Introduction
The basic etiology and pathogenesis of
nasopharyngeal carcinoma is a low hemoglobin level, exhaustion and
coagulation of phlegm, and the incidence rate is 10–30 cases per
100,000 in the general population (1,2). The key
to the recurrence and metastasis of cancer is that immune cells
fail to suppress cancer cells (3).
NPC is curable when diagnosed early and a 5-year survival as high
as 90% may be achieved (4). Once
cancer cells are generated, the body struggles to eliminate them
(4). During pre-surgical resection
of cancer or during the early stage of the disease, there may be
micrometastasis (5). Following
surgical resection and radiotherapy, the micrometastasis can spread
and survive in the lymph system, blood circulation, bone marrow,
liver, lungs and other tissues and organs (6). Micrometastasis often has no obvious
clinical symptoms; however, the residual cancer cells can be the
key prerequisites for the recurrence and metastasis of tumors in
the clinic (6).
The p53 gene has been identified to be closely
associated with several types of human cancer, such as liver and
lung cancer, as well as nasopharyngeal carcinoma (7). The p53 gene was first discovered in
1979 and it has since been demonstrated to serve a number of
different roles, including as an oncogene and a tumor suppressor
gene (8). The p53 gene and the
protein it encodes are associated with cell cycle regulation, cell
growth and apoptosis, which are regulators of cell division
(8). Overexpression of p53 can
induce apoptosis of human cancer cells (9). Following specific inhibition of
caspase-8 and caspase-9, p53 can also be inhibited, which indicates
that p53 serves a role in death-receptor-mediated and
mitochondria-mediated apoptosis (10). Stable expression of the p53 protein
is crucial for the completion of its various functions.
Cyclin Bl serves an important regulatory role in the
G2/M stage of the cell cycle (11).
It has been reported that cell division cycle gene 2 (CDC2) and
cyclin B1 function together in eukaryotes. CDC2/cyclin B1 serves a
role at the G2/M phase of the cell cycle (12). Furthermore, CDC2/cyclin B1 can
accelerate the mitosis of cells, which is a process that is
mediated by phosphorylation and requires numerous different factors
(13). The kinase activity of
CDC2/cyclin B1 promotes mitosis via the G2/M phase (13).
Tanshinone IIA is a composition of active monomers
extracted from the traditional Chinese plant, Salvia
miltiorrhiza, which is commonly used for the treatment of
patients with cardiovascular disease, cerebrovascular disease or
hepatitis (14). Modern medicine has
demonstrated that the main effects of S. μiltiorrhiza
include dilation of blood vessels and the improvement of
microcirculation (15). In recent
years, a number of studies have focused on the use of traditional
Chinese medicines for the treatment of tumors (15–17).
Tanshinone IIA has been studied due to its potential antineoplastic
activity. Studies have demonstrated that tanshinone IIA exhibits a
specific cytotoxic effect on leukemia, hepatocellular carcinoma and
breast cancer cells (16,17). However, to the best of our knowledge,
the effects of tanshinone IIA on human nasopharyngeal carcinoma
cells remain unclear. The present study was designed to investigate
the anti-cancer effects of tanshinone IIA, particularly the
potential inhibition of proliferation and promotion of apoptosis of
human nasopharyngeal carcinoma cells. In addition, the possible
underlying mechanism was discussed.
Materials and methods
Cell culture and reagents
The human nasopharyngeal carcinoma cell line 13-9B
was purchased from the Shanghai Cell Bank of Chinese Academy of
Sciences (Shanghai, China) and cultured in RPMI-1640 complete
medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
containing 10% fetal bovine serum (Gibco; Thermo Fisher Scientific,
Inc.) in a humidified incubator containing 5% CO2 at
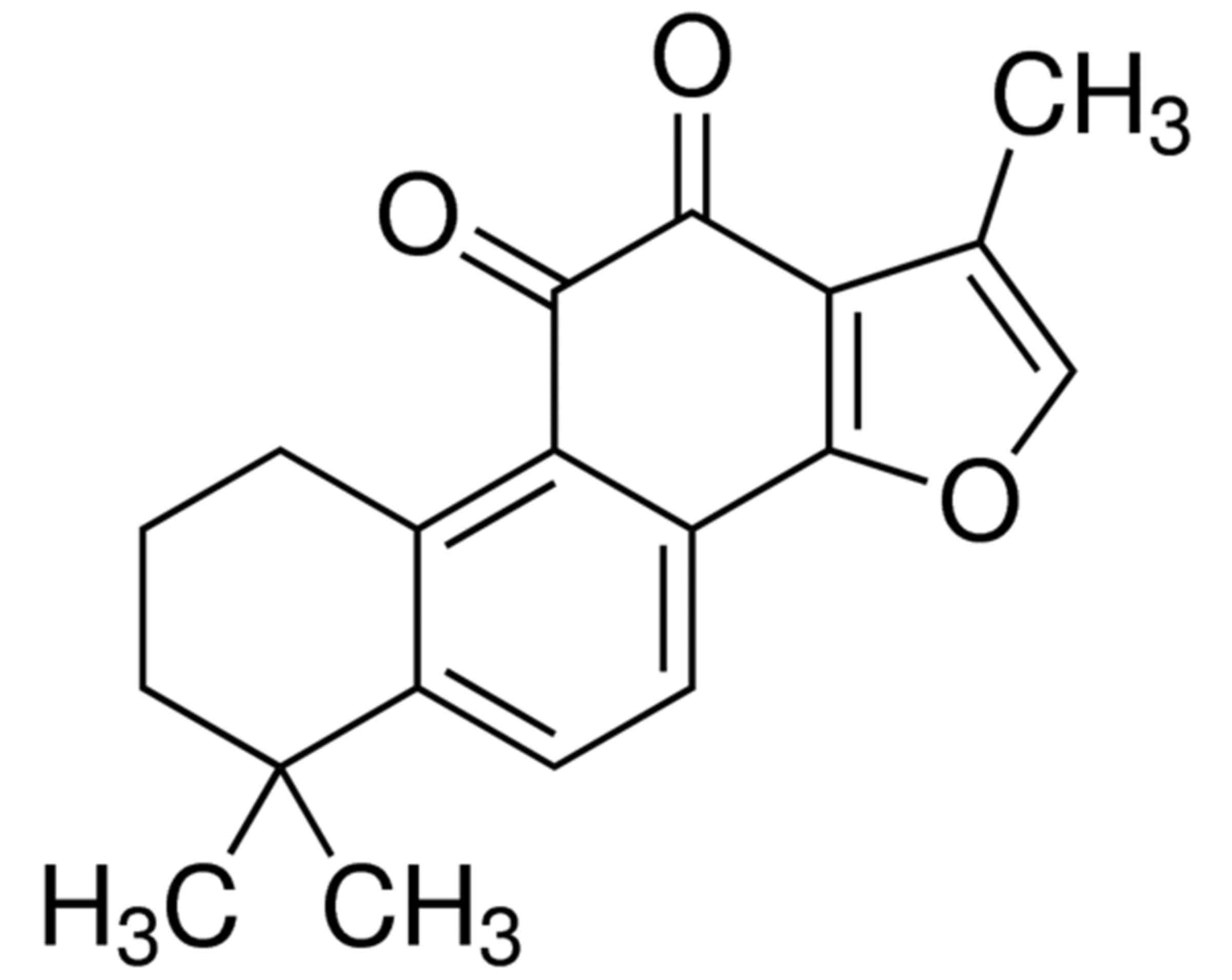
37°C. Tanshinone IIA [≥97% (HPLC); Fig.
