Introduction
Colon cancer is the third most common diagnosed
cancer with 1.8 million new cases in 2018 throughout the world,
with a poor prognosis (1). The
pathogenesis of colorectal cancer is complicate and multifactorial,
and therefore difficult to diagnose in the earlier stage (2). Although surgical resection remains the
only curative treatment for colon cancer, an alternative approach
to reduce the mortality rate is chemotherapy (3). For many years, 5-fluorouracil (5-FU) is
the most common used chemotherapy drug for colon cancer (4). Like other chemotherapeutics, however,
it affects not only the cancer cells but also normal cells. As the
dose increase of 5-FU, the side effects of the drug increase and
resistance to the drug develops frequently. Thus, new strategies
for the use of various natural products of plant origin in
chemotherapy to minimize toxicity and drug side-effects; over 60%
of anticancer drugs in use today are of natural origin (5). In combination with chemotherapy agents,
natural compounds have led to not only reduce risk of drug adverse
effects but can also improve the effectiveness of medication
(5). Natural compounds possess the
ability to modulate signaling pathways and regulated cell
cycle-regulated gene expression, cell differentiation and apoptosis
(5). Moreover, a lot of natural
compounds are well tolerated by humans. Although some natural
compounds have anticancer properties (6), cellular and molecular mechanisms
involved in their anticancer activity is still unclear.
Sasa quelpaertensis Nakai is a species of
bamboo grass native to South Korea and only grown on Halla mountain
of Jeju Island. Like other bamboo species, S. quelpaertensis
Nakai has been used in herbal medicine like other bamboo species
for various pharmacological properties such as antioxidant
(7–9), anticancer (10,11),
antidiabetic (12), inhibition of
tyrosinase and melanin production (13), hepatoprotective (9,14) and
anti-inflammatory (15) properties.
Recently, S. quelpaertensis Nakai has earn commercial
attention for its ability to prohibit human leukemia HL-60 cells
(16,17), gastric adenocarcinoma MKN-74
(17), colon cancer HT-29 (18) cells proliferation by inducing
apoptosis. In those study, an excessive and unregulated nitric
oxide (NO•) synthesis has been implicated to abrogation
of tumorigenicity and induction of apoptosis in tumor cells
(18,19). These results might be possible due to
enhancing effect of iNOS gene stimulated by an S.
quelpaertensis Nakai which may lead to excess cellular levels
of NO• which might be responsible for damage of
proteins, nucleic acids, membranes and organelles, which can lead
to activation of cell death processes such as apoptosis (20,21).
Nevertheless, only few reports are available regarding anticancer
and antiproliferative activity of S. quelpaertensis Nakai,
the precise action of S. quelpaertensis Nakai on apoptotic
mechanism is less well understood.
In the present study, we aimed to investigate the
effects of S. quelpaertensis Nakai to modulate
NO• signaling in human colon cancer cells as a possible
mechanism underlying S. quelpaertensis Nakai-induced
apoptosis. Furthermore, expression analysis of different mRNA and
proteins involved in regulation of S. quelpaertensis
Nakai-induced apoptotic signaling pathway based on p53 status was
also carried out by employing two p53 isogenic HCT116 cell lines,
p53 wildtype (p53-WT) and p53-deficent (p53-null) cells.
Materials and methods
Cell cultures and chemicals
Two isogenic HCT116 human colon carcinomas,
wild-type p53 (p53-WT) and complete knockout of p53 (p53-null)
cells, kindly gifted by Prof. Gerald N. Wogan (Massachusetts
Institute of Technology), were maintained at 37°C with 5% carbon
dioxide in McCoy's 5A medium (cat. no. 12-168F) supplemented with
10% fetal bovine serum (cat. no. 35-015-CV, Corning, NY, USA), 100
units/ml penicillin (cat. no. 17-602E), 100 µg/ml streptomycin
(cat. no. 17-602E) and 2 mM L-glutamine (cat. no. 17-605E).
Reagents and cell culture materials were purchased from the
following sources: Cell culture reagents, Lonza; annexin V-FITC
apoptotic assay kit (cat. no. 630109) and ApoAlert caspase-3
colorimetric assay kit (cat. no. 630217), Clontech Laboratories;
ECL™ western blotting detection reagents (cat. no. RPN2209), GE
Healthcare Bio-Sciences; RIPA lysis buffer (cat. no. R2002),
Biosesang; anti-bcl-2 antibody (cat. no. OP91), Calbiochem;
anti-actin antibody (C4, cat. no. sc-47778) and the secondary goat
anti-rabbit (cat. no. sc-2004) or anti-mouse (cat. no. sc-2005) IgG
conjugated to horseradish peroxidase, Santa Cruz Biotechnology;
anti-survivin antibody (cat. no. 2803), anti-CIAP-1 antibody (cat.
no. 4952), anti-CIAP-2 antibody (cat. no. 3130) and anti-XIAP
antibody (cat. no. R2042), Cell Signaling Technology.
Preparation of S. quelpaertensis Nakai
and its extracts
The leaves of S. quelpaertensis Nakai used
for the present study were plucked during February 2012 from Mt.
Halla on Jeju island in South Korea, and a voucher sample preserved
for reference in the herbarium of Jeju National University. Fresh
leaves were washed with water, drained, dried and powdered. Dried
S. quelpaertensis Nakai leaf (25 g) was extracted with 70%
ethanol (250 ml) on a rotary shaker for 24 h and filtered with a
Sep-Pak C18 cartridge and a 0.45 µm membrane filter
(Waters Corporation). The extracted liquid was concentrated using a
rotary vacuum evaporator (Buchi Rotavapor R-200; Sigma-Aldrich),
freeze dried and kept at −20°C until used for experiment. The
extraction yield of ethanol extracts was determined to be
11.8±0.13%.
Assessment of cell viability
HCT 116 cells were seeded at 1×106
cells/well into 6-well flat-bottom tissue culture plates the day
before treatment. Cells were cultured for 24, 48 and 72 h in
McCoy's 5A medium containing 0, 25, 50, 100 and 200 µg/ml of S.
quelpaertensis Nakai extracts. HCT116 cell viability was
determined 24 h after treatment was determined by trypan blue
exclusion. Cell viability was calculated as relative to control
cells frown in culture medium with DMSO.
Apoptosis analysis
After treatment with 200 µg/ml of S.
quelpaertensis Nakai extracts for 24, 48 and 72 h, cells were
labeled using a FITC Annexin V apoptotic assay kit (Clontech).
Cells were stained for 15 min at room temperature in the dark with
FITC-conjugated annexin V (5 µl) and PI (5 µl). Following
incubation, the cells were analyzed in a Becton Dickinson FACScan
(excitation at 488 nm) equipped with CellQuest software. Early
apoptotic cells were labeled with only annexin V, necrotic cells
were stained with propidium iodide or with both annexin V and
propidium iodide, and living cells were negative for both staining.
Cells treated with argon gas served as negative controls, and those
treated with 2.5 µM etoposide in culture medium for 6 h as served
as positive controls.
