Introduction
Oral squamous cell carcinoma (OSCC) is the most
frequent type of head and neck squamous cell carcinoma, and it is
associated with high morbidity rates and a poor prognosis worldwide
(1,2). OSCC is characterized as an abundance of
blood and lymphatic vessels that favor tumor growth and lymphatic
metastasis (3). Despite the
significant progress in treatment options, including surgery,
radiotherapy and chemotherapy, the five-year survival rate of
patients with OSCC remains relatively low due to its high rate of
recurrence (4). Therefore, it is
important to understand the molecular mechanisms driving OSCC
malignancy to facilitate the development of more appropriate
treatment strategies to improve the prognosis and survival rates of
patients with OSCC.
Special AT-rich sequence-binding protein 2 (SATB2)
is a nuclear matrix attachment region binding protein that
regulates gene expression and chromatin remodeling (5). It was initially identified as a gene
involved in palatogenesis and craniofacial morphogenesis; however,
more recent evidence has revealed that SATB2 may also serve an
important role in tumor development. For example, SATB2
overexpression increased cell proliferation, stem cell-like gene
expression and tumor growth in osteosarcoma (6); and SATB2 was observed to have an
oncogenic role in promoting colorectal cancer tumorigenesis and
progression (7). Nonetheless, the
biological function of SATB2 in OSCC progression and metastasis
remains poorly understood.
It is well established that the hypoxic
microenvironment has a vital role in driving the invasion and
metastasis of OSCC (8,9). Notably, recent evidence has also
suggested that hypoxia may promote stemness and autophagic
properties in cancer cells, which in turn may promote the survival
and malignancy of cancer cells during hypoxic stress that
ultimately leads to poor clinical outcomes (10,11). Our
previous study found that SATB2 was expressed in bone marrow
mesenchymal stem cells, where it served an important role in
regulating the stemness and autophagy of these cells (12). Thus, the present study aimed to
investigate the role of SATB2 in hypoxia-induced stemness and
autophagy in OSCC, as well as its ability to regulate cellular
survival, migration and invasion. The current results revealed that
the expression of SATB2 was significantly increased in the OSCC
cells. Silencing of SATB2 in the OSCC cells suppressed the
hypoxia-induced autophagy and stemness properties, as well as
aggressive behavior of OSCC cells. These findings may provide a
novel target for treating the most common malignancy of the oral
cavity.
Materials and methods
Ethical approval
The current study protocol was reviewed and approved
by the Institutional Review Boards of The First Hospital of Jiaxing
and the Yancheng Hospital Affiliated to Medical School of Southeast
University.
Cell culture and transfection
The SCC9 OSCC cell line was purchased from The Cell
Bank of Type Culture Collection of the Chinese Academy of Sciences.
Cells were cultured in DMEM/F12 (Gibco; Thermo Fisher Scientific,
Inc.) supplemented with 10% FBS (Gibco; Thermo Fisher Scientific,
Inc.) and 1% penicillin/streptomycin (HyClone; GE Healthcare Life
Sciences), and maintained in a humidified atmosphere at 37°C and 5%
CO2.
The SATB2 overexpression plasmid (pcDNA3.1-SATB2)
and empty vector [negative control (NC); pcDNA3.1] were constructed
and purchased from Shanghai GeneChem Co., Ltd. Small interfering
RNAs (siRNAs) targeting SATB2 (si-SATB2) and the NC (si-NC) were
obtained from Shanghai GenePharma Co., Ltd. The sequences of the
siRNAs were as follows: si-SATB2-1, 5′-GUCAGAGAUGAGCUGAAGATT-3′;
si-SATB2-2, 5′-CCAAACACACCAUCAUCAAGU-3′; and si-NC,
5′-UUCUCCGAACGUGUCACGUTT-3′. Transfections of the SCC9 cells with
plasmids (1 µg/well) and siNRAs (50 nM) were performed using a
Lipofectamine® 3000 kit (Gibco; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocols.
To induce hypoxic conditions, cells were transfected
with siRNAs and cultured in normoxic conditions for 24 h, prior to
being exposed to a hypoxic environment of 5% CO2, 1%
O2 and 94% N2 in a hypoxia incubator for 3,
6, 12 and 24 h (Forma Scientific; Thermo Fisher Scientific,
Inc.).
Monodansylcadaverine (MDC)
staining
MDC staining was used to visualize autophagosomes
(13). Briefly, 5×103
SCC9 cells/well were plated into 96-well plates (Corning Inc.) with
DMEM/F12 medium and cultured in the incubator at 37°C for 6 h.
Cells were subsequently cultured with 50 µm MDC (Sigma-Aldrich;
Merck KGaA) for 15 min at 37°C. Then, cells were fixed in 4%
paraformaldehyde (Sigma-Aldrich; Merck KGaA) at room temperature
for 20 min and washed thrice with 1X PBS (pH 7.4). The fluorescence
intensity was visualized and analyzed using an Olympus IX70
inverted fluorescence microscope (magnification, ×100; excitation
wavelength, 340 nm; emission wavelength, 535 nm; Olympus
Corporation). All experiments were repeated in triplicate.
Transmission electron microscopy
(TEM)
Following 24 h of hypoxic treatment, the SCC9 cells
were harvested and fixed with 2.5% glutaraldehyde solution
overnight at 4°C. Then, cells were fixed using 2% osmium tetroxide
for 2 h at room temperature, rehydrated with an increasing series
of ethanol (30, 50, 70, 80, 90 and 100%) and embedded in epoxy
resin. Sections (50–70 nm thick) were cut using an ultramicrotome
and subsequently stained with 2% uranyl acetate and 3% lead citrate
for 15 min each at room temperature. Autophagosomes were observed
under a JEM-1400Plus transmission electron microscope
(magnification, ×6,000; JEOL, Ltd.).
Colony forming assay
The colony forming assay was performed as previously
described (12). Briefly, single
cell suspensions of 1×103 cells/well were plated into
6-well plates (Corning Inc.) and cultured in complete DMEM/F12 at
37°C and 1% CO2 for 12 days; the medium was changed
every 3 days. Upon incubation, the visible colonies were fixed in
4% paraformaldehyde (Sigma-Aldrich) at room temperature for 20 min
and subsequently stained with a 0.1% crystal violet solution
(Sigma-Aldrich; Merck KGaA) for 30 min at room temperature. The
stained cells were visualized under an inverted microscope
(magnification, ×40; Olympus Corporation), and the number of
colonies with ≥50 cells in each well were counted.
