Introduction
At present, ovarian cancer has the third highest
incidence rate (3%) and the highest mortality rate (5%) worldwide
among gynaecological malignancies (1). Epithelial ovarian cancer (EOC) accounts
for >95% of ovarian malignancies and >50% of the patients
with ovarian cancer are not diagnosed at an early stage.
Furthermore, the recurrence rate of EOC is high (>80%) (2), which provides an explanation for the
high mortality rate associated with the disease (3). Moreover, the mechanisms underlying the
origin, pathogenesis and metastasis of EOC are not completely
understood, which makes early diagnosis difficult.
Long non-coding (lnc)RNAs are non-coding RNAs with
transcripts >200 nucleotides in length. lncRNAs are widely
distributed in a number of tissues, for example brain, lung, heart
and ovaries, and their expression is often tissue- and
time-specific (4,5). Although the functions of a number of
lncRNAs are unclear, it has been reported that they are involved
either directly or in the regulation of development, cell
differentiation, metabolism and other biological processes
(6). lncRNAs can regulate gene
expression at the epigenetic, transcriptional and
post-transcriptional levels, which is closely associated with the
occurrence, development and prevention of human diseases (6–8).
Recently, an increasing number of studies have detected significant
differences in the expression levels of lncRNAs between normal and
tumour tissues (9–11). For example, brain cytoplasmic RNA1 is
highly expressed during breast, lung, tongue and ovarian cancer,
and HOX transcript antisense RNA expression is significantly higher
in a number of tumour tissues compared with normal tissues
(12).
MicroRNAs (miRNAs) are small non-coding RNA
molecules, 18–22 nucleotides in length, that are present in
eukaryotic cells. miRNAs bind to the 3′ or 5′ untranslated region
(UTR) of specific target genes, by complete or incomplete
complementary base pairing, in order to inhibit the translation of,
or directly degrade the target mRNA, which ultimately affects
downstream signalling. miRNA expression is important during cell
proliferation, differentiation, apoptosis and autophagy (13). Several studies have demonstrated that
lncRNAs can act as competitive endogenous (ce)RNAs to regulate the
aggregation and biological function of miRNAs (14), thereby affecting mRNA expression
(15,16). Bioinformatics analysis can clarify
the regulatory mechanisms underlying lncRNAs by predicting the
miRNAs that they compete for. Therefore, bioinformatics analysis
can provide a theoretical basis for identifying novel markers for
the early detection, timely diagnosis and clinical intervention of
ovarian cancer (17).
A previous study reported that inorganic
pyrophosphate (PPA1) is closely associated with the
occurrence and development of ovarian cancer (18). Luo et al (19) demonstrated that PPA1 is
associated with several genes [heat shock protein family B (small)
member 1 (HSPB1), tumour protein p53 (TP53), unc-119
lipid binding chaperone (UNC119), small ubiquitin-like
modifier 4 (SUMO4) and SET domain bifurcated histone lysine
methyltransferase 1 (SETDB1)] in ovarian cancer cells.
HSPB1 (20), TP53
(21) and UNC119 (22) are closely related to cell
proliferation and apoptosis, whereas SUMO4 (23) and SETDB1 (24) are associated with regulating
transcriptional activity. Therefore, the aim of the present study
was to compare the lncRNA expression profiles of five ovarian
cancer cell lines and an ovarian epithelial cell line, in order to
identify differentially expressed lncRNAs and their associated
miRNAs.
Materials and methods
Cell culture
The human EOC IGROV1, A2780, SKOV3, ES2 and Hey cell
lines were purchased from the American Type Culture Collection. The
human ovarian epithelial IOSE80 cell line was obtained from Wuxi
Innovate Biomedical Technology Company (www.innovatbio.com/). All cell lines were cultured in
RPMI-1640 medium (Corning, Inc.) supplemented with 10% foetal
bovine serum (Gibco; Thermo Fisher Scientific, Inc.) and 1%
penicillin/streptomycin at 37°C with 5% CO2.
RNA extraction and lncRNA microarray
assay
Total RNA was extracted from EOC and IOSE80 cells
using TRIzol® (Invitrogen; Thermo Fisher Scientific,
Inc.) and quantified using a NanoDrop ND-2000 spectrophotometer
(Thermo Fisher Scientific, Inc.). RNA integrity was assessed using
the Agilent Bioanalyzer 2100 (Agilent Technologies, Inc.). Total
RNA was reverse transcribed into double-stranded cDNAs (ds-cDNAs)
using a Quick Amp Labeling kit (p/n 5190-0442; Agilent
Technologies, Inc.) according to the manufacturer's protocol and
oligo dT primers at 40°C for 2 h and 65°C for 15 min. The ds-cDNAs
were then labelled with Cy3 and hybridized onto the Human lncRNA
Microarray (version 4.0; Shanghai Kangcheng Biological Engineering
Co., Ltd.), which contains probes for 40,173 lncRNAs, as previously
described (25). Following
hybridization, the microarray slides were washed using Gene
Expression Wash Buffer 1 (p/n 5188–5325; Agilent Technologies,
Inc.) and Gene Expression Wash Buffer 2 (p/n 5188–5326; Agilent
Technologies, Inc.), scanned using the Agilent Microarray Scanner
(p/n G2565BA; Agilent Technologies, Inc.), and analysed using
Agilent Feature Extraction software (version 11.0.1.1; Agilent
Technologies, Inc.). Quantile normalization and subsequent data
processing were performed using GeneSpring GX software (version
12.2; Agilent Technologies, Inc). Subsequently, lncRNAs and mRNAs
were selected for further data analysis. Differentially expressed
lncRNAs and mRNAs with statistical significance between the EOC and
IOSE90 cell lines were identified by P<0.05 and |fold change|
>2.
Reverse transcription-quantitative PCR
(RT-qPCR)
qPCR was performed using the SYBR Green RT-PCR
Master mix (Applied Biosystems; Thermo Fisher Scientific, Inc.).
The following thermocycling conditions were used for reverse
transcription: 5 min at 37°C, 60 min at 42°C, and 10 min at 70°C.
