Introduction
Pancreatic cancer (PC) is an aggressive malignant
tumor with a low survival rate; the 5-year survival rate of this
malignancy is <8%, making it the third major cause of
cancer-associated mortality in the USA (1). It has been predicted that PC will
become the second leading cause of cancer-associated mortality
after lung carcinoma by 2030 (2).
Such high mortality is caused by delayed diagnosis due to a lack of
early diagnostic tools. Indeed, the early diagnosis of PC serves a
vital role in disease prognosis and the design of personalized
therapy. Due to the lack of overt symptoms at the early stage of
PC, missed diagnosis rates range between 39 and 70% (3). Patients with advanced stages of PC
demonstrate a more severe physical condition compared with those
diagnosed in the early stage (4). In
addition, certain tumor biomarkers play important roles in the
early diagnosis, therapy and monitoring of PC, such as carbohydrate
antigen (CA)19-9, CA242, vascular endothelial growth factor (VEGF),
CA724 and CA125. However, although the levels of the aforementioned
tumor biomarkers are elevated in certain individuals with PC, this
is not the case for all, and so they have low levels of sensitivity
and specificity (5). More
strikingly, CA19-9 is a biomarker approved by the US Food and Drug
Administration for PC diagnosis, with a sensitivity of 70–81%;
however, it fails to detect PC in the early stages of disease
(6). Therefore, new tumor biomarkers
that serve significant roles in the early diagnosis of PC are
urgently needed.
Currently, proteomics is considered a powerful tool
for accurate monitoring and quantification of protein expression
changes (7). Indeed, several
proteomic technologies have been widely used for the identification
of biomarkers, including two-dimensional electrophoresis, stable
isotope labeling with amino acid in cell culture, two-dimension
difference gel electrophoresis and isobaric tags for relative and
absolute quantification (iTRAQ) (8).
Previously, iTRAQ-based analysis has been used to quantitatively
assess the changes in protein abundance in various biological
samples, with high sensitivity and reproducibility (9). For instance, this approach has been
successfully used to identify diagnostic markers of gastric and
lung cancer (8,9).
The iTRAQ technology is increasingly used in the
field of cancer research; plasma or serum protein analysis via
quantitative proteomics of patients with PC reveals
cancer-associated proteins and polypeptides in comparison with
specimens from non-diseased and chronic pancreatitis controls
(10). Thus, the present study aims
to assess serum proteins in patients with PC and healthy controls,
providing a basis for screening serum biomarkers, which would be
used in the early diagnosis of PC.
Materials and methods
Subjects
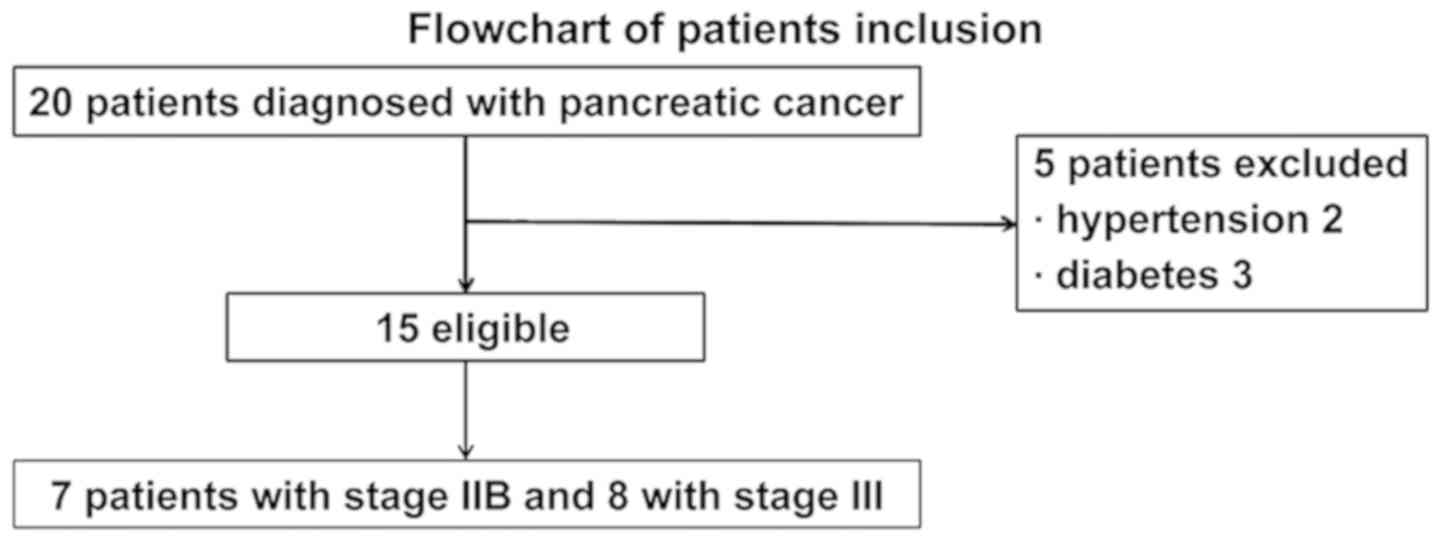
A total of 15 freshly collected serum specimens were
obtained from patients (mean age, 51.70±3.10 years; all male)
diagnosed with PC (without hypertension or diabetes) between
January 2013 and January 2014 at Shanxi Provincial Cancer Hospital
(Taiyuan, China). The samples were obtained prior to treatment
(Fig. 1). The diagnosis criteria
were based on the Guidelines for Diagnosis and Treatment of
Pancreatic Cancer (2014 Edition) from Group of Pancreatic Surgery,
Branch of Surgery, Chinese Medical Association (11). The American Joint Committee on Cancer
(AJCC) Tumor-Node-Metastasis (TNM) staging is the most widely used
cancer staging system worldwide. According to the AJCC 8th edition
staging system for patients with PC, 7 patients had been diagnosed
as stage IIB, and 8 patients had been diagnosed as stage III
(12) (Table I). The tumor status of 15 patients
with PC are shown in Table II.
During the same period, 10 healthy control subjects undergoing
physical examinations at Shanxi Provincial Cancer Hospital (mean
age, 56.40±2.42 years; all male) were enrolled. The present study
was approved by The Ethics Committee of Shanxi Provincial Cancer
Hospital (approval no. 201732), and written informed consent was
obtained from each participant.
 | Table I.Number of patients with pancreatic
cancer at each stage of the 8th edition of the American Joint
Committee on Cancer staging system (12) for pancreatic cancer. |
Table I.