Wound healing assay
The migratory ability of cells was analyzed using a
wound healing assay. Briefly, when the monolayer of cells reached
100% confluence in 6-well plates, a 20-µl sterile pipette tip was
used to create the wound. Cells were washed with 1X PBS three times
to remove the detached and damaged cells. Then, the cells were
incubated under hypoxic conditions for 24 h with serum-free
DMEM/F12. At 0 and 24-h incubation, the cells were imaged using an
inverted light microscope (magnification, ×100; Olympus
Corporation), and the wound closure area was used to calculate the
migratory ability of cells using ImageJ software (version 1.48,
National Institutes of Health). Each experiment was performed in
triplicate.
Cell invasion assay
The invasive ability of cells was determined using a
Transwell assay. A total of 5×104 SCC9 cells were
resuspended in 200 µl serum-free DMEM/F12 and plated in the upper
chambers of Transwell plates (pore size, 8-µm; Costar; Corning
Inc.) which pre-coated with 25 µl Matrigel (BD Biosciences) at 37°C
for 30 min. A total of 500 µl complete DMEM/F12 was plated in the
lower chambers. Following incubation at 37°C in the hypoxia
incubator for 24 h, the cells were fixed in 4% PFA for 20 min at
room temperature and subsequently stained with 0.1% crystal violet
(Sigma-Aldrich; Merck KGaA) for 30 min at room temperature. The
non-invasive cells remaining in the upper chambers were removed
with a swab, and the invasive cells present on the lower surface
were counted in 10 randomly selected fields using an inverted
microscope (magnification, ×100; Olympus Corporation).
Cell proliferation assay
The proliferative ability of SCC9 cells was
evaluated by CCK-8 assay (Beyotime Institute of Biotechnology),
according to the manufacturer's protocols. Briefly,
1×103 SCC9 cells/well with or without siRNA transfection
were seeded into 96-well plates. At 24, 48 and 72 h, 10 µl CCK-8
reagent was added into each well, and the plates were incubated at
37°C for additional 2 h. The absorbance was measured at 450 nm
using a microplate spectrophotometer (Thermo Fisher Scientific
Inc.). Each experiment was performed in triplicate.
Cell cycle assay
Following incubation under hypoxia for 24 h, the
SCC9 cells were harvested, washed with cold PBS and fixed in 70%
ice-cold ethanol overnight at −20°C. The cells were subsequently
stained with 50 µg/ml propidium iodide (PI) and 20 µg/ml RNase A
(BD Biosciences) at 4°C for 30 min. After incubation, the cells
were subjected to DNA content analysis using a BD FACSCalibur flow
cytometer (LSR II; BD Biosciences), and the data were analyzed
using CellQuest™ Pro version 5.2 software (BD Biosciences).
Cell apoptosis assay
Annexin V-fluorescein isothiocyanate
(FITC)/propidium iodide (PI) Apoptosis Detection kit (BD
Biosciences) was performed to detect apoptosis according to the
manufacturer's instructions. Briefly, after washing with PBS, SCC9
cells were collected by trypsinization with 0.25% trypsin solution
without EDTA and resuspended in 500 µl 1X binding buffer from the
kit. Then, 5 µl Annexin V-FITC and 5 µl PI were added for 15 min at
room temperature in the dark. Cell apoptosis was subsequently
detected using a BD FACSCalibur flow cytometer (BD Biosciences),
and data were analyzed using FlowJo version 9.0 software (FlowJo
LLC). Each experiment was performed in triplicate.
Western blotting
Total protein was extracted from SCC9 cells using
RIPA lysis buffer (Beyotime Institute of Biotechnology), containing
a protease inhibitor and phenylmethylsulfonyl fluoride. Following
centrifugation of the lysates at 12,000 × g for 15 min at 4°C, the
supernatants were collected, and total protein was quantified using
a bicinchoninic assay kit (Beyotime Institute of Biotechnology).
Equal amounts of protein from each sample (40 µg/well) were
separated by 10% SDS-PAGE and subsequently transferred onto
polyvinylidene fluoride membranes (EMD Millipore). Membranes were
blocked with 5% skimmed milk (Sigma-Aldrich; Merck KGaA) in
TBS-0.05% Tween-20 (TBST) buffer (Sangon Biotech Co., Ltd.) at room
temperature for 2 h and then incubated with the following primary
antibodies (1:1,000) at 4°C overnight: Anti-hypoxia-inducible
factor 1-α (HIF-1α; cat. no. 3716; Cell Signaling Technology,
Inc.), anti-SATB2 (cat. no. ab34735; Abcam),
anti-microtubule-associated protein light chain 3-II/I (LC3-II/I;
cat. no. 2775; Cell Signaling Technology, Inc.), anti-Beclin-1
(cat. no. 3495; Cell Signaling Technology, Inc.), anti-Oct-4 (cat.
no. ab137427; Abcam), anti-Sox-2 (cat. no. ab171380; Abcam),
anti-Nanog (cat. no. ab70482; Abcam) and anti-β-actin (cat. no.
3700; Cell Signaling Technology, Inc.). Following the primary
antibody incubation, the membranes were washed three times with
TBST and subsequently incubated with horseradish
peroxidase-conjugated secondary antibodies (cat. nos. 7074 and
7076; Cell Signaling Technology, Inc.) for 1 h at room temperature.
Protein bands were visualized using an enhanced chemiluminescence
detection kit (PerkinElmer, Inc.) and a Bio-Rad gel image analysis
system (Bio-Rad Laboratories, Inc.). Protein expression was
quantified using ImageJ version 1.5b software (National Institutes
of Health) and normalized to β-actin as the loading control.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism 5 software (GraphPad Software, Inc.), and all data were
presented as the mean ± SD from ≥3 replicates for each experiment.
Statistical differences amongst groups were determined using
one-way ANOVA, followed by Dunnett's test for multiple comparisons.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Hypoxia induces the expression of
SATB2 in SCC9 cells
Following the exposure of SCC9 cells to hypoxia for
3, 6, 12 or 24 h, western blotting revealed that HIF-1α protein
expression levels began to increase significantly within 3 h,
peaked at 12 h and then decayed at 24 h. A similar pattern was
observed with SATB2 expression levels, indicating that SATB2 may
serve a critical role in OSCC progression under hypoxia (Fig. 1A). To further investigate the role of
SATB2 in OSCC progression, the expression of SATB2 was inhibited by
transfecting si-SATB2 into SCC9 cells under hypoxic conditions. The
western blotting results revealed that SATB2 expression was knocked
down by ~45% in si-SATB2-1-transfected cells and by ~80% in
si-SATB2-2-transfected cells, whereas HIF-1α protein levels
remained unaltered after SATB2 silencing (Fig. 1B). Therefore, the si-SATB2-2 sequence
was used for subsequent experiments.
