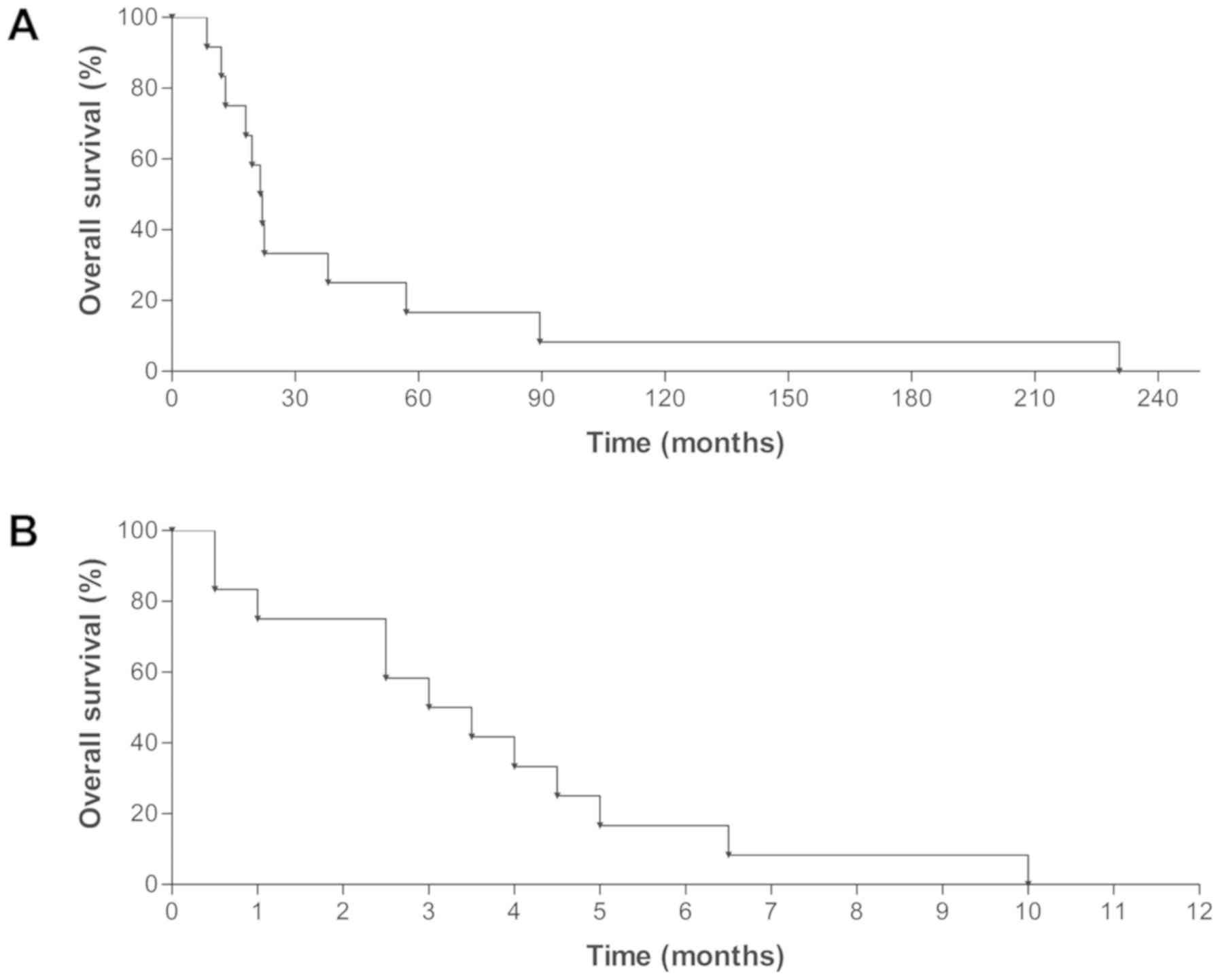
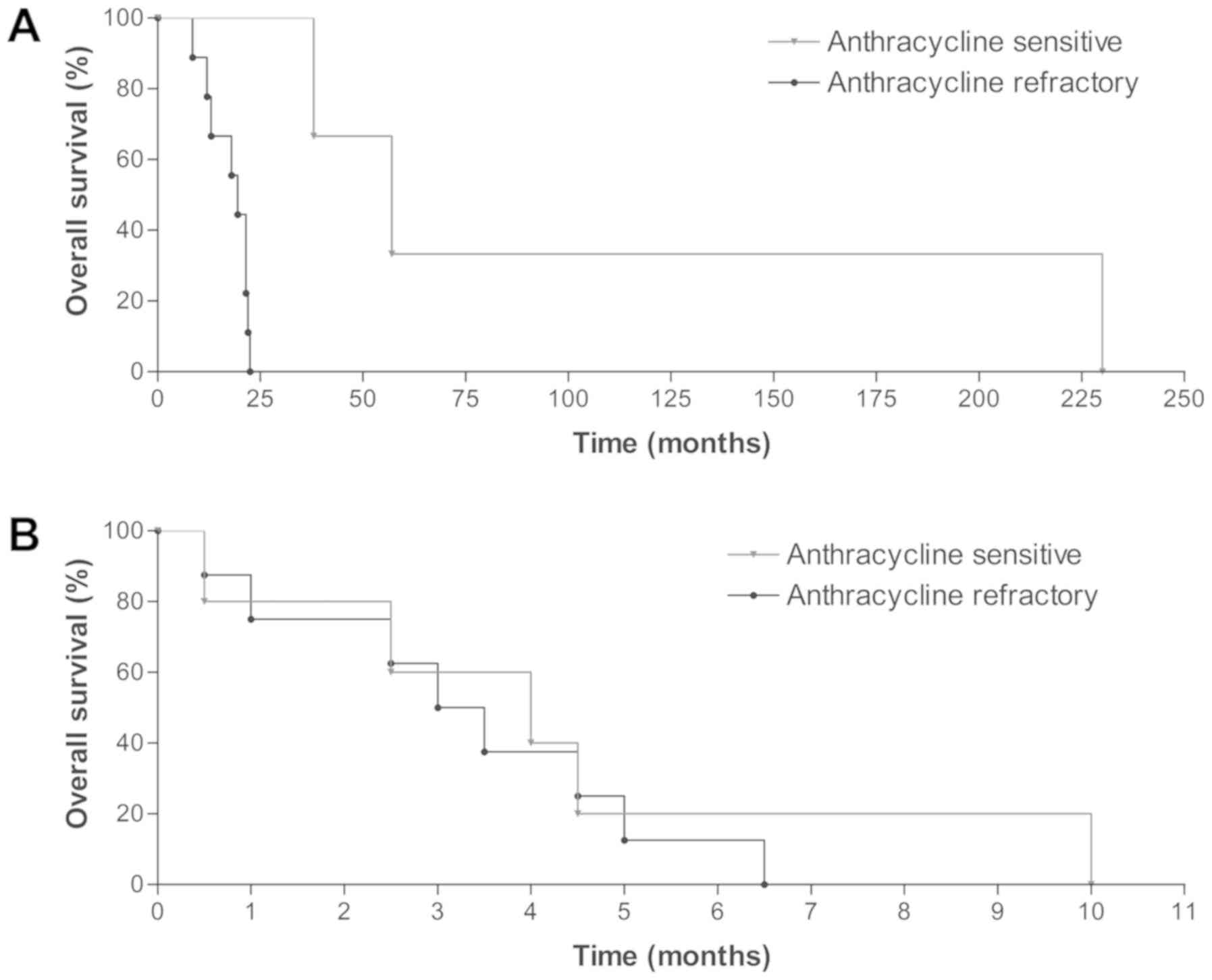
|
1
|
Tilly H, Gomes da Silva M, Vitolo U, Jack
A, Meignan M, Lopez-Guillermo A, Walewski J, André M, Johnson PW,
Pfreundschuh M, et al: Diffuse large B-cell lymphoma (DLBCL): ESMO
Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 26:v116–v125. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Keating GM: Pixantrone: A review in
relapsed or refractory aggressive non-Hodgkin's lymphoma. Drugs.
76:1579–1586. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gregoric B, Zadnik V and Jezersek
Novakovic B: The diffuse large B-cell lymphoma-where do we stand
now in everyday clinical practice. Radiol Oncol. 46:153–159. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Horvat M, Zadnik V, Južnič Šetina T,
Boltežar L, Pahole Goličnik J, Novaković S and Jezeršek Novaković
B: Diffuse large B-cell lymphoma: 10 years' real-world clinical
experience with rituximab plus cyclophosphamide, doxorubicin,
vincristine and prednisolone. Oncol Lett. 15:3602–3609.
2018.PubMed/NCBI
|
|
5
|
Volpetti S, Zaja F and Fanin R: Pixantrone
for the treatment of adult patients with relapsed or refractory
aggressive non-Hodgkin B-cell lymphomas. Onco Targets Ther.
7:865–872. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Krapcho AP, Maresch MJ, Hacker MP, Menta
E, Oliva A, Giuliani FC and Spinelli S: Aza and diaza bioisosteric
anthracene-9,10-diones as antitumor agents. Acta Biochim Pol.