Samples were stored at −20°C until further analysis. The following
thermocycling conditions were used for qPCR: Initial denaturation
at 95°C for 10 min, followed by 40 cycles at 95°C for 10 sec and a
final extension at 60°C for 60 sec. The following primer pairs
(Takara Bio, Inc.) were used for qPCR: Neuropeptides B and W
receptor 1–2 (NPBWR1-2) forward, 5′-TTTTCATTTTTATGTATGGGCA-3′ and
NPBWR1-2 reverse, 5′-ACAACAGAACTCGTTTTAAGTTAC-3′; and β-actin
forward, 5′-GTGGCCGAGGACTTTGATTG3′ and β-actin reverse,
5′-CCTGTAACAACGCATCTCATATT-3′. lncRNA expression levels were
quantified using the 2−ΔΔCq method (26) and normalized to the internal
reference gene β-actin. qPCR was performed in triplicate.
Cell transfection
To induce NPBWR1-2 overexpression, five ovarian
cancer cell lines were transfected with an NPBWR1-2 expression
vector (lnc-NPBWR1-2; Genewiz, Inc.) or with a pcDNA3.1 negative
control (NC) empty vector (Tianjin Saierbio, Inc.). Cells were
plated in 6-well plates (3×105 cells/well) and incubated
overnight at 37°C. Cells (~1.75×105 cells)with the
plasmids (4 µg) were transfected using Lipofectamine®
2000 (Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol. Following transfection for 48 h, cells
were collected and prepared for further experiments.
Cell viability assay
To determine cell viability, cells were examined
using an MTT assay. Cells were seeded (1×103 cells/well;
IGROV1, A2780, SKOV3, ES2, Hey cells and IGROV1, A2780, SKOV3, ES2,
Hey/lnc-NPBWR1-2 cells) into 96-well plates. SKOV3 cell viability
was assessed at different time points (24, 48 and 72 h) and others
was assessed at 48 h using the MTT assay. A total of 100 µl DMSO
was used to dissolve the purple formazan in each well. The optical
density of each well, which represented cell proliferation, was
measured daily for four consecutive days at a wavelength of 570 nm
to estimate the number of viable cells at each time point. The cell
viability rate (%)=experimental group A570 mean value/control group
A570 mean value ×100%.
Cell proliferation assay
Cells were trypsinised using 0.25% trypsin and
resuspended in RPMI-1640 medium (Gibco; Thermo Fisher Scientific,
Inc.) with 10% FBS, seeded 200 cells into 12-well plates and
cultured in a humidified atmosphere containing 5% CO2 at
37°C for 7–14 days. Following incubation, cell colonies were washed
with PBS, fixed with 4% methanol at 4°C for 30 min, and stained
with 0.1% crystal violet (1 mg/ml) for 20 min at room temperature.
Colonies containing >50 cells were counted, and the mean colony
number was calculated.
Cell migration and invasion
assays
Cell migration and invasion were assessed using
Transwell assays. To assess cell invasion, matrigel was dissolved
and plated into each well prior to seeding cells and medium into
the Transwell plates for 1 h at room temperature. Cells were
trypsinised using 0.25% trypsin and subsequently plated
(1×105) in 100 µl serum-free RPMI-1640 medium into the
upper chambers of the Transwell plates. RPMI-1640 medium
supplemented with 20% FBS (500 µl) was plated into the lower
chambers of the Transwell plates. Following incubation for 48
(migration assay) or 72 h (invasion assay), cells on the lower
surface of the Transwell membrane were fixed with 75% methanol [a
mixture of methanol and glacial acetic acid (3:1)] for 30 min at
room temperature, and stained with 0.1% crystal violet for 15 min
at room temperature. Stained cells were counted in three randomly
selected fields of view using an fluorescence inverted microscope
at ×200 magnification.
Cell apoptosis assays
The relative number of apoptotic cells was measured
using an Annexin V-FITC/Propidium Iodide (PI) Apoptosis Detection
kit according to the manufacturer's protocol (Shanghai Kaifeng
Biotechnology). Briefly, cells were seeded (1×105
cells/well) into 6-well plates, 200 µl Annexin V followed by 10 µl
PI was added to each well and cells were incubated for 10–20 min at
room temperature in the dark. Subsequently, cells were washed twice
with cold PBS. Early apoptotic cells were detected and analysed
using a BD LSRFortessa™ flow cytometer (Becton, Dickinson and
Company). Data were analysed using BD FACSDiva software (version
6.0; Becton, Dickinson and Company).
Western blotting
Cells were washed with PBS. Total protein was
extracted using ProteoJET Mammalian Cell Lysis Reagent (Fermentas;
Thermo Fisher Scientific, Inc.), according to the manufacturer's
instructions. Protein levels were determined using a BCA protein
assay kit (Pierce; Thermo Fisher Scientific, Inc.). Subsequently,
proteins (20 µg) were separated via 10% gel by SDS-PAGE and
transferred onto PVDF membranes. The membranes were blocked with
0.05% Starting Block Blocking buffers (Thermo Fisher Scientific,
Inc.) for 10 min at room temperature. Subsequently, the membranes
were incubated at 4°C overnight with primary antibodies targeted
against: IGFBP-7 (cat. no. GTX31152) and GAPDH (cat. no. GTX100118)
(both 1:1,000; both from GeneTex, Inc.). Following primary
incubation, the membranes were washed four times using TBS with
0.1% Tween-20. Membranes were then incubated with goat anti-rabbit
IgG (1:10,000; cat. no. ab205718; Abcam) secondary antibodies for
1.5 h at room temperature. Protein bands were visualized using
Western Lightning™ Chemiluminesence Reagent (PerkinElmer, Inc.) and
photographed using a LabWorks™ gel imaging and analysis system
(Analytik Jena AG). GAPDH was used as the loading control.
Bioinformatics analysis
The microRNA Target Prediction Database (miRDB)
website (mirdb.org/index.html; human) was used
to integrate the lncRNA/miRNA and miRNA-target networks, and
predict miRNA/lncRNA relationships.
Statistical analysis
Data are expressed as the mean ± standard deviation
(unless otherwise shown). Differences in the expression levels of
NPBWR1-2 among the different experimental groups were analysed
using an unpaired Student's t-test. In cell proliferation,
migration, invasion and apoptosis assays, the SKOV3/NC group was
set at 1. Statistical analyses were performed using SPSS software
(version 17.0; SPSS, Inc.). P<0.05 was considered to indicate a
statistically significant difference.