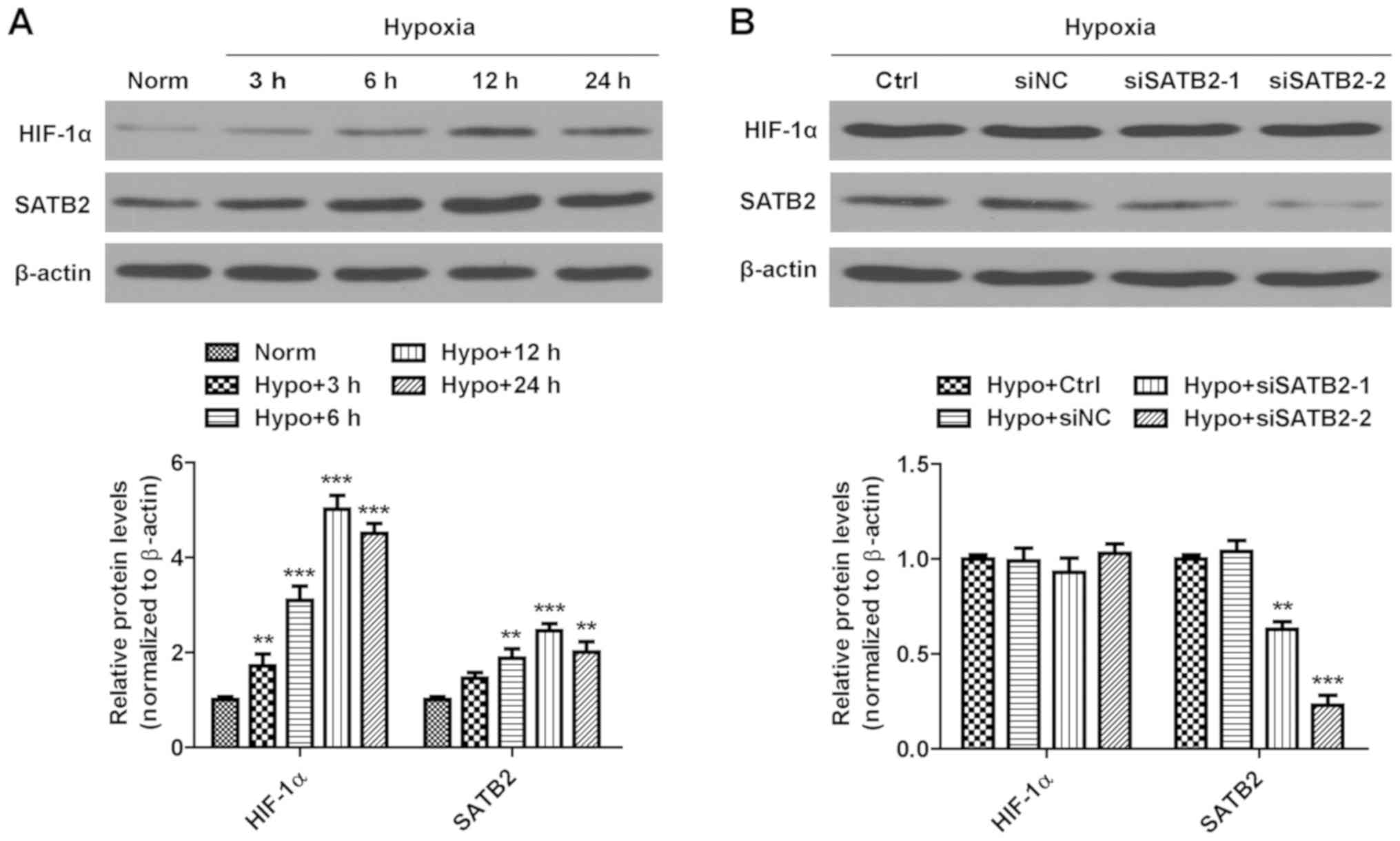 | Figure 1.Expression levels of HIF-1α and SATB2
in SCC9 cells. (A) The relative protein expression levels of HIF-1α
and SATB2 were examined by western blot analysis in SCC9 cells
exposed to normoxic or hypoxic conditions for the indicated times.
**P<0.01, ***P<0.001 vs. normoxia group. (B) The relative
protein expression levels of HIF-1α and SATB2 were detected by
western blot analysis in SCC9 cells transfected with si-NC,
siSATB2-1 and si-SATB2-2, and cultured under hypoxic condition for
24 h. Protein levels were normalized by comparison with β-actin
levels. The results are presented as the mean ± SD of three
independent experiments. **P<0.01, ***P<0.001 vs. hypoxia
control group. HIF-1α, hypoxia-inducible factor 1-α; SATB2, special
AT-rich sequence-binding protein 2; si, small interfering; NC,
negative control; norm, normoxia; hypo, hypoxic; ctrl, control. |
SATB2 knockdown suppresses
hypoxia-induced autophagy in SCC9 cells
To investigate the potential role of SATB2 in
hypoxia-induced autophagy, the expression levels of the autophagy
markers LC3-II/I and Beclin-1 were determined in hypoxia-treated
SCC9 cells. Western blotting revealed that the conversion of
endogenous LC3-I to LC3-II and the expression levels of Beclin-1
were significantly increased in SCC9 cells exposed to hypoxia
compared with cells under normoxia (Fig.
2A). By contrast, the genetic knockdown of SATB2 significantly
decreased the LC3-II/LC3-I ratio and expression levels of Beclin-1
in SCC9 cells compared with the control group under hypoxia
(Fig. 2B).