Cell cycle analysis
For analysis of the cell cycle profile, HCT116 cells
(2×106 cells/100-mm dishes) were cultured for 24 h in
McCoy's 5A medium, and the cells were harvested after 24, 48 or 72
h treatment with 200 µg/ml of S. quelpaertensis Nakai
extracts by trypsinization and by centrifugation at 1,000 rpm for
10 min. The cells were washed twice with ice-cold PBS and then
fixed in 70% (v/v) ethanol overnight. Whole cells were incubated
with 1% BSA (bovine serum albumin)-PBS solution containing 500
µg/ml PI and 10 µg/ml RNasse for 30 min at 37°C. Cellular DNA
content and apoptotic cells based on the PI signal and sub-G1 peak
were measured using a Becton Dickinson FACScan (BD Bioscience). The
percentage of cells in each phase of the cell cycle were determined
by a Becton Dickinson FACScan (BD Bioscience) equipped with
CellQuest Pro™ software (BD Bioscience), and expressed as a
percentage of cells in the respective phases.
Measurement of nitrite production
After each period of exposure, the nitrite levels in
the culture media was assessed by measuring nitrite in media
fractions by the Griess reaction (22). Nitrite concentrations were determined
from a standard curve using sodium nitrite and the values were
expressed as pmoles per 109 viable (trypan
blue-excluding) cells.
RNA isolation and semi-quantitative
RT-PCR analysis
Total RNA was extracted according to the Tri Reagent
(Sigma-Aldrich) supplier's protocol. The RNA was resuspended in
RNase-free buffer, the concentration and purity were measured by UV
spectrophotometer at 260 and 280 nm. Total RNA was reverse
transcribed using the TOP script™ one-step RT PCR kit (Enzynomics)
as described previously (22). In
brief, 1 µg total RNA, forward and reverse oligo(dT)15
primers (20 pmol. each) (Table I), 5
µl one-step RT PCR DyeMix, 25 units of ribonuclease inhibitor,
sterile water were added to a final volume of 20 µl. The conditions
for each PCR were 95°C for 30 sec, 55°C for 30 sec, 72°C for 1 min
for a total of 35 cycles. In all cases RNA samples were tested for
their ability to generate a PCR signal by using positive control
β-actin primers from Bionics. The resulting cDNA was visualized by
1.5% agarose gel electrophoresis following by ethidium bromide. The
primer sequences used in semiquantitative RT-PCR analysis were
listed in Table I.
 | Table I.Oligonucleotides used in
semi-quantitative reverse transcription PCR. |
Table I.
Oligonucleotides used in
semi-quantitative reverse transcription PCR.
| Gene name | Sequence
(5′-3′) |
|---|
| eNOS | Sense:
CCAGCTAGCCAAAGTCACCAT |
|
| Antisense:
GTCTCGGAGCCATACAGGATT |
| iNOS | Sense:
CCAGTGACACAGGATGACCTTCAG |
|
| Antisense:
TGCCATTGTTGGTGG AGTAACG |
| nNOS | Sense:
TTGGGGGCCTGGGATTTCTGG |
|
| Antisense:
GTTGGCATGGGGGAGTGAGC |
| Survivin | Sense:
GCATGGGTGCCCCGACGTTG |
|
| Antisense:
GCTCCGGCCAGAGGCCTCAA |
| CIAP-1 | Sense:
AAGTTCCTACCCCTGTCCAATG |
|
| Antisense:
CAAGTAGATGAGGGTAACTGGC |
| CIAP-2 | Sense:
CCTGTGGTTAAATCTGCCAATG |
|
| Antisense:
CAATTCGGCACCATAACTCTG |
| XIAP | Sense:
ACACCATATACCCGAGGAAC |
|
| Antisense:
CTTGCATACTGTCTTTCTGAGC |
| β-actin | Sense:
GGTCATCTTCTCGCGGTTGGCCTTGGGGT |
|
| Antisense:
CCCCAGGCACCAGGGCGTGAT |
Protein isolation and western blot
analysis
The cells were collected after treatment and lysed
with 450 µl of iced-cold RIPA lysis buffer by incubating for 30–60
min at 4°C. The lysates were centrifuged at 10,000 × g for 10 min
at 4°C, and the protein concentrations were determined using the
Bio-Rad Protein Assay kit (Bio-Rad Laboratories Inc.). Equal
amounts of total proteins (50 µg) were mixed with loading buffer,
senatured, and separated on 15% sodium dodecylsulfate
polyacrylamide gel electrophoresis gels, and then blotted onto
polyvinylidene difluoride (PVDF) membrane (Bio-Rad). The membranes
were blocked with 5% non-fat milk at room temperature for 1 h, and
incubated with specific primary antibodies at room temperature for
2 h. The primary antibodies used in this study were as follows:
Survivin (1:1,000); CIAP-1 (1:1,000); CIAP-2 (1:1,000); XIAP
(1:1,000); Bcl-2 (1:1,000); PARP (1:1,000), or β-actin (1:10,000).
After washing with TBST (TBS containing 0.05% Tween-20, pH 7.6)
three times, the membranes were incubated with the corresponding
peroxidase-conjugated secondary goat anti-rabbit or mouse IgG
(diluted 1:8,000) for 1 h at room temperature. Following washing
with TBST six times, the proteins signal was detected using
Hyperfilm ECL. Densitometric analyses of resultant western blots
were performed with a ChemiDoc MP Imaging System (Bio-Rad
Laboratories).
Caspase 3 activity assay
The enzymatic activity of caspases induced by sodium
butyrate was recorded using an ApoAlert caspase-3 colorimetric
assay kit based on the manufacturer's protocol (Clontech
Laboratories). Briefly, two million cells were lysed in a lysis
buffer for 10 min on an ice bath. The lysed cells were centrifuged
at 16,000 × g for 10 min at 4°C, and 100 µg protein was incubated
with 50 µl of 2× reaction buffer/DTT Mix and 50 µM of caspase 3
substrate DEVD-pNA at 37°C for 1–3 h. The optical density of the
reaction mixture was measured by changes in absorbance at 405 nm
using a µQuant plate reader from Biotek Instruments Inc.
Statistical analysis
The data are presented as the mean ± SD. Statistical
significances were analyzed by one-way analysis of variance with
post hoc Dunnett's test. P<0.05 was considered to indicate a
statistically significant difference (SPSS v.12.0) at a
significance level of P<0.05 and P<0.01.