Results
lncRNA microarray analysis
In order to identify differentially expressed
lncRNAs between EOC and IOSE80 cells, the lncRNA expression
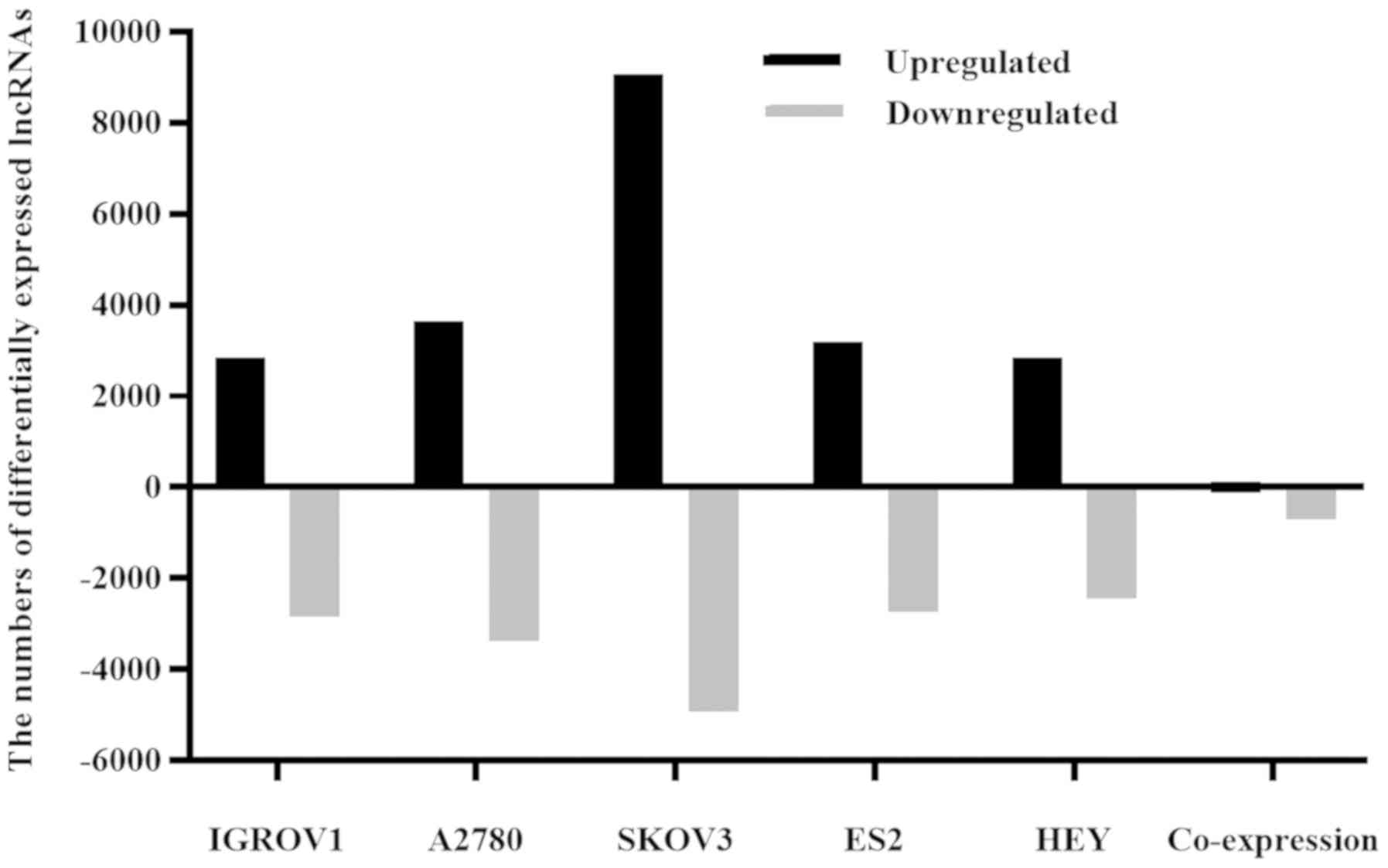
profiles of the two cell types were compared (Fig. 1). Collectively, the five EOC cell
lines displayed 110 upregulated and 699 downregulated lncRNAs,
suggesting that these lncRNAs were associated with EOC formation
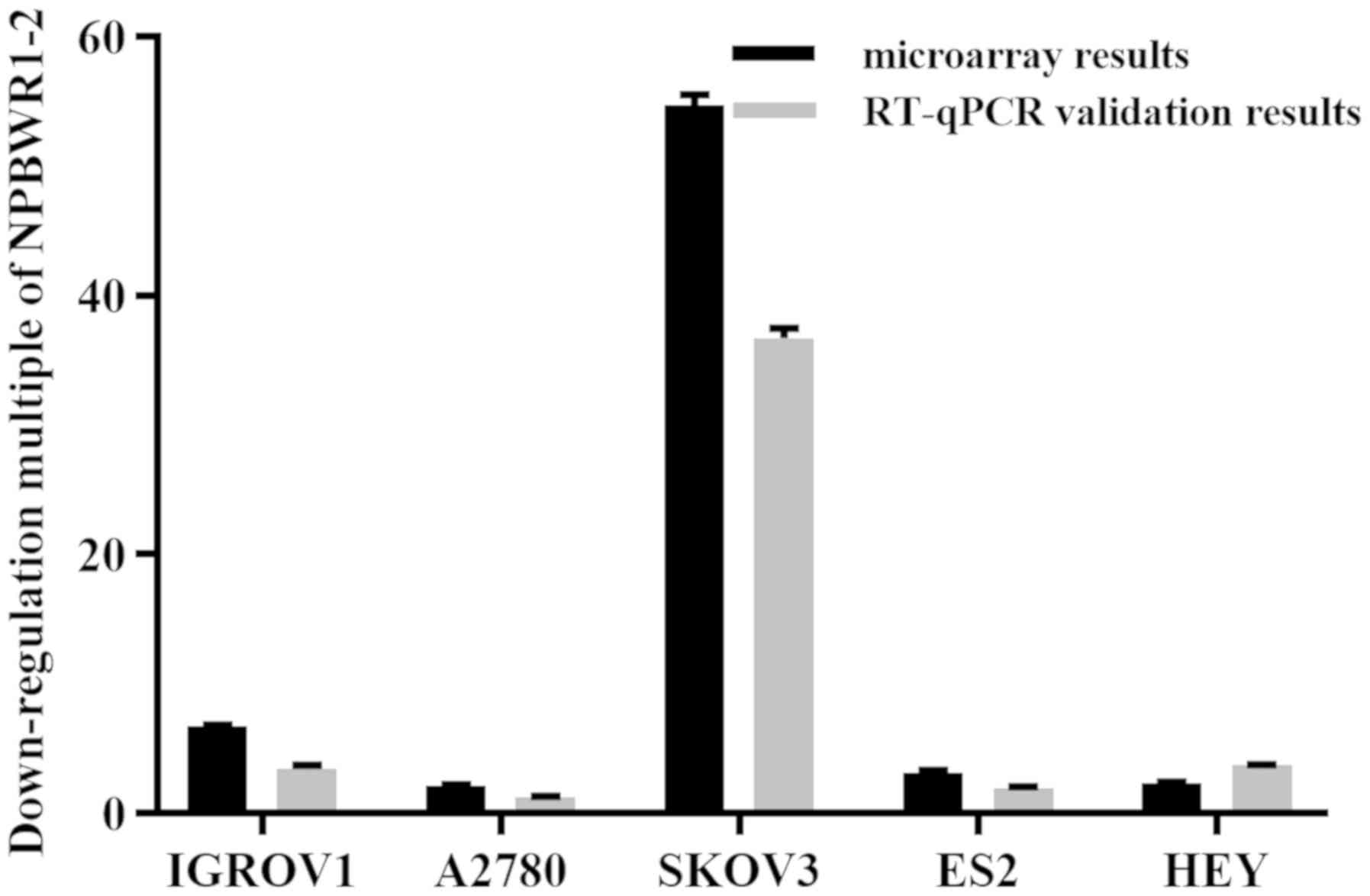
and development. The expression profile of lncRNA NPBWR1-2 was
assessed using RT-qPCR and microarray assays. Compared with IOSE80
cells, NPBWR1-2 was downregulated by more than two-fold in all five
ovarian cancer cell lines, according to the microarray results. The
RT-qPCR results were consistent with the microarray results
(Fig. 2).