1] was purchased from Sigma-Aldrich; Merck KGaA (Darmstadt,
Germany) and dissolved in DMSO to a concentration of 0, 5, 10, 20
or 25 µg/ml.
MTT assay
13-9B cells (1×103 cells/well) were
seeded in 96-well culture plates containing 0, 5, 10, 20 or 25
µg/ml tanshinone IIA with 100 µl growth medium and cultured for 24,
48 and 72 h at 37°C. Subsequently, 20 µl MTT (Sigma-Aldrich; Merck
KGaA) was added to each well and incubated at 37°C for 4 h. DMSO
(20 µl) was added to terminate the reaction and the absorbance was
measured at 490 nm using an automatic microplate reader.
Lactate dehydrogenase (LDH) assay
13-9B cells (1×103 cells/well) were
seeded in 96-well culture plates containing 0, 5, 10, 20 and 25
µg/ml tanshinone IIA with 100 µl growth medium and cultured for 24,
48 and 72 h at 37°C. Subsequently, 100 µl LDH assay substrate (cat.
no. C0017; Beyotime Institute of Biotechnology, Haimen, China) was
added to each well and the cells were further incubated for 30 min.
The absorbance was then measured at 490 nm using an automatic
microplate reader.
Flow cytometry
13-9B cells (1×106 cells/well) were
seeded in 6-well culture plates containing 0, 5, 10 or 20 µg/ml
tanshinone IIA with 2 ml growth medium and cultured for 24 and 48 h
at 37°C. Subsequently, 13-9B cells were stained with an Annexin
V/propidium iodide apoptosis detection kit (Thermo Fisher
Scientific, Inc.) for 15 min at room temperature. Apoptotic cells
were then analyzed using a flow cytometer (FACScalibur; Becton,
Dickinson and Company, Franklin Lakes, NJ, USA) and FlowJo version
7.6.1 software (FlowJo, LLC, Ashland, OR, USA).
Measurement of caspase-3 activity
13-9B cells (1×106 cells/well) were
seeded in 6-well culture plates containing 0, 5, 10 or 20 µg/ml
tanshinone IIA with 2 ml growth medium and cultured for 48 h at
37°C. Subsequently, 50 µg protein extract from 13-9B cells was
incubated and added to a reaction buffer containing Ac-dEVd-pNA
(cat. no. C1116; Beyotime Institute of Biotechnology) at 37°C for 4
h. The absorbance was measured at 405 nm using an automatic
microplate reader.
Western blot analysis
13-9B cells (1×106 cells/well) were
seeded in 6-well culture plates containing 0, 5, 10 or 20 µg/ml
tanshinone IIA with 2 ml growth medium and cultured for 48 h at
37°C. Subsequently, 13-9B cells were harvested and lysed in
ice-cold RIPA buffer (Beyotime Institute of Biotechnology)
containing 20 mM Tris-HCl (pH 7.5) for 5–10 min. The supernatants
were collected following centrifugation at 12,000 × g for 10
minutes at 4°C and the protein concentration was determined using a
Bradford protein assay (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). Equal amount of proteins (50 µg/lane) were subjected to 10%
SDS-PAGE and then electronically transferred onto a PVDF membrane
(EMD Millipore, Billerica, MA, USA). The blot was blocked with TBS
and 0.1% Tween-20 containing 10% non-fat milk at room temperature
for 1 h. The membranes were then incubated with diluted primary
antibodies against PARP (cat. no. sc-136208; 1:500; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), p53 (cat. no. sc-47698;
1:500; Santa Cruz Biotechnology, Inc.), CDC2 (cat. no. sc-8395;
1:3,000; Santa Cruz Biotechnology, Inc.), cyclin B1 (cat. no.
sc-245; 1:500; Santa Cruz Biotechnology, Inc.) and β-actin (cat.
no. sc-8432; 1:5,000; Santa Cruz Biotechnology, Inc.) at 4°C
overnight with gentle agitation. The membranes were incubated with
a horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin
G secondary antibody (cat. no. sc-2004; 1:5,000; Santa Cruz
Biotechnology, Inc.) for 1 h at room temperature. The proteins were
then visualized using chemiluminescent detection reagents (Eastman
Kodak Co., Rochester, NY, USA).
Statistical analysis
Data are presented as the mean ± standard deviation
(N=3). Data were analyzed using SPSS v.17.0 (SPSS, Inc., Chicago,
IL, USA). Student's t-test was used for the pair-wise comparisons
and one-way ANOVA with Tukey's post hoc test was used for multiple
comparisons. P<0.05 was considered to indicate a statistically
significant difference.