Results
S. quelpaertensis Nakai inhibits the
proliferation of human colorectal cancer cells
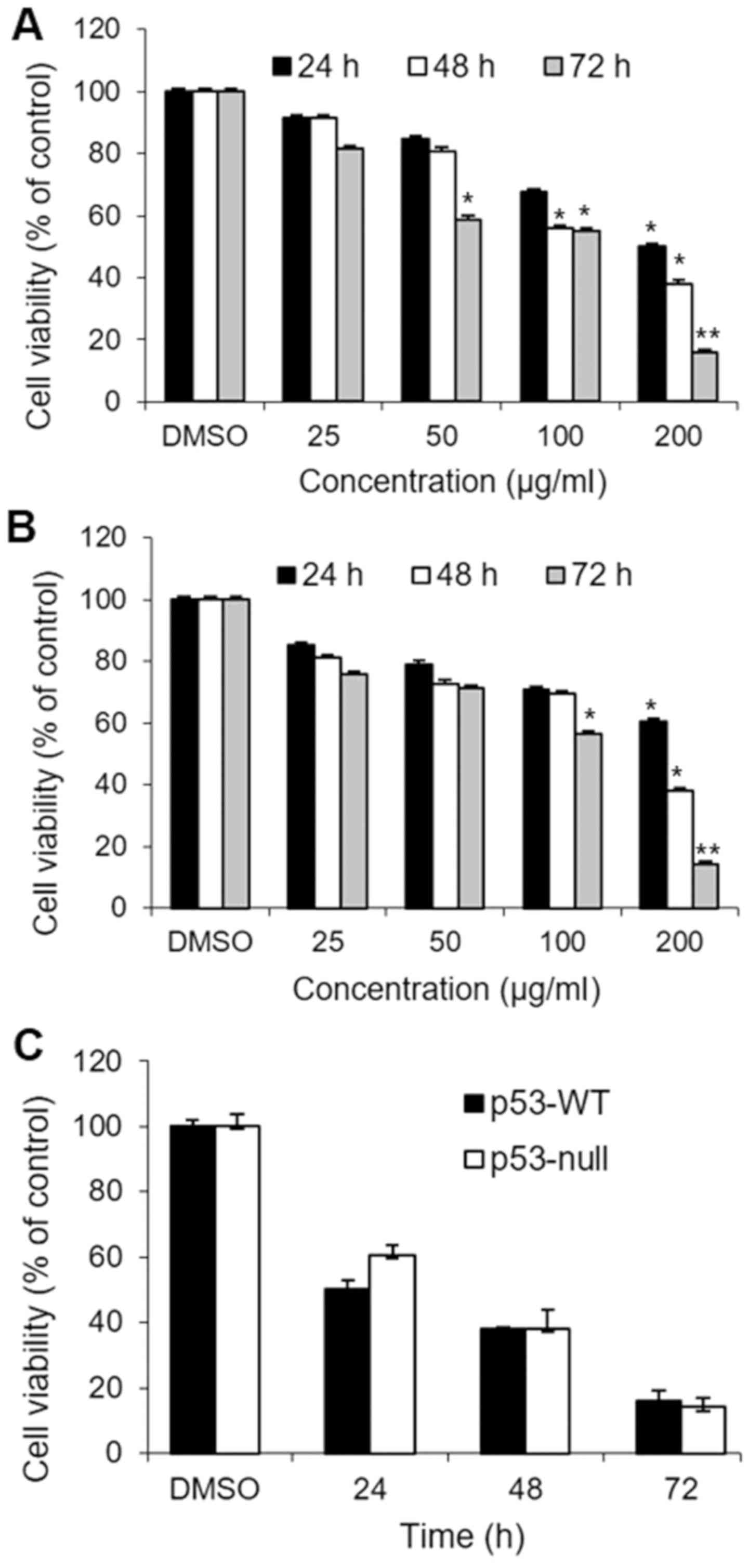
We first evaluated the cytotoxic effects of S.
quelpaertensis Nakai extracts in HCT 116 human colon carcinoma
cells. Both p53-WT and p53-null cells were treated with 0, 25, 50,
100 and 200 µg/ml of S. quelpaertensis Nakai for 24, 48 and
72 h responded similarly, with respect to viability, in that both
treatments decreased the percentage of viable cells dose- and
time-dependently (Fig. 1A and B).
The DMSO vehicle did not affect cell viability relative to colon
cancer cells cultured in medium alone. Maximum curcumin-induced
cytotoxicity was evident after 72 h exposure to 200 µg/ml of S.
quelpaertensis Nakai extracts (P<0.01). Exposure to 200
µg/ml S. quelpaertensis Nakai for 72 h, reduced viability in
p53-WT and p53-null cells to 16 and 14%, respectively, whereas
comparable values after treatment with 200 µg/ml S.
quelpaertensis Nakai extracts for 24 h, were 50 and 60%
(Fig. 1A and B). Fig. 1C shows that there was no significant
difference between S. quelpaertensis Nakai extracts-treated
p53-WT and p53-null cells with respect to cell viability at 200
µg/ml, implying that p53 activation was not required for S.
quelpaertensis Nakai extracts-induced cytotoxicity.
S. quelpaertensis Nakai induces the
apoptosis of p53-WT and p53-null HCT116 cells
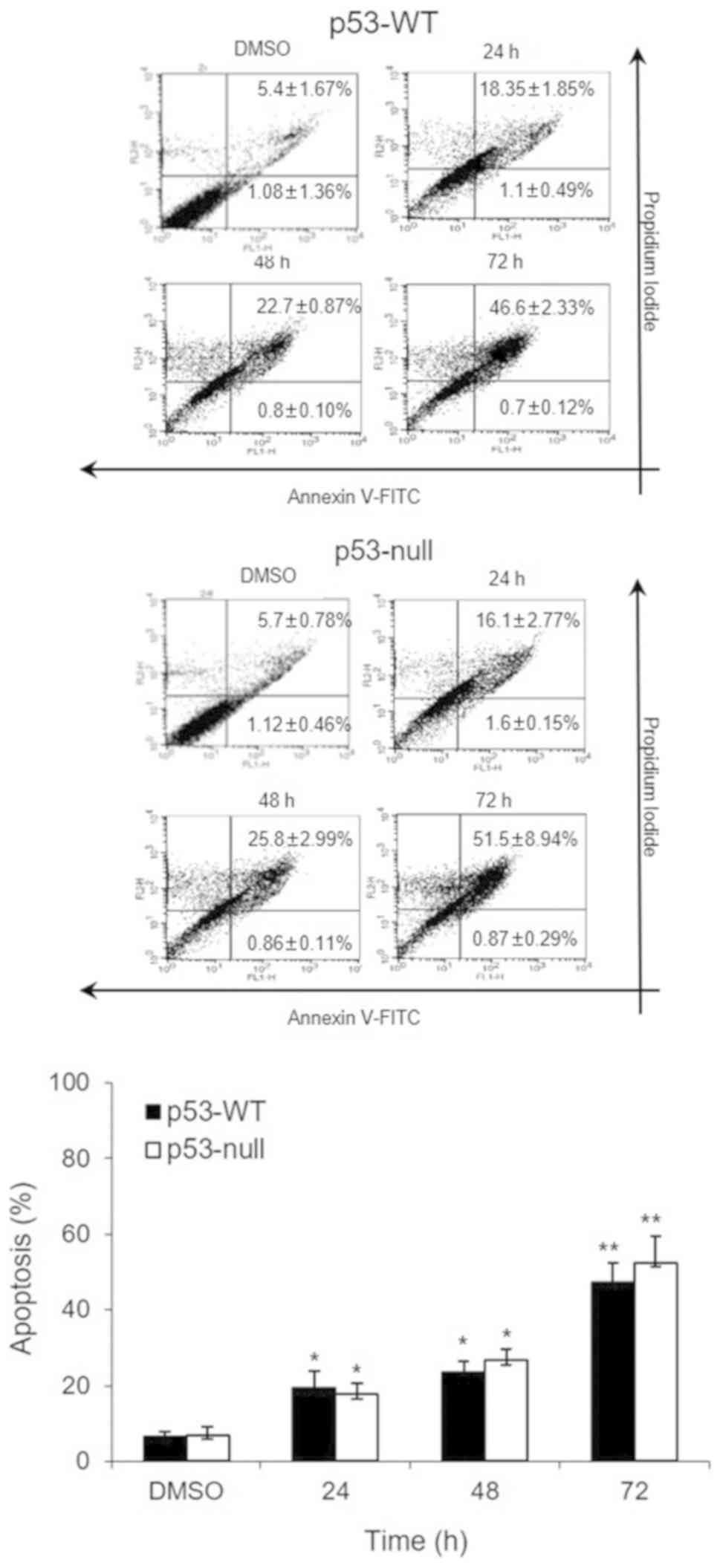
Fig. 2 shows that 200
µg/ml of S. quelpaertensis Nakai extracts-induced apoptosis
in each of the cell types. Approximately 19.5 and 23.5% of p53-WT
cells were apoptotic after S. quelpaertensis Nakai extracts
treatment for 24 and 48 h, respectively (8.6- and 10.4-fold,
respectively, over control level) (Fig.
