Introduction
Cervical squamous cell carcinoma (CSCC) is a leading
cause of cancer-associated mortality in women worldwide and has
significant societal and economic consequences (1). At present, surgery and radiotherapy are
standard treatments for cervical cancer (2). However, the clinical outcomes of these
treatments vary dramatically and are difficult to predict.
Therefore, it is necessary to identify new, effective treatment
approaches, particularly for patients with resistance to
radiotherapy and those susceptible to relapse.
MicroRNAs (miRNAs) are small noncoding RNA molecules
that target protein-coding genes and downregulate their expression
(3). They are of 19–25 nucleotides
in length and are cleaved by 70–100 nt long hairpin precursor
miRNAs (pre-miRNAs) (4). Numerous
essential processes are regulated by miRNAs, including viral
infection, cell growth, apoptosis and cancer development (5). However, few studies have focused on the
role of miRNAs in cervical cancer (6–8), and the
association between miRNAs and cervical cancer transformation
remains poorly understood. Among the miRNAs investigated in
cervical cancer, miR-1246 has oncogenic effects (9), and is elevated in the serum and tumor
tissues isolated from patients with CSCC with lymph node metastasis
(10).
Thrombospondin-2 (TSP2 or THBS2) is a member of the
thrombospondin family. In a previous study in SiHa cervical cancer
cells, THBS2 was shown to be a target of miR-1246
(11). THBS2 regulates cell adhesion
and migration via hydrolysis of the extracellular matrix (ECM)
(12). THBS2 also may serve a role
in inhibiting angiogenesis by regulating matrix metalloproteinases
(MMPs) and ECM proteins (13). While
it is possible that the miR-1246/THBS2/ECM signaling cascade
is involved in cervical cancer metastasis, direct evidence for a
role of this pathway has not been reported.
In the present study, the effects of miR-1246
downregulation via lentiviral transfection, on the THBS2/MMPs/ECM
pathway in the human cervical cancer cell line SiHa were
evaluated.
Materials and methods
Synthesis of recombinant lentiviral
particles (LV-miR-1246-Inh)
Lentivirus particles expressing oligonucleotides
against hsa-miR-1246 (miRBase accession number,
MIMAT0005898) were constructed by Shanghai GeneChem Co., Ltd.
Briefly, double-stranded oligonucleotides encoding a
miR-1246-inhibitor (miR-1246-Inh) was annealed and
inserted into the linearized eukaryotic hU6-MCS-Ubiquitin-EGFR-puro
vector (Shanghai GeneChem Co., Ltd.). The identity of vector was
confirmed by sequencing. An empty vector control was used to
examine if the transfection reagents or the transfection process
itself had any cytotoxic effects on the target cells. The
recombinant vectors or empty expression vector and packaging
vectors (pHelper 1.0 and pHelper 2.0; Shanghai GeneChem Co., Ltd.)
were then co-transfected into 293T cells (Invitrogen; Thermo Fisher
Scientific, Inc.) using Lipofectamine® 2000 (Invitrogen;
Thermo Fisher Scientific, Inc.). The culture supernatants were
collected at 48 h following transfection. All lentiviral vectors
expressed the enhanced green fluorescent protein, which enabled the
measurement and titration of expression efficiency in infected
cells. The cells were screened with puromycin (2 µg/ml) for 3
days.
Lentiviral infection of SiHa
cells
SiHa cells were purchased from the American Type
Culture Collection and cultured in RPMI-1640 (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% fetal bovine serum (FBS;
Gibco; Thermo Fisher Scientific, Inc.), 2 mM glutamine and 0.1 mM
nonessential amino acids at 37°C in a humidified chamber with 5%
CO2. The cells were divided into three groups: No viral
infection (NC), infection with control virus (LV-NC; empty vector)
and infection with lentiviral particles expressing
miR-1246-Inh (LV-miR-1246-Inh; multiplicity of
infection, 50).
Cell proliferation assay
Cell proliferation was assayed using a Cell Counting
Kit-8 (CCK-8; Promega Corporation), according to the manufacturer's
protocol. SiHa cells were seeded in 96-well plates at a density of
1,000–2,000 cells/well in 100 µl complete RPMI-1640 with 10% FBS.
Cell proliferation was assayed every 24 h. Briefly, the CCK-8
solution (10 µl) was added to each well and incubated for 2 h at
37°C. Next, the absorbance of each well was measured
spectrophotometrically at 450 nm using a TriStar microplate reader
(LB941; Berthold Technologies).
Detection of apoptosis by flow
cytometry
Cells (~2.5×106) were trypsinized,
collected, washed with phosphate buffered saline (PBS) and stained
with Annexin V-phycoerythrin (PE) and 7-aminoactinomycin D staining
using an PE Annexin V kit (cat. no. 556421; BD Biosciences) for 10
min at 4°C, according to the manufacturer's protocol. Apoptotic
cells were determined using a flow cytometer (FACScan; Becton
Dickinson; BD Biosciences). Flow cytometry data were analyzed using
ModFit LT software (version 2.0; Verity Software House, Inc.).
Cell cycle assay
Cells were fixed in 70% ethanol at −20°C for 24 h.
After washing with PBS, cells were treated with RNase A (50 µg/ml)
and stained with propidium iodide (25 µg/ml) in PBS for 30 min at
37°C. Samples were analyzed using a flow cytometer, and cell cycle
distribution was determined using ModFit LT software (version 2.0;
Verity Software House, Inc.). The proliferative index was
calculated as the percentage of cells in S/G2/M phase.
Matrigel invasion assay
The invasion activity of SiHa cells was measured
using Transwell chambers (Costar; Corning Inc.) and Matrigel (BD
Biosciences). In total, 1.5–2×104 cells/well in
RPMI-1640 with 10% FBS were seeded in the upper chamber that had
previously been coated with 1 mg/ml Matrigel. The lower chamber was
filled with RPMI-1640 containing 20% serum. Following a 24-h
incubation, the non-migrating cells in the upper chamber were
removed by gentle scraping, and the adherent cells on the lower
surface of the insert were stained with crystal violet for 15 min
at room temperature and imaged using a fluorescence microscope
(Olympus TH4-200). Automated quantification and cell counting were
performed using ImageJ Fiji software (National Institutes of
Health).