Results
Tanshinone IIA inhibits proliferation
of human nasopharyngeal carcinoma cells
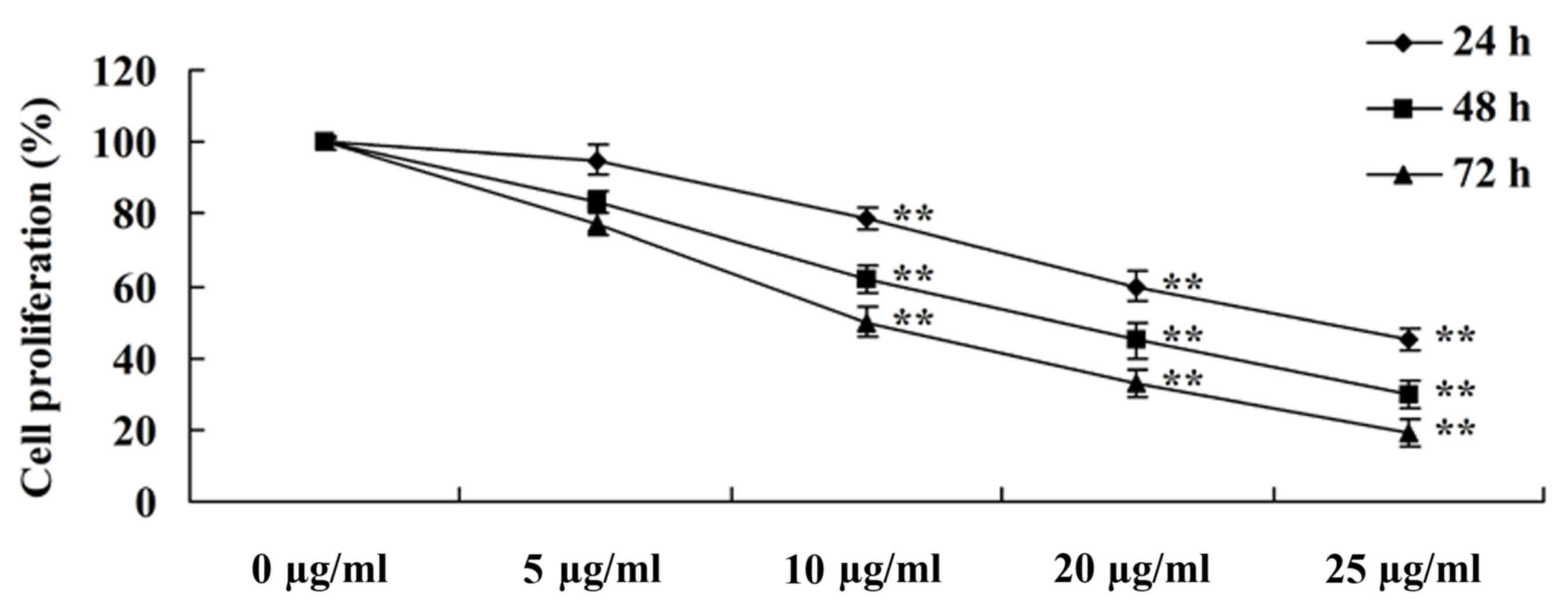
To evaluate the anti-cancer effects of tanshinone
IIA on the proliferation of human nasopharyngeal carcinoma cells,
an MTT assay was performed to evaluate the proliferation of 13-9B
cells. The dose- and time-dependent anti-cancer effects of
tanshinone IIA on the proliferation of 13-9B cells were observed
(Fig. 2). Following treatment with
10, 20 and 25 µg/ml tanshinone IIA for 24, 48 and 72 h, the
proliferation was significantly inhibited compared with cells
treated with 0 µl/ml tanshinone IIA (P<0.01; Fig. 2).
Tanshinone IIA increases the
percentage of cytotoxic human nasopharyngeal carcinoma cells
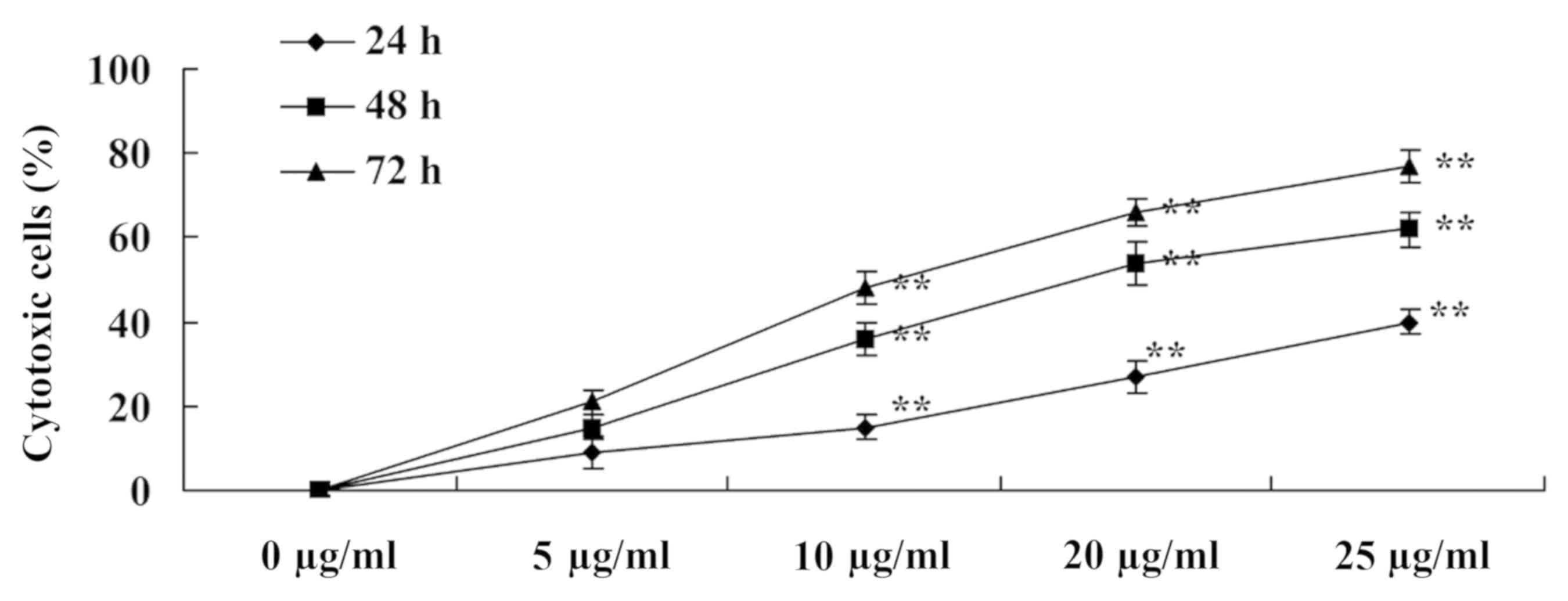
To evaluate the cytotoxic effects of tanshinone IIA
on human nasopharyngeal carcinoma cells, cytotoxic 13-9B cells were
measured using an LDH assay. Following treatment with tanshinone
IIA (10, 20 and 25 µg/ml) for 24, 48 and 78 h, the percentages of
cytotoxic 13-9B cells on days 3, 5 and 7 were significantly
increased compared with untreated cells (P<0.01; Fig. 3).