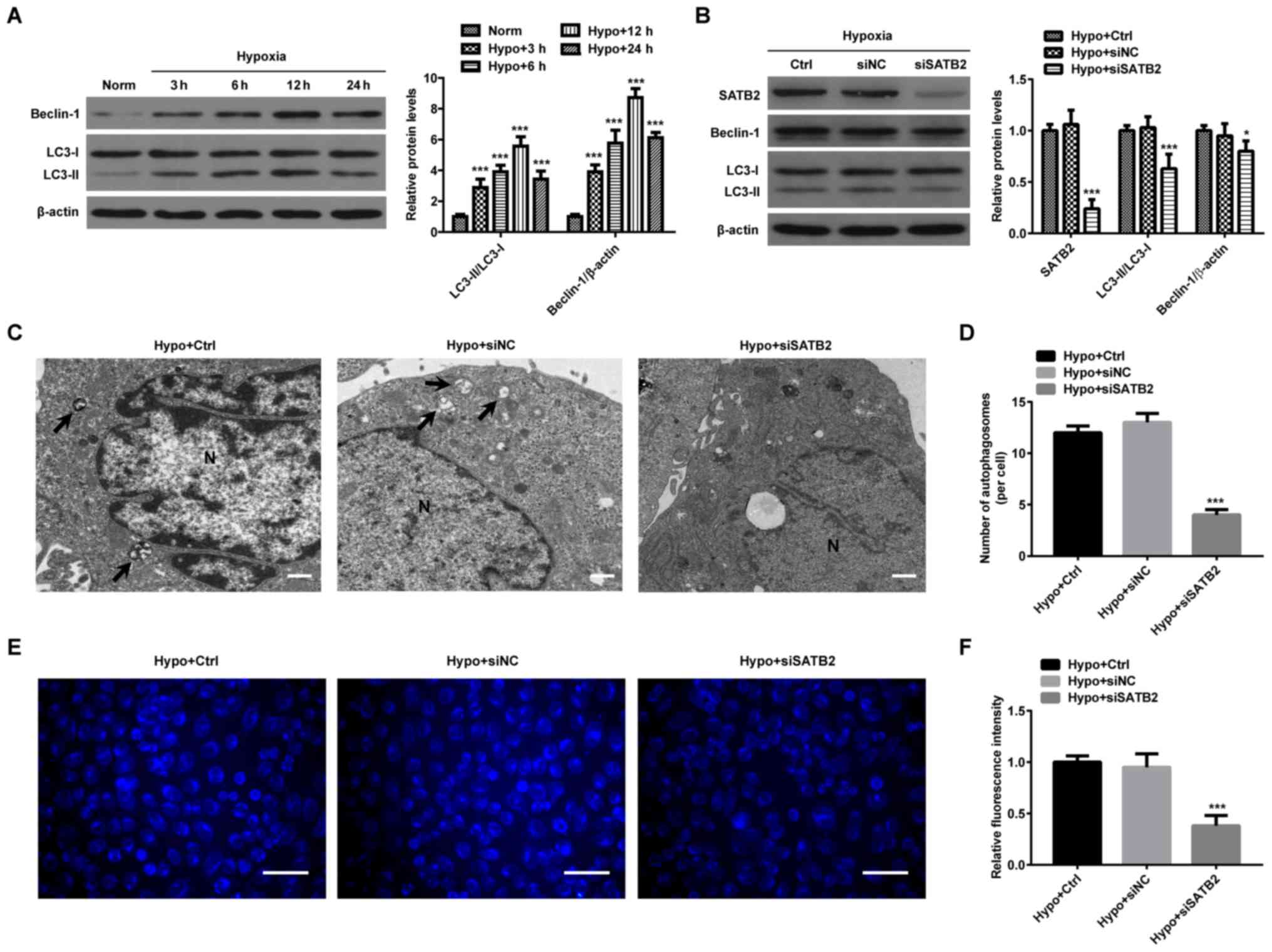 | Figure 2.SATB2 knockdown suppresses
hypoxia-induced autophagy in SCC9 cells. (A) The relative protein
expression levels of LC3-I, LC3-II and Beclin-1 were examined by
western blot analysis in SCC9 cells exposed to normoxic or hypoxic
conditions for the indicated times. Protein levels were normalized
by comparison with β-actin levels. ***P<0.001 vs. normoxia
group. (B) The relative protein expression levels of SATB2, LC3-I,
LC3-II and Beclin-1 were detected by western blot analysis in SCC9
cells transfected with either si-NC or si-SATB2-2, and cultured
under hypoxic conditions for 24 h. Protein levels were normalized
by comparison with β-actin levels. *P<0.05, ***P<0.001 vs.
hypoxia control group. (C) Representative TEM images of
autophagosomes in SCC9 cells transfected with either si-NC or
si-SATB2-2 and cultured under hypoxic conditions for 24 h. Black
arrows indicate autophagy vacuoles. N indicates nuclear. Scale
bar=1 µm. (D) The number of autophagosomes per cross-sectioned cell
was counted (20 cells per group). ***P<0.001 vs. hypoxia control
group. (E) Representative fluorescent images of MDC staining in
SCC9 cells transfected with either si-NC or si-SATB2-2 and cultured
under hypoxic conditions for 24 h. Scale bar=100 µm. (F) ImageJ was
used to calculate the relative fluorescence intensity of MDC. The
results are presented as the mean ± SD of three independent
experiments. ***P<0.001 vs. hypoxia control group. SATB2,
special AT-rich sequence-binding protein 2; LC3,
microtubule-associated protein light chain 3; si, small
interfering; NC, negative control; norm, normoxia; hypo, hypoxic;
ctrl, control; TEM, transmission electron microscopy; MDC,
monodansylcadaverine. |
To further identify the effect of SATB2 on
autophagy, TEM and MDC staining (a marker for autophagic vacuoles)
were also used. TEM observed that a greater number of
autophagosomes accumulated in hypoxic control cells compared with
si-SATB2-transfected cells (Fig. 2C and
D). Similarly, MDC staining demonstrated that the relative
fluorescence intensity of si-SATB2-transfected cells was
significantly decreased when compared with hypoxic control group
(Fig. 2E and F). Together, these
observations indicated that SATB2 may regulate autophagy in SCC9
cells under hypoxic conditions.
SATB2 knockdown suppresses
hypoxia-induced stemness of SCC9 cells
To investigate the effect of SATB2 on the
hypoxia-mediated stemness phenotype, the expression levels of the
stemness-related markers Oct-4, Sox-2 and Nanog were evaluated in
SCC9 cells during hypoxic and normoxic conditions. The protein
expression levels of Oct-4, Sox-2 and Nanog in hypoxic cells were
significantly increased compared with those in normoxic cells
(Fig. 3A). By contrast, knocking
down SATB2 decreased the expression levels of Oct-4, Sox-2 and
Nanog, as well as the colony-forming ratio of SCC9 cells (Fig. 3B and C). Moreover, following the
transfection of empty and SATB2-overexpression plasmids into SCC9
cells under normoxia, the western blotting results revealed that
SATB2 overexpression increased the protein expression levels of
stemness-related markers (Fig. 3D).
Taken together, the results indicated that the genetic silencing of
SATB2 may inhibit the hypoxia-mediated stemness phenotype in SCC9
cells.
