Introduction
The epithelial to mesenchymal transition (EMT)
induces loss of cell epithelial phenotype and has been initially
described in the process of embryonic development (1). The EMT process is also a hallmark of
several human cancers and as EMT progresses tumor cells become
motile and increase their aggressiveness (2–4). Oral
cancer and more specifically oral squamous cell carcinoma (OSCC) is
a very common malignancy. It is characterized by high propensity to
recurrence and metastasis and a relatively modest 5-year survival
rate. Approximately 40 to 50% of OSCC recurrence is believed to
occur in part due to EMT. Differentiated epithelial cancer cells
achieve a dedifferentiated mesenchymal appearance by EMT along with
the capacity to dissociate from each other and migrate (5).
SLUG and SNAIL are the main transcriptional
repressors of E-cadherin and are considered the principal mediators
of EMT (5–7). Upregulation of the mesenchymal
intermediate filament proteins α-SMA and Vimentin is also a typical
event in the course of EMT, leading to disturbed epithelial
integrity (8,9).
EMT occurs only in certain types of cancer cells
that are localized predominantly at the tumor front, while other
cancer cells retain their epithelial traits. Therefore, the
transient nature of EMT and the heterogeneity of the degree of
dedifferentiation pose a challenge for the establishment of
appropriate in vivo studies (10). Recently, a strong association between
EMT and cancer stem cells (CSCs) has also been revealed. It was
shown that CSCs represent a plastic state of tumor cells undergoing
EMT, induced either by cell-intrinsic and/or
microenvironment-associated signals (11). In addition, previous studies have
shown the presence of CSCs in the tumor margins (12). Therefore, the current study examined
whether EMT was present in that specific area. EMT has been studied
in the margin tissue of a limited number of cancers [breast
(13), colon (14), lung (15)] but not in the resection margins of
oral cancer, although it is well known that the histological and
molecular status of the margins are determinants of tumor behavior
(16,17).
The present study aimed to investigate OSCCs and
their resection margins in terms of mRNA expression of the EMT
markers Vimentin, α-SMA, SLUG and SNAIL in tumor and
margin primary cell cultures during specific cell passages.
Moreover, the present study investigated EMT-associated features,
including the clonal, proliferative and migratory potential of
tumor and margin cells.
Materials and methods
Patients and tissues
In order to investigate the incidence of EMT in
OSCC, tumor and margin tissues of 6 patients (2 females and 4
males, average age 59.5±9.33, 3 tongue and 3 floor of the mouth
tumors) diagnosed with OSCC were obtained at the Clinic of
Maxillofacial Surgery of the School of Dental Medicine at the
University of Belgrade. The samples were processed by
immunostaining. Deparaffinization of 5-µm tissue sections was
performed in xylene. The process was repeated two times for 5 min.
The sections were processed by hydration with graded ethanol (100,
96, 80, 70, 50%) 2× for 5 min. Pretreatment was performed in 0.1 ml
citrate buffer (pH 6.0) for 20 min at 98°C. The samples were
incubated in 3% H2O2 for 5 min and rinsed in
Tris-buffered saline solution. This was followed by application of
the UV blocker for 5 min. The samples were incubated with rabbit
polyclonal antibodies for Vimentin, α-SMA, SNAIL and SLUG (Thermo
Fisher Scientific) for 20 min. Following rinsing for 5 min, the
samples were analyzed with Quatro amplifier for 10 min, Quatro
polymer for 10 min, DAB quatro for 5 min and finally counterstained
with hematoxylin for 2 min. Between all these phases, the samples
were rinsed for 5 min. The images were captured by the Olympus DP70
camera and the Olympus BX50 microscope (Olympus).
Cell and tissue culture
OSCC tumor and margin tissues were obtained
immediately prior to the surgery. Tumor margins were obtained 5 mm
from the edges of the tumor. The SCC-25 cancer cell line
(ATCC® CRL-16 28™) was used as the negative control
sample. Fibroblasts isolated from gingiva of heathy donors were
used as the positive control sample. The present study was approved
by the Institutional Ethics Committee (no. 36/31) and conducted in
accordance with the Declaration of Helsinki. The patients were
informed of the study protocol and signed a written informed
consent form. The histopathological diagnosis of OSCC was
established in accordance with the World Health Organization (WHO)
guidelines and the tumor staging was performed using the TNM