Tanshinone IIA induces apoptosis of
human nasopharyngeal carcinoma cells
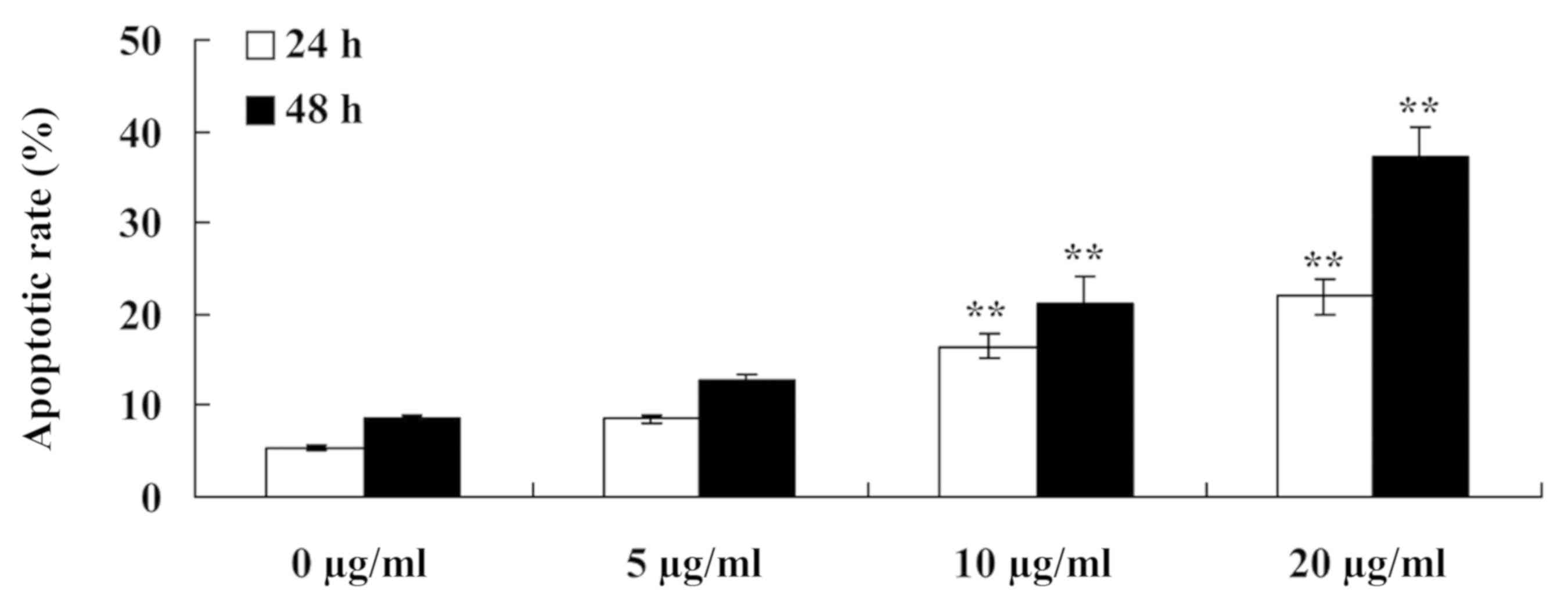
Furthermore, to detect the anti-cancer effects of
tanshinone IIA on the apoptosis of human nasopharyngeal carcinoma
cells, the apoptotic rate of 13-9B cells was measured using flow
cytometry. As presented in Fig. 4,
the apoptotic rate of 13-9B cells was significantly increased
following treatment with tanshinone IIA (10 and 20 µg/ml) for 24
and 48 h, compared with cells treated with 0 µg/ml tanshinone IIA
(P<0.01).
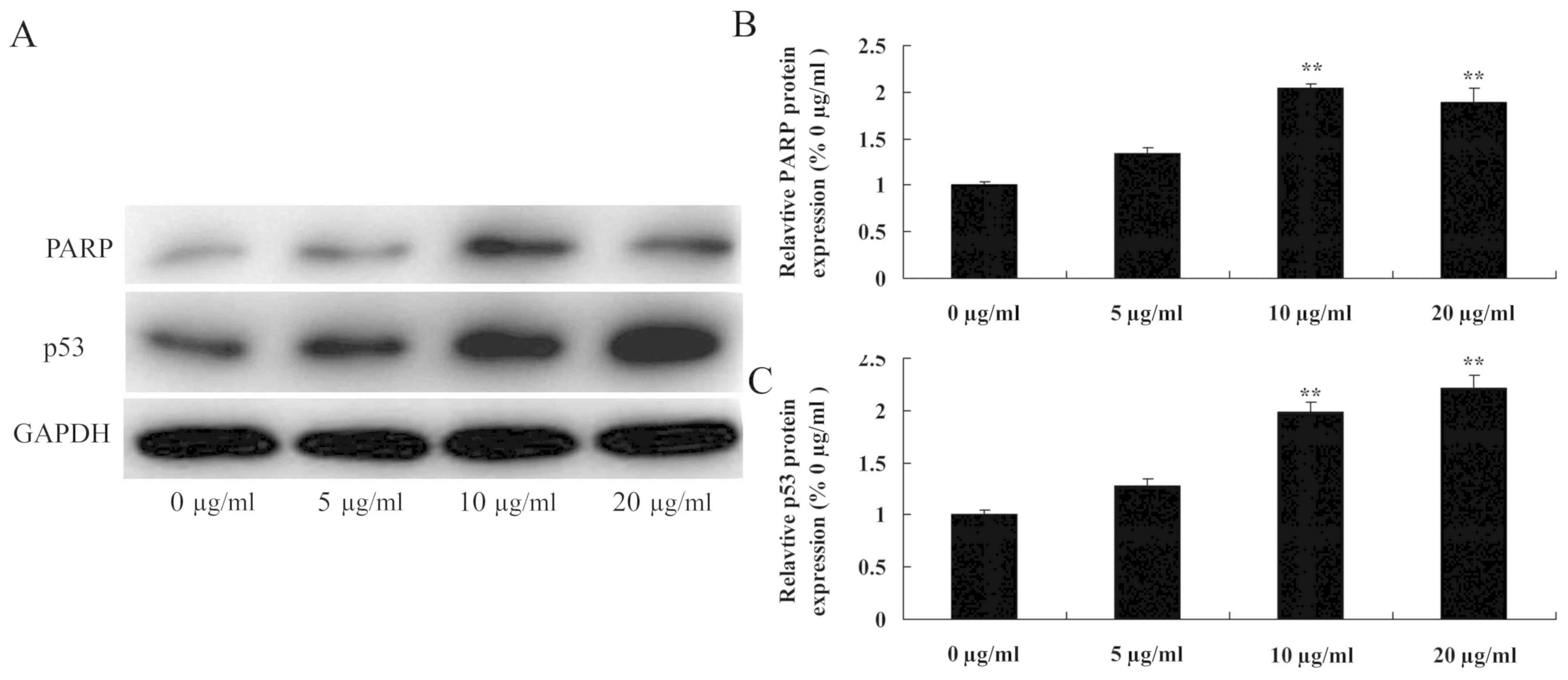
Tanshinone IIA induces PARP and p53
protein expression in human nasopharyngeal carcinoma cells
In the present study, western blot analysis was used
to determine the anti-cancer effects of tanshinone IIA on PARP and
p53 protein expression in human nasopharyngeal carcinoma cells.