NPBWR1-2 overexpression decreases cell
viability, inhibits proliferation, migration and invasion, and
promotes apoptosis
SKOV3 is a serous adenocarcinoma cell line, which
displays one of the highest incidence rates of ovarian cancer
(27). Among the five ovarian cancer
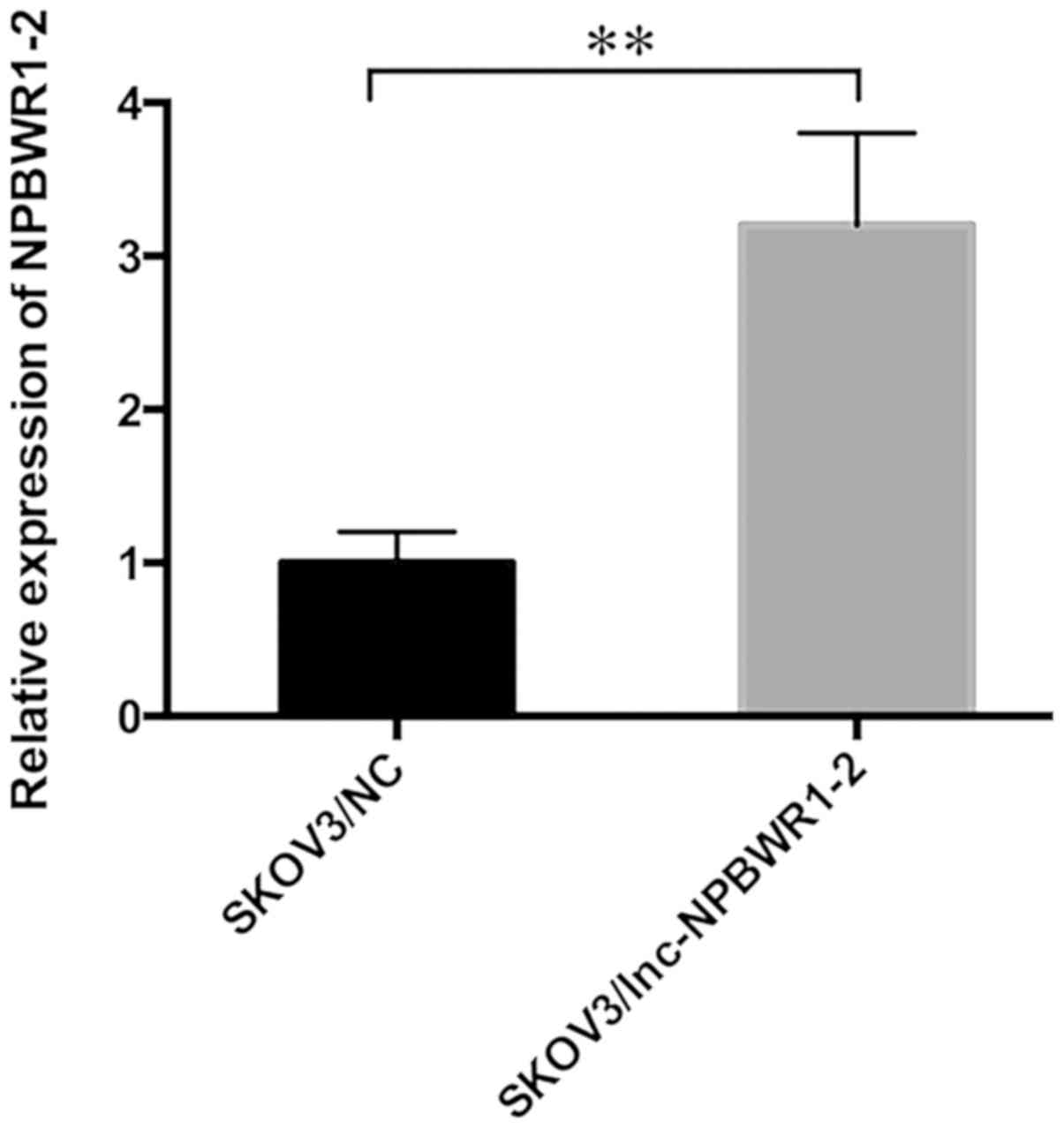
cell lines, the expression levels of NPBWR1-2 were highest in the
SKOV3 cell line; therefore, SKOV3 cells were chosen for subsequent
experiments (Fig. 2). The expression
level of NPBWR1-2 was significantly upregulated in the
SKOV3/lnc-NPBWR1 group compared with the SKOV3/NC group, as
determined by RT-qPCR (Fig. 3).
Similar results were obtained for NPBWR1-2 expression in the other
four ovarian cancer cell lines (Fig.
