|
1
|
Yoshimura T: The production of monocyte
chemoattractant protein-1 (MCP-1)/CCL2 in tumor microenvironments.
Cytokine. 98:71–78. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lee S, Lee E, Ko E, Ham M, Lee HM, Kim ES,
Koh M, Lim HK, Jung J, Park SY and Moon A: Tumor-associated
macrophages secrete CCL2 and induce the invasive phenotype of human
breast epithelial cells through upregulation of ERO1-α and MMP-9.
Cancer Lett. 437:25–34. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Porrello A, Leslie PL, Harrison EB,
Gorentla BK, Kattula S, Ghosh SK, Azam SH, Holtzhausen A, Chao YL,
Hayward MC, et al: Factor XIIIA-expressing inflammatory monocytes
promote lung squamous cancer through fibrin cross-linking. Nat
Commun. 9:19882018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mandal PK, Biswas S, Mandal G, Purohit S,
Gupta A, Majumdar Giri A, Roy Chowdhury S and Bhattacharyya A: CCL2
conditionally determines CCL22-dependent Th2-accumulation during
TGF-beta-induced breast cancer progression. Immunobiology.
223:151–161. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yoshimura T: The chemokine MCP-1 (CCL2) in
the host interaction with cancer: A foe or ally? Cell Mol Immunol.
15:335–345. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liubomirski Y, Lerrer S, Meshel T,
Rubinstein-Achiasaf L, Morein D, Wiemann S, Körner C and Ben-Baruch
A: Tumor-stroma-inflammation networks promote pro-metastatic
chemokines and aggressiveness characteristics in triple-negative
breast cancer. Front Immunol. 10:7572019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rotondi M, Coperchini F, Latrofa F and
Chiovato L: Role of Chemokines in Thyroid Cancer Microenvironment:
Is CXCL8 the Main Player? Front Endocrinol (Lausanne). 9:3142018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Al-Mazidi S, Alotaibi M, Nedjadi T,
Chaudhary A, Alzoghaibi M and Djouhri L: Blocking of cytokines
signalling attenuates evoked and spontaneous neuropathic pain
behaviours in the paclitaxel rat model of chemotherapy-induced
neuropathy. Eur J Pain. 22:810–821. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Khazali AS, Clark AM and Wells A:
Inflammatory cytokine IL-8/CXCL8 promotes tumour escape from
hepatocyte-induced dormancy. Br J Cancer. 118:566–576. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang J and Wang X, Wang Y, Li S and Wang
X: Krüppel like factor 6 splice variant 1 (KLF6-SV1) overexpression
recruits macrophages to participate in lung cancer metastasis by
up-regulating TWIST1. Cancer Biol Ther. 20:680–691. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li F, Kitajima S, Kohno S, Yoshida A,
Tange S, Sasaki S, Okada N, Nishimoto Y, Muranaka H, Nagatani N, et
al: Retinoblastoma inactivation induces a protumoral
microenvironment via enhanced CCL2 secretion. Cancer Res.
79:3903–3915. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng Y, Li H, Deng Y, Tai Y, Zeng K,
Zhang Y, Liu W, Zhang Q and Yang Y: Cancer-associated fibroblasts
induce PDL1+ neutrophils through the IL6-STAT3 pathway that foster
immune suppression in hepatocellular carcinoma. Cell Death Dis.
9:4222018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang H, Yu Y, Zhou L, Ma J, Tang K, Xu P,
Ji T, Liang X, Lv J, Dong W, et al: Circulating tumor
microparticles promote lung metastasis by reprogramming
inflammatory and mechanical niches via a macrophage-dependent
pathway. Cancer Immunol Res. 6:1046–1056. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu X, Jin G, Qian J, Yang H, Tang H, Meng
X and Li Y: Digital gene expression profiling analysis and its
application in the identification of genes associated with improved
response to neoadjuvant chemotherapy in breast cancer. World J Surg
Oncol. 16:822018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mano Y, Yoshio S, Shoji H, Tomonari S,
Aoki Y, Aoyanagi N, Okamoto T, Matsuura Y, Osawa Y, Kimura K, et
al: Bone morphogenetic protein 4 provides cancer-supportive
phenotypes to liver fibroblasts in patients with hepatocellular
carcinoma. J Gastroenterol. 54:1007–1018. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bedini N, Cicchetti A, Palorini F, Magnani
T, Zuco V, Pennati M, Campi E, Allavena P, Pesce S, Villa S, et al:
Evaluation of mediators associated with the inflammatory response
in prostate cancer patients undergoing radiotherapy. Dis Markers.
2018:91281282018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Heiskala M, Leidenius M, Joensuu K and
Heikkila P: High expression of CCL2 in tumor cells and abundant
infiltration with CD14 positive macrophages predict early relapse
in breast cancer. Virchows Arch. 474:3–12. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Izumi K and Mizokami A: Suppressive role
of androgen/androgen receptor signaling via chemokines on prostate
cancer cells. J Clin Med. 8:E3542019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Natsagdorj A, Izumi K, Hiratsuka K,
Machioka K, Iwamoto H, Naito R, Makino T, Kadomoto S, Shigehara K,
Kadono Y, et al: CCL2 induces resistance to the antiproliferative
effect of cabazitaxel in prostate cancer cells. Cancer Sci.