Reverse transcription quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cultured cells using
TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.) and
reverse transcription was performed using a Takara PrimeScript RT
reagent kit (Takara Bio, Inc.). Each RT reaction included 150 ng
template RNA and a pool of RT primers. qPCR was performed using
TaKaRa SYBR® Premix Ex Taq™ II kit (Takara Bio) and the
following primer sequences (Shanghai GenePharma Co., Ltd.):
miR-1246 forward, 5′-TACGAAATGGATTTTTGGAGCAGG-3′ and
U6 forward, 5′-ATTGGAACGATACAGAGAAGATT-3′, along with the
universal reverse primer, 5′-GTCCTTGGTGCCCGAGTG-3′. The reaction
conditions were 40 amplification cycles of 95°C for 3 min, 95°C for
15 sec and 62°C for 30 sec using Applied Biosystems ABI 7500
(Applied Biosystems; Thermo Fisher Scientific, Inc.) quantitative
Real-Time PCR System. U6 was used as a reference for miRNAs.
Each sample was analyzed in triplicate. Comparative threshold cycle
method-fold change (2−ΔΔCq) was used to analyze relative
changes (14).
Western blotting
Xenograft tumor tissues were lysed with
radioimmunoprecipitation assay lysis and extraction buffer (Thermo
Fisher Scientific, Inc.). Proteins (20 µg/lane) were separated by
8% SDS-PAGE and transferred to polyvinylidene difluoride membranes
(EMD Millipore) by electroblotting. Membranes were blocked with 5%
BSA in TBST (10 mM Tris-HCl, pH 8.0, 150 mM NaCl and 0.05%
Tween-20) for 1–1.5 h, and then incubated with the following
primary antibodies (Abcam) overnight at 4°C: THBS2 (1:500 dilution;
cat. no. ab84469), MMP2 (1:5,000 dilution; cat. no. ab37150), MMP9
(1:5,000 dilution; cat. no. ab38898), ECM components (1:1,000
dilution; cat. no. ab130585) and β-actin (1:5,000 dilution; cat.
no. ab228001). Subsequent to washing, the blots were incubated with
goat anti-rabbit immunoglobulin G (H+L)-horseradish peroxidase
secondary antibodies (1:5,000 dilution; cat. no. BS13278; Bioworld
Technology, Inc.) and visualized using super enhanced
chemiluminescence detection reagent (Amersham; GE Healthcare). Band
intensity was scanned using Bio-Rad Universal Hood II (Bio-Rad
Laboratories, Inc.) and analyzed using the Image Lab™ software
(version 5.1; Bio-Rad Laboratories, Inc.) and normalized to the
expression of β-actin.
Tumor xenograft model
Athymic BALB/c nude female mice (4–6 weeks old; body
weight, 15–18 g) were obtained and raised in the Animal
Experimental Center of Guangxi Medical University (Guangxi, China)
in solid-bottomed cages. Cages were sanitized once weekly.
Environmental conditions were maintained at 21±2°C with 50±20%
relative humidity and 15 air changes hourly. Animals were kept on a
12-h light/dark cycle and provided ad libitum access to
water and food. Mice were randomly assigned to one of three groups
(16 mice/group). SiHa cells (NC, LV-NC or LV-miR-1246-Inh)
were trypsinized and resuspended to a final concentration of
5×106 cells/0.1 ml PBS. The cells were then injected
subcutaneously into the left flanks of the mice. Four weeks later,
mice were euthanized by exposure to carbon dioxide with a flow rate
of 9 l/min (volume of the chamber, 30 l). Death was confirmed by
physical examination. Lack of breath, heartbeat and eyelid reflex
was observed for 10 min. Tumor size was monitored every 2 days by
measuring length and width with calipers, and volume was calculated
according to the formula: Volume (mm3)=width2
(mm2) × length (mm)/2 (15). Tumor growth rate is expressed as the
change in tumor volume over the number of days from the initial
cell injection. Animal experiments were conducted according to the
institutional guidelines of the National Research Council's Guide
for the Care and Use of Laboratory Animals (China) and were
approved by the Institutional Experimental Animals Review Board of
Guangxi Medical University (Guangxi, China).
Immunohistochemistry
The Ki-67 protein is expressed in all phases of the
cell cycle except G0 and serves as a good marker for
proliferation (16). The mice were
sacrificed and the tumors were fixed in 10% formalin at 4°C
overnight. Immunohistochemistry was performed using 3-µm-thick
paraffin sections. Paraffin sections were dewaxed and rehydrated
through xylene and a graded alcohol series. Endogenous peroxidase
activity was blocked with 3% hydrogen peroxide for 15 min at
20–25°C. Following washing in water, nonspecific binding sites were
blocked with 5% bovine serum albumin (cat. no. A2153;
Sigma-Aldrich; Merck KGaA) in PBS for 10 min at 20–25°C. The slides
were then incubated with a primary polyclonal antibody against Ki67
(cat. no. ab15580; 1:1,000; Abcam) at 4°C overnight. The slides
were then gently rinsed with PBS and developed using the Envision
system/HRP (cat. no. K400911; Dako; Agilent Technologies, Inc.) for
30 min and substrate-chromogen (3,3′-diaminobenzidine) at room
temperature. The nuclei were counterstained with Mayer's
hematoxylin for 3 min at room temperature. The percentage of
positive stains under a fluorescent microscope (Olympus X71) was
recorded. The Ki-67 proliferation index was automatically evaluated
using the ImageJ plugin ‘IHC profiler’ (17).
To quantify angiogenesis, microvessel density (MVD)
was assessed by immunostaining with an anti-CD31 antibody (no
dilution; cat. no. MAB-0031; Fuzhou Maixin Biotech Co., Ltd.), as
previously described (18). The
sections were observed at a low magnification (×100), and the
densest area of microvessels was selected and counted at a high
magnification. Microvessel number was recorded by counting any
positively stained endothelial cell or endothelial cell cluster as
a single, countable microvessel in a high-power field (HPF; ×200).
The 10 most neovascularized regions were selected for each sample.
The mean of the top three counts was used as the microvessel count
for each case.
Statistical analysis
Data are expressed as the mean ± standard deviation
of three independent experiments, each performed in triplicate.