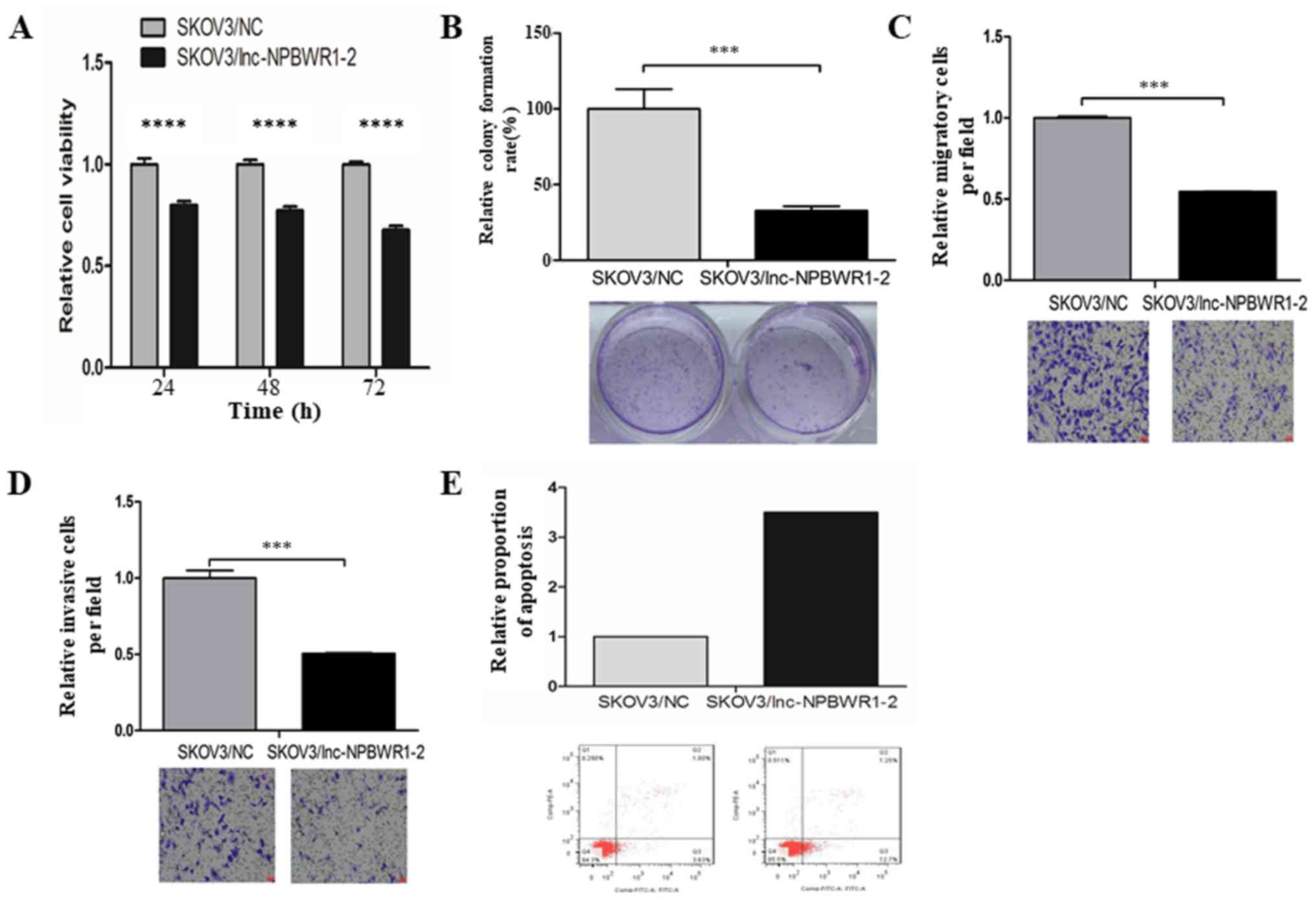
S1). Cell viability was significantly decreased in the
lnc-NPBWR1-2 group compared with the NC group for each of the five
cell lines (IGROV1, P<0.0001; A2780, P<0.001; SKOV3,
P<0.0001; ES2, P<0.0001; and Hey, P<0.0001, Fig. S2). The results of the cell viability
assay in SKOV3 cells are displayed in Fig. 4A. The mean OD570 values of
the SKOV3/NC and SKOV3/lnc-NPBWR1-2 groups were 0.328±0.018 and
0.41±0.030, 0.431±0.019 and 0.557±0.021, and 0.590±0.019 and
0.87±0.011 at 24, 48 and 72 h, respectively (P<0.0001; Fig. 4A). Cell proliferation was
significantly decreased in the SKOV3/lnc-NPBWR1-2 group compared
with the SKOV3/NC group (P<0.001; Fig. 4B), indicating that NPBWR1-2
overexpression inhibited SKOV3 cell proliferation in vitro.
The mean number of migratory cells in the SKOV3/lnc-NPBWR1-2 and
SKOV3/NC groups was 249.667±25.541 and 457.333±4.933, respectively
(P<0.001; Fig. 4C). The number of
invading cells was significantly lower in the SKOV3/lnc-NPBWR1-2
group compared with the SKOV3/NC group (P<0.001; Fig. 4D). Furthermore, the relative number
of apoptotic cells was increased in the SKOV3/lnc-NPBWR1-2 group
compared with the SKOV3/NC group (Fig.
4E).