2). These apoptotic cell deaths by S. quelpaertensis
Nakai extracts were observed to be approximately the same in
p53-null cells as well (Fig. 2).
Treatment with 200 µg/ml of S. quelpaertensis Nakai extracts
for 24 and 48 h resulted in 17.7 and 26.6% of apoptosis,
respectively (6.8- and 10.2-fold increases, P<0.05 and
P<0.01; Fig. 2). A stronger
apoptotic response was induced by S. quelpaertensis Nakai
extracts treatment for 72 h, inducing maximum frequencies of 47.3
and 52.4% in both cell lines (20.8- and 20.1-fold elevation over
controls, P<0.01; Fig. 2).
S. quelpaertensis Nakai regulates cell
cycle arrest along with increase of sub-G1 population
To examine the appearance of the sub-G1 fraction, an
indicator of apoptotic cell death, PI staining of DNA and flow
cytometry were done in 200 µg/ml of S. quelpaertensis Nakai
extracts-treated p53-WT and p53-null cells. S.
quelpaertensis Nakai extracts significantly increase the
appearance of sub-G1 fraction from 2.1 and 1.9% under control
conditions to 30.5 and 31% after 72 h of treatment in p53-WT and
p53-null cells (P<0.01), respectively, indicating an increase of
S. quelpaertensis Nakai extracts-induced apoptotic cell
death (Table II). Furthermore, the
stage at which growth inhibition induced by with S.
quelpaertensis Nakai extracts occurs in the p53-WT and p53-null
HCT116 cell cycle progressions were determined, with cellular
distribution in the different phases the treatment (Table II). In DMSO controls, flow cytometry
analysis showed 76.6 and 75.4% of cells in G0/G1 phase, 4.8 and
5.6% of cells in S phase, and 13.6 and 13.5% of cells in G2/M phase
in p53-WT and p53-null HCT116 cells, respectively. In contrast, in
cells treated with 200 µg/ml of S. quelpaertensis Nakai
extracts for 72 h, the proportions of cells in G0/G1, S, and G2/M
phases were 15.7 and 12%, 14.3 and 16.6%, and 35.7 and 29.87% in
p53-WT and p53-null cells, respectively. These results showed that
the percentage of S and G2/M phases cells increased, while those in
the G1 phase decreased after treatment with S.
quelpaertensis Nakai (P<0.05 and P<0.01), suggesting that
it promotes cell growth inhibition by inducing S and G2/M phase
arrests in both p53-WT and p53-null HCT116 cells (Table II).
 | Table II.Effect of S. quelpaertensis
Nakai extracts (200 µg/ml) on cell cycle distribution in p53-WT and
p53-null HCT116 cells. |
Table II.
Effect of S. quelpaertensis
Nakai extracts (200 µg/ml) on cell cycle distribution in p53-WT and
p53-null HCT116 cells.
|
|
| Non-apoptotic cells
(%) |
|---|
|
|
|
|
|---|
| Treatment | Apoptotic cells (%)
(sub-G1) | G0/G1 | S | G2/M |
|---|
| p53-WT |
|
DMSO | 2.1±0.33 | 76.6±6.31 | 4.8±2.28 | 13.6±11.24 |
| 24
h |
10.0±0.97a |
48.9±1.83a | 8.1±0.33 | 27.0±2.25 |
| 48
h |
20.2±4.31a |
37.0±2.91a | 7.7±0.37 |
28.0±2.00a |
| 72
h |
30.5±4.9b |
15.7±0.38b |
14.3±0.54a |
35.7±2.26a |
| P53-null |
|
DMSO | 1.9±0.39 | 75.4±2.29 | 5.6±0.51 | 13.5±0.95 |
| 24
h |
12.0±0.70a |
47.1±1.90a | 6.6±0.52 | 23.9±1.97 |
| 48
h |
19.2±1.25a |
34.2±2.45a | 8.4±0.20 |
24.3±8.06a |
| 72
h |
31.0±2.07b |
12.0±3.00b |
16.6±0.72a |
29.8±9.75a |
Effects of S. quelpaertensis Nakai on
nitrite production and NOS isoenzymes expression
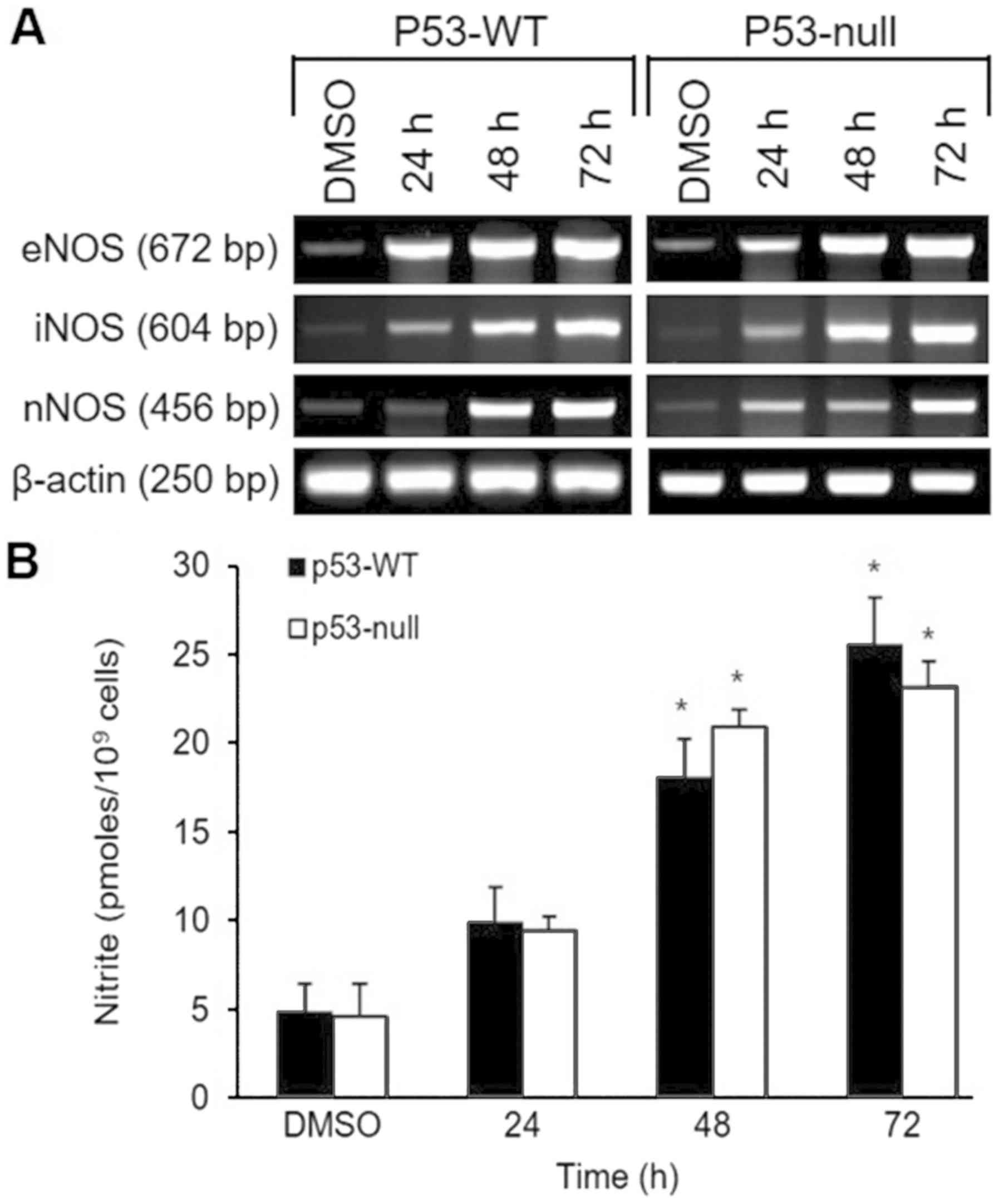
The effect of S. quelpaertensis Nakai on
three isoforms of NOS (eNOS, iNOS and nNOS) mRNA expression was
evaluated by use of semiquantitative RT-PCR (Fig. 3A). RT-PCR analysis showed that after
48 and 72 h treatment with 200 µg/ml of S. quelpaertensis
Nakai induced increases in each NOS isoform in p53-WT and p53-null
cells, with apparently equal potency (Fig. 3A). Expression of the β-actin gene and
production of its mRNA was not altered during any of the treatments
(Fig. 3A). These findings are
consistent with the time-dependent increases in nitrite production
were observed in both cell type in response to S.