Data were expressed as means with standard error. SPSS software was
used for statistical analysis (version 23.0; IBM Corp.).
Kolmogorov-Smirnov was used to test for normality. Differences
between groups were assessed by two-way repeated measures ANOVA and
multiple comparisons by least significant difference (LSD) post hoc
test when the data had a normal distribution (tumor xenograft
model). The remaining data did not display normal distribution, and
was therefore analyzed with non-parametric methods; Kruskal-Wallis
H test comparing three groups and Mann-Whitney U test as a post hoc
test between any two groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
Downregulation of miR-1246 inhibits
proliferation, and induces apoptosis of SiHa cells
SiHa cells were infected with LV-miR-1246-Inh
and a stable anti-miR-1246 SiHa cell line was established.
Control lentiviruses were used to infect SiHa cells as control.
Compared with the expression in the control group,
LV-miR-1246-Inh-infected SiHa cells showed significant
downregulation of miR-1246 (P=0.001; Fig. 1A), indicating stable knockdown of
miR-1246.
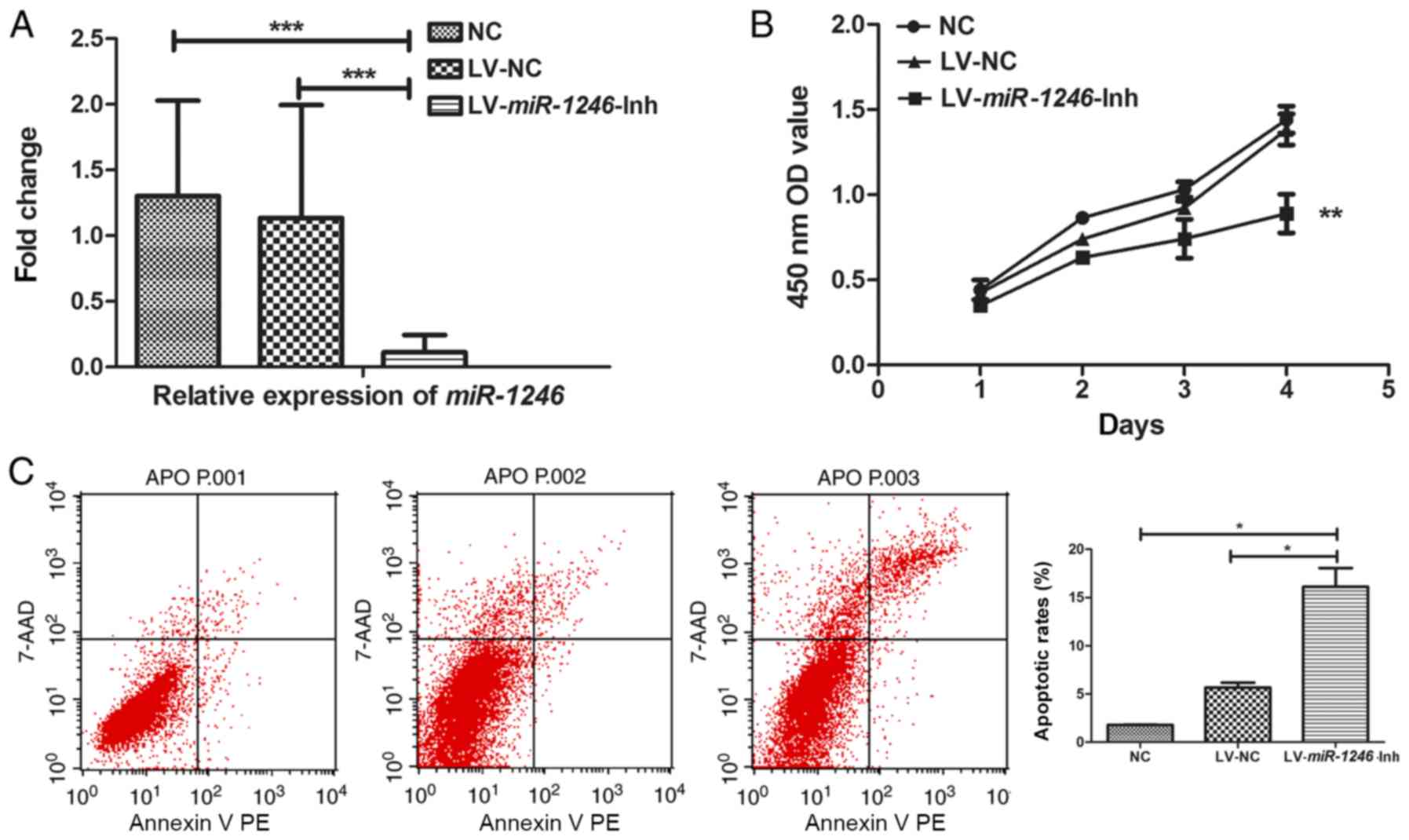 | Figure 1.SiHa cell growth, cell cycle and
invasion following miR-1246 downregulation. (A)
miR-1246 expression in SiHa cells following infection with
LV-miR-1246-Inh or LV-NC compared with untreated NC. (B)
Proliferation of SiHa cells following miR-1246
downregulation. (C) Cell apoptosis detected by Annexin V-PE and
7-AAD labelling and flow cytometry. (D) Matrigel cell invasion
assay showing the effects of LV-miR-1246-Inh on cell
invasion. Magnification, ×200. (E and F) Cell cycle distribution
determined by propidium iodide staining and flow cytometry
highlighting the effects of LV-miR-1246-Inh on cell
populations in the G1 and S phases. *P<0.05, **P<0.01,
***P<0.001. miR-1246, microRNA-1246; LV, lentivirus; Inh,
inhibitor; NC, negative control; PE, phycoerythrin; 7-AAD,
7-aminoactinomycin D; OD, optical density. |
Next, the role of miR-1246 in various cell
processes, including proliferation, apoptosis and invasion was
evaluated. In miR-1246-Inh-infected SiHa cells,
proliferation was significantly suppressed compared with that in
the control groups (NC and LV-NC; Fig.