Following exposure to different concentrations of tanshinone IIA
(10 and 20 µg/ml) for 48 h, PARP and p53 protein expression in
13-9B cells was significantly increased compared with cells treated
with 0 µg/ml tanshinone IIA (P<0.01; Fig. 5). However, 5 µg/ml tanshinone IIA did
not significantly increase PARP and p53 protein expression levels
in 13-9B cells.
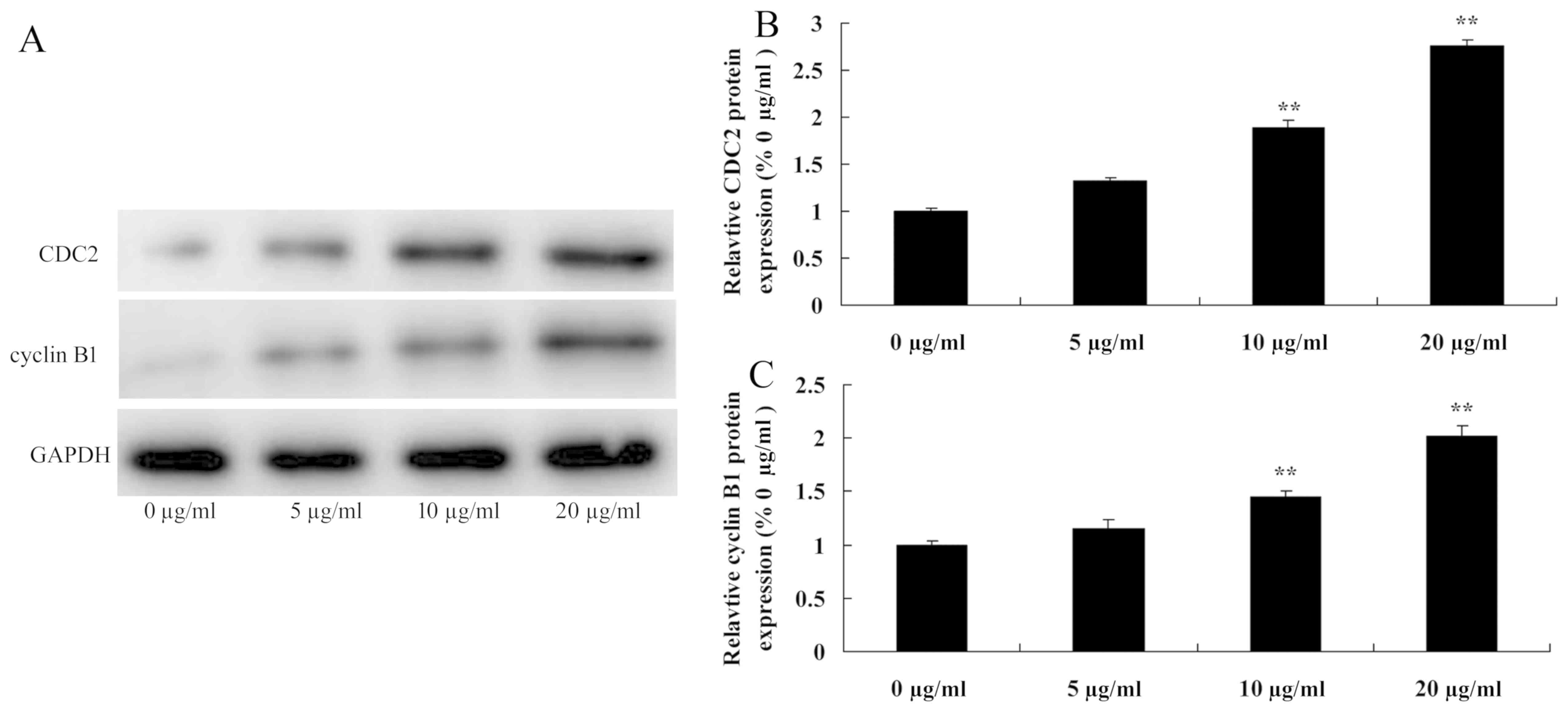
Tanshinone IIA induces CDC2 and cyclin
B1 protein expression in human nasopharyngeal carcinoma cells
Subsequently, the anti-cancer effects of tanshinone
IIA on CDC2 and cyclin B1 protein expression in human
nasopharyngeal carcinoma cells were evaluated by western blot
analysis (Fig. 6). When 13-9B cells
were treated with tanshinone IIA (10 and 20 µg/ml) for 48 h the
CDC2 and cyclin B1 protein expression levels were significantly
increased compared with cells treated with 0 µg/ml of tanshinone
IIA (P<0.01; Fig. 6). However, 5
µg/ml tanshinone IIA did not significantly increase CDC2 and cyclin
B1 protein expression in 13-9B cells.