quelpaertensis Nakai extracts treatments (Fig. 3B).
S. quelpaertensis Nakai regulated
expression of inhibitors of apoptosis (IAP) family
In this study, we subsequently investigated
mechanism underlying cell death induced by S. quelpaertensis
Nakai by RT-PCR and western blotting, with a focus upon
antiapoptotic activity of IAP family member (Fig. 4). Compared with controls, the CIAP-1
and CIAP-2 mRNA expression were remarkably down-regulated after
treated with S. quelpaertensis Nakai extracts for 24, 48 and
72 h, while the level of Survivin and XIAP mRNA expression did not
decrease until both p53-WT and p53-null cells were exposed to S.
quelpaertensis Nakai extracts for 72 h (Fig. 4A). Moreover, treatment of the cells
with S. quelpaertensis Nakai extracts for 24, 48 and 72 h
led to a reduction of Survivin, CIAP-1 and CIAP-2 proteins in both
p53-WT and p53-null HCT116 cells (Fig.
4B). In contrast, no apparent changes of XIAP mRNA and protein
expressions were found in S. quelpaertensis Nakai-treated
group compared with DMSO controls (Fig.
4A and B).
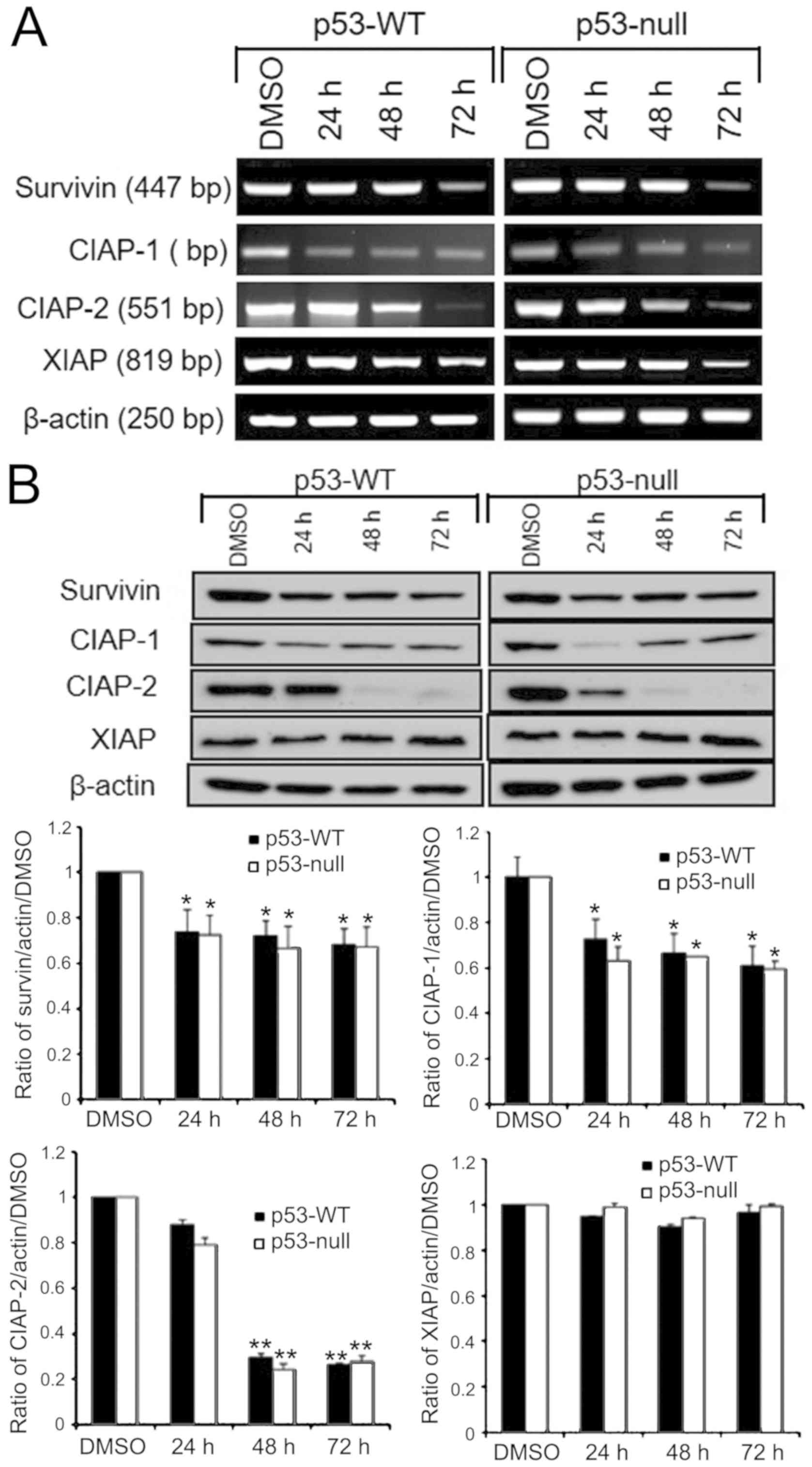 | Figure 4.mRNA and protein expression levels of
IAP in human colon cancer cells after treatment with S.