110:279–288. 2019.PubMed/NCBI
|
|
20
|
Wang X, Yang X, Tsai Y, Yang L, Chuang KH,
Keng PC, Lee SO and Chen Y: IL-6 mediates macrophage infiltration
after irradiation via up-regulation of CCL2/CCL5 in non-small cell
lung cancer. Radiat Res. 187:50–59. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
He S and Zhang X: The rs1024611 in the
CCL2 gene and risk of gynecological cancer in Asians: A
meta-analysis. World J Surg Oncol. 16:342018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Guan X, Liu Z, Zhang J and Jin X:
Myeloid-derived suppressor cell accumulation in renal cell
carcinoma is correlated with CCL2, IL-17 and IL-18 expression in
blood and tumors. Adv Clin Exp Med. 27:947–953. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Amann B, Perabo FG, Wirger A, Hugenschmidt
H and Schultze-Seemann W: Urinary levels of monocyte
chemo-attractant protein-1 correlate with tumour stage and grade in
patients with bladder cancer. Br J Urol. 82:118–121. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Saik OV, Nimaev VV, Usmonov DB, Demenkov
PS, Ivanisenko TV, Lavrik IN and Ivanisenko VA: Prioritization of
genes involved in endothelial cell apoptosis by their implication
in lymphedema using an analysis of associative gene networks with
ANDSystem. BMC Med Genomics. 12 (Suppl 2):S472019. View Article : Google Scholar