1B). Flow cytometry revealed average apoptotic rates of 1.78,
6.00 and 16.10% in the NC, LV-NC and LV-miR-1246-Inh groups,
respectively. The apoptotic rate of the LV-miR-1246-Inh group was
increased compared with that in the NC and LV-NC groups (both
P=0.040; Fig. 1C), indicating that
miR-1246 downregulation significantly increased apoptosis in
this cervical cancer cell line. Furthermore, in the
LV-miR-1246-Inh group, the number of cells that invaded the
Transwell Matrigel-coated membrane was 71.3±4.27, while that in the
NC and LV-NC groups were 187.5±4.79 and 162.5±4.78, respectively
(Fig. 1D), demonstrating altered
cell invasion upon miR-1246 knockdown (P=0.013; Fig. 1D).
Downregulation of miR-1246 induces
cell cycle arrest in the G1/S phase
Given that miR-1246 downregulation inhibited
SiHa cell proliferation, the effect of decreased miR-1246 on
cell cycle progression was determined. Analysis of the cell cycle
distribution of SiHa cells indicated that the average number of
cells in G1 phase increased in cells infected with
LV-miR-1246-Inh (78.59%) compared with LV-NC (71.52%,
P=0.042) and NC (61.79%, P=0.042; Fig.
1E). Additionally, the cell population in the S phase was
reduced sharply in the LV-miR-1246-Inh group (10.48%)
compared with the LV-NC (24.21%, P=0.027) and NC groups (15.49%,
P=0.027; Fig. 1F).
Downregulation of miR-1246 inhibits
tumor growth
To elucidate the role of miR-1246 in tumor
growth, SiHa cells of each treatment group (NC, LV-NC and
LV-miR-1246-Inh) were injected into nude mice, and the
growth of the xenograft tumors was monitored. Each subgroup
contained 5–6 mice, and the experiment was repeated three times
(n=16) for each group. Mice were euthanized when the average tumor
volume was >500 mm3. During the four weeks of
observation, three mice in the NC group and one mouse in the LV-NC
group <5 weeks old died of anorexia and injury in the third
week, although the skin was disinfected and the mice were shifted
to another cage to ensure sufficient access to food. The tumor
volume in the NC group was lower on day 17 following inoculation
compared with that in the LV-miR-1246-Inh group (Fig. 2B; P<0.05). The xenograft tumors
derived from cells with miR-1246 downregulation grew slowly
compared with those in the two control groups, indicating that
downregulation of miR-1246 markedly inhibited tumor
development (Fig. 2A and B).
Antiproliferative and antiangiogenic
effect of miR-1246 downregulation in xenograft tumors
To investigate the growth of the xenograft tumors
from the different groups, the Ki-67 proliferative index was used
as a measure of the proliferative activity. Fig. 3A shows representative immunostaining
images for the three groups. In the LV-miR-1246-Inh group,
Ki67 level (60.93±13.36%) was significantly lower compared with
that in the control groups (NC, 89.8±2.76%; LV-NC, 86.54±6.03%;
P<0.001).
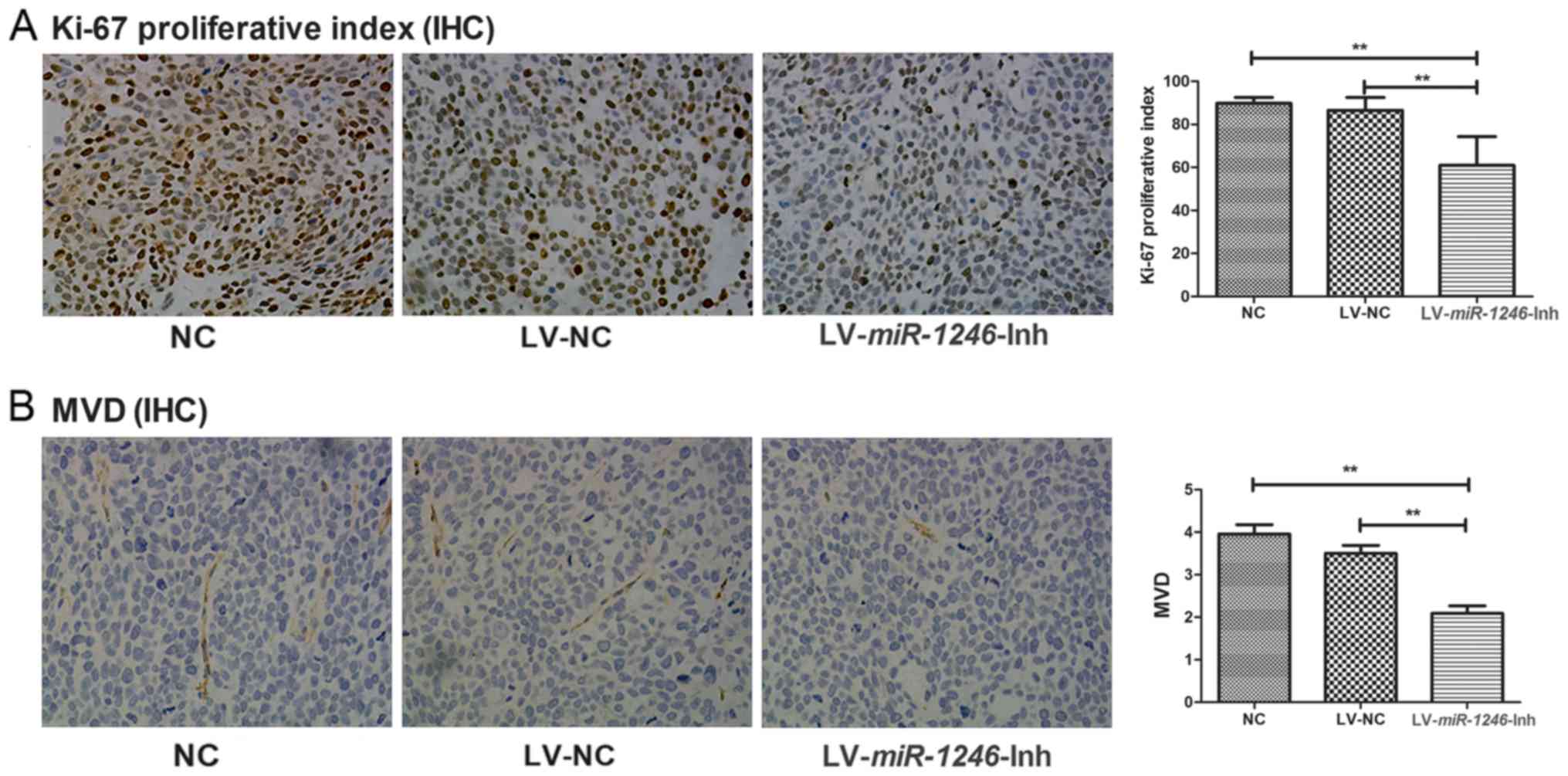 | Figure 3.Antiproliferative and antiangiogenic
effects of miR-1246 downregulation in tumor xenografts. (A)
Expression of Ki67 proliferation marker in the following groups:
NC, LV-NC and LV-miR-1246-Inh. Magnification, ×200. (B)
CD-31 expression in the following groups: NC, LV-NC and
LV-miR-1246-Inh. Magnification, ×200. **P<0.01.