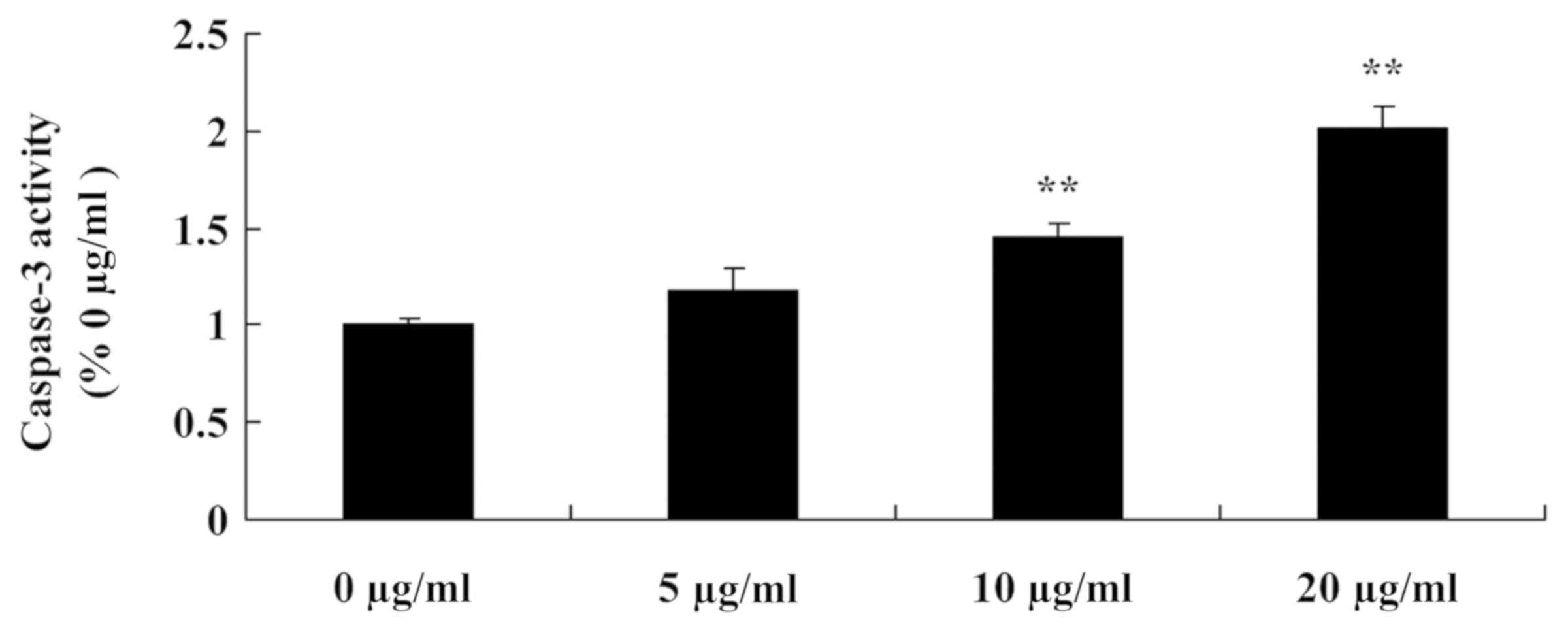
Tanshinone IIA induces caspase-3
activity in human nasopharyngeal carcinoma cells
To investigate the anti-cancer effects of tanshinone
IIA on caspase-3 activity in human nasopharyngeal carcinoma cells,
caspase-3 activity in 13-9B cells was measured using an ELISA kit.
The results indicated that caspase-3 activity in 13-9B cells was
significantly increased following treatment with 10 and 20 µg/ml
tanshinone IIA compared with 0 µg/ml tanshinone IIA (P<0.01;
Fig. 7); the results of 5 µg/ml
tanshinone IIA treatment were not statistically significant.
Discussion
Currently, the predominant clinical treatments for
nasopharyngeal carcinoma are surgery, radiotherapy and chemotherapy
(18). Patients who are eligible for
receiving surgery typically choose surgical treatment; however,
certain patients may lose pronunciation function (19). Following surgery, radiotherapy and
chemotherapy can be performed according to the condition of the
disease (20). Patients with
advanced tumor, who lose the opportunity of receiving surgery, can
select radiotherapy and chemotherapy directly; however, a
tracheostomy is often required to relieve laryngeal obstruction
(19). In summary, a combined
application of surgery, radiotherapy and chemotherapy demonstrates
an improved treatment effect; however, these treatments may cause
different degrees of throat injury, hyperemia and edema (21). Considering the present treatment
options, there is as requirement to investigate Chinese medicines
that may be administered to patients following surgery,
radiotherapy and chemotherapy to reduce postoperative recurrence
and metastasis, decrease the side effects of chemotherapy, and
improve the quality of life and the survival rate (22). The present results demonstrated that
tanshinone IIA could inhibit cell proliferation and induce
apoptosis of human nasopharyngeal carcinoma cells.
The p53 gene and the protein it encodes are
associated with cell growth and apoptosis in the regulation of cell
cycle (9). Following the damage of
DNA in cells, p53 induces arrest of the cell cycle and activates
repair of the DNA damage in order to maintain genomic stability
(23). The C-terminal of the p53
protein can detect and bind to regions of DNA damage, and regulate
and activate a gene cluster that participates in repairing the DNA
(24). At the same time, the p53
protein itself also exhibits exonuclease activity, which can
directly serve a role in the process of DNA repair (25). If DNA damage cannot be repaired, p53
will activate the transcription of apoptotic genes, which initiates
programmed cell death, also termed apoptosis. p53 protein can
promote the expression of numerous apoptosis-associated genes
(25). The present results indicate
that tanshinone IIA induces PARP and p53 protein expression in
human nasopharyngeal carcinoma cells. Similarly, Munagala et
al (26) suggested that
tanshinone IIA induces apoptosis of cervical cancer cells via p53
and PARP.
There are six subtypes of cyclin B, of which cyclin
B1 is most widely studied. Cyclin B1 is the most closely associated
with tumors and has therefore received the most attention (27). Previous studies have used in
situ hybridization and polymerase chain reaction to
demonstrated that cyclin B1 serves key role in the process of yeast
mitosis (28,29). Cyclin B1 can combine with CDC2 to
form a complex. Once activated, this complex can initiate cells to
progress from the G1/S phase to the G2/M phase (29). A previous study has reported that
drugs can act on and inhibit the cyclin B1/CDC2 complex, which
delays the G2/M phase and inhibits cell growth (30). In the present study, tanshinone IIA
was demonstrated to induce CDC2 and cyclin B1 protein expression,
and increase the activity of caspase-3 in human nasopharyngeal
carcinoma cells. Similarly, Su (31)
reported that tanshinone IIA inhibits gastric carcinoma AGS cells
via CDC2 and cyclin B1 expression. The present study only used
13-9B cells as a model of nasopharyngeal carcinoma, which was a
limitation to the study; a wider range of models needs to be used
in future research.