Number of patients with pancreatic
cancer at each stage of the 8th edition of the American Joint
Committee on Cancer staging system (12) for pancreatic cancer.
| Variables | Number of
patients |
|---|
| Tumor depth |
|
| T1 | 4 |
| T2 | 7 |
| T3 | 4 |
| Lymph node
metastasis |
|
| N1 | 7 |
| N2 | 8 |
| Distant
metastasis |
|
| M0 | 15 |
| Tumor stage |
|
| IIB | 7 |
| III | 8 |
 | Table II.Tumor status of 15 patients with
pancreatic cancer included in the present study. |
Table II.
Tumor status of 15 patients with
pancreatic cancer included in the present study.
| Patient no. | Sex | Tumor size, cm | Tumor site | Degree of
differentiation | Lymph node Metastasis
numbers | T | N | M | Stages |
|---|
| 1 | Male | 3×2.5×2.5 | Head of pancreas | Moderately | 3 | 2 | 1 | 0 | IIB |
| 2 | Male | 1.8×1.8 | Tail of pancreas | Moderately | 1 | 1 | 1 | 0 | IIB |
| 3 | Male | 3×2×0.5 | Tail of pancreas |
Moderately/poorly | 2 | 2 | 1 | 0 | IIB |
| 4 | Male | 1.8×1.8 | Head of
pancreas | Moderately | 2 | 1 | 1 | 0 | IIB |
| 5 | Male | 2.5×2.5×2 | Head of
pancreas | Moderately | 3 | 2 | 1 | 0 | IIB |
| 6 | Male | 1.5×1.5 | Head of
pancreas | Moderately | 1 | 1 | 1 | 0 | IIB |
| 7 | Male | 2×2×1.5 | Body of
pancreas | Moderately | 2 | 1 | 1 | 0 | IIB |
| 8 | Male | 3×3×3 | Body of
pancreas |
Highly/moderately | 5 | 2 | 2 | 0 | III |
| 9 | Male | 3×3×3 | Head of
pancreas | Moderately | 4 | 2 | 2 | 0 | III |
| 10 | Male | 3×2.5×2.5 | Head of
pancreas |
Moderately/poorly | 6 | 2 | 2 | 0 | III |
| 11 | Male | 5×3.7×3.5 | Head of
pancreas | Moderately | 8 | 3 | 2 | 0 | III |
| 12 | Male | 4×3×3 | Head of
pancreas | Moderately | 4 | 2 | 2 | 0 | III |
| 13 | Male | 4×3.5×3.5 | Body of
pancreas | Moderately | 5 | 2 | 2 | 0 | III |
| 14 | Male | 5×3×3 | Head of
pancreas | Poorly | 9 | 3 | 2 | 0 | III |
| 15 | Male | 5×4×4 | Head of
pancreas | Highly | 8 | 3 | 2 | 0 | III |
Serum collection and preservation
Blood samples were collected in the morning before
the patients had eaten, placed at 4°C for 1.5 h, and centrifuged at
3,000 × g for 10 min at 4°C. The resulting supernatant (serum) was
collected and stored at −80°C.
Depletion of highly and lowly abundant
proteins
The serum samples from 15 patients and 10 healthy
controls collected at baseline were immuno-affinity depleted of
highly abundant proteins using a Multiple Affinity Removal Column
Human 14 (4.6×50 mm; Agilent), according to the manufacturer's
protocol. Lowly abundant proteins were collected on a
high-performance liquid chromatography (HPLC) system (1260 Infinity
II LC System; Agilent) using dilution, stripping and neutralization
buffers provided, according to the manufacturer's protocol. The
detailed conditions were: Column, Agilent HC-C18 (4.6×150 mm, 5
µm); mobile phase, methanol/water 20:80 (v/v) containing 0.05 M
potassium dihydrogen phosphate (pH 3.5); detection wavelength, 238
nm; flow rate, 1.0 ml/min; column temperature, 35°C; injection
volume, 20 µl. To 200 µl plasma, 600 µl 10% perchloric acid was
added; after mixing, the sample was centrifuged at 16,000 × g for
10 min on a GL-20G-II high-speed desktop refrigerated centrifuge
(Flying Pigeon; Shanghai Anting Scientific Instrument Factory,
Shanghai). Following this, 20 µl of the resulting supernatant was
used for quantitative analysis.
Protein quantification and SDS-PAGE
electrophoresis
The serum original samples of patients with
pancreatic cancer and healthy controls were analyzed by SDS-PAGE.
Serum protein levels were determined using a Bradford assay kit
(Beyotime Institute of Biotechnology). The depletion efficiency of
each approach was evaluated by SDS-PAGE. A Coomassie Blue Stain kit
PH0351 (Feijing Scientific Research Reagent Store) was used
containing 100 ml Coomassie Blue stain and 500 ml Coomassie Blue
decoloring solution. Coomassie blue staining was performed at 23°C
for 2–4 h followed by decolorizing at 23°C for 4–8 h. The type of
gel was SDS-PAGE gels. No two-dimensional separation was used.
Then, equal amounts (15 µg) of total protein were separated by 12%
SDS-PAGE electrophoresis.
Peptide extraction and iTRAQ
labeling
In total, 200 µg of highly abundant protein depleted
serum was denatured with 200 µl dithiothreitol (20 mmol/l) at 37°C
for 1 h, and alkylated with iodoacetamide (50 mmol/l) at room
temperature for 45 min. Urea (8 mol/l) was used for elution, and
ammonium bicarbonate (25 mmol/l) for washing. Finally, trypsin was
used overnight to digest the serum proteins at 37°C, and the
reactions were terminated by addition of formic acid at a final
concentration of 1%. Next, iTRAQ labeling was performed for
peptides from healthy controls and PC samples using an iTRAQ
reagent 8-plex kit (Applied Biosystems; Thermo Fisher Scientific,
Inc.), according to the manufacturer's protocol. Peptides were
desalted, dried and labeled with different isobaric tags for 2 h at
25°C. Peptides enriched in serum samples from healthy controls and
patients with PC were labeled with 116 and 117 tags, respectively.
Finally, the labeled peptides were dried under vacuum and stored at
−80°C.
Separation of iTRAQ-labeled peptides
under high pH reversed phase (RP) conditions
Digestion and iTRAQ labeled serum samples were
reconstituted in 400 µl of 20 mm ammonium formate/2% acetonitrile
(pH 10). Following this, the samples were loaded on an RP C18
capillary column (3, 4.6 µmx250 mm; Shimadzu Corporation) and
fractionated on an Agilent 1100 series HPLC instrument by basic RP
chromatography at a flow rate of 1.0 ml/min. The mobile phase
consisted of 90% acetonitrile (pH 10), used with a gradient of 5 to
30% solvent B (formic acid), for 0–60 min. Fractions were collected
every 1 min for a total of 60 fractions; early and late fractions
were pooled, resulting in a total of 30 fractions. The pooled
samples were reconstituted in 0.1% formic acid for liquid
chromatography-tandem mass spectrometry (LC-MS/MS) analysis.