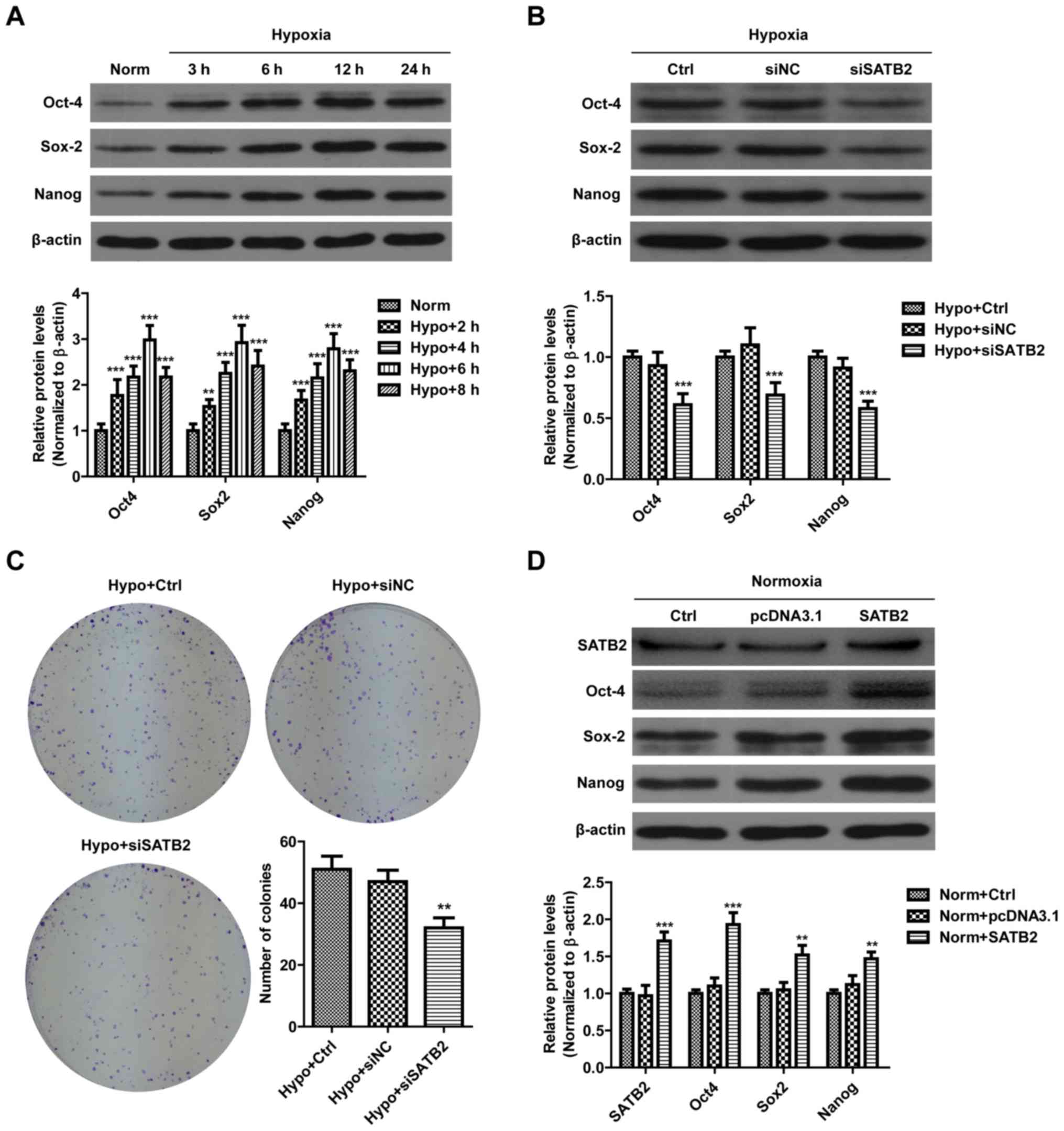 | Figure 3.SATB2 knockdown suppresses
hypoxia-induced stemness of SCC9 cells. (A) The relative protein
expression levels of Oct-4, Sox-2 and Naong were examined by
western blot analysis in SCC9 cells exposed to normoxic or hypoxic
conditions for the indicated times. Protein levels were normalized
by comparison with β-actin levels. **P<0.01, ***P<0.001 vs.
normoxia group. (B) Relative protein expression levels of Oct-4,
Sox-2 and Nanog were detected by western blot analysis in SCC9
cells transfected with either si-NC or si-SATB2-2 and cultured
under hypoxic conditions for 24 h. Protein levels were normalized
by comparison with β-actin levels. ***P<0.001 vs. hypoxia
control group. (C) Images of colonies stained with crystal violet,
and quantification of the number of colonies formed in SCC9 cells
transfected with either si-NC or si-SATB2-2 and cultured under
hypoxic conditions for 12 days. **P<0.01 vs. hypoxia control
group. (D) The relative protein expression levels of Oct-4, Sox-2
and Nanog were detected by western blot analysis in SCC9 cells
transfected with either empty (pcDNA3.1) or SATB2-overexpression
plasmids and cultured under normoxic conditions. **P<0.01,
***P<0.001 vs. normoxia group. The results are presented as the
mean ± SD of three independent experiments. SATB2, special AT-rich
sequence-binding protein 2; si, small interfering; NC, negative
control; norm, normoxia; hypo, hypoxic; ctrl, control. |
SATB2 knockdown inhibits the
proliferation while promoting the apoptosis of hypoxia-exposed SCC9
cells
To verify the effect of SATB2 on the proliferative
ability of SCC9 cells, both CCK-8 and cell cycle assays were
performed. The results from the CCK-8 assay demonstrated that the
knockdown of SATB2 significantly suppressed the proliferative
ability of SCC9 cells under hypoxia (Fig. 4A). Furthermore, as anticipated, cells
transfected with si-SATB2 induced G0/G1 cell cycle arrest, which
manifested through a markedly higher percentage of cells in the
G0/G1 phase and a lower percentage of cells in the S phase
(Fig. 4B and D). Subsequently,
Annexin V-FITC/PI assays were used to investigate the effects of
SATB2 knockdown on SCC9 cell apoptosis. The si-SATB2-transfected
group exhibited a significantly increased number of Annexin
V-positive cells compared with the control or si-NC group under
hypoxic conditions (Fig. 4C and E).
Overall, these findings indicated that SATB2 knockdown may inhibit
cell growth and induce cell apoptosis in hypoxic SCC9 cells.