|
|
25
|
Roblek M, Protsyuk D, Becker PF,
Stefanescu C, Gorzelanny C, Glaus Garzon JF, Knopfova L,
Heikenwalder M, Luckow B, Schneider SW and Borsig L: CCL2 is a
vascular permeability factor inducing CCR2-dependent endothelial
retraction during lung metastasis. Mol Cancer Res. 17:783–793.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yumimoto K, Sugiyama S, Mimori K and
Nakayama KI: Potentials of C-C motif chemokine 2-C-C chemokine
receptor type 2 blockers including propagermanium as anticancer
agents. Cancer Sci. 110:2090–2099. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Triulzi T, Forte L, Regondi V, Di Modica
M, Ghirelli C, Carcangiu ML, Sfondrini L, Balsari A and Tagliabue
E: HER2 signaling regulates the tumor immune microenvironment and
trastuzumab efficacy. Oncoimmunology. 8:e15129422019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Song M, Sasazuki S, Camargo MC, Shimazu T,
Charvat H, Yamaji T, Sawada N, Kemp TJ, Pfeiffer RM, Hildesheim A,
et al: Circulating inflammatory markers and colorectal cancer risk:
A prospective case-cohort study in Japan. Int J Cancer.
143:2767–2776. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Grossman JG, Nywening TM, Belt BA, Panni
RZ, Krasnick BA, DeNardo DG, Hawkins WG, Goedegebuure SP, Linehan
DC and Fields RC: Recruitment of CCR2+ tumor associated