miR-1246, microRNA-1246; LV, lentivirus; IHC,
immunohistochemistry; Inh, inhibitor; MVD, microvessel density; NC,
negative control. |
To investigate the effect of miR-1246
downregulation on angiogenesis, MVD was assessed following CD31
staining. The MVD of the NC and LV-NC group were similar (3.95±1.48
and 3.58±1.19 microvessels/HPF, respectively; Fig. 3B). However, the MVD was significantly
lower in the LV-miR-1246-Inh group (2.08±1.20
microvessels/HPF; P<0.001) than in the NC and LV-NC groups.
Downregulation of miR-1246 increases
THBS2 protein expression and induces changes in the THBS2/MMP
signaling pathway
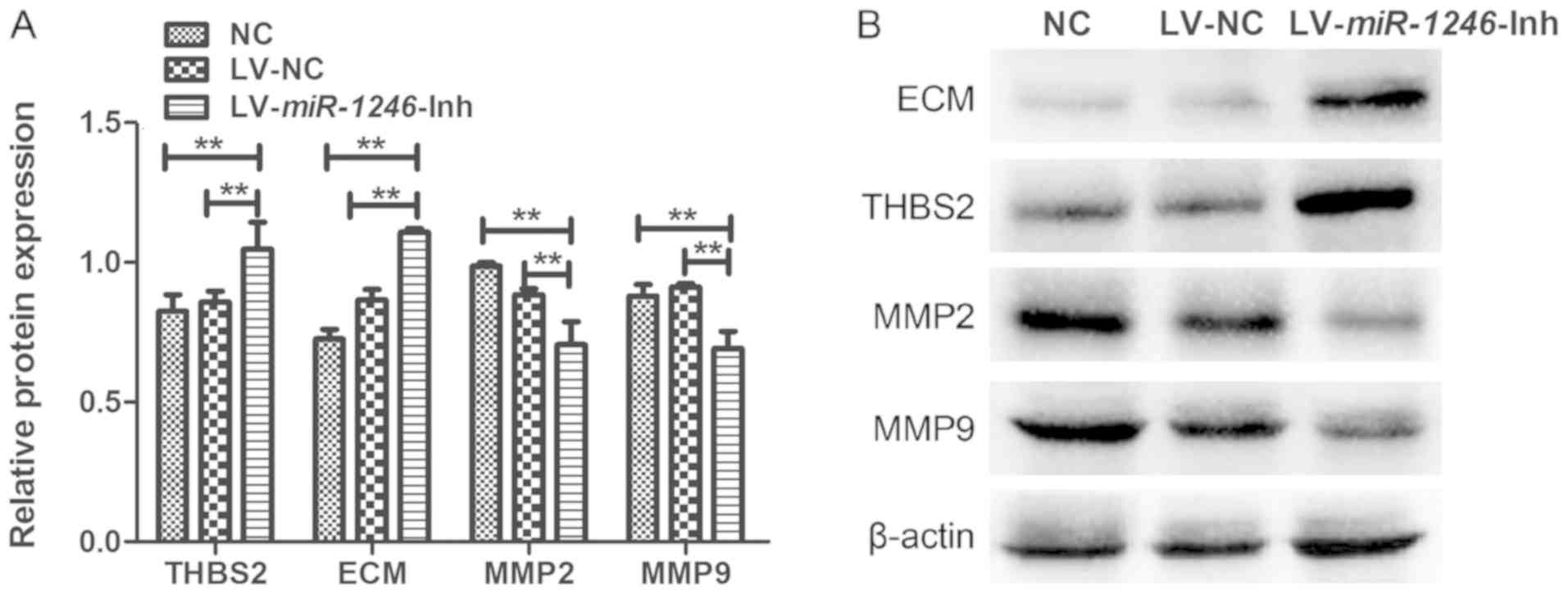
THBS2 is a predicted miR-1246 target gene
(10). To evaluate whether THBS2 was
affected by miR-1246 downregulation, the protein level of
THBS2 was examined by western blotting. THBS2 protein expression in
cells infected with LV-miR-1246-Inh was significantly higher
than that observed in the control groups (Fig. 4A). These data suggest that
downregulation of miR-1246 leads to increased expression of
THBS2 in SiHa cells.
The expression levels of ECM, MMP2 and MMP9 proteins
were also examined to further investigate the role of
miR-1246 in regulating THBS2 signaling (Fig. 4). The expression of ECM components
was increased following miR-1246 knockdown compared with
that in the LV-NC and NC groups. In contrast, MMP2 and MMP9 levels
were decreased in the miR-1246-silenced cells compared with
the LV-NC and NC groups. There were no significant differences in
the expression levels of these proteins between the NC and LV-NC
groups. Thus, downregulation of miR-1246 in SiHa cervical
cancer cells significantly elevated the expression of THBS2 and
ECM, and decreased expression of MMP2 and MMP9, suggesting that
miR-1246 inhibits the THBS2 signaling pathway.
Discussion
A number of tumor-suppressor genes and oncogenes are
aberrantly expressed in CSCC, including the pro-apoptotic protein
Bax, which is frequently inactivated in carcinoma, and tumor
protein p53, which serves a key role in cervical cancer (19). Previous studies (20–22) have
provided significant new information concerning the function of
noncoding RNAs, including miRNAs, in cervical cancer pathogenesis.