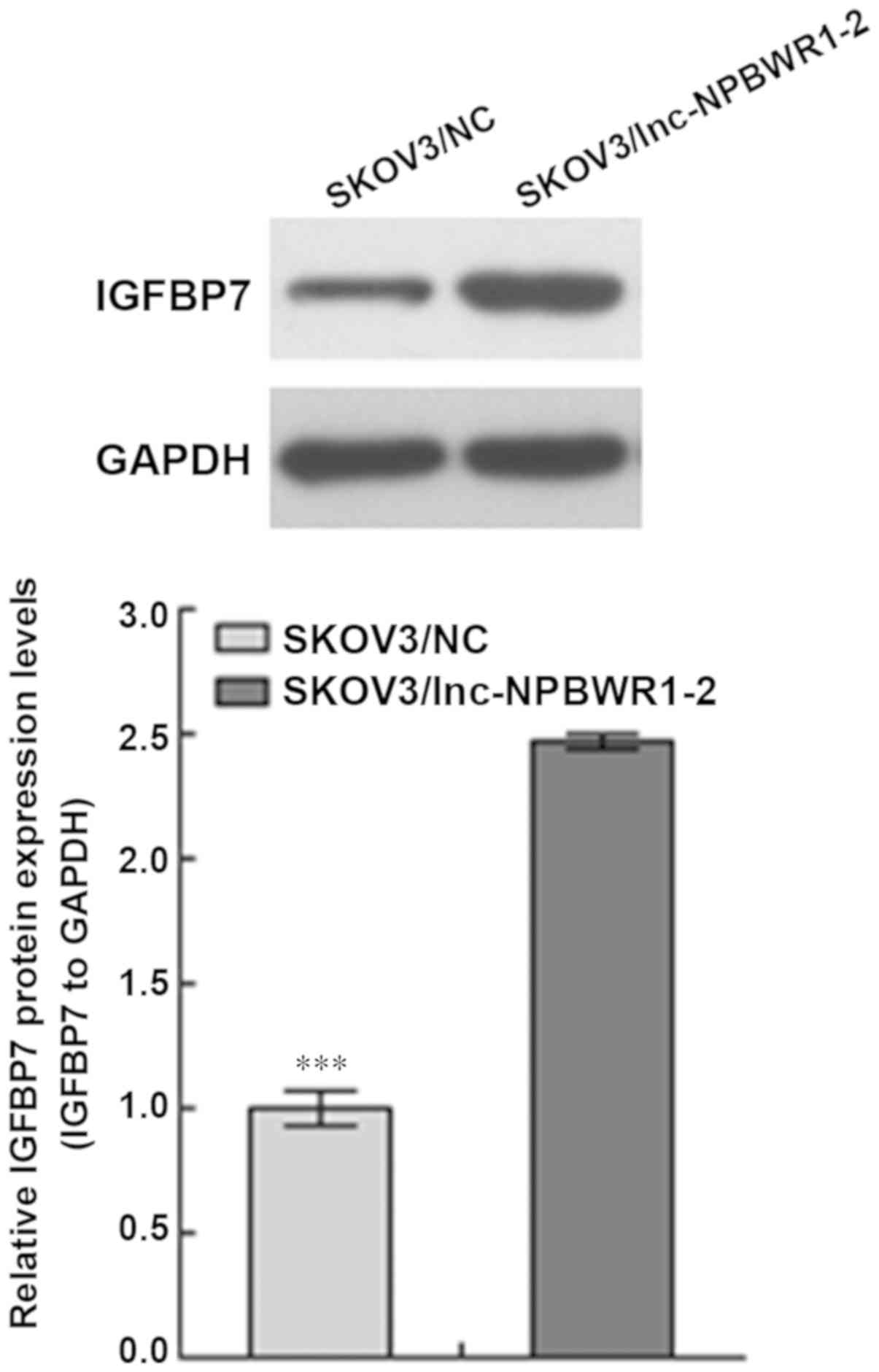
NPBWR1-2 overexpression affects IGFBP7
expression
The expression level of IGFBP7 in the SKOV3/NC group
was significantly lower compared with the SKOV3/lnc-NPBWR1-2 group
(P<0.001; Fig. 5).
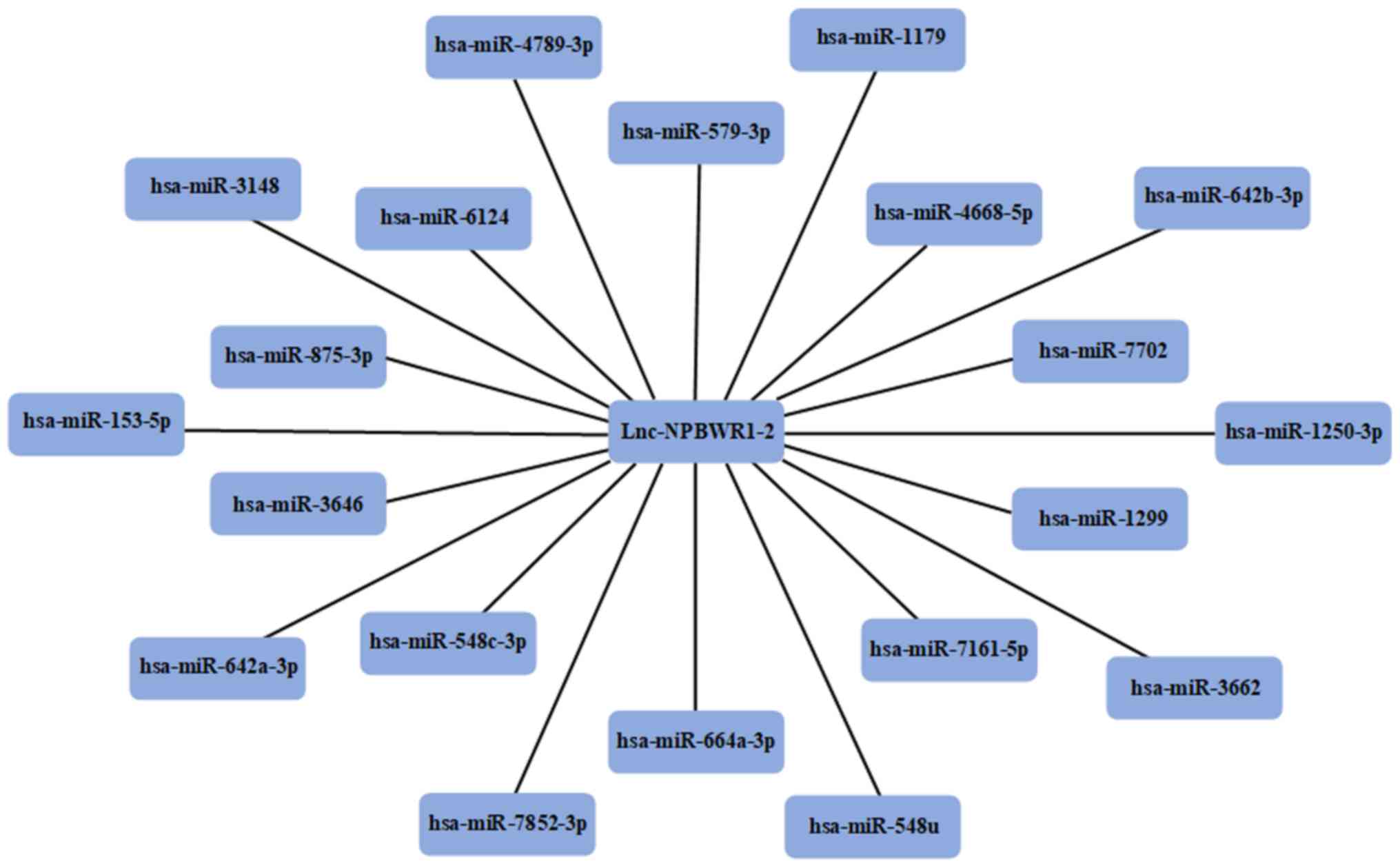
miRNA prediction
Using the miRDB, the miRNAs that competed with
lncRNA NPBWR1-2 were predicted. A total of 20 miRNAs were
identified as potential targets for NPBWR1-2 (Fig. 6).