macrophage to sites of liver metastasis confers a poor prognosis in
human colorectal cancer. Oncoimmunology. 7:e14707292018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Coniglio SJ: Role of tumor-derived
chemokines in osteolytic bone metastasis. Front Endocrinol
(Lausanne). 9:3132018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bauer D, Redmon N, Mazzio E and Soliman
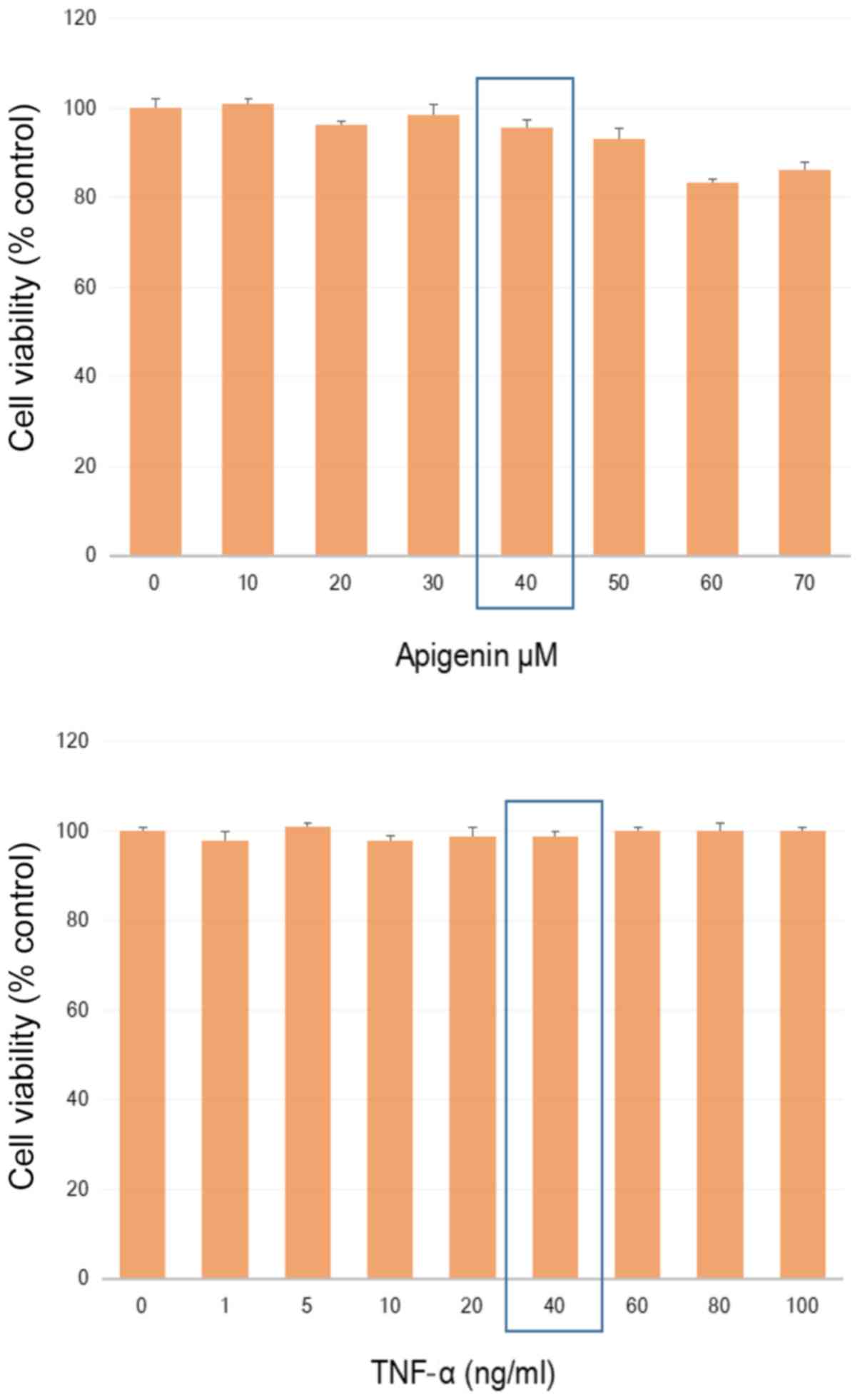
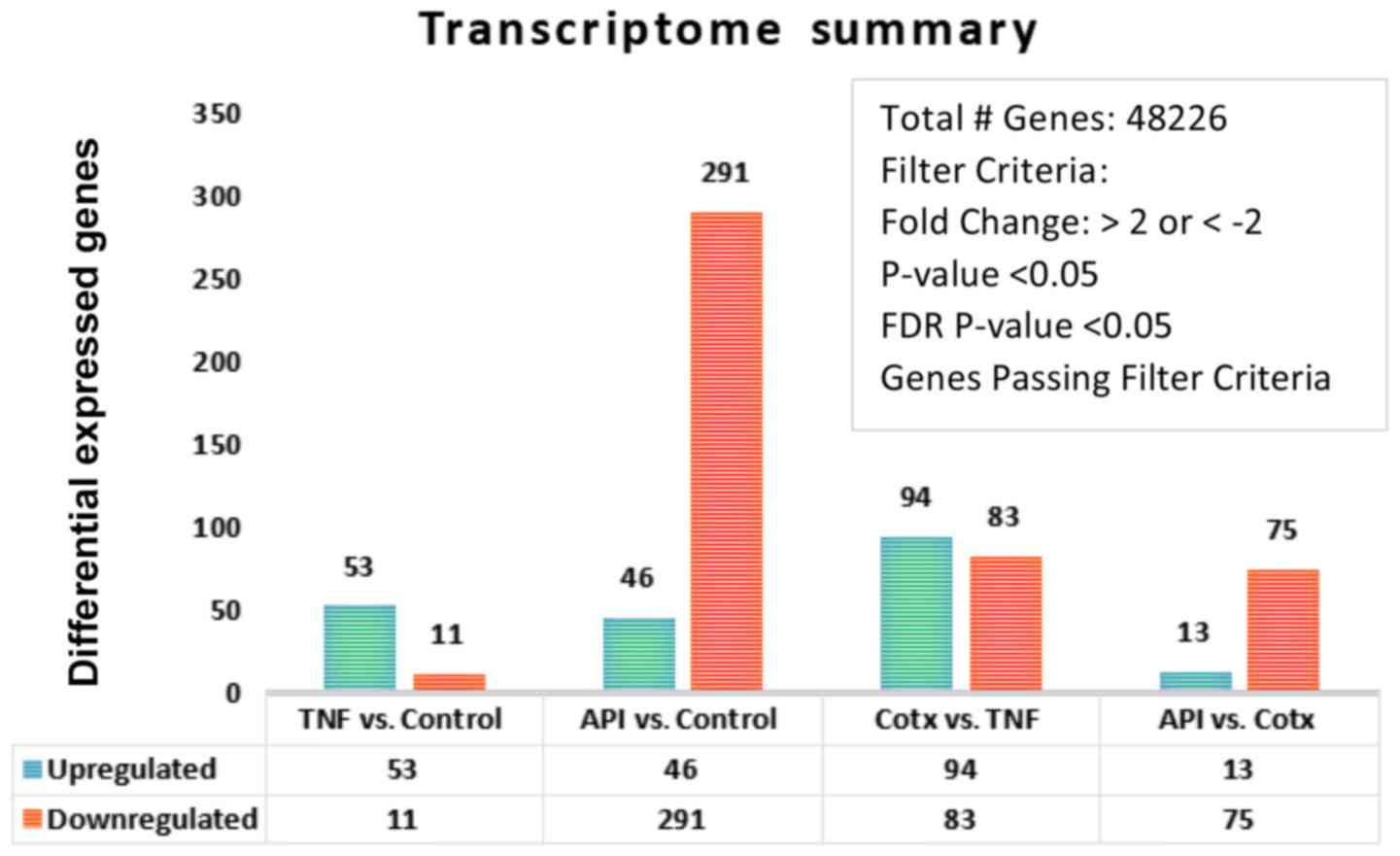
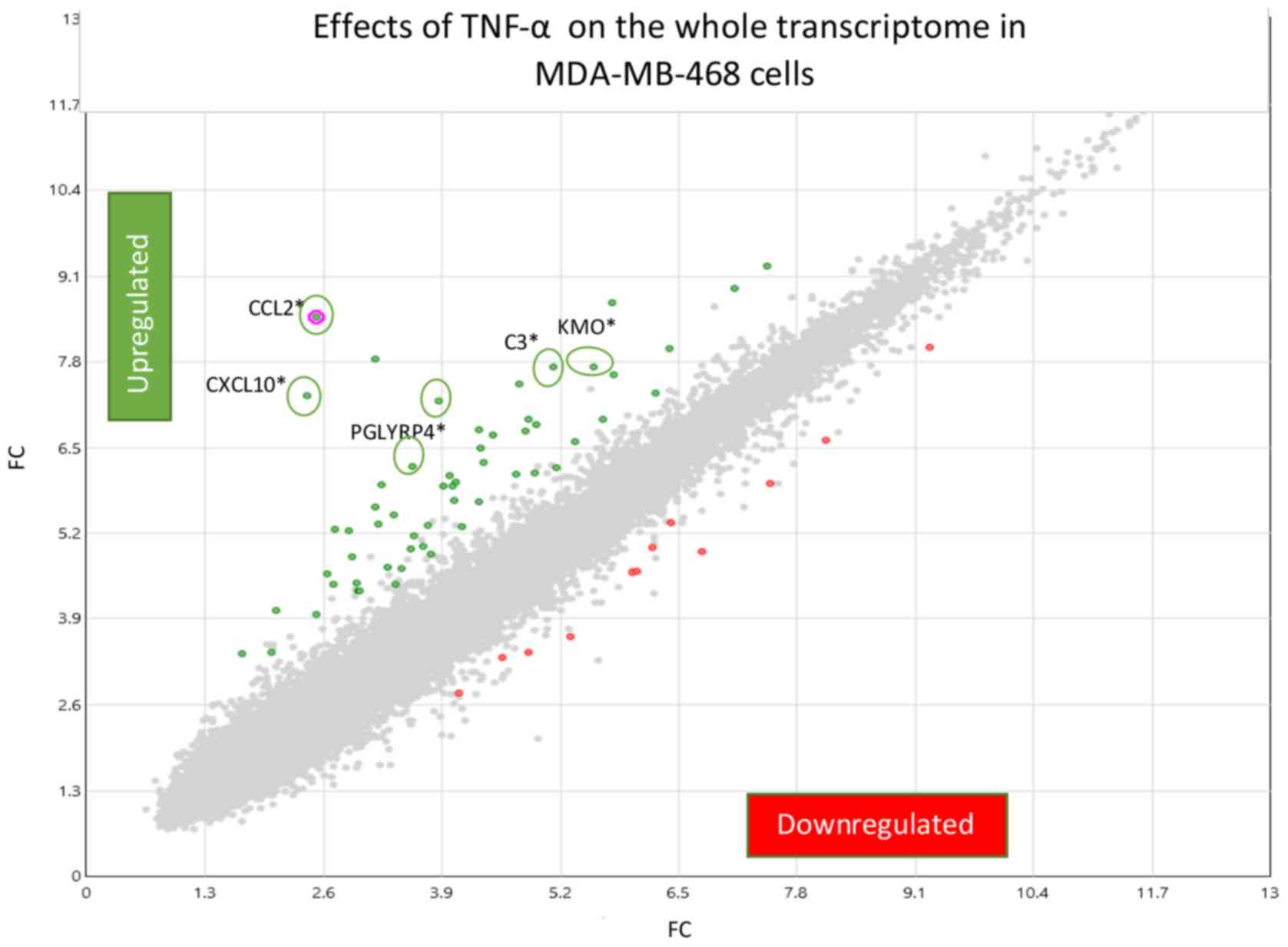
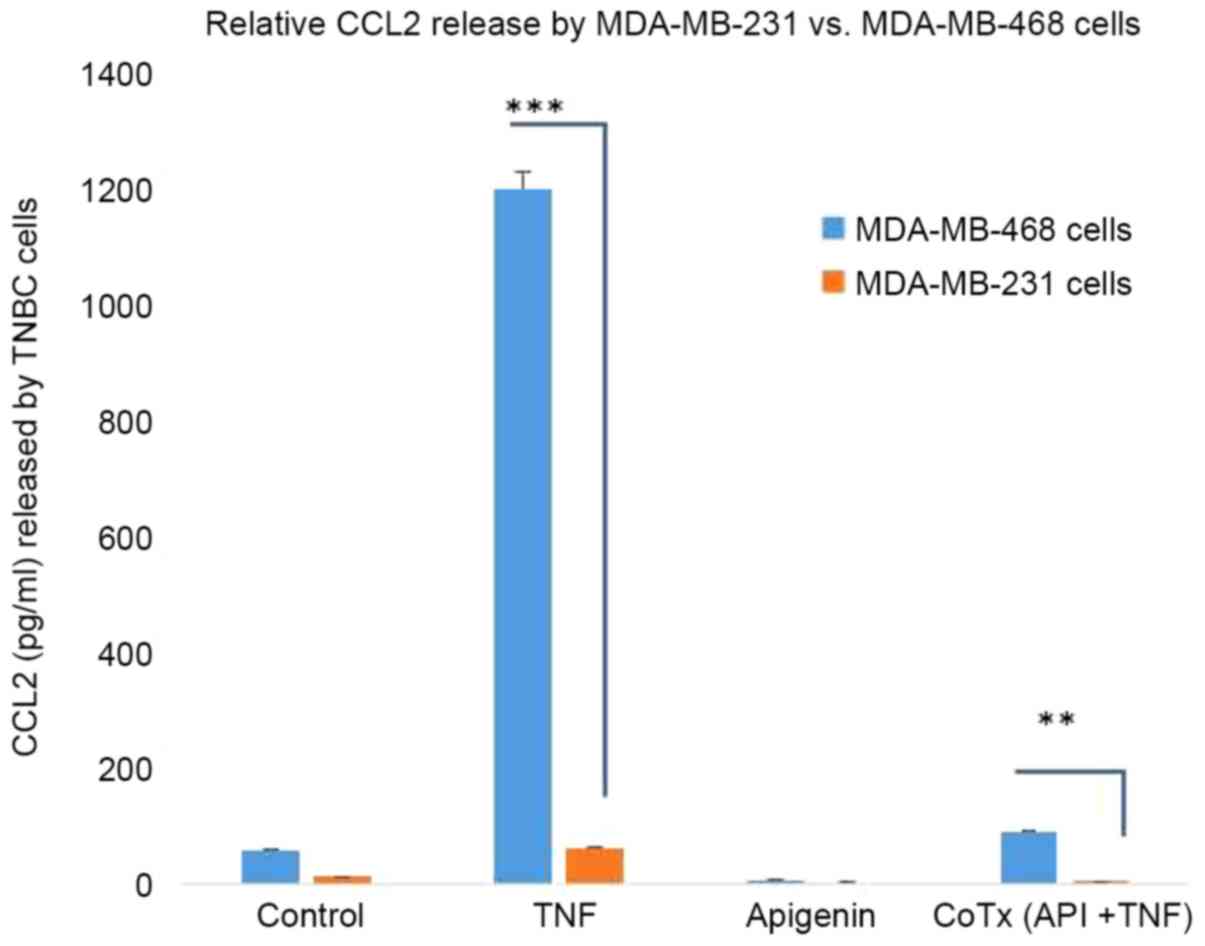
KF: Apigenin inhibits TNFalpha/IL-1alpha-induced CCL2 release
through IKBK-epsilon signaling in MDA-MB-231 human breast cancer
cells. PLoS One. 12:e01755582017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mazzio EA, Lewis CA and Soliman KFA:
Transcriptomic profiling of MDA-MB-231 cells exposed to boswellia
serrata and 3-O-acetyl-B-boswellic acid; ER/UPR mediated programmed
cell death. cancer genomics proteomics. 14:409–425. 2017.PubMed/NCBI
|
|
33
|
Garlapati C, Joshi S, Sahoo B, Kapoor S
and Aneja R: The persisting puzzle of racial disparity in triple
negative breast cancer: Looking through a new lens. Front Biosci
(Schol Ed). 11:75–88. 2019. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Newman LA, Jenkins B, Chen Y, Oppong JK,