LC-MS/MS analysis
Peptides in each fraction were separated on an RP
C18 capillary column (3, 75 µmx100 mm) and eluted using a linear
gradient of 5 to 30% solvent B1 (0.1% v/v) formic acid in
acetonitrile for 40 min, at a flow rate of 300 nl/min). MS data
were acquired by the shotgun proteomics method (LTQ-Orbitrap mass
spectrometer; Sanofi S.A.); in each cycle, a full scan was acquired
at a resolution of 30,000 dpl with a mass range of 380–1,600 m/z.
Up to 10 of the most intense precursor ions with charge range of +2
to +4 were selected with an isolation window of 2 Da, and
subsequently fragmented by higher-energy collisional dissociation
with a normalized collision energy of 40%. The MS/MS scans were
acquired at a resolution of 7,500 dpl. Precursor ions were
dynamically excluded from reselection for 60 sec.
Database analysis
Raw data from the mass spectrometer were analyzed by
the Mascot (version 2.3.02; http://www.matrixscience.com/) and Scaffold (version
4.4.3; Proteome Software, Inc.) programs. The search database was
Swiss-Prot (uniprot.org) (human species). Tyrosine
served as a variable modifier protein. The search parameters were:
Trypsin allowed P enzyme specificity, with up to two missing
cleavages; precursor ion mass tolerance, ±10 ppm; fragment ion mass
tolerance, 0.05 Da; fixed modification, carbamidomethylation and no
variable modification. The identified proteins were quantified
according to the iTRAQ of the specific polypeptide. Differential
proteins in serum proteome: Differential multiple >1.5,
P<0.05.
Gene ontology (GO) and
Igenuity® Pathway Analysis (IPA) analyses of
differentially expressed proteins
Differentially expressed proteins (ratio >1.5 or
<0.5; P<0.05) were imported into the Panther database
(pantherdb.org/) and matched with the human genome
data. Following this, the proteins were classified into molecular
function, biological process and cellular component according to
gene ontology (GO). Canonical pathway, upstream regulation and
protein-protein interaction analyses were performed by the IPA
software (Ingenuity Systems; Qiagen, Inc.).
Statistical analysis
Data are presented as mean ± standard deviation.
SPSS 19.0 software (SPSS; IBM Corp.) was used to analyze the
results. Student's t-test was applied to compare mean values
between the two groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
Characteristics of patients with
PC
As presented in Table
III, serum CA19-9, CA242, CA724, CA50 and VEGF levels in
patients with PC were significantly increased compared with the
values of healthy controls. In addition, these diagnostic factors
were higher than normal levels, except VEGF.
 | Table III.Clinical data and carbohydrate
antigen levels in healthy controls (male) and patients with
pancreatic cancer (male) (mean ± SD) |
Table III.
Clinical data and carbohydrate
antigen levels in healthy controls (male) and patients with
pancreatic cancer (male) (mean ± SD)
| Variable | Healthy control
group (n=10) | Patients with
pancreatic cancer group (n=15) | Reference
values | P-value |
|---|
| Age, years | 51.70±3.10 | 56.40±2.42 | − | − |
| Complications | None | None | – | – |
| CA19-9 | 5.05±2.01 U/ml | 251.3±63.87
U/ml | <20 U/ml | P<0.01 |
| CA242 | 2.12±0.71 U/ml | 28.76±13.82
U/ml | <12 U/ml | P<0.01 |
| CA724 | 4.31±1.29 U/ml | 12.76±7.62
U/ml | <10 U/ml | P<0.05 |
| CA50 | 5.48±1.46 U/ml | 176.0±60.71
U/ml | <20 U/ml | P<0.05 |
| VEGF | 185.0±60.36
pg/ml | 365.0±39.67
pg/ml | 62–707 pg/ml | P<0.05 |
| Total, n | 10 | 15 | – | – |
| Age, years | 51.70±3.10 | 56.40±2.42 | – | – |
| Complications | None | None | – | – |
Qualitative and quantitative analyses
of serum proteins following immuno-affinity depletion of highly
abundant proteins
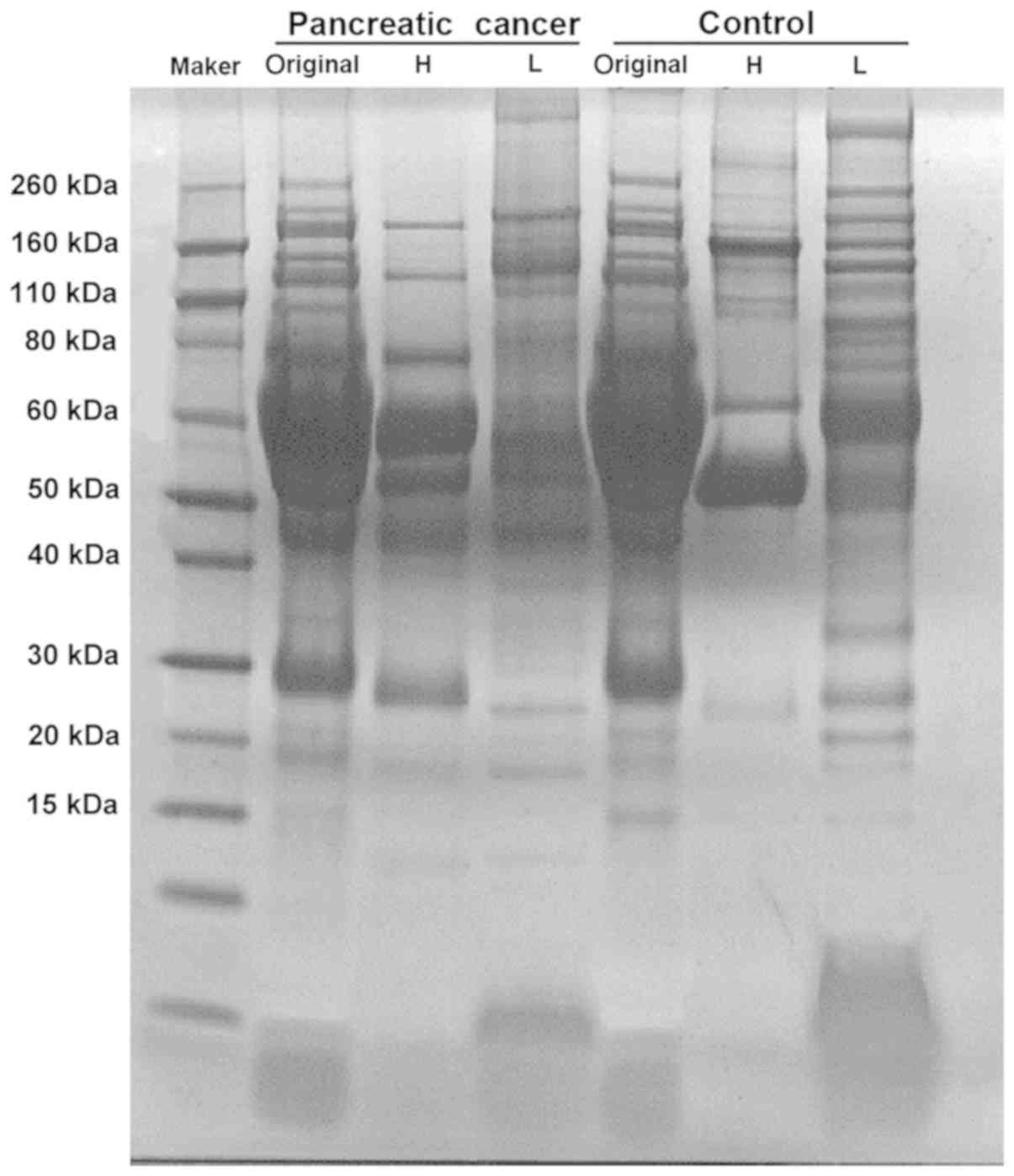
Serum samples from patients with PC and control
patients were immuno-affinity depleted of the 14 most abundant
proteins using Multiple Affinity Removal Column Human 14, and
SDS-PAGE was performed to assess various serum samples in the two
patient groups. As presented in Fig.