quelpaertensis Nakai. (A) Reverse transcription PCR and (B)
western blot analyses of the IAP family (Survivin, CIAP-1, CIAP-2
and XIAP) mRNA and protein levels in p53-WT and p53-null HCT116
cells treated with 200 µg/ml S. quelpaertensis Nakai extract
for 24, 48 and 72 h. Semi-quantitative PCR was performed using
primers specific to survivin, CIAP-1, CIAP-2 and XIAP or a β-actin
control on 1 µg total RNA prepared. Additionally, cell lysates were
prepared and subjected to western blot analysis using specific
antibodies. Band intensities were calculated by densitometric
analysis and normalized to actin levels. Typical results from three
independent experiments are shown. The statistical significance of
the results was analyzed by one-way ANOVA and post hoc Dunnett's
test. *P<0.05 and **P<0.01 vs. DMSO. S. quelpaertensis,
Sasa quelpaertensis; WT, wild type; CIAP, cellular inhibitor of
apoptosis; XIAP, X-linked inhibitor of apoptosis. |
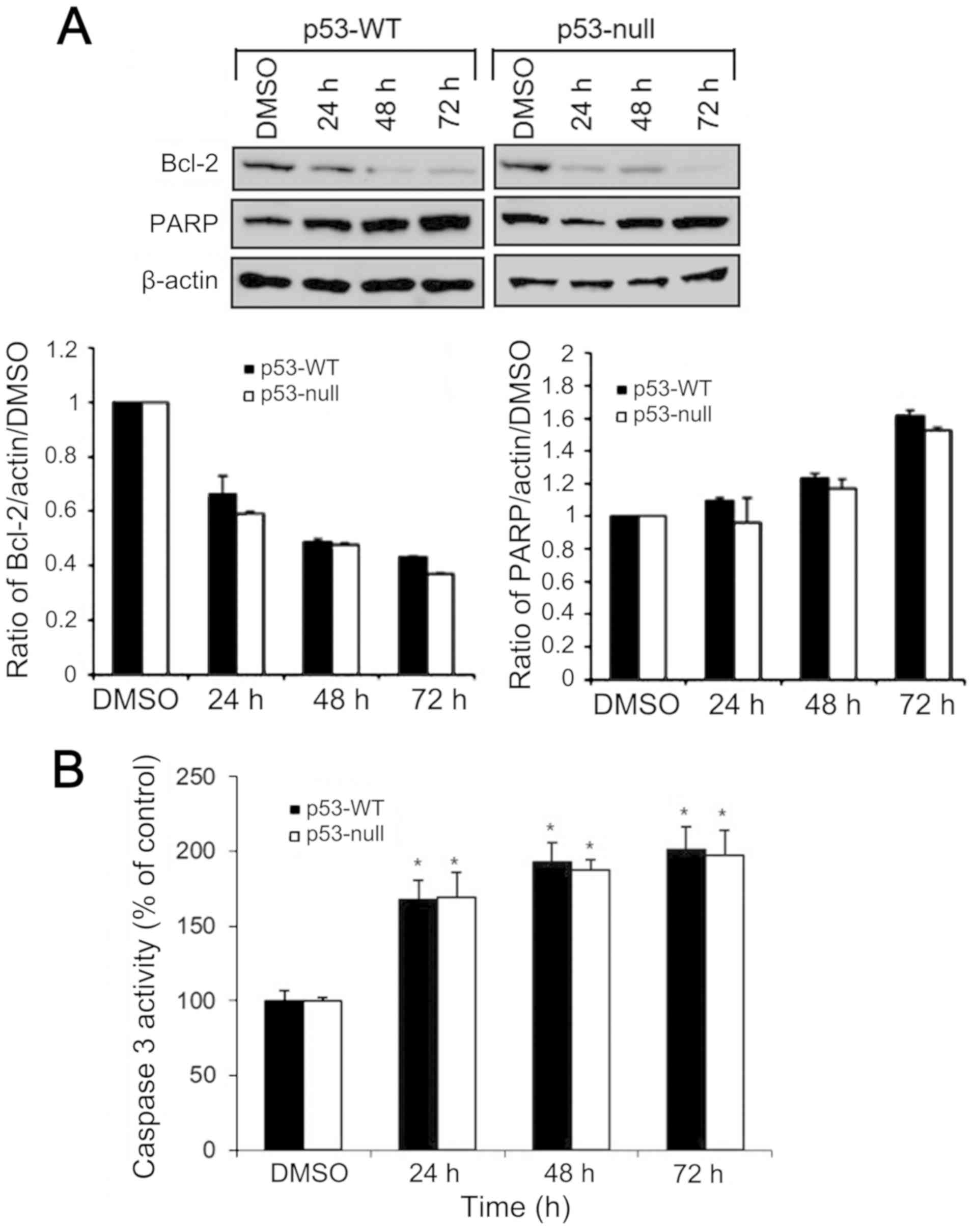
Effects of S. quelpaertensis Nakai
treatment on Bcl-2. PARP expression and caspase 3 activity
The levels of Bcl-2 and PARP protein expression in
HCT116 cells were analyzed by western blotting. The p53-WT and
p53-null cells were treated with 200 µg/ml of S.
quelpaertensis Nakai extracts for 24, 48 and 72 h. Compared
with the control group, treatment with S. quelpaertensis
Nakai downregulated the expression of Bcl-2, whereas the levels of
PARP were upregulated in both cells (Fig. 5A). To further determine the apoptotic
cell death induced by S. quelpaertensis Nakai, HCT116 cells
were left untreated or treated with 200 µg/ml of S.
quelpaertensis Nakai extracts for 24, 48 and 72 h, and
caspase-3 activity assay in a colorimetric assay based on the
cleavage of the synthetic peptide Ac-DEVD-pNA was done (Fig. 5B). S. quelpaertensis Nakai
increased caspase-3 activity by approximately 2-fold (Fig. 5B).
Discussion
Cancer is affecting millions of people every year
and our emphasis is to explore appropriate natural sources and to
suggest a novel anticancer candidate that can combat cancer in a
better way. We have recently reported antioxidant and anticancer
activities of the ethanol and water extracts of Sasa
quelpaertensis Nakai leaves (19,23). The
results showed that both extracts showed antioxidant activities
with different magnitudes of potency. The ethanol extracts of S.
quelpaertensis Nakai contained a larger quantity of phenolics
and flavonoids (2.1- and 4.6-fold, respectively) and exhibited
stronger radical scavenging, ferrous chelating and reducing power
abilities, and an anti-proliferative effect on HCT116 colon cancer
cells when compared to water extracts (23). In addition, other studies revealed
that S. quelpaertensis Nakai extracts showed
antiproliferation and apoptosis induction properties in human
leukemia HL-60 cells (17), as well
as in human and lung cancer A549 and H1299 cells. Despite previous
reports demonstrated that potential clinical application of dwarf
bamboo S. quelpaertensis Nakai extracts for prevention or
treatment of neoplastic disease (19,23,24), the
underlying mechanism has not been clarified.
The aim of the present study was to investigate
molecular mechanisms of cell death induced by S.
quelpaertensis Nakai extracts in HCT116 colon carcinoma cells.