Adjei E, Jibril AS, Hoda S, Cheng E, Chitale D, Bensenhaver JM, et
al: Hereditary susceptibility for triple negative breast cancer
associated with western sub-saharan african ancestry: Results from
an international surgical breast cancer collaborative. Ann Surg.
270:484–492. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hossain F, Danos D, Prakash O, Gilliland
A, Ferguson TF, Simonsen N, Leonardi C, Yu Q, Wu XC, Miele L and
Scribner R: Neighborhood social determinants of triple negative
breast cancer. Front Public Health. 7:182019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jiagge E, Jibril AS, Davis M,
Murga-Zamalloa C, Kleer CG, Gyan K, Divine G, Hoenerhoff M,
Bensenhave J, Awuah B, et al: Androgen receptor and ALDH1
expression among internationally diverse patient populations. J
Glob Oncol. 4:1–8. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bassey-Archibong BI, Hercules SM, Rayner
LGA, Skeete DHA, Smith Connell SP, Brain I, Daramola A, Banjo AAF,
Byun JS, Gardner K, et al: Kaiso is highly expressed in TNBC
tissues of women of African ancestry compared to Caucasian women.
Cancer Causes Control. 28:1295–1304. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Telonis AG and Rigoutsos I: Race
disparities in the contribution of miRNA isoforms and tRNA-derived
fragments to triple-negative breast cancer. Cancer Res.