2, low abundance proteins demonstrated improved separation and
exhibited higher concentrations. In addition, an increased number
of low abundance proteins was observed; however, the albumin band
disappeared. In the serum proteome, 251,479 spectra were obtained.
Proteomics revealed 531 proteins, of which 442 were
quantifiable.
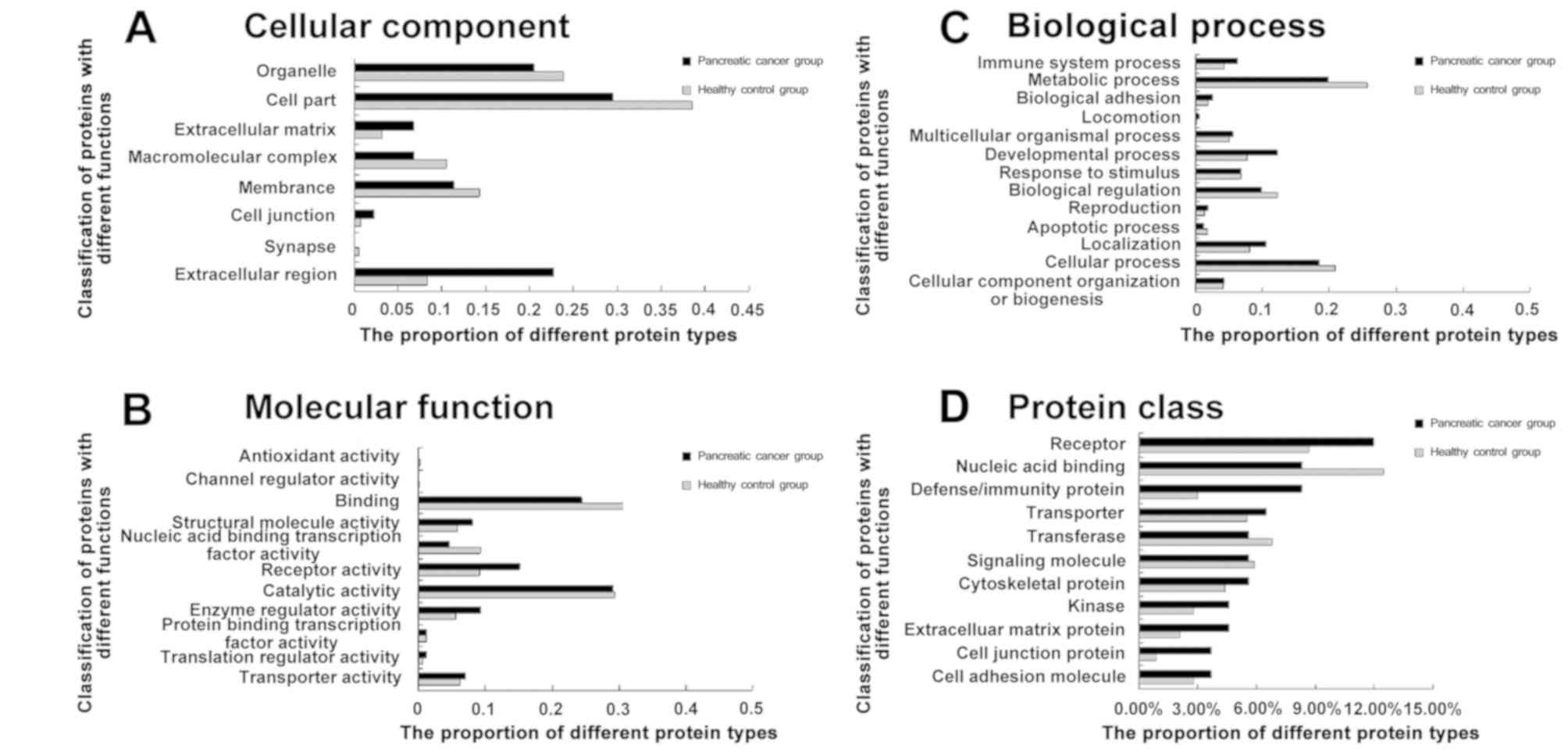
GO analysis
Based on the aforementioned 442 quantifiable
proteins, more than two peptide segments were identified by Mascot
and Scaffold. There were 76 differentially expressed proteins in
samples from patients with PC compared with control specimens
(Table IV). Among them, 70 proteins
were downregulated, with 24 demonstrating a coordination ratio of
0.4 or more. Meanwhile, six proteins were upregulated by 2–3 fold.
To comprehensively assess the biological significance of the 76
differentially expressed proteins, analysis was performed by the
Panther gene classification system, which includes biology
processes, cellular components, molecular functions and protein
classes. Compared with the normal serum proteome database
(pantherdb.org/chart/summary/pantherChart.jsp?filterLevel=1&chartType=1&listType=1&type=5&species=Homo%20sapiens),
proteins associated with the extracellular matrix, receptor
activity and enzyme regulator activity were upregulated in patients
with PC, and those involved in the extracellular matrix accounted
for 6.8% of all proteins (Fig. 3).
However, proteins involved in nucleic acid binding transcription
factor activity and metabolic processes were significantly
downregulated.
 | Table IV.Analysis of the 76 differentially
expressed proteins identified in the serum of patients with
pancreatic cancer and healthy controls. |
Table IV.