In the present study, we observed that S. quelpaertensis
Nakai decrease of cell viability with increasing concentrations at
different treatment times in p53-WT and p53-null HCT116 cells. Flow
cytometric analysis suggested that apoptosis was a major
contributor to cell death induced by S. quelpaertensis Nakai
in both cell types; nearly 47% of p53-WT and 52% of p53-null cells
stained with PI and annexin V 72 h after S. quelpaertensis
Nakai treatment (P<0.01). DNA content measurement by Cell cycle
analysis showed a remarkable accumulation of subploid cells in the
sub-G1 area in both p53-WT and p53-null cells after treatment with
S. quelpaertensis Nakai for 72 h when compared with the DMSO
control group (P<0.01; Table
II). Since accumulation of the sub-G1 peaks indicated
characteristics of apoptosis, our results provide strong evidence
for cytotoxicity induced by with S. quelpaertensis Nakai
resulting in the decrease of the number of viable cells.
Nitric oxide (NO•) is a free radical
messenger molecule that plays a crucial role in controlling various
physiological functions in vivo (25,26).
This molecule is produced by three different isoforms of the enzyme
nitric oxide synthase (NOS) which can regulate biological activity
in a variety of cells (25).
NO• has also been shown to be involved in many of the
pathophysiological processes that contribute to the development and
progression of cancer (27). Based
on the existing literature, it is clear that NO• may be
viewed as a double-edged sword in cancer (27,28).
High concentrations of NO• may mediate cancer cell
apoptosis and the inhibition of cancer growth, whereas cancer
growth and proliferation is promoted at low concentrations of
NO•. The regulation of cancer growth by NO•
represents an important player in cancer research, including colon
cancer (25,27,28). In
this study, S. quelpaertensis Nakai extracts caused
upregulation of all endogenous NOS activities and, in turn, an
increase in NO• production leading to cell death.
Seventy two h after treatment with S. quelpaertensis Nakai
induced the highest level of NOS expression and 4.7- to 5.5-fold
higher NO• production, compared with compared with DMSO
control, indicating a direct relationship between increased
NO• production and the loss of cell viability, caused by
S. quelpaertensis Nakai.
Because mechanisms through which S.
quelpaertensis Nakai extracts induce cell death are poorly
understood, we here investigated their effects on apoptotic
signaling pathways. In the current study, treatment of the cells
with 200 µg/ml of S. quelpaertensis Nakai for 24, 48 and 72
h led to a reduction of survivin, CIAP-1, CIAP-2 and XIAP in both
p53-WT and p53-null HCT116 cells, suggesting the high expression of
these IAPs in human colon cancer cells may act as a contributing
factor to resistance by S. quelpaertensis Nakai. Activation
of the nuclear factor κB (NF-κB) transcription factor plays an
important role in inhibition of apoptotic pathway (29). Apoptosis-regulatory IAP family such
as survivin, CIAP-1, CIAP-2 and XIAP is transcriptionally regulated
by NF-κB (30). These IAPs have been
reported to block apoptosis by direct binding to caspases such as
caspase-3 and caspase-9, indicating that expression of IAPs under
the control of NF-κB plays an important role in the anti-apoptotic
pathway (30).
Here we also observed an increase in the PARP and
caspase 3 activity in S. quelpaertensis Nakai
extracts-treated colon cancer cells while suppressing expression of
anti-apoptotic proteins such as Bcl-2, implying apoptosis induction
that involved downregulation of Bcl-2 and cleavage of PARP, and its
mechanism may be associated with the Bcl-2/caspase-3 signaling
pathway. Caspase-3 is a family of cysteine proteases and plays a
crucial role in the execution phase of apoptosis, and that its
activation often marks the commitment to apoptosis (31,32).
Bcl-2 inhibits cytochrome c release from mitochondria as well as
caspase-3 (32,33). PARP is cleaved by caspase-3, which
causes apoptosis (32,34). Our findings are therefore consistent
with other reports that S. quelpaertensis Nakai triggers
apoptosis in human leukemia HL-60 (16,17) and
gastric cancer MKN-74 (17) cells by
up-regulation of Bax, caspase 3 and PARP as well as down-regulation
of anti-apoptotic proteins such as survivin and Bcl-2.
The p53 tumor suppressor gene is a critical
regulator of cell survival and proliferation, activated by cellular
stresses including DNA damage, oncogene activation and cytotoxic
agents (35–37). The loss of p53 activity promotes
tumorigenesis in various organs including colon (38). Generally, tumor therapy including
radiotherapy and chemotherapy, which can induce p53-mediated
promotion in tumor cells (35–37).
Although disruption of p53 expression generally facilitates cancer
cell resistance to chemotherapy in some studies, it might not be
concluded that p53 negative tumors are always less sensitive to
these drugs (38,39). The effects of p53 on chemosensitivity
can be dependent both on external stimulation types and on internal
genetic environment of the cells (38). In the present study, we
systematically explored the possible role of p53 in the
proapoptotic activity of S. quelpaertensis Nakai using
p53-isogenic pair of colon cancer cell-lines and demonstrated that
exposure to S. quelpaertensis Nakai caused apoptosis in
colon cancer cells indiscriminately of p53 status since a similar
cytotoxic and apoptotic effects were observed in p53-WT and
p53-null HCT116 cells, demonstrating that p53 is not the only
determinant of the fate of S. quelpaertensis Nakai-treated
colon cancer cells. This might be possibly explained by the
induction of apoptosis proceeds through a caspase-mediated
mitochondria amplification regardless of p53 status.
Taken together, our results indicate that S.
quelpaertensis Nakai-induced apoptosis in HCT 116 colon cancer
cells was independent of p53 expression. Furthermore, this study
demonstrates that oxidative stress as a result of NO•
production triggered p53-independent apoptosis. Therapeutic
application of S. quelpaertensis Nakai is therefore
predicted to be effective against colon cancers cells irrespective
of their p53 status.
Acknowledgements
The author would like to thank Professor Gerald
Norman Wogan (Department of Biological Engineering, Massachusetts
Institute of Technology) for providing human colon cancer cell
lines.
Funding
The present study was supported by the Basic Science
Research Program (grant nos. 2017R1D1A1B03028849,
2016R1A6A1A03012862 and 2014R1A1A2056292) through the National
Research Foundation of Korea (NRF) funded by the Ministry of
Education, Science and Technology, Republic of Korea.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
MYK conceived the study, designed the experiments,
performed the experiments, analyzed the data, and wrote and
approved the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The author declares that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Palma S, Zwenger AO, Croce MV, Abba MC and
Lacunza E: From molecular biology to clinical trials: Toward
personalized colorectal cancer therapy. Clin Colorectal Cancer.
15:104–115. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Janne PA and Mayer RJ: Chemoprevention of
colorectal cancer. N Engl J Med. 342:1960–1968. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
de Gramont A, Figer A, Seymour M, Homerin
M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer
G, et al: Leucovorin and fluorouracil with or without oxaliplatin
as first-line treatment in advanced colorectal cancer. J Clin
Oncol. 18:2938–2947. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rejhová A, Opattová A, Čumová A, Slíva D
and Vodička P: Natural compounds and combination therapy in
colorectal cancer treatment. Eur J Med Chem. 144:582–594. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lichota A and Gwozdzinski K: Anticancer
activity of natural compounds from plant and marine environment.
Int J Mol Sci. 19(pii): E35332018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hu C, Zhang Y and Kitts DD: Evaluation of
antioxidant and prooxidant activities of bamboo Phyllostachys nigra
var. Henonis leaf extract in vitro. J Agric Food Chem.