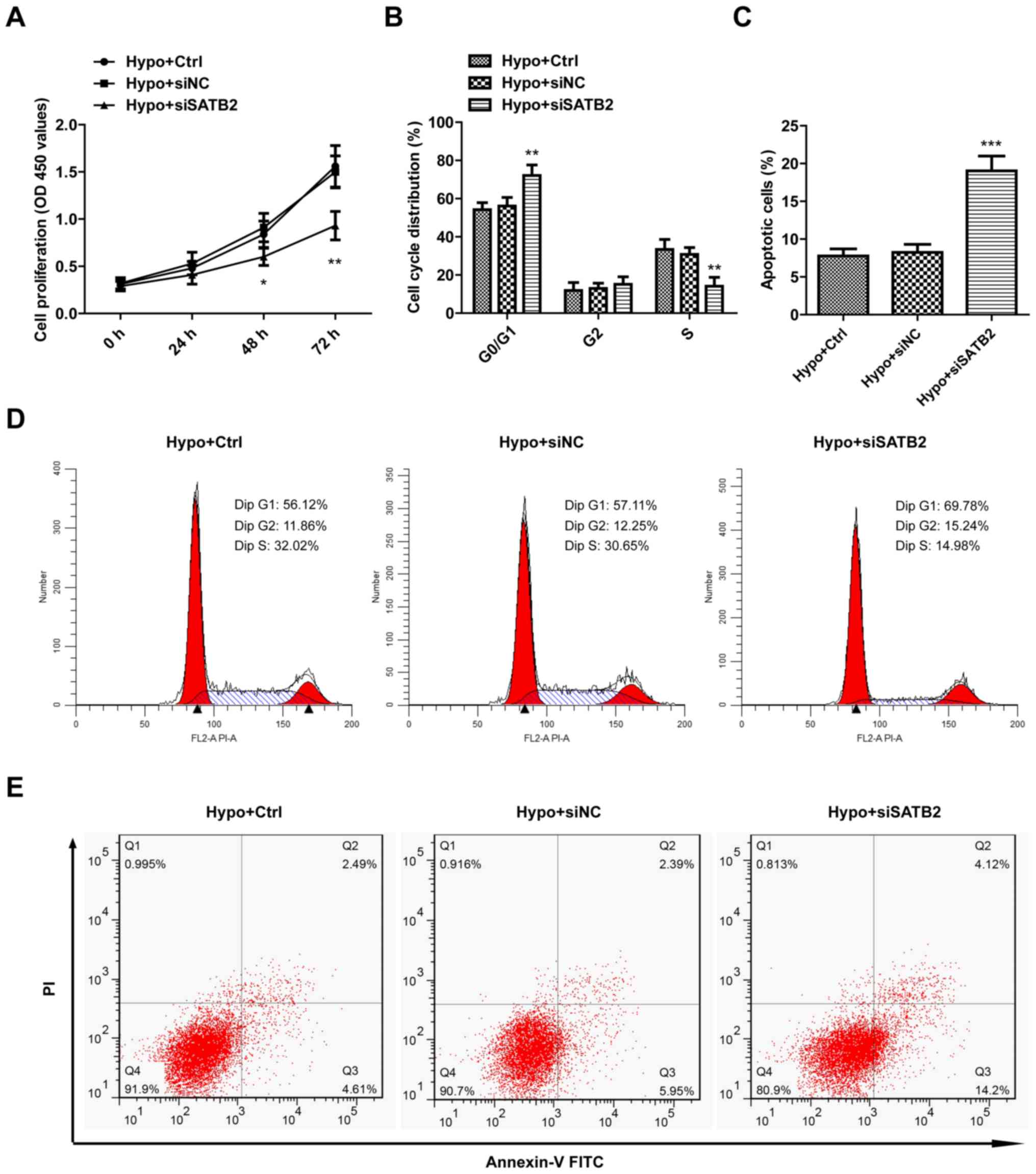 | Figure 4.SATB2 knockdown inhibits the
proliferation while promoting the apoptosis of hypoxia-exposed SCC9
cells. (A) The cell proliferation capacity was determined by CCK-8
assay in SCC9 cells transfected with either si-NC or si-SATB2-2,
and cultured under hypoxic conditions for 24, 48 and 72 h. (B and
D) Cell cycle was detected by flow cytometry with PI staining in
SCC9 cells transfected with either si-NC or si-SATB2-2 and cultured
under hypoxic conditions for 24 h. (C and E) Cell apoptosis was
detected by flow cytometry with Annexin V-FITC and PI double
staining in SCC9 cells transfected with either si-NC or si-SATB2-2
and cultured under hypoxic conditions for 24 h. The results are
presented as the mean ± SD of three independent experiments.
*P<0.05, **P<0.01, ***P<0.001 vs. hypoxia control group.
SATB2, special AT-rich sequence-binding protein 2; si, small
interfering; NC, negative control; hypo, hypoxic; ctrl, control;
PI, propidium iodide. |
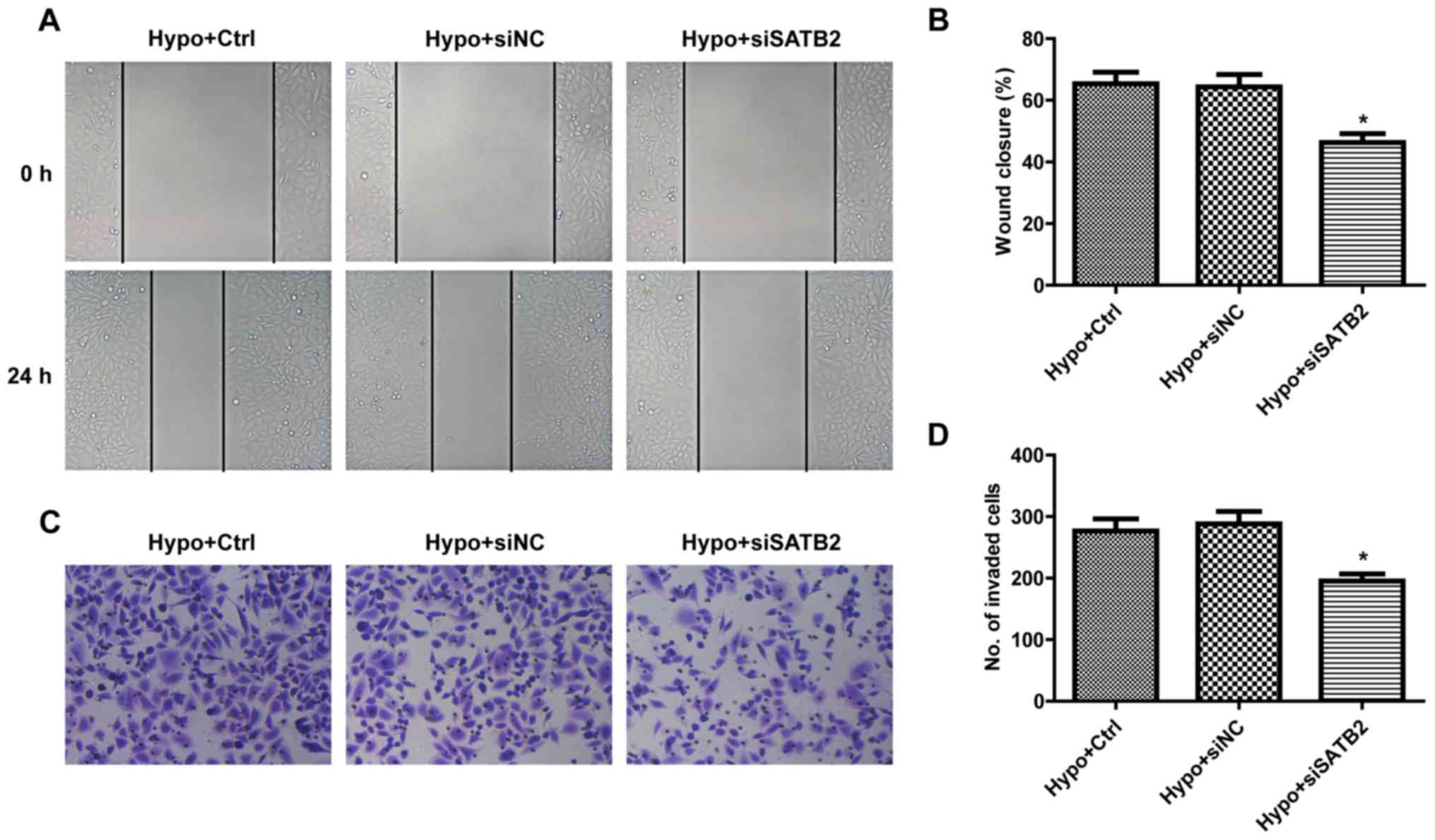
SATB2 knockdown inhibits the migration
and invasion of hypoxia-exposed SCC9 cells
To determine the role of SATB2 in SCC9 cell
migration under hypoxic conditions, wound healing assays were used
to measure the cell migratory ability. In SATB2 knockdown cells,
the migration rate was significantly reduced (Fig. 5A and B). The effect of SATB2 on cell
invasion was detected using Transwell assays. Compared with the
hypoxic control group, the number of invasive cells in the si-SATB2
group was significantly decreased (Fig.
5C and D). These data suggested that SATB2 silencing may
inhibit migratory and invasive processes in SCC9 cells under
hypoxic conditions.
Discussion
An increasing number of studies have observed that
SATB2 is aberrantly expressed in numerous types of malignant tumor,
including OSCC (6,7,14–16). For
example, Chung et al (16)
previously reported that SATB2 was preferentially expressed in
advanced-stage primary OSCC, and that the knockdown of SATB2
re-sensitized OSCC cells to chemotherapy-induced apoptosis.
However, the role of SATB2 in regulating autophagic and stemness
properties of cancer cells remains relatively unclear, and, to the
best of our knowledge, it has yet to be investigated in OSCC cells.
In the present study, the expression levels of SATB2 were
significantly increased in SCC9 cells under hypoxic conditions,
whereas the genetic silencing of SATB2 did not regulate the
expression of HIF-1α, suggesting that SATB2 is one of the
downstream molecules of HIF-1α. Moreover, SATB2 knockdown
suppressed the hypoxia-induced autophagy and stemness properties of
SCC9 cells, and consequently suppressed their proliferative,
migratory and invasive ability, while stimulating cell cycle arrest
and apoptosis in SCC9 cells under hypoxia. These findings suggested
that SATB2 may be a novel target for the treatment of OSCC.
Apoptosis and autophagy are two crucial processes
that maintain cellular homeostasis in physiological and
pathological conditions, in which crosstalk between the two
