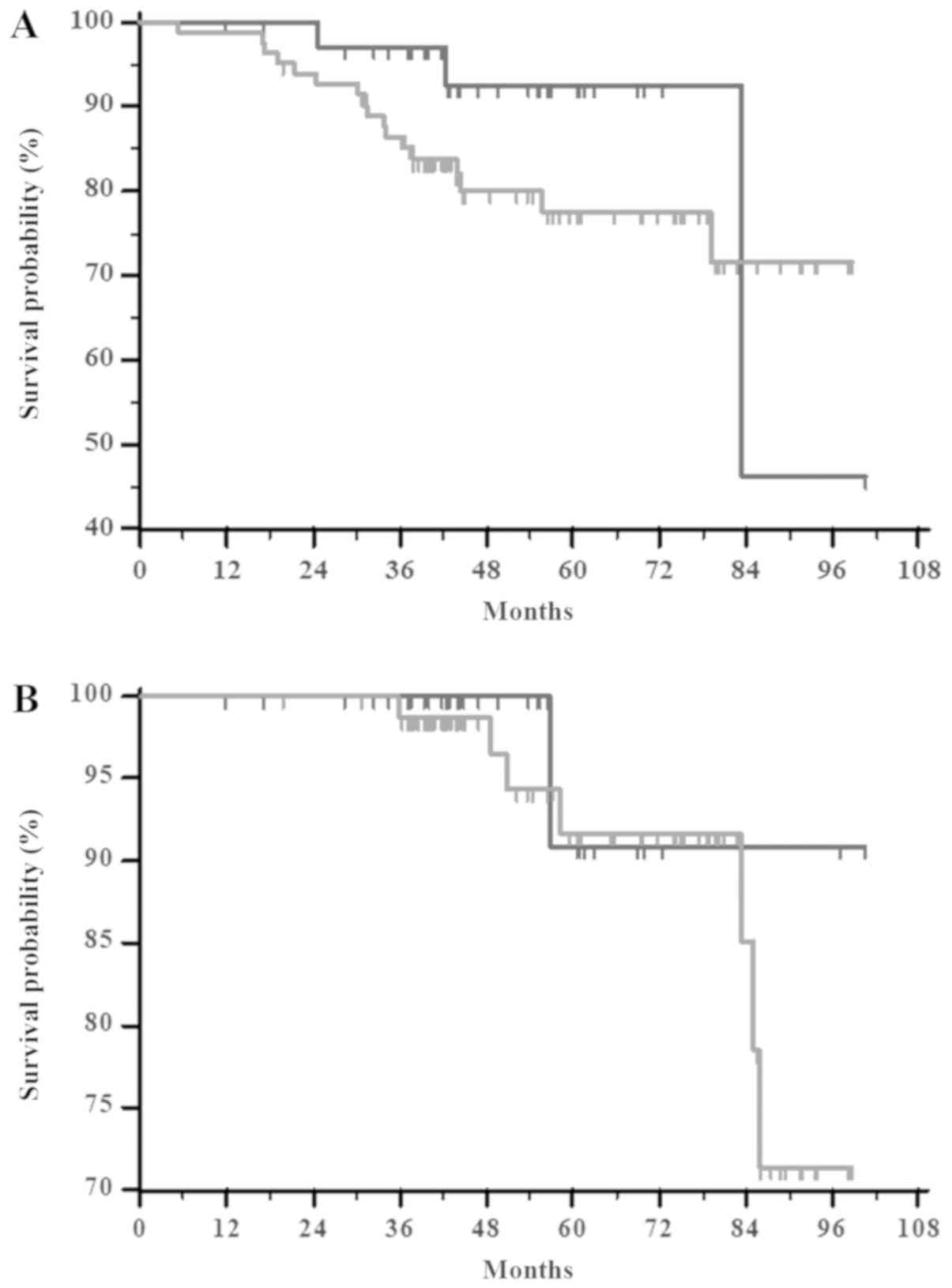
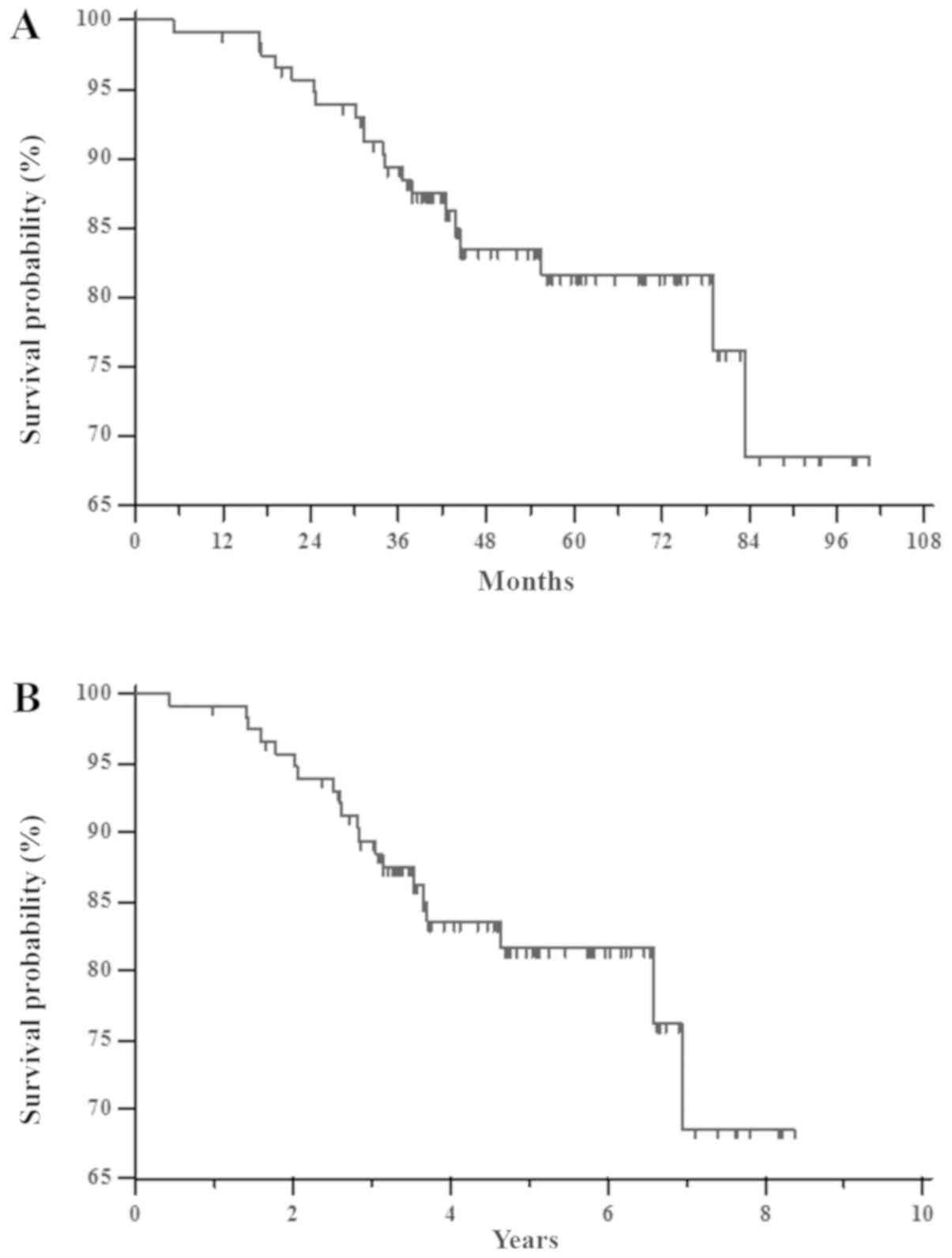
|
1
|
Thompson AM and Moulder-Thompson SL:
Neoadjuvant treatment of breast cancer. Ann Oncol. 23 (Suppl
10):x231–x236. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Untch M, Konecny GE, Paepke S and von
Minckwitz G: Current and future role of neoadjuvant therapy for
breast cancer. Breast. 23:526–537. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rubovszky G and Horváth Z: Recent advances
in the neoadjuvant treatment of breast cancer. J Breast Cancer.
20:119–131. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen Y, Shi XE, Tian JH, Yang XJ, Wang YF
and Yang KH: Survival benefit of neoadjuvant chemotherapy for
resectable breast cancer: A meta-analysis. Medicine (Baltimore).