classification. Margin samples were obtained at least 5 mm from the
edges of the surgical defects following primary tumor excision and
the absence of neoplastic cells was histologically confirmed.
Dulbecco's modified Eagles medium (DMEM) supplemented with 20%
fetal bovine serum (FBS) and 100 U/ml penicillin-100 µg/ml
streptomycin (Sigma-Aldrich; Merck KGaA) was used for tissue
culture. The tissue samples were homogenized with blades into 1
mm3 pieces and washed 3 times with PBS to remove loosely
bound cells, as previously described. An explant-cell culture
system (12,18) was carried out with periodical removal
of fibroblasts using differential trypsinization (19). The cells were grown in DMEM
supplemented with 10% FBS and 100 U/ml penicillin-100 µg/ml
streptomycin in T75 cell culture flasks. A 1:1 mixture of DMEM and
Ham's F12 medium supplemented with 400 ng/ml hydrocortisone and 10%
FBS was used for the SCC-25 cell culture. SCC-25 cells used for the
study were at the 10th passage. The cells were preserved at 37°C in
a humidified atmosphere containing 5% CO2. The medium
was changed every 2–3 days and the cells were passaged prior to
reaching 80% confluence. A total of 5×105 cells were
plated for the next passage. The tissue cells were designated as
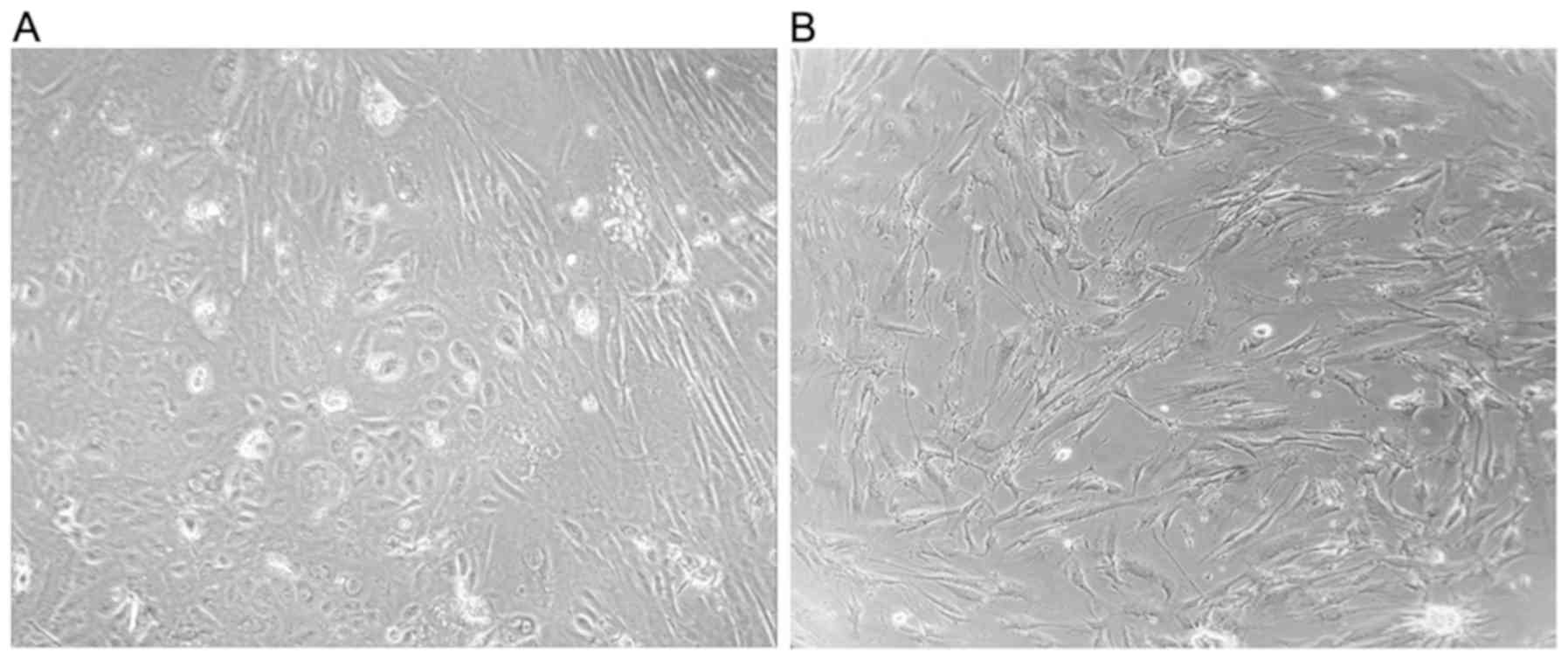
tumor tissue (Tu) and margin tissue (M) cells. These cells were
obtained following the first (P1) and the fifth (P5) passage
(Fig. 1). All experiments were
performed in triplicate and repeated for three times.
RNA extraction and reverse
transcription-quatintitative (RT-q) PCR
Total RNA was extracted from OSCC and margin cells,
SCC-25 and fibroblasts (106 cells per tube) using TRIzol
(Invitrogen; Thermo Fisher Scientific, Inc.). Complementary DNA was
prepared using the Revert Aid First Strand cDNA synthesis kit
(Thermo Fisher Scientific, Inc.) according to the manufacturer's
instructions. Subsequently, RT-qPCR analysis was performed on a
Line Gene-K Fluorescence Real-time PCR Detection System (Bioer
Technology, Inc.) using Maxima™ SYBR-Green/ROX qPCR Master Mix
(Thermo Fisher Scientific Inc.). The expression of GAPDH
(housekeeping gene) was used for normalization. The
2−ΔΔct method was used for the relative quantification
of gene expression as described by Livak and Schmittgen (20). All oligonucleotide primers were
purchased from Sigma-Aldrich; Merck KGaA and their sequences are
provided in Table I.
 | Table I.Primers used for expression
analysis. |
Table I.
Primers used for expression
analysis.
| Primer name | Sequence
(5′→3′) |
|---|
| Vimentin |
|
|
Forward |
TCTACGAGGAGGAGATGCGG |
|
Reverse |
GGTCAAGACGTGCCAGAGAC |
| α-SMA |
|
|
Forward |
CAATGGCTCTGGGCTCTGTAAG |
|
Reverse |
TGTTCTATCGGGTACTTCAGGGTC |
| SNAIL |
|
|
Forward |
ACCACTATGCCGCGCTCTT |
|
Reverse |
GGTCGTAGGGCTGCTGGAA |
| SLUG |
|
|
Forward |
TGTTGCAGTGAGGGCAAGAA |
|
Reverse |
GACCCTGGTTGCTTCAAGGA |
| GAPDH |
|
|
Forward |
TCATGACCACAGTCCATGCCATCA |
|
Reverse |
CCCTGTTGCTGTAGCCAAATTCGT |
Protein extraction and western
blotting
The cells were resuspended in RIPA lysis buffer (50
mM Tris-HCl pH 7.6, 150 mM sodium chloride, 1% Triton X-100, 1%
sodium deoxycholate, 0.1% sodium dodecyl sulphate, 2 mM EDTA and 50
mM sodium fluoride). A protease inhibitor cocktail (Pierce
Biotechnology, Inc.) and sodium orthovanadate (Sigma-Aldrich; Merck
KGaA) were added to the lysis buffer prior to use. Western blotting
was conducted by running equal amount protein samples on
polyacrylamide gels. The proteins were transferred from the gels to
polyvinylidene difluoride (PVDF) membrane. The experiment was
performed in duplicate. The membranes were first probed with
primary antibodies against anti-vimentin (Santa Cruz Biotechnology,
Inc.; cat. no. sc-32322, 1:200), then stripped with Restore Western
Blot Stripping Buffer (cat. no. 21059, Thermo Fisher Scientific,
Inc.) according to manufacture's instructions. After that, the
membranes were probed with anti-alpha smooth muscle actin antibody
(Abcam; cat. no. ab7817, 1:300). After stripping again with same
buffer, membranes were probed with β-actin (R&D; cat. no.
MAB8929, 1:6,000). Peroxidase-conjugated goat antimouse
immunoglobulin (Thermo Fisher Scientific, Inc.; cat. no. 31430,
1:5,000) was used as the secondary antibody. Hyperfilm was used to
visualize the protein expression by a chemiluminescent reagent kit
(Abcam; cat. no. ab79907) according to the manufacturer's
instructions. The content of Vimentin and α-SMA in the tumor and
margin cell cultures was estimated by densitometry of the scanned
immunoblot bands using the Image Lab (Bio-Rad) software and
normalized to the β-actin protein density.