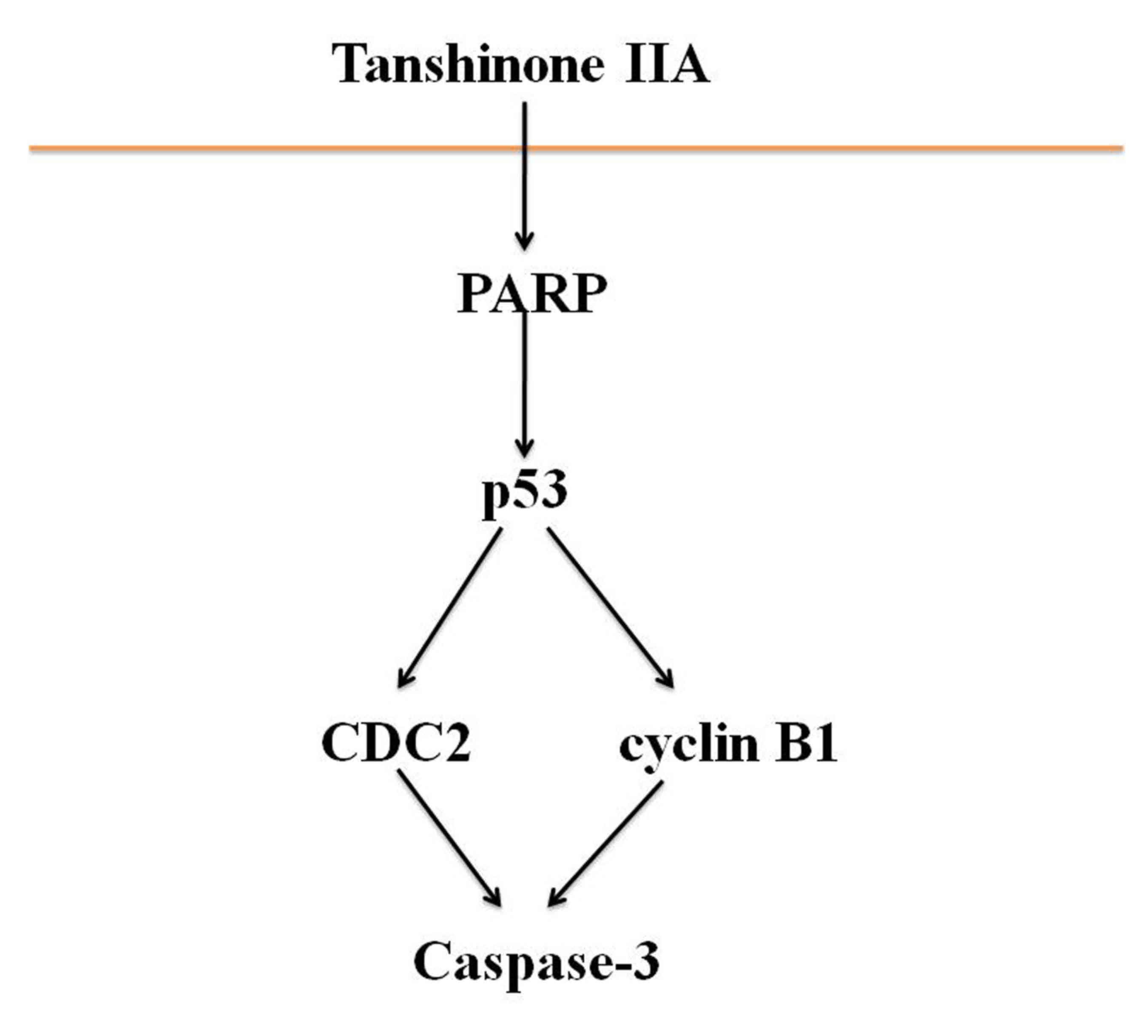
In conclusion, the results of the present study
indicate that tanshinone IIA inhibits proliferation and induces
apoptosis of human nasopharyngeal carcinoma cells via activation of
PARP, p53, cyclin B1/CDC2 and caspase-3-mediated signaling
(Fig. 8). The present study provides
experimental evidence that supports the use of tanshinone IIA in
the clinical treatment of human nasopharyngeal carcinoma.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The data sets generated and/or analyzed during the
present study are available from the corresponding author on
reasonable request.
Authors' contributions
LL designed the experiments. BL, AZ, ZS, HY, ZW, YS,
TM and YZ performed the experiments. LL and BL analyzed the data.
LL wrote the manuscript. All authors have read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hui-Yuen JS, Li XQ and Askanase AD:
Belimumab in systemic lupus erythematosus: A perspective review.
Ther Adv Musculoskelet Dis. 7:115–121. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ellis JS, Wan X and Braley-Mullen H:
Transient depletion of CD4+ CD25+ regulatory T cells results in
multiple autoimmune diseases in wild-type and B-cell-deficient NOD
mice. Immunology. 139:179–186. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Venegas-Pont M, Manigrasso MB, Grifoni SC,
LaMarca BB, Maric C, Racusen LC, Glover PH, Jones AV, Drummond HA
and Ryan MJ: Tumor necrosis factor-alpha antagonist etanercept
decreases blood pressure and protects the kidney in a mouse model
of systemic lupus erythematosus. Hypertension. 56:643–649. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Song B, Wang Z, Liu Y, Xu S, Huang G,
Xiong Y, Zhang S, Xu L, Deng X and Guan S: Immunosuppressive
activity of daphnetin, one of coumarin derivatives, is mediated
through suppression of NF-κB and NFAT signaling pathways in mouse T
cells. PLoS One. 9:e965022014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yu WW, Lu Z, Zhang H, Kang YH, Mao Y, Wang
HH, Ge WH and Shi LY: Anti-inflammatory and protective properties
of daphnetin in endotoxin-induced lung injury. J Agric Food Chem.
62:12315–12325. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shu K, Kuang N, Zhang Z, Hu Z, Zhang Y, Fu
Y and Min W: Therapeutic effect of daphnetin on the autoimmune
arthritis through demethylation of proapoptotic genes in synovial
cells. J Transl Med. 12:2872014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liao MJ, Lin LF, Zhou X, Zhou XW, Xu X,
Cheng X, Gao Q and Luo HM: Daphnetin prevents chronic unpredictable
stress-induced cognitive deficits. Fundam Clin Pharmacol.
