and Lee WH: miR-495
is upregulated by E12/E47 in breast cancer stem cells, and promotes
oncogenesis and hypoxia resistance via downregulation of E-cadherin
and REDD1. Oncogene. 30:2463–2474. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Xu XH, Wu XB, Wu SB, Liu HB, Chen R and Li
Y: Identification of miRNAs differentially expressed in clinical
stages of human colorectal carcinoma-an investigation in Guangzhou,
China. PLoS One. 9:e940602014. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tan YY, Xu XY, Wang JF, Zhang CW and Zhang
SC: miR-654-5p attenuates breast cancer progression by targeting
EPSTI1. Am J Cancer Res. 6:522–532. 2016.PubMed/NCBI
|
|
67
|
Geraldo MV, Nakaya HI and Kimura ET:
Down-regulation of 14q32-encoded miRNAs and tumor suppressor role
for miR-654-3p in papillary thyroid cancer. Oncotarget.
8:9597–9607. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wang L, Liu JL, Yu L, Liu XX, Wu HM, Lei
FY, Wu S and Wang X: Downregulated miR-495 [Corrected] inhibits the
G1-S phase transition by targeting Bmi-1 in breast cancer. Medicine
(Baltimore). 94:e7182015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Yan L, Yao J and Qiu J: miRNA-495
suppresses proliferation and migration of colorectal cancer cells
by targeting FAM83D. Biomed Pharmacother. 96:974–981. 2017.
View Article : Google Scholar : PubMed/NCBI
|