48:3170–3176. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kweon MH, Hwang HJ and Sung HC:
Identification and antioxidant activity of novel chlorogenic acid
derivatives from bamboo (Phyllostachys edulis). J Agric Food Chem.
49:4646–4655. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ra JH, Nakamura M, Herath KHINM, Jee Y and
Kim JS: Antioxidant and hepatoprotective effects of different
ethanol concentrations in extraction from leaves of Sasa
quelpaertensis Nakai. South African J Botany. 112:376–382. 2017.
View Article : Google Scholar
|
|
10
|
Seki T and Maeda H: Cancer preventive
effect of Kumaizasa bamboo leaf extracts administered prior to
carcinogenesis or cancer inoculation. Anticancer Res. 30:111–118.
2010.PubMed/NCBI
|
|
11
|
Sakai S, Saito G, Sugayama J, Kamasuka T,
Takano T and Takada S: Anticancer effect of polysaccharide fraction
prepared from bamboo grass. Gan. 55:197–203. 1964.PubMed/NCBI
|
|
12
|
Yoon SA, Kang SI, Shin SH, Ko HC and Kim
SJ: Anti-diabetic potential of a Sasa quelpaertensis Nakai extract
in L6 skeletal muscle cells. Food Sci Biotechnol. 23:1335–1339.
2014. View Article : Google Scholar
|
|
13
|
An SM, Lee SI, Choi SW, Moon SW and Boo
YC: p-Coumaric acid, a constituent of Sasa quelpaertensis Nakai,
inhibits cellular melanogenesis stimulated by alpha-melanocyte
stimulating hormone. Br J Dermatol. 159:292–299. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Madushani Herath KHIN, Bing SJ, Cho J, Kim
A, Kim G, Kim JS, Kim JB, Doh YH and Jee Y: Sasa quelpaertensis
leaves ameliorate alcohol-induced liver injury by attenuating
oxidative stress in HepG2 cells and mice. Acta Histochem.
120:477–489. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ryou SH, Kang MS, Kim KI, Kang YH and Kang
JS: Effects of green tea or Sasa quelpaertensis bamboo leaves on
plasma and liver lipids, erythrocyte Na efflux, and platelet
aggregation in ovariectomized rats. Nutr Res Pract. 6:106–112.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jang MG, Park SY, Lee SR, Choi SY, Hwang
JH, Ko HC, Park JG, Chung WS and Kim SJ: Sasa quelpaertensis leaf
extracts induce apoptosis in human leukemia HL-60 cells. Food Sci
Biotechnol. 17:188–190. 2008.
|
|
17
|
Jang MG, Ko HC and Kim SJ: Effect of Sasa
quelpaertensis Nakai extracts and its constituent p-coumaric acid
on the apoptosis of human cancer cell lines. J Food Biochem.
24:293–297. 2018.
|
|
18
|
Byun JH and Kim MY: Apoptotic effect of
Sasa quelpaertensis Nakai in human colon cancer HT-29 cells. J Life
Sci. 24:1012–1018. 2014. View Article : Google Scholar
|
|
19
|
Kim JH and Kim MY: Antiproliferative and
apoptotic effects of Sasa quelpaertensis Nakai in human cancer
cells. J Life Sci. 24:903–909. 2014. View Article : Google Scholar
|
|
20
|
Redza-Dutordoir M and Averill-Bates DA:
Activation of apoptosis signalling pathways by reactive oxygen
species. Biochim Biophys Acta. 1863:2977–2992. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tang D, Kang R, Berghe TV, Vandenabeele P
and Kroemer G: The molecular machinery of regulated cell death.
Cell Res. 29:347–364. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Moon SH, Cho MH and Kim MY: Cellular
inactivation of nitric oxide induces p53-dependent apoptosis in
human melanoma cells. Trop J Pharm Res. 15:1595–1603. 2016.
View Article : Google Scholar
|
|
23
|
Kim JY, Kim JH, Byun JH, Lee YJ, Im SJ,
Lee D, Moon SH and Kim MY: Antioxidant and anticancer activities of
water and ethanol extracts obtained from Sasa quelpaertensis Nakai.
Life Sci J. 10:1250–1254. 2013.
|
|
24
|
Kim M, Kim YS, Kim KM, Ko HC, Kim SJ, Kim
JH and Kim Y: Combination of Sasa quelpaertensis Nakai leaf extract
and cisplatin suppresses the cancer stemness and invasion of human
lung cancer cells. Integr Cancer Ther. 13:529–540. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xu W, Liu LZ, Loizidou M, Ahmed M and
Charles IG: The role of nitric oxide in cancer. Cell Res.
12:311–320. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wink DA, Ridnour LA, Hussain SP and Harris
CC: The reemergence of nitric oxide and cancer. Nitric Oxide.
19:65–67. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rao CV: Nitric oxide signaling in colon
cancer chemoprevention. Mutat Res. 555:107–119. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li CQ and Wogan GN: Nitric oxide as a
modulator of apoptosis. Cancer Lett. 226:1–15. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Karin M and Lin A: NF-kappaB at the
crossroads of life and death. Nat Immunol. 3:221–227. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Salvesen GS and Duckett CS: IAP proteins:
Blocking the road to death's door. Nat Rev Mol Cell Biol.
3:401–410. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fiandalo MV and Kyprianou N: Caspase
control: Protagonists of cancer cell apoptosis. Exp Oncol.
34:165–175. 2012.PubMed/NCBI
|
|
32
|
Jin SJ, Yang Y, Ma L, Ma BH, Ren LP, Guo
LC, Wang WB, Zhang YX, Zhao ZJ and Cui M: In vivo and in
vitro induction of the apoptotic effects of oxysophoridine on
colorectal cancer cells via the Bcl-2/Bax/caspase-3 signaling
pathway. Oncol Lett. 14:8000–8006. 2017.PubMed/NCBI
|
|
33
|
Zhao Y, Jing Z, Lv J, Zhang Z, Lin J, Cao
X, Zhao Z, Liu P and Mao W: Berberine activates
caspase-9/cytochrome c-mediated apoptosis to suppress
triple-negative breast cancer cells in vitro and in vivo. Biomed
Pharmacother. 95:18–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Koh DW, Dawson TM and Dawson VL: Mediation
of cell death by poly(ADP-ribose) polymerase-1. Pharmacol Res.
52:5–14. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ozaki T and Nakagawara A: p53: The
attractive tumor suppressor in the cancer research field. J Biomed
Biotechnol. 2011:6039252011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Levine AJ, Momand J and Finlay CA: The p53
tumour suppressor gene. Nature. 351:453–456. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zilfou JT and Lowe SW: Tumor suppressive
functions of p53. Cold Spring Harb Perspect Biol. 1:a0018832009.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen L, Jiang J, Cheng C, Yang A, He Q, Li
D and Wang Z: P53 dependent and independent apoptosis induced by
lidamycin in human colorectal cancer cells. Cancer Biol Ther.
6:965–973. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mahyar-Roemer M and Roemer K: p21
Waf1/Cip1 can protect human colon carcinoma cells against
p53-dependent and p53-independent apoptosis induced by natural
chemopreventive and therapeutic agents. Oncogene. 20:3387–3398.
2001. View Article : Google Scholar : PubMed/NCBI
|