Analysis of the 76 differentially
expressed proteins identified in the serum of patients with
pancreatic cancer and healthy controls.
| A, Upregulated
proteins |
|---|
|
|---|
| Protein | ID
(Swiss-Prot) | Change fold |
|---|
| Cluster of
transmembrane protease serine 13 | Q9BYE2 | 3 |
| Triose-phosphate
isomerase | P60174 | 2.5555556 |
| Complement factor
H-related protein 2 | P36980 | 2.5 |
| Lysozyme C | P61626 | 2.2222222 |
| DNA repair protein
RAD50 | Q92878 | 2.2 |
| WD
repeat-containing protein 67 | Q96DN5 | 2 |
|
| B, Downregulated
proteins |
|
| Protein | ID
(Swiss-Prot) | Change
fold |
|
| Cadherin-related
family member 2 | Q9BYE9 | 0.4545455 |
| Apolipoprotein
C-I | P02654 | 0.4545455 |
| Cluster of membrane
primary amine oxidase | Q16853 | 0.4545455 |
| A-kinase anchor
protein 13 | Q12802 | 0.4444444 |
| Cluster of Ig κ
chain V–III region WOL | P01623 | 0.4166667 |
| Titin | Q8WZ42 | 0.4 |
| Ig κ chain V–I
region EU | P01598 | 0.4 |
| Polymeric
immunoglobulin receptor | P01833 | 0.4 |
| Mast/stem cell
growth factor receptor kit | P10721 | 0.4 |
| Collagen α-1(I)
chain | P02452 | 0.4 |
| Roundabout homolog
4 | Q8WZ75 | 0.4 |
| G-protein coupled
receptor 126 | Q86SQ4 | 0.4 |
| Protein RRP5
homolog | Q14690 | 0.4 |
| Plasminogen
activator inhibitor 1 | P05121 | 0.4 |
| 4F2 cell-surface
antigen heavy chain | P08195 | 0.4 |
| Plexin-B1 | O43157 | 0.4 |
| Sushi, nidogen and
EGF-like domain-containing protein 1 | Q8TER0 | 0.4 |
| Multiple epidermal
growth factor-like domains protein 8 | Q7Z7M0 | 0.4 |
| Androgen
receptor | P10275 | 0.4 |
| TRAF3-interacting
protein 1 | Q8TDR0 | 0.4 |
| NACHT, LRR and PYD
domains-containing protein 6 | P59044 | 0.4 |
| Ankyrin repeat
domain-containing protein 30A | Q9BXX3 | 0.4 |
| Collagen α-1(XVI)
chain | Q07092 | 0.4 |
| Protein QN1
homolog | Q5TB80 | 0.4 |
| α-amylase 1 | P04745 | 0.375 |
| Ig heavy chain
V–III region GAL | P01781 | 0.3636364 |
| A disintegrin and
metalloproteinase with thrombospondin motifs 13 | Q76LX8 | 0.3636364 |
| Proteasome subunit
β type-4 | P28070 | 0.3636364 |
| Endoglin | P17813 | 0.3636364 |
| Leukocyte elastase
inhibitor | P30740 | 0.3636364 |
| Sodium channel
protein type 8 subunit α | Q9UQD0 | 0.3636364 |
| Desmoglein-2 | Q14126 | 0.3333333 |
| Msx2-interacting
protein | Q96T58 | 0.3 |
| Unconventional
myosin-XV | Q9UKN7 | 0.3 |
| Serum amyloid A-4
protein | P35542 | 0.3 |
| Apolipoprotein
C-IV | P55056 | 0.3 |
| Golgi-associated
plant pathogenesis-related protein 1 | Q9H4G4 | 0.3 |
| Vascular
endothelial growth factor receptor 3 | P35916 | 0.3 |
| Kinesin-like
protein KIF20B | Q96Q89 | 0.3 |
| Centromere protein