pathways can occur. Previously, hypoxia-induced autophagy was
demonstrated to promote tumor cell survival by eliminating
potentially toxic macromolecules and damaged organelles (17,18).
Moreover, several previous studies in OSCC have reported that the
inhibition of autophagy enhances apoptotic cell death, suggesting
that a combination treatment of anticancer drugs and autophagy
inhibitors may be an effective strategy for OSCC treatment
(19–21). In the present study, hypoxia-induced
classic hallmarks of autophagy in SCC9 cells were observed,
including accumulation of autophagosomes, conversion of LC3-I to
LC3-II and increased expression levels of Beclin-1. Moreover, the
knockdown of SATB2 using RNA interference was found to suppress
hypoxia-induced autophagy and promote apoptosis in SCC9 cells.
Overall, our findings indicate that SATB2 may inhibit cellular
apoptosis partially through promoting autophagy in OSCC.
It has been suggested that the acquisition of
stem-like properties by cancer cells markedly contributes to cancer
recurrence and poor prognosis (22,23).
With this in mind, it has been previously reported by Yu et
al (14) that the overexpression
of SATB2 in human pancreatic normal ductal epithelial cells
increased the expression levels of the stem cell markers CD44, CD24
and CD133, and the transcription factors Oct-4, Sox-2 and Nanog.
However, Li et al (24) found
that SATB2 directly bound to the regulatory elements of stem cell
markers such as CD133, CD44, meis homeobox 2 and axin 2, and
consequently inhibited the progression of colorectal cancer by
negatively regulating the stemness of colorectal cancer cells.
Therefore, the roles of SATB2 on the biological function of cancer
cells are dependent on the tumor cell line. Based on
loss-of-function experiments, the results of the current study were
consistent with those found by Yu et al (14); the present findings demonstrated that
the knockdown of SATB2 inhibited the expression of the
hypoxia-induced stemness factors Oct-4, Sox-2 and Nanog, in
addition to preventing colony formation, which suggested that the
stemness phenotype was inhibited following SATB2 knockdown. Due to
the strong association between epithelial-to-mesenchymal transition
(EMT) and stemness in OSCC cells (25), further studies are required to
investigate the effect of SATB2 on EMT processes.
In the present study, SATB2 knockdown was observed
to inhibit cell proliferation and facilitate cell cycle arrest,
demonstrating an increased G0/G1 phase fraction and a decreased S
phase fraction in SCC9 cells that were exposed to hypoxia.
Considering that cell cycle progression is one of the main causes
of the proliferation of tumor cells, multiple studies have
suggested that cell cycle-associated genes may be primary targets
for cancer treatment (26,27). In addition, a significant decrease in
both the cellular migratory and invasive ability was observed
following SATB2 gene silencing. Thus, to the best of our knowledge,
this is the first study to suggest that SATB2 may exert multiple
biological functions in OSCC.