Colony formation assay
The cells were seeded in the 12-well plate at a
concentration of 104 cells per well in DMEM supplemented
with 10% FBS. Tumor and margin cells were incubated (37°C, 5%
CO2) and the number of live cells was counted following
3, 5, and 7 days of incubation period.
A total of 200 cells were plated per well in 32
mm-wide plates and cultured in 1.5 ml of DMEM supplemented with 10%
FBS for 7 and 14 days. The cell colonies were washed with PBS,
fixed in formalin for 5 min and stained with 0.05% crystal violet
for 30 min at room temperature. Following removal of the dye, the
colonies with more than 50 cells were counted as positive using the
ImageJ software. The results are presented as colony formation
efficiency (21) and as the ratio
between the number of colonies formed and the number of seeded
cells.
Wound healing assay
A wound healing assay was conducted to detect cell
motility (22). Single-cell
suspensions of margins and tumors were added to 24-well plates
(2×105 cells/well in 0.7 ml of DMEM supplemented with
10% FBS) and cultured for 4–5 days to a confluence of approximately
80%. The monolayer was scratched with a sterile 1.2 mm-wide pippete
tip across the center of the well in a straight line to cause a
wound in the confluent cell monolayer. Subsequently, the wells were
washed with PBS and grown in the incubator with serum-free DMEM
(37°C, 5% CO2). A BIB-100/T inverted microscope and
HDCE-90D camera with Scope Image 9.0 software (BOECO Germany) were
used to measure the closest area of the scratch. Cell migration was
calculated by monitoring the entire movement of the monolayer. The
shortest distance was measured between the separated monolayers and
divided with time. The cell speed, measured in µm/h, was calculated
for all 24 h intervals until the scratch area was closed, as
follows:
S(Iday)=d(0h)-d(24h)24S(IIday)=d(24h)-d(48h)24S(IIIday)=d(48h)-d(72h)24
The average cell speed was
S=S(Iday)+S(IIday)+S(IIIday)3
Statistical analysis
One-way or two-way ANOVA tests, with Tukey's post
hoc comparison were performed in the present study, after checking
the distribution normality by Kolmogorov-Smirnov normality test.
The values are presented as mean ± SD. Statistical significance was
set at P<0.05. The software package GraphPad Prism version 6 was
used for the analyses (GraphPad Software, Inc.).
Results
Vimentin, α-SMA, SLUG and SNAIL are
expressed in tumor and margin samples
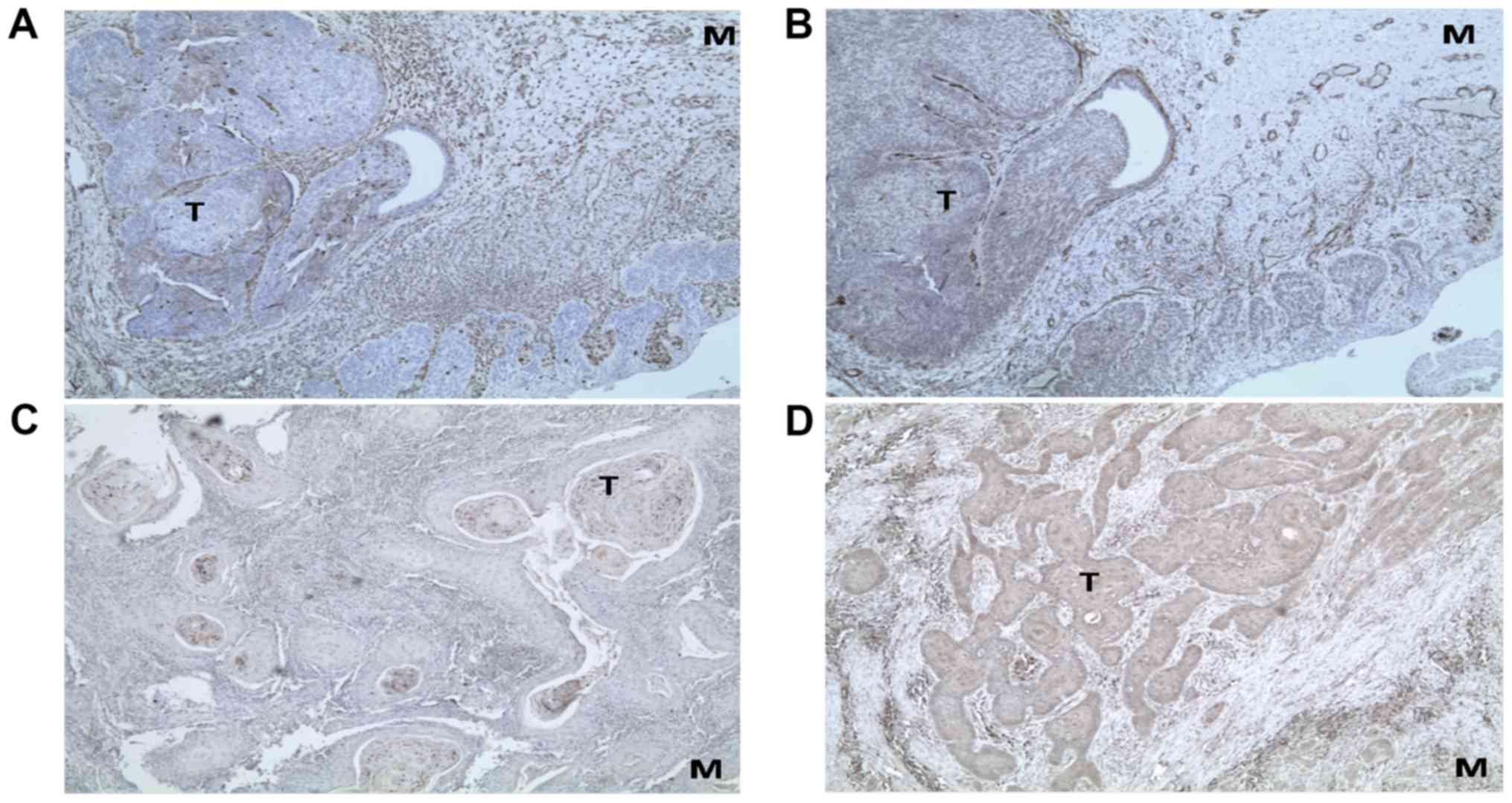
Prior to undertaking mRNA expression analyses in
cell cultures, the presence of EMT markers was determined by
immunohistochemical analysis of fixed tissue specimens. Tumors and
resection margins were used from patients for establishing cell
cultures. A clear immunostaining could be observed for all the
markers (Vimentin, α-SMA, SLUG and SNAIL) in the tumors as well as
in their margins. The representative micrographs are shown in
Fig. 2.