F | P49454 | 0.3 |
| Ig heavy chain
V–III region JON | P01780 | 0.3 |
| Leukemia inhibitory
factor receptor | P42702 | 0.3 |
| Cingulin-like
protein 1 | Q0VF96 | 0.3 |
| G protein-coupled
receptor kinase 5 | P34947 | 0.3 |
| Plasma serine
protease inhibitor | P05154 | 0.2727273 |
| Plexin-D1 | Q9Y4D7 | 0.2727273 |
|
Lactotransferrin | P02788 | 0.25 |
| Pericentrin | O95613 | 0.25 |
| Dystonin | Q03001 | 0.25 |
| Serine-protein
kinase ATM | Q13315 | 0.2307692 |
| Pyruvate
dehydrogenase (lipoamide) kinase isozyme 2, mitochondrial | Q15119 | 0.2307692 |
| Serine/arginine
repetitive matrix protein 1 | Q8IYB3 | 0.2222222 |
| Apolipoprotein
C-III | P02656 | 0.2 |
| Hornerin | Q86YZ3 | 0.2 |
| Protein
shroom2 | Q13796 | 0.2 |
| Transmembrane
protein 74 | Q96NL1 | 0.2 |
|
Serine/threonine-protein kinase WNK4 | Q96J92 | 0.2 |
| RING finger protein
214 | Q8ND24 | 0.1818182 |
| Midasin | Q9NU22 | 0.1666667 |
| Dynein heavy chain
11, axonemal | Q96DT5 | 0.1666667 |
|
Regulatory-associated protein of mTOR | Q8N122 | 0.1538462 |
| Zinc finger ZZ-type
and EF-hand domain-containing protein 1 | O43149 | 0.1538462 |
| Poly (ADP-ribose)
polymerase 1 | P09874 | 0.125 |
| Probable
ATP-dependent RNA helicase DDX46 | Q7L014 | 0.125 |
| Peregrin | P55201 | 0.125 |
| Semaphorin-4B | Q9NPR2 | 0.117647 |
| Nesprin-1 | Q8NF91 | 0.1 |
| Canalicular
multispecific organic anion transporter 1 | Q92887 | 0.1 |
| Apoptotic
protease-activating factor 1 | O14727 | 0.1 |
| Sodium/hydrogen
exchanger 2 | Q9UBY0 | 0.1 |
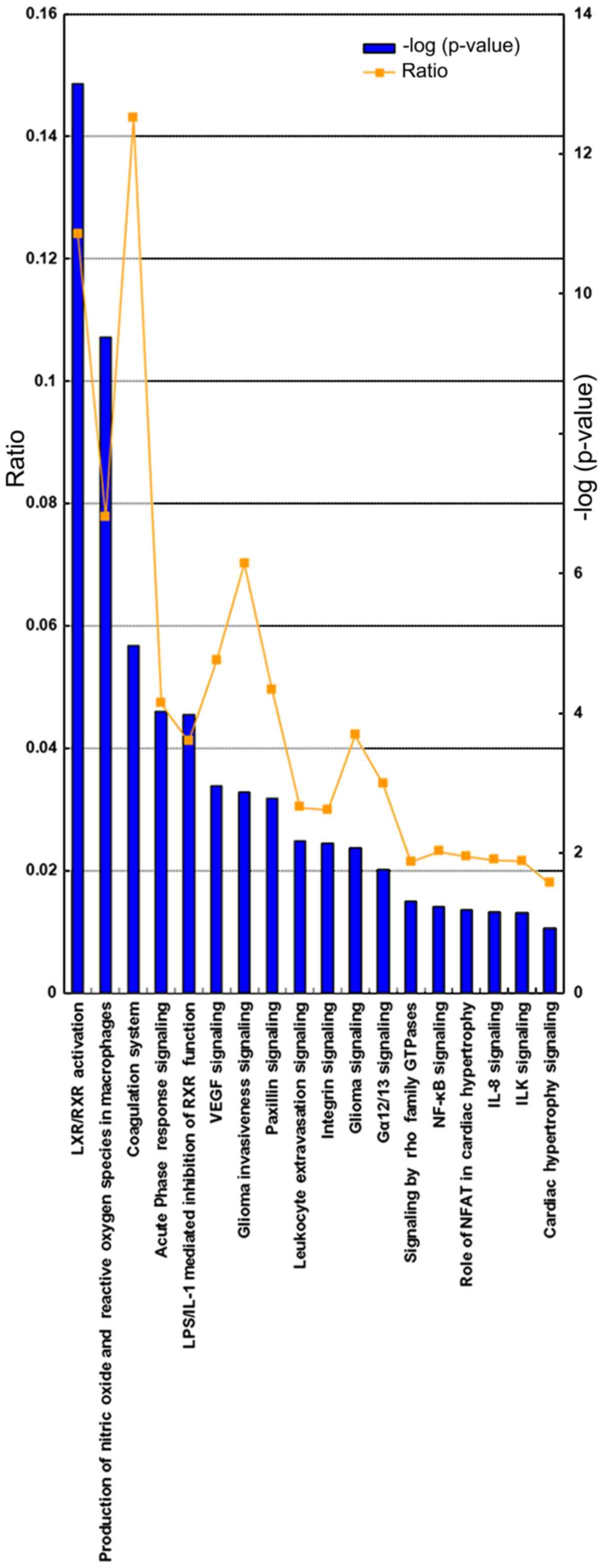
Pathways identified by IPA
A bioinformatics analysis of the identified
differentially expressed proteins was performed using the IPA
software. As presented in Fig. 4,
certain pathways were significantly altered. Proteins in the
coagulation system (P=4.97; ratio, 1.43×10−1) and
lipopolysaccharide/interleukin-1 mediated inhibition of retinoid X
receptor (RXR) function (P=3.98; ratio, 4.11×10−2) were
significantly upregulated. In comparison, liver X receptor/RXR
activation (P=13.00; ratio, 1.24×10−1), production of
nitric oxide and reactive oxygen species in macrophages (P=9.38;
ratio, 7.78×10−2), paxillin signaling (P=2.78; ratio,
4.95×10−2), and integrin signaling (P=2.14; ratio,
2.99×10−2) were significantly downregulated. In
addition, some differentially expressed proteins were involved in
cell survival, molecular translocation, viral infection and lipid
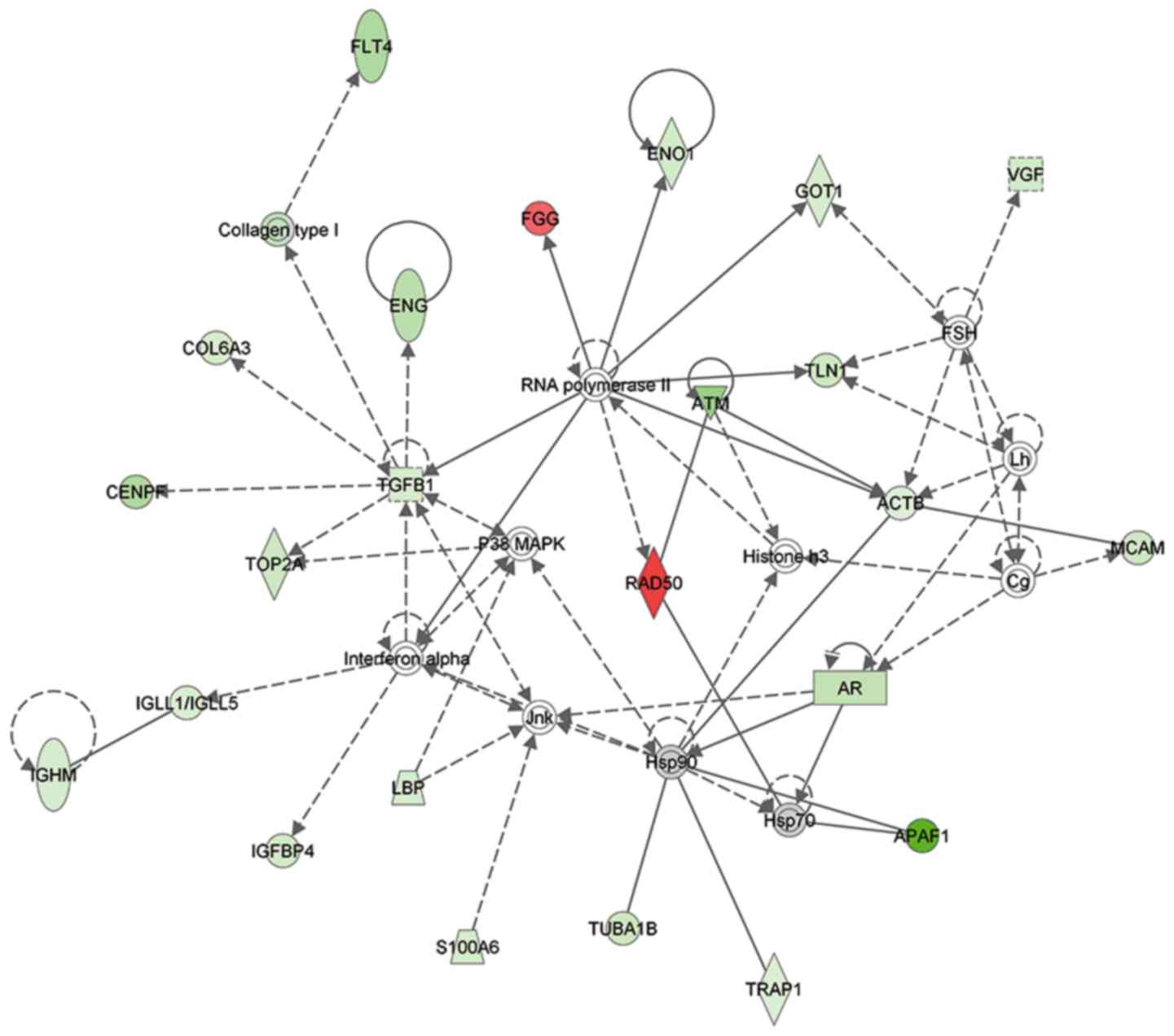
efflux, revealing decreased levels. A protein interaction network
analysis also revealed that certain associated proteins were
upregulated or downregulated (Fig.