42:427–432. 1995.PubMed/NCBI
|
|
7
|
Singal PK and Iliskovic N:
Doxorubicin-induced cardiomyopathy. N Engl J Med. 339:900–905.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mordente A, Meucci E, Silvestrini A,
Martorana GE and Giardina B: New developments in
anthracycline-induced cardiotoxicity. Curr Med Chem. 16:1656–1672.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Borchmann P, Morschhauser F, Parry A,
Schnell R, Harousseau JL, Gisselbrecht C, Rudolph C, Wilhelm M,
Günther H, Pfreundschuh DM, et al: Phase-II study of the new
aza-anthracenedione, BBR 2778, in patients with relapsed aggressive
non-Hodgkin's lymphomas. Haematologica. 88:888–894. 2003.PubMed/NCBI
|
|
10
|
Hasinoff BB, Wu X, Patel D, Kanagasabai R,
Karmahapatra S and Yalowich JC: Mechanisms of action and reduced
cardiotoxicity of pixantrone; a topoisomerase II targeting agent
with cellular selectivity for the topoisomerase IIα isoform. J
Pharmacol Exp Ther. 356:397–409. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pettengell R, Coiffier B, Narayanan G, de
Mendoza FH, Digumarti R, Gomez H, Zinzani PL, Schiller G, Rizzieri
D, Boland G, et al: Pixantrone dimaleate versus other
chemotherapeutic agents as a single-agent salvage treatment in
patients with relapsed or refractory aggressive non-Hodgkin
lymphoma: A phase 3, multicentre, open-label, randomised trial.
Lancet Oncol. 13:696–706. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pettengell R, Coiffier B, Egorov A, Singer
J and Sivcheva L: Long-term response and remission with pixantrone
in patients with relapsed or refractory aggressive non-Hodgkin
lymphoma: Post-hoc analysis of the multicenter, open-label,
randomized PIX301 trial. Clin Drug Investig. 38:527–533. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Eyre TA, Linton KM, Rohman P, Kothari J,
Cwynarski K, Ardeshna K, Bailey C, Osborne WL, Rowntree C, Eden D,
et al: Results of a multicentre UK-wide retrospective study
evaluating the efficacy of pixantrone in relapsed, refractory
diffuse large B cell lymphoma. Br J Haematol. 173:896–904. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
d'Amore F, Leppä S, Larsen TS, Brown P,
Relander T, Mannisto S, Jørgensen JM, Holte H, Lauritzsen GF,
Enblad G, et al: A phase 1/2 study of pixantrone, etoposide,
bendamustine and, in CD20+ tumors, rituximab in patients
with relapsed aggressive B- or T-cell lymphomas-the P[R]EBEN study.
Hematol Oncol. 35:423–424. 2017. View Article : Google Scholar
|
|
15
|
Mareschal S, Lanic H, Ruminy P, Bastard C,
Tilly H and Jardin F: The proportion of activated B-cell like
subtype among de novo diffuse large B-cell lymphoma increases with
age. Haematologica. 96:1888–1890. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scott DW, Mottok A, Ennishi D, Wright GW,
Farinha P, Ben-Neriah S, Kridel R, Barry GS, Hother C, Abrisqueta
P, et al: Prognostic significance of diffuse large B-cell lymphoma
cell of origin determined by digital gene expression in
formalin-fixed paraffin-embedded tissue biopsies. J Clin Oncol.
33:2848–2856. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Iqbal J, Meyer PN, Smith LM, Johnson NA,
Vose JM, Greiner TC, Connors JM, Staudt LM, Rimsza L, Jaffe E, et
al: BCL2 predicts survival in germinal center B-cell-like diffuse
large B-cell lymphoma treated with CHOP-like therapy and rituximab.
Clin Cancer Res. 17:7785–7795. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Iqbal J, Neppalli VT, Wright G, Dave BJ,
Horsman DE, Rosenwald A, Lynch J, Hans CP, Weisenburger DD, Greiner
TC, et al: BCL2 expression is a prognostic marker for the activated
B-cell-like type of diffuse large B-cell lymphoma. J Clin Oncol.
24:961–968. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Petrella T, Copie-Bergman C, Brière J,
Delarue R, Jardin F, Ruminy P, Thieblemont C, Figeac M, Canioni D,
Feugier P, et al: BCL2 expression but not MYC and BCL2 coexpression
predicts survival in elderly patients with diffuse large B-cell
lymphoma independently of cell of origin in the phase 3 LNH03-6B
trial. Ann Oncol. 28:1042–1049. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Song MK, Chung JS, Lee JJ, Yang DH, Kim
IS, Shin DH and Shin HJ: High Ki-67 expression in involved bone
marrow predicts worse clinical outcome in diffuse large B cell
lymphoma patients treated with R-CHOP therapy. Int J Hematol.
101:140–147. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li ZM, Huang JJ, Xia Y, Zhu YJ, Zhao W,
Wei WX, Jiang WQ, Lin TY, Huang HQ and Guan ZZ: High Ki-67
expression in diffuse large B-cell lymphoma patients with
non-germinal center subtype indicates limited survival benefit from
R-CHOP therapy. Eur J Haematol. 88:510–517. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tang YL, Zhou Y, Cheng LL, Su YZ and Wang
CB: BCL2/Ki-67 index predict survival in germinal center
B-cell-like diffuse large B-cell lymphoma. Oncol Lett.
14:3767–3773. 2017. View Article : Google Scholar : PubMed/NCBI
|