RT-qPCR analysis was used to detect the expression
levels of the epithelial to mesencymal transition markers
(Vimentin, α-SMA, SLUG and SNAIL) in both tumor and margin cells.
The expression levels in these two types of cells were different.
However, no significanct difference was noted (P>0.05). Vimentin
was the marker that exhibited the highest levels of expression in
both OSCC and margin cells and as expected in fibroblast cultures.
However, these results were not noted in the oral cancer cell line
SCC-25, which maintained its epithelial characteristics during
cultivation. The levels of Vimentin mRNA were higher in
tumor and margin cells at the beginning of cultivation (1st
passage) than in the cells of the 5th passage. However, the
differences noted were not significant. In contrast, the relative
gene expression levels of α-SMA (in margin cultures) and SNAIL (in
tumor cultures) were higher in the 5th passage compared with those
of the 1st passage (P<0.05) (Fig.
3). This trend was also noted in the expression levels of SLUG
in tumor cells.
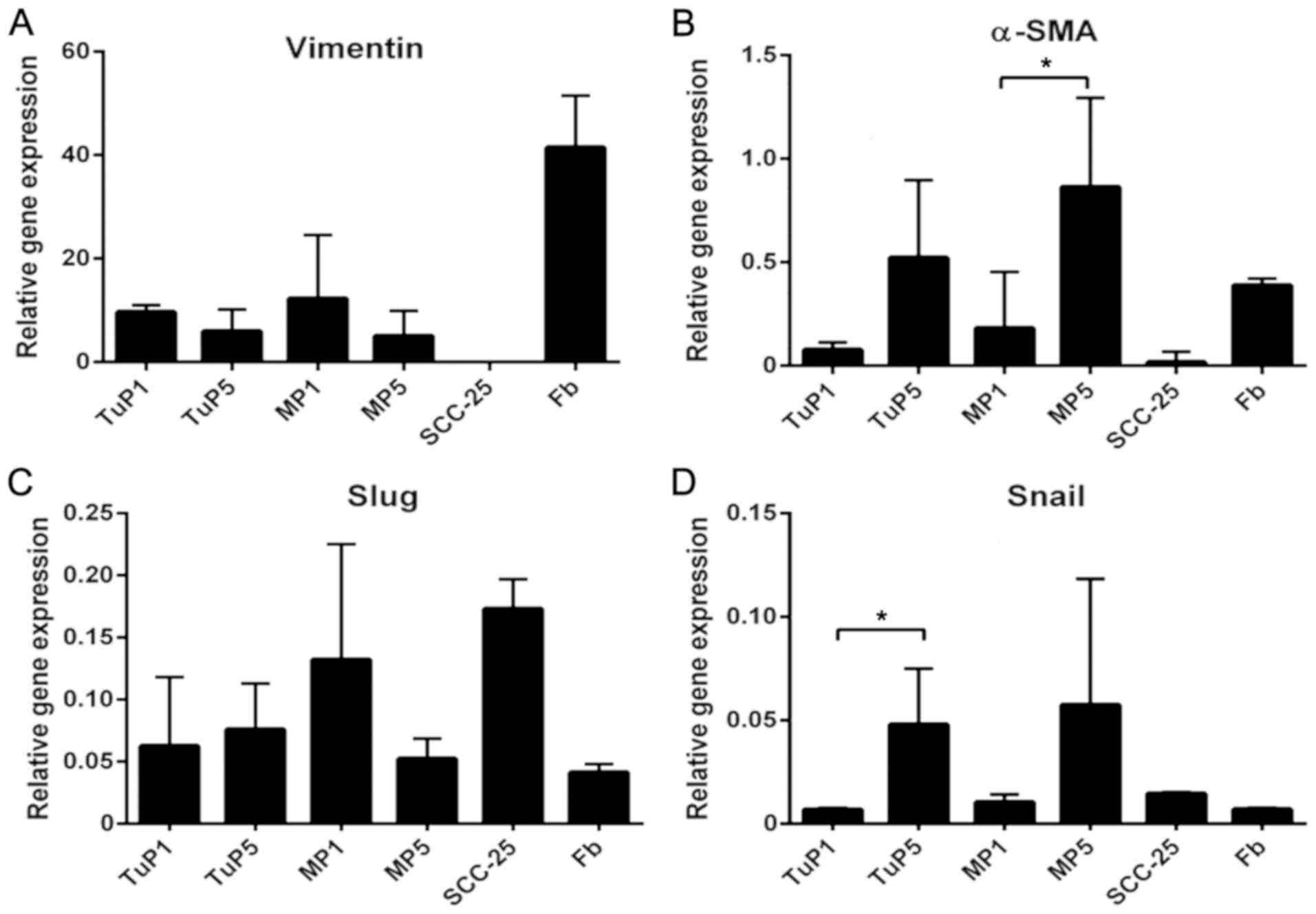 | Figure 3.Expression of epithelial to