Discussion
Recently, an increasing number of studies
investigating the roles of lncRNAs associated with cancer, immune
signalling and the maintenance of stem cell biological
characteristics have been conducted (12,28,29).
lncRNAs display ‘one-to-many’ and ‘many-to-one’ regulatory
functions and can regulate gene expression at multiple levels,
including epigenetic, transcriptional and post-transcriptional
levels (30). Previous studies have
indicated that abnormal lncRNA expression is associated with tumour
development, recurrence and metastasis (31–33). In
clinical practice, patients administered with the same treatment
often display different clinical responses, which may be explained
in part by the differential expression of lncRNAs among patients
(34). lncRNAs also function as
competitive endogenous RNAs to regulate miRNA expression; however,
the association between lncRNAs and miRNAs in ovarian cancer is not
completely understood (35).
In patients with ovarian cancer, it is rarely
possible to identify the histological type before surgery, and the
lack of effective available biomarkers is a challenge for the
detection and diagnosis of early-stage EOC without obvious symptoms
(3,36). By identifying a marker that is
sensitive to all types of EOC, ovarian cancer could be detected and
diagnosed at an earlier stage, allowing patients to receive early
treatment to maximize survival time. In the present study, five
different ovarian cancer cell lines, which represent the most
common ovarian cancer tissue types, were selected. Combined with
the results of the present study, NPBWR1-2 was identified as a
candidate lncRNA for ovarian cancer. The gene encoding NPBWR1-2 is
located on chromosome 8, and to the best of our knowledge, the
expression profile of NPBWR1-2 in ovarian cancer has not been
previously reported (37). By
performing a series of in vitro experiments, NPBWR1-2
expression levels in SKOV3 cells, following transfection with an
NPBWR1-2 overexpression vector, were detected. The increased
expression of NPBWR1-2 reduced the proliferation, invasion and
migration of SKOV3 cells, suggesting that NPBWR1-2 overexpression
inhibited the proliferation, invasion and migration of ovarian
cancer cells. The results of the lncRNA microarray and in
vitro experiments indicated that NPBWR1-2 was associated with
ovarian cancer.
In various types of cancer, IGFBP7 is involved in a
number of processes, including cell differentiation, cell adhesion,
angiogenesis, cell proliferation and survival, aging and apoptosis
(38). Moreover, it has been
reported that IGFBP7 acts as a tumour suppressor gene (39). The western blotting results indicated
that NPBWR1-2 overexpression significantly increased the expression
of IGFBP7, which further suggested that NPBWR1-2 was associated
with the occurrence and development of ovarian cancer.
Using bioinformatics analysis, 20 miRNAs that were
predicted to bind to lncRNA NPBWR1-2 were identified, most of which
are associated with the occurrence and development of tumours,
including miR-153-5p, miR-548c-3p, miR-664a, miR-1299 and miR-1179.
Recently, miRNA-153-5p was identified as an anticancer factor,
which regulates tumour suppressor genes and participates in the
growth, metastasis and infiltration of tumours (40). Some studies have reported that
miRNA-153-5p is negatively regulated in hepatocellular carcinoma
cell lines and tissues, and inhibits cell migration and invasion by
binding to the 3′ UTR of Snail (41–43). Niu
et al (44) demonstrated that
miRNA-153 overexpression decreases the proliferation and invasion
of osteosarcoma cells by inhibiting the transforming growth
factor-β signalling pathway. Zhou et al (45) reported that miR-153 inhibits cell
proliferation, suppresses epithelial-mesenchymal transition and
reduces cell invasion in ovarian cancer cells by downregulating SET
domain containing 7, histone lysine methyltransferase and zinc
finger E-box binding homeobox 2, suggesting that miR-153 may serve
as a therapeutic target for ovarian cancer.
The role of miR-548c-3p has been investigated in
various types of cancer, including breast, prostate and
Helicobacter pylori-negative gastric cancer (46–48). It
has been hypothesized that miR-548c-3p mutations may drive
tumorigenesis (49). In breast
cancer tissues, miR-548c-3p expression is low; however, miR-548c-3p
overexpression can induce apoptosis, which ultimately inhibits
breast cancer cell proliferation (50). Furthermore, miR-548c-3p inhibits
glioma tumorigenesis via MYB proto-oncogene, transcription factor
(51). The results of the
aforementioned studies indicated that miR-548-3p may serve as a
therapeutic target in various types of cancer.
A number of studies have reported that miR-664a
participates in the regulation of cancer cell proliferation and
migration, primarily via increasing miR-664a expression (52–54).
Sahin et al (55)
demonstrated that miR-664a binds to and alters the expression of
lncRNA maternally expressed 3, which alters the migration of
osteosarcoma cells.
Previous studies have suggested that miRNA-1299 is
associated with the occurrence and development of a number of
different tumours; however, its precise role has not been
previously reported. During prostate cancer, miR-1299 regulates the
Pim-1 proto-oncogene, serine/threonine kinase-STAT3 signalling
pathway (56). miR-1299 is also
downregulated in retinoblastoma, alcoholic hepatitis and
hepatocellular carcinoma tissues (57–59).
miRNA-1179 is located on chromosome 15q26.1, which
has been identified as a cancer susceptibility locus (60). Several studies have reported the
aberrant expression of miR-1179 in various types of human cancer,
such as colorectal, familial breast, pancreatic and thyroid cancer,
as well as glioma (61–65); however, the role of miR-1179 during
cancer progression is not completely understood.
Although the results of the present study further
indicated that numerous potential molecular markers were closely
associated with the development of ovarian cancer, further
investigation is required. To provide a theoretical basis for the
development of novel clinical treatments, future studies should
verify the molecular markers identified in the present study,
perform functional studies on lncRNA NPBWR1-2 and explore the
molecular mechanism underlying the interaction between lncRNA
NPBWR1-2 and its associated miRNAs.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the State Key
Laboratory of Medicinal Chemical Biology (grant. no. 2018003).