97:e106342018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), . Long-term outcomes for neoadjuvant
versus adjuvant chemotherapy in early breast cancer: Meta-analysis
of individual patient data from ten randomised trials. Lancet
Oncol. 19:27–39. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kaufmann M, Hortobagyi GN, Goldhirsch A,
Scholl S, Makris A, Valagussa P, Blohmer JU, Eiermann W, Jackesz R,
Jonat W, et al: Recommendations from an international expert panel
on the use of neoadjuvant (primary) systemic treatment of operable
breast cancer: An update. J Clin Oncol. 24:1940–1949. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang L, Zhong X, Pu T, Qiu Y, Ye F and Bu
H: Clinical significance and prognostic value of receptor
conversion in hormone receptor positive breast cancers after
neoadjuvant chemotherapy. World J Surg Oncol. 16:512018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yoshida A, Hayashi N, Suzuki K, Takimoto
M, Nakamura S and Yamauchi H: Change in HER2 status after
neoadjuvant chemotherapy and the prognostic impact in patients with
primary breast cancer. J Surg Oncol. 116:1021–1028. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Straver ME, Rutgers EJ, Rodenhuis S, Linn
SC, Loo CE, Wesseling J, Russell NS, Oldenburg HS, Antonini N and
Vrancken Peeters MT: The relevance of breast cancer subtypes in the
outcome of neoadjuvant chemotherapy. Ann Surg Oncol. 17:2411–2418.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Curigliano G, Burstein HJ, P Winer E,
Gnant M, Dubsky P, Loibl S, Colleoni M, Regan MM, Piccart-Gebhart
M, Senn HJ, et al: De-escalating and escalating treatments for
early-stage breast cancer: The St. Gallen International expert
consensus conference on the primary therapy of early breast cancer
2017. Ann Oncol. 28:1700–1712. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Debiasi M, Polanczyk CA, Ziegelmann P,
Barrios C, Cao H, Dignam JJ, Goss P, Bychkovsky B, Finkelstein DM,
Guindalini RS, et al: Efficacy of anti-HER2 agents in combination
with adjuvant or neoadjuvant chemotherapy for early and locally
advanced HER2-positive breast cancer patients: A network
meta-analysis. Front Oncol. 8:1562018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Swain SM, Ewer MS, Viale G, Delaloge S,
Ferrero JM, Verrill M, Colomer R, Vieira C, Werner TL, Douthwaite
H, et al: Pertuzumab, trastuzumab, and standard anthracycline- and
taxane-based chemotherapy for the neoadjuvant treatment of patients
with HER2-positive localized breast cancer (BERENICE): A phase II,
open-label, multicenter, multinational cardiac safety study. Ann
Oncol. 29:646–653. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gleeson JP, Keegan NM and Morris PG:
Adding pertuzumab to trastuzumab and taxanes in HER2 positive
breast cancer. Expert Opin Biol Ther. 18:251–262. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nami B, Maadi H and Wang Z: Mechanisms
underlying the action and synergism of trastuzumab and pertuzumab
in targeting HER2-positive breast cancer. Cancers (Basel). 10(pii):
E3422018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chang JC, Wooten EC, Tsimelzon A,
Hilsenbeck SG, Gutierrez MC, Elledge R, Mohsin S, Osborne CK,
Chamness GC, Allred DC and O'Connell P: Gene expression profiling
for the prediction of therapeutic response to docetaxel in patients
with breast cancer. Lancet. 362:362–369. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Burcombe RJ, Makris A, Richman PI, Daley
FM, Noble S, Pittam M, Wright D, Allen SA, Dove J and Wilson GD:
Evaluation of ER, PgR, HER-2 and Ki-67 as predictors of response to
neoadjuvant anthracycline chemotherapy for operable breast cancer.
Br J Cancer. 92:147–155. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu YL, Connolly EP and Kalinsky K:
Obesity's impact on survival is independent of dose adjustments in
neoadjuvant chemotherapy in women with breast cancer. Breast Cancer
Res Treat. 168:2852018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu YL, Saraf A, Catanese B, Lee SM, Zhang
Y, Connolly EP and Kalinsky K: Obesity and survival in the
neoadjuvant breast cancer setting: Role of tumor subtype in an
ethnically diverse population. Breast Cancer Res Treat.
167:277–288. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Karatas F, Erdem GU, Sahin S, Aytekin A,
Yuce D, Sever AR, Babacan T, Ates O, Ozisik Y and Altundag K:
Obesity is an independent prognostic factor of decreased
pathological complete response to neoadjuvant chemotherapy in
breast cancer patients. Breast. 32:237–244. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fontanella C, Lederer B, Gade S, Vanoppen
M, Blohmer JU, Costa SD, Denkert C, Eidtmann H, Gerber B, Hanusch
C, et al: Impact of body mass index on neoadjuvant treatment
outcome: A pooled analysis of eight prospective neoadjuvant breast
cancer trials. Breast Cancer Res Treat. 150:127–139. 2015.
View Article : Google Scholar : PubMed/NCBI
|