In the present study, the role of miR-1246 in SiHa cervical
cancer cells and tumor xenografts was investigated. Lentiviruses
expressing shRNA against miR-1246 were used to downregulate
miR-1246 expression, and the effects of miR-1246
downregulation on cellular processes and tumor growth were
evaluated, with particular emphasis on the THBS2/ECM/MMP signaling
pathway.
miR-1246 was identified in 2008 during
microRNA profiling of human stem cells (23). In 2015, Krissansen et al
(24) revealed that miR-1246
was significantly upregulated in the sera of patients with
inflammatory bowel disease, Crohn's disease and ulcerative colitis
compared with healthy individuals. Furthermore, in a study
evaluating 168 patients with cancer and 65 healthy controls,
Todeschini et al (25)
identified miR-1246 as a potential diagnostic biomarker for
high-grade serous ovarian carcinoma, with a sensitivity of 87%, a
specificity of 77%, and an accuracy of 84%. The link between
miR-1246 and cancer pathogenesis has been investigated in
other studies. Zhang et al (26) showed that miR-1246 confers
tumorigenicity and is required for lung cancer metastasis in
non-small cell lung cancer (NSCLC) via the tumor-initiating cell
model. Inhibition of miR-1246 appeared to decrease stemness
and epithelial-mesenchymal transition in NSCLC, in addition to
suppressing proliferation, sphere-formation, colony formation and
invasion (27). Additionally,
Hasegawa et al (28)
demonstrated that miR-1246 increased the tumor-initiating
potential of cells and induced drug resistance in vivo, and
it was showed that high miR-1246 expression correlated with
poor prognosis in patients.
Notably, the present study is not the first to
investigate the molecular mechanism of miR-1246 in cervical
cancer. In our own laboratory, the full miRNA profile of patients
with CSCC was screened and it was demonstrated that miR-1246
is expressed at similar levels in the serum and cervical tumor
tissues of patients with lymph node metastasis (10). These data are consistent with the
study by Takeshita et al (29), which reported that miR-1246
expression can be used to differentiate patients with esophageal
squamous cell carcinoma from healthy individuals, indicating that
this miRNA may also have applications as a marker for lymph node
metastasis in CSCC. Despite significant efforts to understand
miR-1246 function in CSCC and other type of cancer, its
mechanism remains unclear.
In the present study, miRNA knockdown was performed
to evaluate the role of miR-1246 in CSCC tumorigenesis and
progression. The role of miR-1246 in these processes has
been previously investigated in a similar study using the same cell
line. The expression of miR-1246 has been transiently
upregulated or downregulated by transfection of analogs and
antagonists with Lipofectamine® 2000 (11). THBS2 has been characterized as a
functional target of miR-1246 by luciferase reporter gene assays
and western blot analysis (11).
However, in the present study, the effects of
LV-miR-1246-Inh infection in SiHa cells were evaluated in
more depth, focusing on cell proliferation, apoptosis and invasion
following stable miR-1246 knockdown. Additionally, the
present study investigated the role of the THBS2/ECM signaling
pathway in cervical cancer using a tumor xenograft model. The
results of the current study indicated that, compared with control
cells, the growth rate of cells infected with
LV-miR-1246-Inh was reduced, as was the capacity for
invasion. Apoptosis of the cervical cells was increased when
miR-1246 was knocked down. Moreover, downregulation of
miR-1246 arrested the cell cycle in the G1 phase.
Further studies of the molecular mechanisms underlying this
phenomenon are required to identify the changes in regulatory
proteins that control the G1/S transition. These results
indicate that miR-1246 may function as an oncogene in CSCC
development and progression, and that miR-1246
downregulation inhibits tumor growth and impairs cervical cancer
cell invasion.
Notably, the identification of
miR-1246-specific target genes has provided new insights
into the mechanisms underlying its role in cancer development.
THBS2 (also called TSP2) is an miR-1246 target
gene (11). THBS gene and/or protein
expression is significantly correlated with decreased microvessel
count in oral cancer (30), gastric
cancer (31), colorectal cancer
(32), NSCLC (33), glioma (34) and pancreatic cancer (35). In cervical cancer, the microvessel
count in patients lacking THBS2 expression is higher than
that observed in patients expressing it, although this difference
is not statistically significant (36). In the present study, inhibition of
miR-1246 was associated with THBS2 upregulation. The tumor
growth index was decreased in the xenograft tumors of the
LV-miR-1246-Inh group, consistent with the in vitro
results showing increased apoptosis and cell cycle arrest in SiHa
cells with miR-1246 knocked down. In addition, in the
LV-miR-1246-Inh group, MVD was decreased and THBS2
expression was increased. It has been suggested that THBS2 serves a
unique role in the control of angiogenesis, as well as tumor growth
and metastasis, and downregulation of THBS2 has been associated
with tumor progression and poor prognosis in cervical cancer
(36). In the present study,
miR-1246 downregulation resulted in increased expression of
THBS2, but decreased expression of MMP2 and MMP9. These data are
consistent with a previous study showing that THBS2 overexpression
downregulated MMP2 and MMP9 expression in a human colon carcinoma
cell line (37). However, a study by
Chen et al (38) found
conflicting results, as THBS2 was observed to promote prostate
cancer bone metastasis via MMP2 upregulation. These data suggest
that the role of THBS2 may differ among different cancer types.
Degradation of the basement membrane and remodeling
of the ECM by matrix metalloproteinases, including MMP2 and MMP9 is
essential in tumor invasion and metastasis (39). Hirose et al (40) previously demonstrated that decreased
THBS2 affects the levels of MMP2 and MMP9, and the THBS2/MMP
interaction directly modulates the metabolism of the ECM. This
observation was supported by the results of the present study, as
ECM proteins levels, potentially upregulated due to THBS2/MMP
interaction, were altered with miR-1246 downregulation.
Taken together, these data show that miR-1246 may serve a
distinct role in cervical cancer cell growth and tumor pathogenesis
by regulating THBS2 expression, which subsequently alters the
expression and function of MMPs. These changes have a profound
impact on the ECM and ECM-related processes, including invasion,
proliferation and apoptosis.