mesenchymal transition-associated markers in tumor, margin and
control cell cultures. (A) Vimentin, (B) α-SMA, (C) Slug and (D)
Snail expression. The SCC-25 cancer cell line was used as a
negative control sample, whereas Fb isolated from gingiva were used
as positive control samples. Data are presented as the mean ±
standard deviation. *P<0.05 as indicated. EMT, epithelial to
mesenchymal transition; Fb, fibroblasts; SMA, smooth muscle actin;
TuP1, tumor cells derived from the first passage; TuP5, tumor cells
of the fifth passage; MP1, margin cells of the first passage; MP5,
margin cells of the fifth passage. |
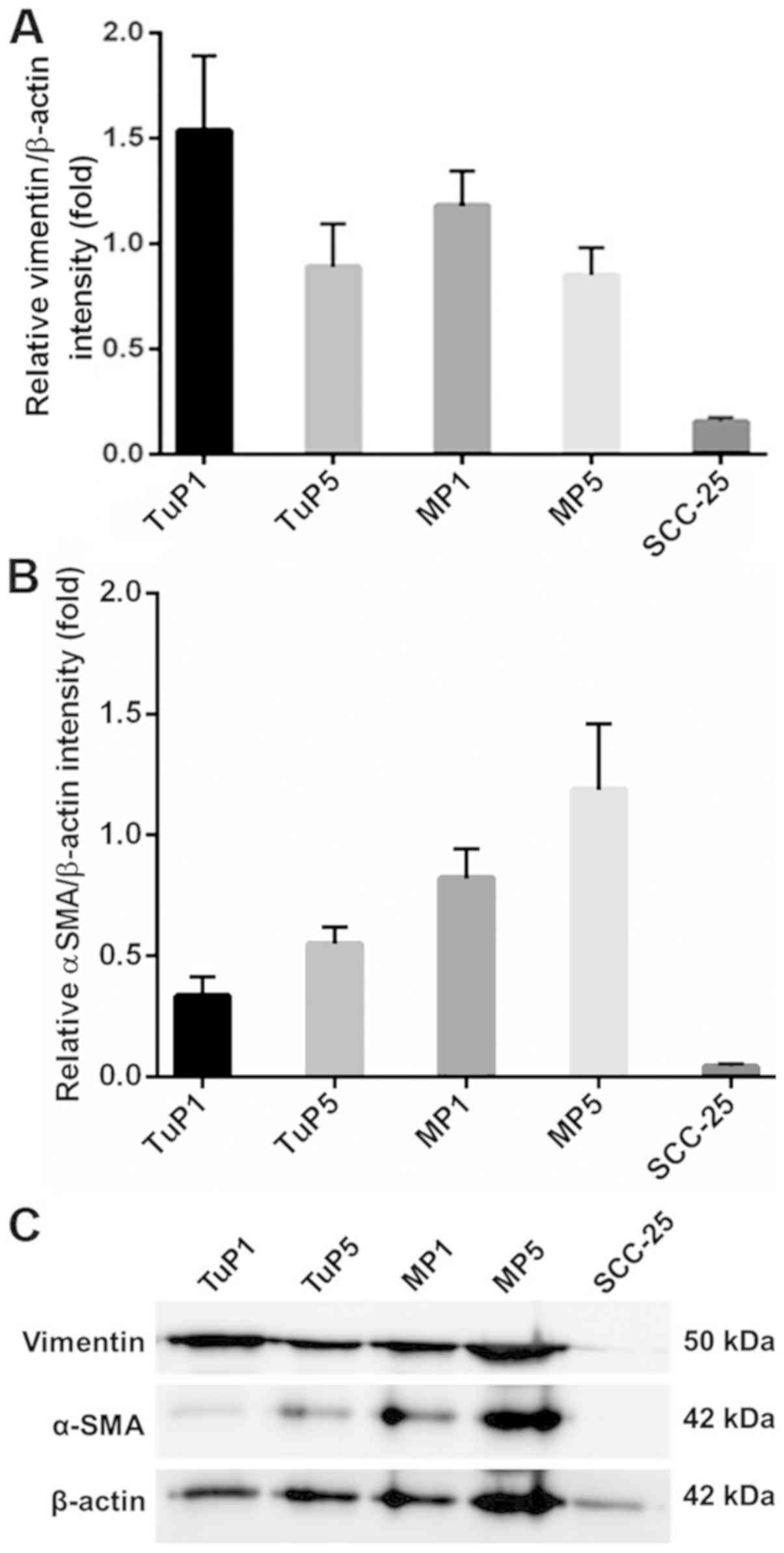
In order to confirm the findings obtained by qPCR
analysis, the protein expression levels of the two EMT markers
Vimentin and α-SMA were examined. Protein expression was in line
with mRNA levels, although without statistically significant
differences between passages (Fig.
4).
The proliferation rate of tumor and
margin cells depends on their passage number
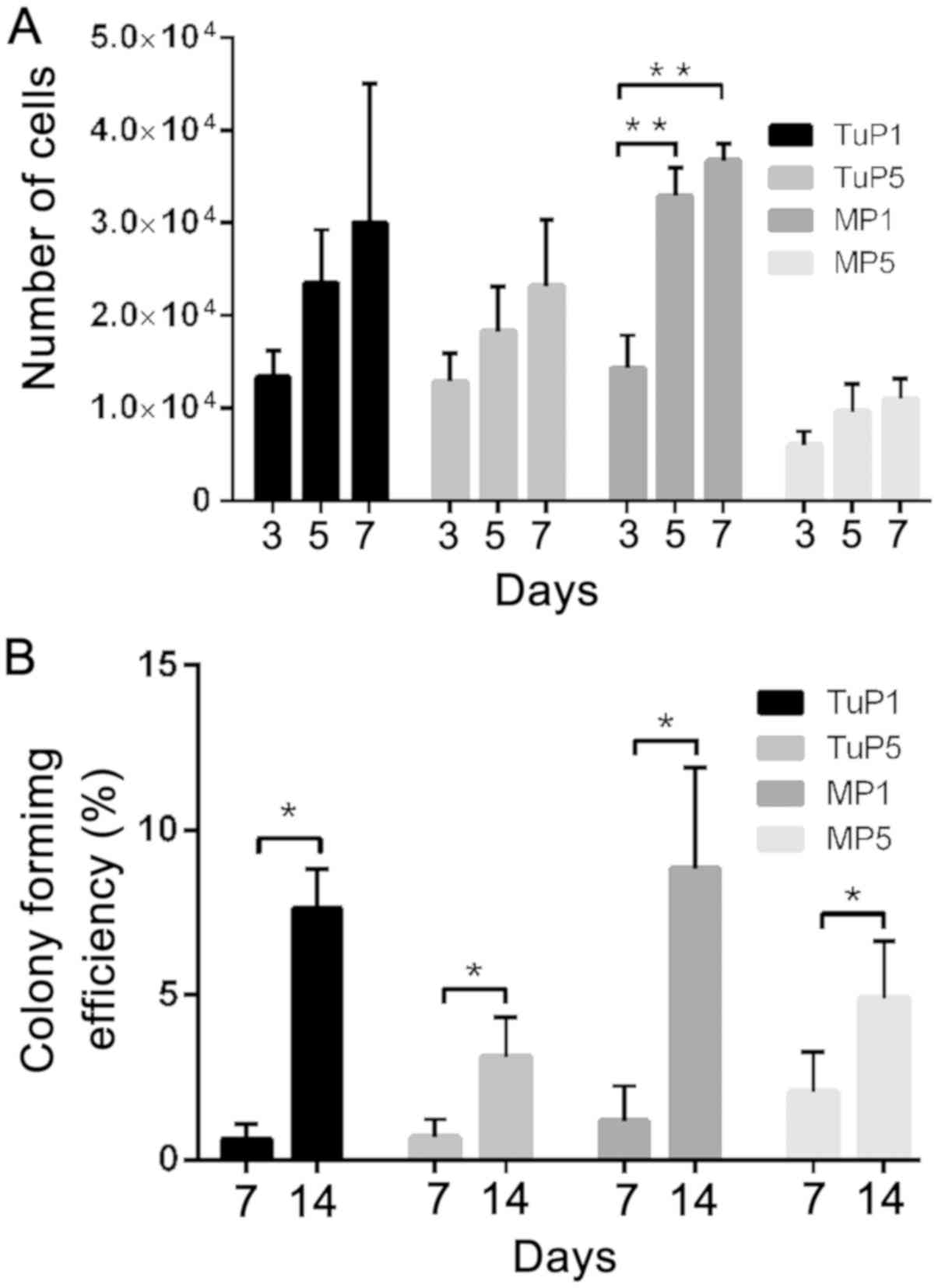
The number of cells was increased during the 7-day
assay duration, with the exception of the 5th passage margin cells.