In conclusion, the present study demonstrated that
SATB2 may serve as an oncogene in SCC9 cells by accelerating
autophagy and stemness processes. The silencing of SATB2 gene
expression inhibited the proliferative, migratory and invasive
ability of OSCC cells, which are all important processes that are
required for OSCC progression. In future studies, OSCC animal
models should be used to further investigate the function of SATB2
and its relevance in OSCC.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the
Science and Technology Plan Project of Jiaxing, Zhejiang, China
(grant no. 2016BY28004) and the Medical and Health Science and
Technology Project of Zhejiang Province, China (grant no.
2017196633).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WD and CW designed the study and wrote the initial
draft of the article. YC, NQ, GS, JZ and ZG contributed to analysis
and interpretation of data and assisted in the preparation of the
article. WD, YC, NQ and GS performed the experiments. All authors
contributed to data collection and interpretation, and critically
reviewed the article. All authors read and approved the final
version of the manuscript and agree to be accountable for all
aspects of the work in ensuring that questions related to the
accuracy or integrity of any part of the work are appropriately
investigated and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Krishna Rao SV, Mejia G, Roberts-Thomson K
and Logan R: Epidemiology of oral cancer in Asia in the past
decade-an update (2000–2012). Asian Pac J Cancer Prev.
14:5567–5577. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ausoni S, Boscolo-Rizzo P, Singh B, Da
Mosto MC, Spinato G, Tirelli G, Spinato R and Azzarello G:
Targeting cellular and molecular drivers of head and neck squamous
cell carcinoma: Current options and emerging perspectives. Cancer
Metastasis Rev. 35:413–426. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zanoni DK, Montero PH, Migliacci JC, Shah
JP, Wong RJ, Ganly I and Patel SG: Survival outcomes after
treatment of cancer of the oral cavity (1985–2015). Oral Oncol.
90:115–121. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dobreva G, Dambacher J and Grosschedl R:
SUMO modification of a novel MAR-binding protein, SATB2, modulates
immunoglobulin mu gene expression. Genes Dev. 17:3048–3061. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xu HY, Fang W, Huang ZW, Lu JC, Wang YQ,
Tang QL, Song GH, Kang Y, Zhu XJ, Zou CY, et al: Metformin reduces
SATB2-mediated osteosarcoma stem cell-like phenotype and tumor
growth via inhibition of N-cadherin/NF-κB signaling. Eur Rev Med
Pharmacol Sci. 21:4516–4528. 2017.PubMed/NCBI
|
|
7
|
Yu W, Ma Y, Shankar S and Srivastava RK:
SATB2/β-catenin/TCF-LEF pathway induces cellular transformation by
generating cancer stem cells in colorectal cancer. Sci Rep.
7:109392017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Joseph JP, Harishankar MK, Pillai AA and
Devi A: Hypoxia induced EMT: A review on the mechanism of tumor
progression and metastasis in OSCC. Oral Oncol. 80:23–32. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pérez-Sayáns M, Suárez-Peñaranda JM, Pilar
GD, Barros-Angueira F, Gándara-Rey JM and García-García A:
Hypoxia-inducible factors in OSCC. Cancer Lett. 313:1–8. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Daskalaki I, Gkikas I and Tavernarakis N:
Hypoxia and selective autophagy in cancer development and therapy.
Front Cell Dev Biol. 6:1042018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vadde R, Vemula S, Jinka R, Merchant N,
Bramhachari PV and Nagaraju GP: Role of hypoxia-inducible factors
(HIF) in the maintenance of stemness and malignancy of colorectal
cancer. Crit Rev Oncol Hematol. 113:22–27. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dong W, Zhang P, Fu Y, Ge J, Cheng J, Yuan
H and Jiang H: Roles of SATB2 in site-specific stemness, autophagy
and senescence of bone marrow mesenchymal stem cells. J Cell
Physiol. 230:680–690. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mizushima N: Methods for monitoring
autophagy. Int J Biochem Cell Biol. 36:2491–2502. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yu W, Ma Y, Shankar S and Srivastava RK:
Role of SATB2 in human pancreatic cancer: Implications in
transformation and a promising biomarker. Oncotarget.
7:57783–57797. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fukuhara M, Agnarsdóttir M, Edqvist PH,
Coter A and Ponten F: SATB2 is expressed in Merkel cell carcinoma.
Arch Dermatol Res. 308:449–454. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chung J, Lau J, Cheng LS, Grant RI,
Robinson F, Ketela T, Reis PP, Roche O, Kamel-Reid S, Moffat J, et
al: SATB2 augments ΔNp63α in head and neck squamous cell carcinoma.
EMBO Rep. 11:777–783. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song S, Tan J, Miao Y, Li M and Zhang Q:
Crosstalk of autophagy and apoptosis: Involvement of the dual role
of autophagy under ER stress. J Cell Physiol. 232:2977–2984. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Thorburn A: Apoptosis and autophagy:
Regulatory connections between two supposedly different processes.
Apoptosis. 13:1–9. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Park BS, Choi NE, Lee JH, Kang HM, Yu SB,
Kim HJ, Kang HK and Kim IR: Crosstalk between fisetin-induced
apoptosis and autophagy in human oral squamous cell carcinoma. J
Cancer. 10:138–146. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang X, Liu W, Wang P and Li S: RNA
interference of long noncoding RNA HOTAIR suppresses autophagy and
promotes apoptosis and sensitivity to cisplatin in oral squamous
cell carcinoma. J Oral Pathol Med. 47:930–937. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sophia J, Kowshik J, Dwivedi A, Bhutia SK,
Manavathi B, Mishra R and Nagini S: Nimbolide, a neem limonoid
inhibits cytoprotective autophagy to activate apoptosis via
modulation of the PI3K/Akt/GSK-3β signalling pathway in oral
cancer. Cell Death Dis. 9:10872018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee S and Schmitt CA: The dynamic nature
of senescence in cancer. Nat Cell Biol. 21:94–101. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lathia JD and Liu H: Overview of cancer
stem cells and stemness for community oncologists. Target Oncol.
12:387–399. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li Y, Liu YH, Hu YY, Chen L and Li JM:
Special AT-rich sequence-binding protein 2 acts as a negative
regulator of stemness in colorectal cancer cells. World J
Gastroenterol. 22:8528–8539. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu LF, Hu Y, Yang CC, Xu XH, Ning TY,
Wang ZL, Ye JH and Liu LK: Snail overexpression induces an
epithelial to mesenchymal transition and cancer stem cell-like
properties in SCC9 cells. Lab Invest. 92:744–752. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mills CC, Kolb EA and Sampson VB:
Development of chemotherapy with cell-cycle inhibitors for adult
and pediatric cancer therapy. Cancer Res. 78:320–325. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Molinari M: Cell cycle checkpoints and
their inactivation in human cancer. Cell Prolif. 33:261–274. 2000.
View Article : Google Scholar : PubMed/NCBI
|