In summary, the present study provides evidence of
the essential biological role of miR-1246 in cervical cancer
pathogenesis and identifies the THBS2/MMP/ECM pathway as a
potential target in cervical cancer cells. These findings provide a
hypothesis for the future use of miR-1246 suppression in the
treatment of cervical cancer. Although large-scale and long-term
follow-up studies are needed to confirm the role of miR-1246
and THBS2 in cervical cancer, the present study firmly supports the
essential role of miR-1246 in cervical cancer cell invasion
and metastasis.
Acknowledgements
Not applicable.
Funding
The study was supported by the National Natural
Science Foundation of China (grant no. 81460398).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
DSY, JYC and PD contributed to the study design and
conception. PD, YHL and ND contributed to the collection, analysis
and interpretation of data, and PD contributed to the writing of
the manuscript.
Ethics approval and consent to
participate
Animal experiments were conducted according to the
institutional guidelines of the National Research Council's Guide
for the Care and Use of Laboratory Animals (China) and were
approved by the Institutional Experimental Animals Review Board of
Guangxi Medical University (Guangxi, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CSCC
|
cervical squamous cell carcinoma
|
|
THBS2
|
TSP2, thrombospondin-2
|
|
MMPs
|
matrix metalloproteinases
|
|
FBS
|
fetal bovine serum
|
|
NC
|
negative control
|
|
NSCLC
|
non-small cell lung cancer
|
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cibula D, Abu-Rustum NR, Fischerova D,
Pather S, Lavigne K, Slama J, Alektiar K, Ming-Yin L, Kocian R,
Germanova A, et al: Surgical treatment of ‘intermediate risk’ lymph
node negative cervical cancer patients without adjuvant
radiotherapy-A retrospective cohort study and review of the
literature. Gynecol Oncol. 151:438–443. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schwarz DS, Hutvagner G, Du T, Xu ZS,
Aronin N and Zamore PD: Asymmetry in the assembly of the RNAi
enzyme complex. Cell. 115:199–208. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gregory RI, Chendrimada TP, Cooch N and
Shiekhattar R: Human RISC couples microRNA biogenesis and
posttranscriptional gene silencing. Cell. 123:631–640. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pedroza-Torres A, Lopez-Urrutia E,
Garcia-Castillo V, Jacobo-Herrera N, Herrera LA, Peralta-Zaragoza
O, López-Camarillo C, De Leon DC, Fernández-Retana J, Cerna-Cortés
JF, et al: MicroRNAs in cervical cancer: Evidences for a miRNA
profile deregulated by HPV and its impact on radio-resistance.
Molecules. 19:6263–6281. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hu X, Schwarz JK, Lewis JS Jr, Huettner
PC, Rader JS, Deasy JO, Grigsby PW and Wang X: A microRNA
expression signature for cervical cancer prognosis. Cancer Res.
70:1441–1448. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ribeiro J and Sousa H: MicroRNAs as
biomarkers of cervical cancer development: A literature review on
miR-125b and miR-34a. Mol Biol Rep. 41:1525–1531. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fiannaca A, La Rosa M, La Paglia L, Rizzo
R and Urso A: Analysis of miRNA expression profiles in breast
cancer using biclustering. BMC Bioinformatics. 16 (Suppl 4):S72015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liao JM, Zhou X, Zhang Y and Lu H:
MiR-1246: A new link of the p53 family with cancer and Down
syndrome. Cell Cycle. 11:2624–2630. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen J, Yao D, Li Y, Chen H, He C, Ding N,
Lu Y, Ou T, Zhao S, Li L and Long F: Serum microRNA expression
levels can predict lymph node metastasis in patients with
early-stage cervical squamous cell carcinoma. Int J Mol Med.
32:557–567. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen J, Yao D, Zhao S, He C, Ding N, Li L
and Long F: MiR-1246 promotes SiHa cervical cancer cell
proliferation, invasion, and migration through suppression of its
target gene thrombospondin 2. Arch Gynecol Obstet. 290:725–732.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bao Y, Wang L, Shi L, Yun F, Liu X, Chen
Y, Chen C, Ren Y and Jia Y: Transcriptome profiling revealed
multiple genes and ECM-receptor interaction pathways that may be
associated with breast cancer. Cell Mol Biol Lett. 24:382019.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang Z, Kyriakides TR and Bornstein P:
Matricellular proteins as modulators of cell-matrix interactions:
Adhesive defect in thrombospondin 2-null fibroblasts is a
consequence of increased levels of matrix metalloproteinase-2. Mol
Biol Cell. 11:3353–3364. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Naito S, von Eschenbach AC, Giavazzi R and
Fidler IJ: Growth and metastasis of tumor cells isolated from a
human renal cell carcinoma implanted into different organs of nude
mice. Cancer Res. 46:4109–4115. 1986.PubMed/NCBI
|
|
16
|
Menon SS, Guruvayoorappan C, Sakthivel KM
and Rasmi RR: Ki-67 protein as a tumour proliferation marker. Clin
Chim Acta. 491:39–45. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Varghese F, Bukhari AB, Malhotra R and De
A: IHC Profiler: An open source plugin for the quantitative
evaluation and automated scoring of immunohistochemistry images of
human tissue samples. PLoS One. 9:e968012014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Weidner N, Semple JP, Welch WR and Folkman
J: Tumor angiogenesis and metastasis-correlation in invasive breast
carcinoma. N Engl J Med. 324:1–8. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
He L, He X, Lowe SW and Hannon GJ:
microRNAs join the p53 network-another piece in the
tumour-suppression puzzle. Nat Rev Cancer. 7:819–822. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang Y, Liu Y, Li G, Li L, Geng P and Song
H: microRNA-214 suppresses the growth of cervical cancer cells by
targeting EZH2. Oncol Lett. 16:5679–5686. 2018.PubMed/NCBI
|
|
21
|
Pedroza-Torres A, Campos-Parra AD,
Millan-Catalan O, Loissell-Baltazar YA, Zamudio-Meza H, Cantú de
León D, Montalvo-Esquivel G, Isla-Ortiz D, Herrera LA,
Ángeles-Zaragoza Ó, et al: MicroRNA-125 modulates radioresistance
through targeting p21 in cervical cancer. Oncol Rep. 39:1532–1540.