78:1140–1154. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Torres-Luquis O, Madden K, N'Dri N M, Berg
R, Olopade OF, Ngwa W, Abuidris D, Mittal S, Lyn-Cook B and
Mohammed SI: LXR/RXR pathway signaling associated with
triple-negative breast cancer in African American women. Breast
Cancer (Dove Med Press). 11:1–12. 2018.PubMed/NCBI
|
|
40
|
Austin D, Hamilton N, Elshimali Y, Pietras
R, Wu Y and Vadgama J: Estrogen receptor-beta is a potential target
for triple negative breast cancer treatment. Oncotarget.
9:33912–33930. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu Y, Sarkissyan M, Clayton S, Chlebowski
R and Vadgama JV: Association of vitamin D3 Level with breast
cancer risk and prognosis in African-American and hispanic women.
Cancers (Basel). 9:E1442017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ma H, Ursin G, Xu X, Lee E, Togawa K, Duan
L, Lu Y, Malone KE, Marchbanks PA, McDonald JA, et al: Reproductive
factors and the risk of triple-negative breast cancer in white
women and African-American women: A pooled analysis. Breast Cancer
Res. 19:62017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Siddharth S and Sharma D: Racial disparity
and triple-negative breast cancer in African-American women: A
multifaceted affair between obesity, biology, and socioeconomic
determinants. Cancers (Basel). 10:E5142018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Dietze EC, Chavez TA and Seewaldt VL:
Obesity and triple-negative breast cancer: Disparities,
controversies, and biology. Am J Pathol. 188:280–290. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao X, Qu J, Sun Y, Wang J, Liu X, Wang
F, Zhang H, Wang W, Ma X, Gao X and Zhang S: Prognostic
significance of tumor-associated macrophages in breast cancer: A
meta-analysis of the literature. Oncotarget. 8:30576–30586.
2017.PubMed/NCBI
|
|
46
|
Jeong H, Hwang I, Kang SH, Shin HC and
Kwon SY: Tumor-associated macrophages as potential prognostic
biomarkers of invasive breast cancer. J Breast Cancer. 22:38–51.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Valeta-Magara A, Gadi A, Volta V, Walters
B, Arju R, Giashuddin S, Zhong H and Schneider RJ: Inflammatory
breast cancer promotes development of M2 tumor-associated
macrophages and cancer mesenchymal cells through a complex
chemokine network. Cancer Res. 79:3360–3371. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang N, Liu W, Zheng Y, Wang S, Yang B, Li
M, Song J, Zhang F, Zhang X, Wang Q and Wang Z: CXCL1 derived from
tumor-associated macrophages promotes breast cancer metastasis via
activating NF-κB/SOX4 signaling. Cell Death Dis. 9:8802018.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Liu Y, Zheng H, Li Q, Li S, Lai H, Song E,
Li D and Chen J: Discovery of CCL18 antagonist blocking breast
cancer metastasis. Clin Exp Metastasis. 36:243–255. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Little AC, Pathanjeli P, Wu Z, Bao L, Goo
LE, Yates JA, Oliver CR, Soellner MB and Merajver SD: IL-4/IL-13
stimulated macrophages enhance breast cancer invasion via
Rho-GTPase regulation of synergistic VEGF/CCL-18 signaling. Front
Oncol. 9:4562019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cassetta L, Fragkogianni S, Sims AH,
Swierczak A, Forrester LM, Zhang H, Soong DYH, Cotechini T, Anur P,
Lin EY, et al: Human tumor-associated macrophage and monocyte
transcriptional landscapes reveal cancer-specific reprogramming,
biomarkers, and therapeutic targets. Cancer Cell. 35:588–602.e510.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Gupta S, Jain A, Syed SN, Pflüger-Müller
B, Leisegang MS, Weigert A, Brandes RP, Ebersberger I, Brüne B and
Namgaladze D: IL-6 augments IL-4-induced polarization of primary
human macrophages through synergy of STAT3, STAT6 and BATF
transcription factors. Oncoimmunology. 7:e14941102018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Xu X, Ye J, Huang C, Yan Y and Li J: M2
macrophage-derived IL6 mediates resistance of breast cancer cells
to hedgehog inhibition. Toxicol Appl Pharmacol. 364:77–82. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tandon I and Sharma NK: Macrophage
flipping from foe to friend: A matter of interest in breast
carcinoma heterogeneity driving drug resistance. Curr Cancer Drug
Targets. 19:189–198. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sudhakaran M, Sardesai S and Doseff AI:
Flavonoids: New frontier for immuno-regulation and breast cancer
control. Antioxidants (Basel). 8:E1032019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Han R, Gu S, Zhang Y, Luo A, Jing X, Zhao
L, Zhao X and Zhang L: Estrogen promotes progression of
hormone-dependent breast cancer through CCL2-CCR2 axis by
upregulation of Twist via PI3K/AKT/NF-κB signaling. Sci Rep.
8:95752018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Dutta P, Sarkissyan M, Paico K, Wu Y and
Vadgama JV: MCP-1 is overexpressed in triple-negative breast
cancers and drives cancer invasiveness and metastasis. Breast
Cancer Res Treat. 170:477–486. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yao M, Fang W, Smart C, Hu Q, Huang S,
Alvarez N, Fields P and Cheng N: CCR2 chemokine receptors enhance
growth and cell-cycle progression of breast cancer cells through
SRC and PKC Activation. Mol Cancer Res. 17:604–617. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ling Z, Yang X, Chen X, Xia J, Cheng B and
Tao X: CCL2 promotes cell migration by inducing
epithelial-mesenchymal transition in oral squamous cell carcinoma.