The differences noted between tumor and margin cells were evident
in the 5th passage of cells. Tumor cells exhibited higher
proliferative potential (Fig. 5A).
Generally, in both tumor and margin cells, the proliferation rates
decreased from the 1st to the 5th passage.
The differences noted with regard to the colony
formation ability were statistically significant from the 7th to
the 14th day (P<0.05). The number of colonies formed by the
tumor cells (1.25 and 15.25 following 7 and 14 days of incubation,
respectively), and margin cells (2.33 and 17.67 following 7 and 14
days of incubation, respectively) was similar. The colony formation
efficiency is provided in Fig. 5B.
No significant differences were noted in that parameter during
different passages. The motility of the tumor and margin cells did
not exhibit significant differences between cell passages (Fig. 6). The average value of cell speed
during the 72 h period is given in Fig.
6Q.
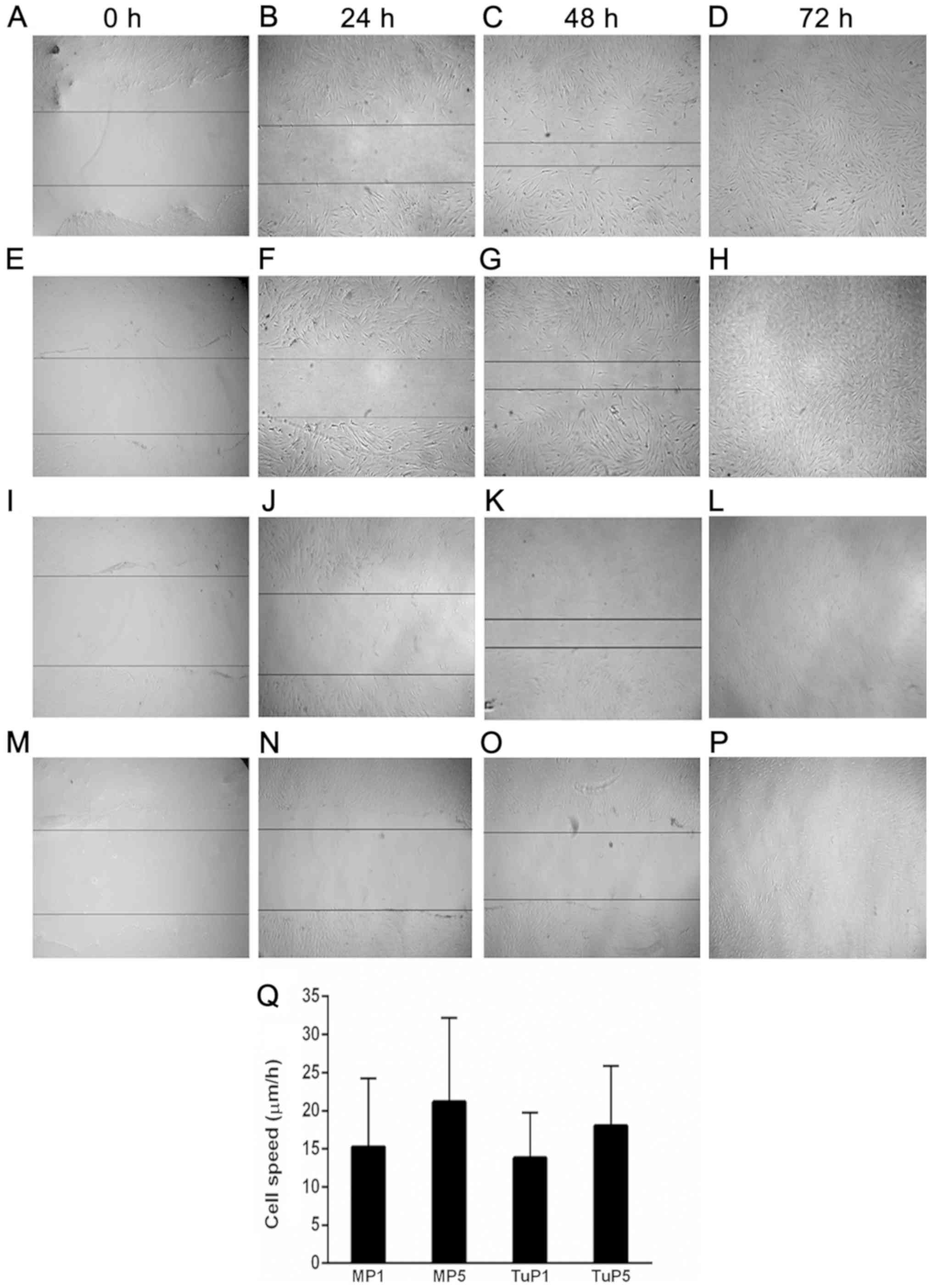 | Figure 6.Representative micrographs of cell
migration. Tumor cells of the first passage at (A) 0 h, (B) 24 h,
(C) 48 h and (D) 72 h after the scratch. Tumor cells of the fifth
passage at (E) 0 h, (F) 24 h, (G) 48 h and (H) 72 h after the
scratch. Margin cells of the first passage at (I) 0 h, (J) 24 h,
(K) 48 h and (L) 72 h after the scratch. Margin cells of the fifth
passage at (M) 0 h, (N) 24 h, (O) 48 h and (P) 72 h after the
scratch. (Q) Average migration was subsequently calculated.