2018.PubMed/NCBI
|
|
22
|
González-Quintana V, Palma-Berré L,
Campos-Parra AD, López-Urrutia E, Peralta-Zaragoza O, Vazquez-Romo
R and Pérez-Plasencia C: MicroRNAs are involved in cervical cancer
development, progression, clinical outcome and improvement
treatment response (Review). Oncol Rep. 35:3–12. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Morin RD, O'Connor MD, Griffith M,
Kuchenbauer F, Delaney A, Prabhu AL, Zhao Y, McDonald H, Zeng T,
Hirst M, et al: Application of massively parallel sequencing to
microRNA profiling and discovery in human embryonic stem cells.
Genome Res. 18:610–621. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krissansen GW, Yang Y, McQueen FM, Leung
E, Peek D, Chan YC, Print C, Dalbeth N, Williams M and Fraser AG:
Overexpression of miR-595 and miR-1246 in the sera of patients with
active forms of inflammatory bowel disease. Inflamm Bowel Dis.
21:520–530. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Todeschini P, Salviato E, Paracchini L,
Ferracin M, Petrillo M, Zanotti L, Tognon G, Gambino A, Calura E,
Caratti G, et al: Circulating miRNA landscape identifies miR-1246
as promising diagnostic biomarker in high-grade serous ovarian
carcinoma: A validation across two independent cohorts. Cancer
Lett. 388:320–327. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang WC, Chin TM, Yang H, Nga ME, Lunny
DP, Lim EK, Sun LL, Pang YH, Leow YN, Malusay SR, et al:
Tumour-initiating cell-specific miR-1246 and miR-1290 expression
converge to promote non-small cell lung cancer progression. Nat
Commun. 7:117022016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim G, An HJ, Lee MJ, Song JY, Jeong JY,
Lee JH and Jeong HC: Hsa-miR-1246 and hsa-miR-1290 are associated
with stemness and invasiveness of non-small cell lung cancer. Lung
Cancer. 91:15–22. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hasegawa S, Eguchi H, Nagano H, Konno M,
Tomimaru Y, Wada H, Hama N, Kawamoto K, Kobayashi S, Nishida N, et
al: MicroRNA-1246 expression associated with CCNG2-mediated
chemoresistance and stemness in pancreatic cancer. Br J Cancer.
111:1572–1580. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Takeshita N, Hoshino I, Mori M, Akutsu Y,
Hanari N, Yoneyama Y, Ikeda N, Isozaki Y, Maruyama T, Akanuma N, et
al: Serum microRNA expression profile: miR-1246 as a novel
diagnostic and prognostic biomarker for oesophageal squamous cell
carcinoma. Br J Cancer. 108:644–652. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kishi M, Nakamura M, Nishimine M, Ishida
E, Shimada K, Kirita T and Konishi N: Loss of heterozygosity on
chromosome 6q correlates with decreased thrombospondin-2 expression
in human salivary gland carcinomas. Cancer Sci. 94:530–535. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ao R, Guan L, Wang Y and Wang JN:
Silencing of COL1A2, COL6A3, and THBS2 inhibits gastric cancer cell
proliferation, migration, and invasion while promoting apoptosis
through the PI3k-Akt signaling pathway. J Cell Biochem.
119:4420–4434. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tokunaga T, Nakamura M, Oshika Y, Abe Y,
Ozeki Y, Fukushima Y, Hatanaka H, Sadahiro S, Kijima H, Tsuchida T,
et al: Thrombospondin 2 expression is correlated with inhibition of
angiogenesis and metastasis of colon cancer. Br J Cancer.
79:354–359. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Weng TY, Wang CY, Hung YH, Chen WC, Chen
YL and Lai MD: Differential expression pattern of THBS1 and THBS2
in lung cancer: Clinical outcome and a systematic-analysis of
microarray databases. PLoS One. 11:e1610072016. View Article : Google Scholar
|
|
34
|
Fears CY, Grammer JR, Stewart JE Jr, Annis
DS, Mosher DF, Bornstein P and Gladson CL: Low-density lipoprotein
receptor-related protein contributes to the antiangiogenic activity
of thrombospondin-2 in a murine glioma model. Cancer Res.
65:9338–9346. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nakamura M, Oida Y, Abe Y, Yamazaki H,
Mukai M, Matsuyama M, Chijiwa T, Matsumoto H and Ueyama Y:
Thrombospondin-2 inhibits tumor cell invasion through the
modulation of MMP-9 and uPA in pancreatic cancer cells. Mol Med
Rep. 1:423–427. 2008.PubMed/NCBI
|
|
36
|
Kodama J, Hashimoto I, Seki N, Hongo A,
Yoshinouchi M, Okuda H and Kudo T: Thrombospondin-1 and −2
messenger RNA expression in invasive cervical cancer: Correlation
with angiogenesis and prognosis. Clin Cancer Res. 7:2826–2831.
2001.PubMed/NCBI
|
|
37
|
Kamochi J, Tokunaga T, Tomii Y, Abe Y,
Hatanaka H, Kijima H, Yamazaki H, Watanabe N, Matsuzaki S, Ueyama Y
and Nakamura M: Overexpression of the thrombospondin 2 (TSP2) gene
modulated by the matrix metalloproteinase family expression and
production in human colon carcinoma cell line. Oncol Rep.
10:881–884. 2003.PubMed/NCBI
|
|
38
|
Chen PC, Tang CH, Lin LW, Tsai CH, Chu CY,
Lin TH and Huang YL: Thrombospondin-2 promotes prostate cancer bone
metastasis by the up-regulation of matrix metalloproteinase-2
through down-regulating miR-376c expression. J Hematol Oncol.
10:332017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Davies KJ: The complex interaction of
matrix metalloproteinases in the migration of cancer cells through
breast tissue stroma. Int J Breast Cancer. 2014:8390942014.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hirose Y, Chiba K, Karasugi T, Nakajima M,
Kawaguchi Y, Mikami Y, Furuichi T, Mio F, Miyake A, Miyamoto T, et
al: A functional polymorphism in THBS2 that affects alternative
splicing and MMP binding is associated with lumbar-disc herniation.
Am J Hum Genet. 82:1122–1129. 2008. View Article : Google Scholar : PubMed/NCBI
|