J Oral Pathol Med. 48:477–482. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Xu W, Wei Q, Han M, Zhou B, Wang H, Zhang
J, Wang Q, Sun J, Feng L, Wang S, et al: CCL2-SQSTM1 positive
feedback loop suppresses autophagy to promote chemoresistance in
gastric cancer. Int J Biol Sci. 14:1054–1066. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wang T, Zhan Q, Peng X, Qiu Z and Zhao T:
CCL2 influences the sensitivity of lung cancer A549 cells to
docetaxel. Oncol Lett. 16:1267–1274. 2018.PubMed/NCBI
|
|
62
|
Sarin N, Engel F, Rothweiler F, Cinatl J,
Michaelis M, Frötschl R, Fröhlich H and Kalayda GV: Key players of
cisplatin resistance: towards a systems pharmacology approach. Int
J Mol Sci. 19:E7672018. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Regan DP, Coy JW, Chahal KK, Chow L,
Kurihara JN, Guth AM, Kufareva I and Dow SW: The angiotensin
receptor blocker losartan suppresses growth of pulmonary metastases
via AT1R-independent inhibition of CCR2 signaling and monocyte
recruitment. J Immunol. 202:3087–3102. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Lu J, Zhong H, Chu T, Zhang X, Li R, Sun
J, Zhong R, Yang Y, Alam MS, Lou Y, et al: Role of
anlotinib-induced CCL2 decrease in anti-angiogenesis and response
prediction for nonsmall cell lung cancer therapy. Eur Respir J.
53:18015622019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Yao Z, Zhang J, Zhang B, Liang G, Chen X,
Yao F, Xu X, Wu H, He Q, Ding L and Yang B: Imatinib prevents lung
cancer metastasis by inhibiting M2-like polarization of
macrophages. Pharmacol Res. 133:121–131. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Liu H, Wang SH, Chen SC, Chen CY and Lin
TM: Zoledronic acid blocks the interaction between breast cancer
cells and regulatory T-cells. BMC Cancer. 19:1762019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ku WT, Tung JJ, Lee TJ and Lai KC:
Long-term exposure to oroxylin a inhibits metastasis by suppressing
CCL2 in oral squamous cell carcinoma Cells. Cancers (Basel).
11:E3532019. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wang D, Yue DL, Wang D, Chen XF, Yin XY,
Wang YP, Yang L and Zhang Y: Aspirin inhibits cell stemness of
esophageal cancer by downregulation of chemokine CCL2. Zhonghua
Zhong Liu Za Zhi. 40:744–749. 2018.(In Chinese). PubMed/NCBI
|
|
69
|
Iwamoto H, Izumi K, Natsagdorj A, Naito R,
Makino T, Kadomoto S, Hiratsuka K, Shigehara K, Kadono Y, Narimoto
K, et al: Coffee diterpenes kahweol acetate and cafestol
synergistically inhibit the proliferation and migration of prostate
cancer cells. Prostate. 79:468–479. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ishii N, Araki K, Yokobori T, Hagiwara K,
Gantumur D, Yamanaka T, Handa T, Tsukagoshi M, Igarashi T, Watanabe
A, et al: Conophylline suppresses pancreatic cancer desmoplasia and
cancer-promoting cytokines produced by cancer-associated
fibroblasts. Cancer Sci. 110:334–344. 2019.PubMed/NCBI
|
|
71
|
Shen H, He M, Lin R, Zhan M, Xu S, Huang
X, Xu C, Chen W, Yao Y, Mohan M and Wang J: PLEK2 promotes
gallbladder cancer invasion and metastasis through EGFR/CCL2
pathway. J Exp Clin Cancer Res. 38:2472019. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Zhou L, Jiang Y, Liu X, Li L, Yang X, Dong
C, Liu X, Lin Y, Li Y, Yu J, et al: Promotion of tumor-associated
macrophages infiltration by elevated neddylation pathway via
NF-κB-CCL2 signaling in lung cancer. Oncogene. 38:5792–5804. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Ding M, He SJ and Yang J: MCP-1/CCL2
Mediated by autocrine loop of PDGF-BB promotes invasion of lung
cancer cell by recruitment of macrophages via CCL2-CCR2 axis. J
Interferon Cytokine Res. 39:224–232. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Tong J, Shen Y, Zhang Z, Hu Y, Zhang X and
Han L: Apigenin inhibits epithelial-mesenchymal transition of human
colon cancer cells through NF-κB/Snail signaling pathway. Biosci
Rep. 39:BSR201904522019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Lee HH, Jung J, Moon A, Kang H and Cho H:
Antitumor and anti-invasive effect of apigenin on human breast
carcinoma through suppression of IL-6 expression. Int J Mol Sci.
20:E31432019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Xu M, Li D, Yang C and Ji JS: MicroRNA-34a
inhibition of the TLR signaling pathway Via CXCL10 suppresses
breast cancer cell invasion and migration. Cell Physiol Biochem.
46:1286–1304. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhang J, Chen J, Guan GW, Zhang T, Lu FM
and Chen XM: Expression and clinical significance of chemokine
CXCL10 and its receptor CXCR3 in hepatocellular carcinoma. Beijing
Da Xue Xue Bao Yi Xue Ban. 51:402–408. 2019.(In Chinese).