Magnification, ×40. TuP1, tumor cells derived from the first
passage; TuP5, tumor cells of the fifth passage; MP1, margin cells
of the first passage; MP5, margin cells of the fifth passage; d,
day. |
Discussion
The poor prognosis of OSCC is mainly caused due to
the high recurrence and metastasis rates and remains a significant
medical challenge, regardless of the advances in diagnostic and
therapeutic procedures (23,24). Recent studies have recognized EMT as
a critical process during tumor cell invasion of the surrounding
stroma, which may explain specific essential steps leading to
metastasis and tumor recurrence.
The present study hypothesized that EMT occurs
preferentially at the tumor margin, which is the site of vessel
-invasion (13,14,25).
Consequently, margin tissue samples were used in addition to
resected OSCC samples in order to establish cell cultures and
assess their corresponding EMT-associated features. All EMT markers
(Vimentin, α-SMA, SNAIL, SLUG) were expressed in both types of
cells (tumor and margin) at various levels. The expression levels
of these markers did not exhibit significant differences between
cancer and margin cells, although, they were significantly higher
in the margin tissues.
In the present study, high levels of Vimentin
mRNA were detected in both tumor and margin cell cultures. The
migratory potential of the two cell types was highly concordant,
probably due to similar expression levels of Vimentin. Increased
expression levels of Vimentin are associated with cancer
progression. This protein is an indicator of high cell migratory
activity (8,26) which is in agreement with the results
of the present study.
One of the major factors affecting EMT marker
expression was cell passaging. The expression levels of the markers
were increased during cell passages and significant differences
were established for α-SMA and SNAIL in both types of cells. This
finding can tentatively be explained by cell culture enrichment
with cancer stem cells during passages as determined by the
increase in the expression levels of CSC markers, such as CD44,
CD133, Oct4, Sox2 and Nanog (12).
This in turn reflects to a certain extent the clonal evolution of
tumors and the increase of aggressiveness during tumor progression.
It was previously shown that SNAIL exerted an important role in
inducing and maintaining CSC-like properties by EMT induction in
OSCC (27). Alternatively, the cells
in the culture may undergo transdifferentiation during passaging,
progressively acquiring the mesenchymal phenotype. This phenomenon
has been previously described in ovarian cancer cells, which can
synthesize collagen I and II, while gradually losing the expression
of cytokeratin during passaging (28).
The proliferation rates of tumor and margin cells
was decreased over the passaging period and this finding was in
accordance with Vega et al (29) and Mejlvang et al (30) who demonstrated that the activation of
EMT reduces cell proliferation. In the present study, tumor and
tumor margin cells exhibited similar abilities to form colonies,
which was also accompanied by relevant EMT-associated features
(31). This similar capacity to form
colonies was in accordance with the relative homogenous expression
of the EMT markers analyzed in tumor and margin samples. These
findings provide additional evidence in favor of the concept that
EMT is present at multiple sites within the tumor, as previously
hypotesized (6,32). A recent study further demonstrated a
characteristic pattern of expression for the proteins TWIST1,
SNAI1, SNAI2 and ZEB1 in the center of primary breast tumors and
their margins (33). Cancer
‘self-seeding’ ability may be another explanation for the
remarkable phenotypic similarities of cancer and margin cells
(34).
The data presented in the present study suggest that
the EMT markers Vimentin, α-SMA, SNAIL and SLUG were similarly
expressed in tumor and margin cells of OSCC and that cell passaging
increased their expression levels. With the exception of certain
minor differences, the parameters proliferation rate, clonal
ability, and migratory capacity were similar in the cells
originating from tumors and resection margins, which suggested the
importance of margin pathological status in terms of tumor
aggressiveness.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Ministry of
Education, Science and Technological Development, Republic of
Serbia (grant nos. 175075 and III 41027) and the Ministry of
Foreign Affairs of Italy (grant no. PGR02954).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
ML and JM conceived and designed the current study.
ML, MM, BT, DJ and FB performed RT-qPCR. SM, ZT and TS performed
histology, immunohistochemistry and western blotting. ML, MM, BT,
DJ and DT performed cell culture. ML, MM, SM, ZT, DJ, GD and FB
analyzed and interpreted the data. LM, JD and MD aquired clinical
data and samples. ML, GD and JM wrote the manuscript. All authors
revised the manuscript.
Ethics approval and consent to
participate
The current study was approved by the Institutional
Ethical Committee of the School of Dental Medicine (approval no.
36/31). Written informed consent was obtained from all
patients.
Patient consent for publication
All patients signed an informed consent to
participate in the study that included patients' agreement to the
publication of their data/images on condition of anonymity.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Maeng YI, Kim KH, Kim JY, Lee SJ, Sung WJ,
Lee CK, Park JB and Park KK: Transcription factors related to
epithelial mesenchymal transition in tumor center and margin in
invasive lung adenocarcinoma. Int J Clin Exp Pathol. 7:4095–4103.
2014.PubMed/NCBI
|
|
2
|
Acloque H, Adams MS, Fishwick K,
Bronner-Fraser M and Nieto MA: Epithelial-mesenchymal transitions:
The importance of changing cell state in development and disease. J
Clin Invest. 119:1438–1449. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lim J and Thiery JP:
Epithelial-mesenchymal transitions: Insights from development.
Development. 139:3471–3486. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Thiery JP: Epithelial-mesenchymal
transitions in tumour progression. Nat Rev Cancer. 2:442–454. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Moreno-Bueno G, Portillo F and Cano A:
Transcriptional regulation of cell polarity in EMT and cancer.