5).
Discussion
CA199 is a tumor-associated antigen (13). It is present in the majority of
healthy individuals. Not all individuals are able to produce
similar levels of CA19-9; the levels are closely associated with
physiological characteristics and genetic status (13). The physiological level of CA19-9
varies from individual to individual due to their different
genotypes. In the serum of those with Lewis (Le) antigen Le-a (+)
and Le-b (−) genotypes, CA19-9 is maintained at a certain level
(14). The serum CA19-9 level of
Le-a (−), Le-b (+) genotype was relatively low. CA19-9 could not be
detected in the serum of individuals with Le-a (−), Le-b (−)
genotype. In individuals who are negative for Lewis antigen, CA19-9
was not significantly elevated despite the PC tumor being large.
Despite CA19-9 being a PC-specific antigen, its specificity and
sensitivity to the PC tumor are low, which makes it difficult to
use CA19-9 as a biomarker in screening for the early diagnosis of
PC, although it is a common method (14). CA19-9 has been detected in various
other types of cancer, including pancreatic (84%), gallbladder
(69%), colorectal (39%) and ovarian (35%) cancer (15). Pancreatitis, hepatitis, biliary
inflammation and obstructive diseases have been reported as the
most common benign diseases exhibiting elevated levels of CA19-9
(15). The diagnostic sensitivity of
CA19-9 for PC decreased when the critical value of CA19-9 increased
and when the specificity level increased. When the critical value
of CA19-9 reached 100 ku/l, the sensitivity and specificity were 68
and 98%, respectively (14). Even
though CA19-9 is considered a good diagnostic marker for PC, it
should not be used for screening of the early stages of PC, as the
levels of CA19-9 are rarely elevated in this stage (14). Even when the PC lesions have been
detected using CT, but the tumor size was <3 cm, only 50% of the
patients had elevated CA19-9 (14).
The most important role that CA19-9 serves in PC is to evaluate the
recurrence of PC and the efficacy of radiotherapy and chemotherapy
following surgery via assessing the changes in the levels of
CA19-9. Despite the level of CA19-9 in patients with PC being
increased >10 times in the present study, it could not be used
as a direct indicator for the diagnosis of PC.
The present study assessed serum proteins in
patients with PC and healthy controls and identified a total of 531
proteins using iTRAQ technology. Among them, 442 were
quantitatively analyzed. Several differentially expressed proteins
were observed in subjects with PC in comparison with healthy
controls, including APAF-1, RAD50 and TGF-β1. GO and IPA analyses
demonstrated that the differentially expressed proteins mainly
participated in the extracellular matrix component, receptor
function, enzyme regulation, nucleic acid binding transcription
factor activity and metabolic-associated biological process. In
addition, protein-protein interaction analysis using the IPA
software also revealed marked alterations of APAF-1, RAD50 and
TGF-β1 in patients with PC.
As presented in Fig.
5 and Table IV, APAF-1 was
significantly downregulated by 10-fold in serum samples from
patients with PC compared with the normal group. It has previously
been demonstrated that APAF-1 is an essential and necessary
component of apoptotic bodies in the intrinsic apoptotic pathway
(16). APAF-1 serves an important
role in the mitochondrial apoptotic pathway, primarily affecting
the normal apoptosis of cells (17).
Loss of heterozygosity and DNA methylation can inactivate APAF-1,
which is a common phenomenon in numerous types of cancer in humans,
including PC (18,19). It has also been reported that when
APAF-1 is inhibited in the pancreatic duct, it can result in the
development of PC (17).
As an important component of the Mre11-Rad50-Nbs1
complex (20), RAD50 is primarily
involved in maintaining chromosome stability. In the present study,
RAD50 was increased by 2.2-fold in serum samples from patients with
PC compared with the normal group. A mutation analysis of 32
double-stranded DNA damage repair genes in breast cancer and PC
revealed that RAD50 inactivation was more likely to promote the
development of PC (21); however,
high expression of RAD50 is conserved (22). It is not yet understood whether RAD50
protein upregulation in patients with PC is a protected molecular
mechanism or whether the upregulation is maintained via negative
feedback regulation; further research is required in order for the
underlying mechanism to be elucidated.
TGF-β plays an important role in cell cycle arrest,
apoptosis, homeostasis, wound healing and immune regulation. In the
case of cancer, TGF-β plays a dual role in different situations,
functioning as a tumor suppressor or oncogene in the early disease
stages (23). TGF-β has three
isoforms, including TGF-β1, TGF-β2 and TGF-β3; among them, TGF-β1
is the most abundant in humans. The TGF-β signaling pathway
consists of several stages, starting with TGF-β1 activation and
release, followed by binding to three high affinity receptors
(24). TGF-β binds to TGF-βR1 and
TGF-βR2 receptors on the cell surface, forming dimeric-SMADs
following activation. A previous study demonstrated that TGF-βR2 is
one of the 16 most commonly mutated genes in PC (25). In multiple types of human cancer, the
TGF-β signaling pathway is activated (26). As presented in Table IV, TGF-β1 was 1.67-fold less
expressed in serum samples from patients with PC compared with
control values. Through IPA analysis of upstream regulatory genes
of TGF-β1, it was also revealed that significantly downregulated
TGF-β1 may be due to the suppression of hypoxia inducible factor 1
(HIF1) α. According to a previous report, HIF1α plays important
roles in the growth, invasion and metastasis of PC (27). Significant changes in TGF-β1
expression indicate that TGF-β1 may be one of the diagnostic
indices for PC.
The present study demonstrated APAF-1, RAD50 and
TGF-β1 should be considered candidate biomarkers for PC diagnosis.
However, due to the limited sample size, further studies with
larger sample sizes are required in order to confirm the identified
differentially expressed proteins and validate their values in the
diagnosis of PC. The results of the present study could aid in
identifying future therapeutic drugs for the treatment of PC.
Acknowledgements
Not applicable.
Funding
The present study was supported by The Scientific
and Technological Project in Shanxi (grant no. 100311099-3).
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article.
Authors' contributions
KJ and XD conceived and designed the study. KJ and
XZ performed the experiments and data analyses. KJ wrote the
manuscript. KJ and XD helped to revise the manuscript. XZ
contributed to discussions and provided reagents/materials/analysis
tools important for the completion of this work.
Ethics approval and consent to
participate
All procedures performed in the present study that
involved human participants were in accordance with the ethical
standards of the institutional and/or national Ethics Committee and
with the 1964 Helsinki declaration and its later amendments or
comparable ethical standards. The present study was approved by the
ethics committee of Shanxi Provincial Cancer Hospital (approval no.