27:510–516. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Manhas A, Khanna V, Prakash P, Goyal D,
Malasoni R, Naqvi A, Dwivedi AK, Dikshit M and Jagavelu K: Curcuma
oil reduces endothelial cell-mediated inflammation in
postmyocardial ischemia/reperfusion in rats. J Cardiovasc
Pharmacol. 64:228–236. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Casella GT, Bunge MB and Wood PM: Improved
immunocytochemical identification of neural, endothelial, and
inflammatory cell types in paraffin-embedded injured adult rat
spinal cord. J Neurosci Methods. 139:1–11. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang EB, Zhao YN, Zhang K and Mack P:
Daphnetin, one of coumarin derivatives, is a protein kinase
inhibitor. Biochem Biophys Res Commun. 260:682–685. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu L, Fu J, Li T, Cui R, Ling J, Yu X, Ji
H and Zhang Y: NG, a novel PABA/NO-based oleanolic acid derivative,
induces human hepatoma cell apoptosis via a ROS/MAPK-dependent
mitochondrial pathway. Eur J Pharmacol. 691:61–68. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tessitore A, Cicciarelli G, Mastroiaco V,
Del Vecchio F, Capece D, Verzella D, Fischietti M, Vecchiotti D,
Zazzeroni F and Alesse E: Therapeutic Use of MicroRNAs in Cancer.
Anticancer Agents Med Chem. 16:7–19. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu J, Zheng L, Ma L, Wang B, Zhao Y, Wu
N, Liu G and Lin X: Oleanolic acid inhibits proliferation and
invasiveness of Kras-transformed cells via autophagy. J Nutr
Biochem. 25:1154–1160. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang HX, Wang ZT, Lu XX, Wang YG, Zhong J
and Liu J: NLRP3 gene is associated with ulcerative colitis (UC),
but not Crohn's disease (CD), in Chinese Han population. Inflamm
Res. 63:979–985. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cummings JR, Cooney RM, Clarke G, Beckly
J, Geremia A, Pathan S, Hancock L, Guo C, Cardon LR and Jewell DP:
The genetics of NOD-like receptors in Crohn's disease. Tissue
Antigens. 76:48–56. 2010.PubMed/NCBI
|
|
16
|
Villani AC, Lemire M, Fortin G, Louis E,
Silverberg MS, Collette C, Baba N, Libioulle C, Belaiche J, Bitton
A, et al: Common variants in the NLRP3 region contribute to Crohn's
disease susceptibility. Nat Genet. 41:71–76. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Roberts RL, Topless RK, Phipps-Green AJ,
Gearry RB, Barclay ML and Merriman TR: Evidence of interaction of
CARD8 rs2043211 with NALP3 rs35829419 in Crohn's disease. Genes
Immun. 11:351–356. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Palejwala NV, Walia HS and Yeh S: Ocular
manifestations of systemic lupus erythematosus: A review of the
literature. Autoimmune Dis 2012. 2908982012.(Epub ahead of print).
doi: 10.1155/2012/290898.
|
|
19
|
Jolly CA, Muthukumar A, Reddy Avula CP and
Fernandes G: Maintenance of NF-kappaB activation in T-lymphocytes
and a naive T-cell population in autoimmune-prone (NZB/NZW)F(1)
mice by feeding a food-restricted diet enriched with n-3 fatty
acids. Cell Immunol. 213:122–133. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wong HK, Kammer GM, Dennis G and Tsokos
GC: Abnormal NF-kappa B activity in T lymphocytes from patients
with systemic lupus erythematosus is associated with decreased
p65-RelA protein expression. J Immunol. 163:1682–1689.
1999.PubMed/NCBI
|
|
21
|
Liu J, Zheng L, Zhong J, Wu N, Liu G and
Lin X: Oleanolic acid induces protective autophagy in cancer cells
through the JNK and mTOR pathways. Oncol Rep. 32:567–572. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lu Y, Zhu M and Chen W, Yin L, Zhu J, Chen
N and Chen W: Oleanolic acid induces apoptosis of MKN28 cells via
AKT and JNK signaling pathways. Pharm Biol. 52:789–795. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ghasemlou N, Lopez-Vales R, Lachance C,
Thuraisingam T, Gaestel M, Radzioch D and David S:
Mitogen-activated protein kinase-activated protein kinase 2 (MK2)
contributes to secondary damage after spinal cord injury. J
Neurosci. 30:13750–13759. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li XQ, Cao XZ, Wang J, Fang B, Tan WF and
Ma H: Sevoflurane preconditioning ameliorates neuronal deficits by
inhibiting microglial MMP-9 expression after spinal cord
ischemia/reperfusion in rats. Mol Brain. 7:692014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen J, Zhang X, Lentz C, Abi-Daoud M,
Paré GC, Yang X, Feilotter HE and Tron VA: miR-193b Regulates Mcl-1
in Melanoma. Am J Pathol. 179:2162–2168. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Munagala R, Aqil F, Jeyabalan J and Gupta
RC: Tanshinone IIA inhibits viral oncogene expression leading to
apoptosis and inhibition of cervical cancer. Cancer Lett 356 (2 Pt
B). 536–546. 2015. View Article : Google Scholar
|
|
27
|
Kaukoniemi KM, Rauhala HE, Scaravilli M,
Latonen L, Annala M, Vessella RL, Nykter M, Tammela TL and
Visakorpi T: Epigenetically altered miR-193b targets cyclin D1 in
prostate cancer. Cancer Med. 4:1417–1425. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wahdan-Alaswad RS, Cochrane DR, Spoelstra
NS, Howe EN, Edgerton SM, Anderson SM, Thor AD and Richer JK:
Metformin-induced killing of triple-negative breast cancer cells is
mediated by reduction in fatty acid synthase via miRNA-193b. Horm
Cancer. 5:374–389. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Blick C, Ramachandran A, McCormick R,
Wigfield S, Cranston D, Catto J and Harris AL: Identification of a
hypoxia-regulated miRNA signature in bladder cancer and a role for
miR-145 in hypoxia-dependent apoptosis. Br J Cancer. 113:634–644.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang S, Guo Y, Zhang C, Gao W, Wen S,
Huangfu H and Wang B: Primary laryngeal cancer-derived miR-193b
induces interleukin-10-expression monocytes. Cancer Invest.
33:29–33. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Su CC: Tanshinone IIA inhibits gastric
carcinoma AGS cells through increasing p-p38, p-JNK and p53 but
reducing p-ERK, CDC2 and cyclin B1 expression. Anticancer Res.
34:7097–7110. 2014.PubMed/NCBI
|