PubMed/NCBI
|
|
78
|
Guo J, Xiao Y, Iyer R, Lu X, Lake M,
Ladror U, Harlan J, Samanta T, Tomlinson M, Bukofzer G, et al:
Empowering therapeutic antibodies with IFN-α for cancer
immunotherapy. PLoS One. 14:e02198292019. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wu Y, Yuan L, Lu Q, Xu H and He X:
Distinctive profiles of tumor-infiltrating immune cells and
association with intensity of infiltration in colorectal cancer.
Oncol Lett. 15:3876–3882. 2018.PubMed/NCBI
|
|
80
|
Fang S, Xu T, Xiong M, Zhou X, Wang Y,
Haydu LE, Ross MI, Gershenwald JE, Prieto VG, Cormier JN, et al:
Role of immune response, inflammation, and tumor immune
response-Related cytokines/chemokines in melanoma progression. J
Invest Dermatol. 139:2352–2358.e3. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Nieto JC, Zamora C, Porcel JM, Mulet M,
Pajares V, Muñoz-Fernandez AM, Calvo N, Espinosa I, Pascual-García
M, Bielsa S and Vidal S: Migrated T lymphocytes into malignant
pleural effusions: An indicator of good prognosis in lung
adenocarcinoma patients. Sci Rep. 9:29962019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Kang SH, Keam B, Ahn YO, Park HR, Kim M,
Kim TM, Kim DW and Heo DS: Inhibition of MEK with trametinib
enhances the efficacy of anti-PD-L1 inhibitor by regulating
anti-tumor immunity in head and neck squamous cell carcinoma.
Oncoimmunology. 8:e15150572019. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Tokunaga R, Zhang W, Naseem M, Puccini A,
Berger MD, Soni S, McSkane M, Baba H and Lenz HJ: CXCL9, CXCL10,
CXCL11/CXCR3 axis for immune activation-A target for novel cancer
therapy. Cancer Treat Rev. 63:40–47. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Morrison K, Challita-Eid PM, Raitano A, An
Z, Yang P, Abad JD, Liu W, Lortie DR, Snyder JT, Capo L, et al:
Development of ASG-15ME, a novel antibody-drug conjugate targeting
SLITRK6, a new urothelial cancer biomarker. Mol Cancer Ther.
15:1301–1310. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lin JK, Chen YC, Huang YT and Lin-Shiau
SY: Suppression of protein kinase C and nuclear oncogene expression
as possible molecular mechanisms of cancer chemoprevention by
apigenin and curcumin. J Cell Biochem Suppl. 28-29:39–48. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Alhalabi O, Rafei H, Shah A,
Siefker-Radtke A, Campbell M and Gao J: Targeting advanced
urothelial carcinoma-developing strategies. Curr Opin Oncol.
31:207–215. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Sanford T, Porten S and Meng MV: Molecular
analysis of upper tract and bladder urothelial carcinoma: Results
from a microarray comparison. PLoS One. 10:e01371412015. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Birt DF, Walker B, Tibbels MG and Bresnick
E: Anti-mutagenesis and anti-promotion by apigenin, robinetin and
indole-3-carbinol. Carcinogenesis. 7:959–963. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Birt DF, Mitchell D, Gold B, Pour P and
Pinch HC: Inhibition of ultraviolet light induced skin
carcinogenesis in SKH-1 mice by apigenin, a plant flavonoid.
Anticancer Res. 17:85–91. 1997.PubMed/NCBI
|
|
90
|
Sharma A, Ghani A, Sak K, Tuli HS, Sharma
AK, Setzer WN, Sharma S and Das AK: Probing into therapeutic
anti-cancer potential of apigenin: Recent trends and future
directions. recent pat inflamm Allergy Drug Discov. Aug
16–2019.doi: 10.2174/1872213X13666190816160240 (Epub ahead of
print). View Article : Google Scholar
|
|
91
|
Ginwala R, Bhavsar R, Chigbu DI, Jain P
and Khan ZK: Potential role of flavonoids in treating chronic
inflammatory diseases with a special focus on the anti-inflammatory
activity of apigenin. Antioxidants (Basel). 8:E352019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chen Z, Tian D, Liao X, Zhang Y, Xiao J,
Chen W, Liu Q, Chen Y, Li D, Zhu L and Cai S: Apigenin combined
with gefitinib blocks autophagy flux and induces apoptotic cell
death through inhibition of HIF-1α, c-Myc, p-EGFR, and glucose
metabolism in EGFR L858R+T790M-mutated H1975 cells. Front
Pharmacol. 10:2602019. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Chen X, Xu H, Yu X, Wang X, Zhu X and Xu
X: Apigenin inhibits in vitro and in vivo tumorigenesis in
cisplatin-resistant colon cancer cells by inducing autophagy,
programmed cell death and targeting m-TOR/PI3K/Akt signalling
pathway. J BUON. 24:488–493. 2019.PubMed/NCBI
|
|
94
|
Gao AM, Zhang XY, Hu JN and Ke ZP:
Apigenin sensitizes hepatocellular carcinoma cells to doxorubic
through regulating miR-520b/ATG7 axis. Chem Biol Interact.
280:45–50. 2018. View Article : Google Scholar : PubMed/NCBI
|