Oncogene. 27:6958–6969. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peinado H, Olmeda D and Cano A: Snail, ZEB
and bHLH factors in tumour progression: An alliance against the
epithelial phenotype? Nat Rev Cancer. 7:415–428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Misra A, Pandey C, Sze SK and Thanabalu T:
Hypoxia activated EGFR signaling induces epithelial to mesenchymal
transition (EMT). PLoS One. 7:497662012. View Article : Google Scholar
|
|
9
|
Liang L, Zeng M, Pan H, Liu H and He Y:
Nicotinamide N-methyltransferase promotes epithelial-mesenchymal
transition in gastric cancer cells by activating transforming
growth factor-β1 expression. Oncol Lett. 15:4592–4598.
2018.PubMed/NCBI
|
|
10
|
Krisanaprakornkit S and Iamaroon A:
Epithelial-mesenchymal transition in oral squamous cell carcinoma.
ISRN Oncol. 2012:6814692012.PubMed/NCBI
|
|
11
|
Zhang Z, Dong Z, Lauxen IS, Filho MS and
Nor JE: Endothelial cell-secreted EGF induces epithelial to
mesenchymal transition and endows head and neck cancer cells with
stem-like phenotype. Cancer Res. 74:2869–2881. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lazarevic M, Milosevic M, Trisic D, Toljic
B, Simonovic J, Nikolic N, Mikovic N, Jelovac D, Petrovic M,
Vukadinovic M and Milasin J: Putative cancer stem cells are present
in surgical margins of oral squamous cell carcinoma. J BUON.
23:1686–1692. 2018.PubMed/NCBI
|
|
13
|
Nassar A, Radhakrishnan A, Cabrero IA,
Cotsonis GA and Cohen C: Intratumoral heterogeneity of
immunohistochemical marker expression in breast carcinoma: A tissue
microarray-based study. Appl Immunohistochem Mol Morphol.
18:433–441. 2010.PubMed/NCBI
|
|
14
|
Brabletz T, Jung A, Reu S, Porzner M,
Hlubek F, Kunz-Schughart LA, Knuechel R and Kirchner T: Variable
beta-catenin expression in colorectal cancers indicates tumor
progression driven by the tumor environment. Proc Natl Acad Sci.
98:10356–10361. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zacharias M, Brcic L, Eidenhammer S and
Popper H: Bulk tumour cell migration in lung carcinomas might be
more common than epithelial-mesenchymal transition and be
differently regulated. BMC Cancer. 18:7172018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Eljabo N, Nikolic N, Carkic J, Jelovac D,
Lazarevic M, Tanic N and Milasin J: Genetic and epigenetic
alterations in the tumour, tumour margins, and normal buccal mucosa
of patients with oral cancer. Int J Oral Maxillofac Surg.
47:976–982. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jelovac DB, Tepavčević Z, Nikolić N, Ilić
B, Eljabo N, Popović B, Čarkić J, Konstantinović V, Vukadinović M,
Miličić B and Milašin J: The amplification of c-erb-B2 in
cancer-free surgical margins is a predictor of poor outcome in oral
squamous cell carcinoma. Int J Oral Maxillofac Surg. 45:700–705.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mitra A, Mishra L and Li S: Technologies
for deriving primary tumor cells for use in personalized cancer
therapy. Trends Biotechnol. 31:347–354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jones JC: Reduction of contamination of
epithelial cultures by fibroblasts. CSH Protoc. pdb.prot4478.
2008.
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using r, eal-time quantitative PCR
and the 2(T)(-Delta Delta C) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Trivanović D, Kocić J, Mojsilović S,
Krstić A, Ilić V, Djordjević IO, Santibanez JF, Jovcić G, Terzić M
and Bugarski D: Mesenchymal stem cells isolated from peripheral
blood and umbilical cord Wharton's jelly. Srp Arh Celok Lek.
141:178–186. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wong K, Rubenthiran U and Jothy S:
Motility of colon cancer cells: Modulation by CD44 isoform
expression. Exp Mol Pathol. 75:124–130. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Feller LL, Khammissa RR, Kramer BB and
Lemmer JJ: Oral squamous cell carcinoma in relation to field
precancerisation: Pathobiology. Cancer Cell Int. 13:31–39. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zaravinos A: The regulatory role of
MicroRNAs in EMT and cancer. J Oncol. 2015:8658162015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kurihara K, Isobe T, Yamamoto G, Tanaka Y,
Katakura A and Tachikawa T: Expression of BMI1 and ZEB1 in
epithelial-mesenchymal transition of tongue squamous cell
carcinoma. Oncol Rep. 34:771–778. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Qin X, Yan M, Li R, Ye D, Zhang J, Xu Q,
Feng Y, Sun Q, Jiang C and Chen W: Identification and
characterization of a highly metastatic epithelial cancer cell line
from rat tongue cancer. Arch Oral Biol. 95:58–67. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ota I, Masui T, Kurihara M, Yook JI,
Mikami S, Kimura T, Shimada K, Konishi N, Yane K, Yamanaka T and
Kitahara T: Snail-induced EMT promotes cancer stem cell-like
properties in head and neck cancer cells. Oncol Rep. 35:261–266.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Van Marck VL and Bracke ME:
Epithelial-mesenchymal transitions in human cancer. Madame Curie
Bioscience Database. (Landes Bioscience, Austin). 2000.
|
|
29
|
Vega S, Morales AV, Ocaña OH, Valdés F,
Fabregat I and Nieto MA: Snail blocks the cell cycle and confers
resistance to cell death. Genes Dev. 18:1131–1143. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mejlvang J, Kriajevska M, Vandewalle C,
Chernova T, Sayan AE, Berx G, Mellon JK and Tulchinsky E: direct
repression of cyclin D1 by sip1 attenuates cell cycle progression
in cells undergoing an epithelial mesenchymal transition. Mol Biol
Cell. 18:4615–4624. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Martin TA, Goyal A, Watkins G and Jiang
WG: Expression of the transcription factors snail, slug, and twist
and their clinical significance in human breast cancer. Ann Surg
Oncol. 12:488–496. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Alkatout I, Wiedermann M, Bauer M, Wenners
A, Jonat W and Klapper W: Transcription factors associated with
epithelial-mesenchymal transition and cancer stem cells in the
tumor centre and margin of invasive breast cancer. Exp Mol Pathol.
94:168–173. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim MY, Oskarsson T, Acharyya S, Nguyen
DX, Zhang XH, Norton L and Massagué J: Tumor self-seeding by
circulating cancer cells. Cell. 139:1315–1326. 2009. View Article : Google Scholar : PubMed/NCBI
|