201732). All the patients included in the present study provided
written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
PC
|
pancreatic cancer
|
|
iTRAQ
|
isobaric tags for relative and
absolute quantification
|
|
GO
|
gene ontology
|
References
|
1
|
American Cancer Society, . Cancer Facts
and Figs 2017. American Cancer Society. (New York, NY). 2017.
|
|
2
|
Rahib L, Smith BD, Aizenberg R, Rosenzweig
AB, Fleshman JM and Matrisian LM: Projecting cancer incidence and
deaths to 2030: The unexpected burden of thyroid, liver, and
pancreas cancers in the United States. Cancer Res. 74:2913–2921.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ross PL, Huang YN, Marchese JN, Williamson
B, Parker K, Hattan S, Khainovski N, Pillai S, Dey S, Daniels S, et
al: Multiplexed protein quantitation in Saccharomyces cerevisiae
using amine-reactive isobaric tagging reagents. Mol Cell
Proteomics. 3:1154–1169. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Blaney JM, Crawford G, Elder TR, Johnston
G and Gavin AT: Hospital cancer deaths: Late diagnosis and missed
opportunity. BMJ Support Palliat Care. 1:135–139. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goh SK, Gold G, Christophi C and
Muralidharan V: Serum carbohydrate antigen 19-9 in pancreatic
adenocarcinoma: A mini review for surgeons. ANZ J Surg. 87:987–992.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Su SB, Qin SY, Chen W, Luo W and Jiang HX:
Carbohydrate antigen 19-9 for differential diagnosis of pancreatic
carcinoma and chronic pancreatitis. World J Gastroenterol.
21:4323–4333. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen J, Ge L, Liu A, Yuan Y, Ye J, Zhong
J, Liu L and Chen X: Identification of pathways related to FAF1/H.
Pylori-associated gastric carcinogenesis through an integrated
approach based on iTRAQ quantification and literature review. J
Proteomics. 131:163–176. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang X, Li Y, Xu G, Liu M, Xue L, Liu L,
Hu S, Zhang Y, Nie Y, Liang S, et al: Mechanism study of peptide
GMBP1 and its receptor GRP78 in modulating gastric cancer MDR by
iTRAQ-based proteomic analysis. BMC Cancer. 15:3582015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Martin-Bernabe A, Cortes R, Lehmann SG,
Seve M, Cascante M and Bourgoin-Voillard S: Quantitative proteomic
approach to understand metabolic adaptation in non-small cell lung
cancer. J Proteome Res. 13:4695–4704. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pan S, Brentnall TA and Chen R: Proteomics
analysis of bodily fluids in pancreatic cancer. Proteomics.
15:2705–2715. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pancreatic Surgery Group, : Guidelines for
Diagnosis and Treatment of Pancreatic Cancer (2014 Edition). Chin J
Prac Surg. 34:1011–1017. 2014.
|
|
12
|
Amin MB, Edge S, Greene F, Brookland RK,
Washington MK, Gershenwald JE, Compton CC, Hess KR, Sullivan DC,
Jessup JM, Brierley JD, Gaspar LE, Schilsky RL, Balch CM,
Winchester DP, Asare EA, Madera M, Gress DM and Meyer LR: AJCC
Cancer Staging Manual (8th). Springer. New York, NY: 2016.
|
|
13
|
Kondo N, Murakami Y, Uemura K, Hayashidani
Y, Sudo T, Hashimoto Y, Nakashima A, Sakabe R, Shigemoto N, Kato Y,
et al: Prognostic impact of perioperative serum CA19-9 levels
inpatients with resectable pancreatic cancer. Ann Surg Oncol.
17:2321–2329. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Luo G, Liu C, Guo M, Long J, Liu Z, Xiao
Z, Jin K, Cheng H, Lu Y, Ni Q and Yu X: CA19-9-Low&Lewis (+)
pancreatic cancer: A unique subtype. Cancer Lett. 385:46–50. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Poruk KE, Gay DZ, Brown K, Mulvihill JD,
Boucher KM, Scaife CL, Firpo MA and Mulvihill SJ: The clinical
utility of CA19-9 in pancreatic adenocarcinoma: Diagnostic and
prognostic updates. Curr Mol Med. 13:340–351. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wu CC and Bratton SB: Regulation of the
intrinsic apoptosis pathway by reactive oxygen species. Antioxid
Redox Signal. 19:546–558. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu N, Sun YY, Zhang XW, Chen S, Wang Y,
Zhang ZX, Song SW, Qiu GB and Fu WN: Oncogenic miR-23a in
Pancreatic Ductal Adenocarcinogenesis Via Inhibiting APAF1. Dig Dis
Sci. 60:2000–2008. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Umetani N and Hoon DS: Frequent LOH at
chromosome 12q22-23 and Apaf-1 inactivation in glioblastoma. Brain
Pathol. 14:2242004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fu WN, Bertoni F, Kelsey SM, McElwaine SM,
Cotter FE, Newland AC and Jia L: Role of DNA methylation in the
suppression of Apaf-1 protein in human leukaemia. Oncogene.
22:451–455. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Uziel T, Lerenthal Y, Moyal L, Andegeko Y,
Mittelman L and Shiloh Y: Requirement of the MRN complex for ATM
activation by DNA damage. EMBO J. 22:5612–5621. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang X, Szabo C, Qian C, Amadio PG,
Thibodeau SN, Cerhan JR, Petersen GM, Liu W and Couch FJ:
Mutational analysis of thirty-two double-strand DNA break repair
genes in breast and pancreatic cancers. Cancer Res. 68:971–975.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dzikiewicz-Krawczyk A: The importance of
making ends meet: Mutations in genes and altered expression of
proteins of the MRN complex and cancer. Mutat Res. 659:262–273.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tian M and Schiemann WP: The TGF-beta
paradox in human cancer: An update. Future Oncol. 5:259–271. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bernabeu C, Lopez-Novoa JM and Quintanilla
M: The emerging role of TGF-beta superfamily coreceptors in cancer.
Biochim Biophys Acta. 1792:954–973. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Biankin AV, Waddell N, Kassahn KS, Gingras
MC, Muthuswamy LB, Johns AL, Miller DK, Wilson PJ, Patch AM, Wu J,
et al: Pancreatic cancer genomes reveal aberrations in axon
guidance pathway genes. Nature. 491:399–405. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wakefield LM and Hill CS: Beyond TGFbeta:
Roles of other TGFbeta superfamily members in cancer. Nat Rev
Cancer. 13:328–341. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Niu F, Li Y, Lai FF, Ni L, Ji M, Jin J,
Yang HZ, Wang C, Zhang DM and Chen XG: LB-1 exerts antitumor
activity in pancreatic cancer by inhibiting HIF-1α and Stat3
Signaling. J Cell Physiol. 230:2212–2223. 2015. View Article : Google Scholar : PubMed/NCBI
|