Availability of data and materials
The datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SL, CL and PQ made substantial contributions to the
conception and design of the work. QD performed the experiments. YR
collected, analysed and interpreted the data. SL and CL drafted the
work and revised it critically for important intellectual content.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
lncRNA
|
long non-coding RNA
|
|
EOC
|
epithelial ovarian cancer
|
|
miRNAs
|
microRNAs
|
|
ds-cDNAs
|
double-stranded cDNAs
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Korkmaz T, Seber S and Basaran G: Review
of the current role of targeted therapies as maintenance therapies
in first and second line treatment of epithelial ovarian cancer; In
the light of completed trials. Crit Rev Oncol Hematol. 98:180–188.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li H, Xiao N, Li Z and Wang Q: Expression
of inorganic pyrophosphatase (PPA1) correlates with poor prognosis
of epithelial ovarian cancer. Tohoku J Exp Med. 241:165–173. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mercer TR, Dinger ME, Sunkin SM, Mehler MF
and Mattick JS: Specific expression of long noncoding RNAs in the
mouse brain. Proc Natl Acad Sci USA. 105:716–721. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dinger ME, Pang KC, Mercer TR and Mattick
JS: Differentiating protein-coding and noncoding RNA: Challenges
and ambiguities. PLOS Comput Biol. 4:e10001762008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bonasio R and Shiekhattar R: Regulation of
transcription by long noncoding RNAs. Annu Rev Genet. 48:433–455.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mercer TR and Mattick JS: Structure and
function of long noncoding RNAs in epigenetic regulation. Nat
Struct Mol Biol. 20:300–307. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yoon JH, Abdelmohsen K and Gorospe M:
Posttranscriptional gene regulation by long noncoding RNA. J Mol
Biol. 425:3723–3730. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Avgeris M, Tsilimantou A, Levis PK,
Rampias T, Papadimitriou MA, Panoutsopoulou K, Stravodimos K and
Scorilas A: Unraveling UCA1 lncRNA prognostic utility in urothelial
bladder cancer. Carcinogenesis. 40:965–974. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang Y, Liu YT, Tang H, Xie WQ, Yao H, Gu
WT, Zheng YZ, Shang HB, Wang Y, Wei YX, et al: Exosome-transmitted
lncRNA H19 inhibits the growth of pituitary adenoma. J Clin
Endocrinol Metab. 104:126345–6356. 2019. View Article : Google Scholar
|
|
11
|
Li Y, Zhu G, Ma Y and Qu H: lncRNA CCAT1
contributes to the growth and invasion of gastric cancer via
targeting miR-219-1. J Cell Biochem. Dec 12–2017.(Epub ahead of
print).
|
|
12
|
Liu S, Zhang M and Qu P: Expression level
and clinical significance of HOX transcript antisense intergenic
RNA in cervical cancer: A meta-analysis. Sci Rep. 6:380472016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jun GJ, Zhong GG and Ming ZS: miR-218
inhibits the proliferation of glioma U87 cells through the
inactivation of the CDK6/cyclin D1/p21Cip1/Waf1 pathway. Oncol
Lett. 9:2743–2749. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cesana M, Cacchiarelli D, Legnini I,
Santini T, Sthandier O, Chinappi M, Tramontano A and Bozzoni I: A
long noncoding RNA controls muscle differentiation by functioning
as a competing endogenous RNA. Cell. 147:358–369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou RS, Zhang EX, Sun QF, Ye ZJ, Liu JW,
Zhou DH and Tang Y: Integrated analysis of lncRNA-miRNA-mRNA ceRNA
network in squamous cell carcinoma of tongue. BMC Cancer.
19:7792019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fan CN, Ma L and Liu N: Systematic
analysis of lncRNA-miRNA-mRNA competing endogenous RNA network
identifies four-lncRNA signature as a prognostic biomarker for
breast cancer. J Transl Med. 16:2642018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lin C, Yuan G, Hu Z, Zeng Y, Qiu X, Yu H
and He S: Bioinformatics analysis of the interactions among lncRNA,
miRNA and mRNA expression, genetic mutations and epigenetic
modifications in hepatocellular carcinoma. Mol Med Rep.
19:1356–1364. 2019.PubMed/NCBI
|
|
18
|
Niu H, Zhou W, Xu Y, Yin Z, Shen W, Ye Z,
Liu Y, Chen Y, Yang S, Xiang R, et al: Silencing PPA1 inhibits
human epithelial ovarian cancer metastasis by suppressing the
Wnt/β-catenin signaling pathway. Oncotarget. 8:76266–76278. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Luo D, Wang G, Shen W, Zhao S, Zhou W, Wan
L, Yuan L, Yang S and Xiang R: Clinical significance and functional
validation of PPA1 in various tumors. Cancer Med. 5:2800–2812.
2016. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim JA, Lee S, Kim DE, Kim M, Kwon BM and
Han DC: Fisetin, a dietary flavonoid, induces apoptosis of cancer
cells by inhibiting HSF1 activity through blocking its binding to
the hsp70 promoter. Carcinogenesis. 36:696–706. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bodnar M, Luczak M, Bednarek K, Szylberg
L, Marszalek A, Grenman R, Szyfter K, Jarmuz-Szymczak M and Giefing
M: Proteomic profiling identifies the inorganic pyrophosphatase
(PPA1) protein as a potential biomarker of metastasis in laryngeal
squamous cell carcinoma. Amino Acids. 48:1469–1476. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lei B, Chai W, Wang Z and Liu R: Highly
expressed UNC119 promotes hepatocellular carcinoma cell
proliferation through Wnt/β-catenin signaling and predicts a poor
prognosis. Am J Cancer Res. 5:3123–3134. 2015.PubMed/NCBI
|
|
23
|
Hwang KW, Won TJ, Kim H, Chun HJ, Chun T
and Park Y: Erratum to ‘Characterization of the regulatory roles of
the SUMO’. Diabetes Metab Res Rev. 28:196–202. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rivière L, Gerossier L, Ducroux A, Dion S,
Deng Q, Michel ML, Buendia MA, Hantz O and Neuveut C: HBx relieves
chromatin-mediated transcriptional repression of hepatitis B viral
cccDNA involving SETDB1 histone methyltransferase. J Hepatol.
63:1093–1102. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou M, Ye Z, Gu Y, Tian B, Wu B and Li J:
Genomic analysis of drug resistant pancreatic cancer cell line by
combining long non-coding RNA and mRNA expression profiling. Int J
Clin Exp Pathol. 8:38–52. 2015.PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tudrej P, Olbryt M, Zembala-Nożyńska E,
Kujawa KA, Cortez AJ, Fiszer-Kierzkowska A, Pigłowski W, Nikiel B,
Głowala-Kosińska M, Bartkowska-Chrobok A, et al: Establishment and
characterization of the novel High-grade serous ovarian cancer cell
line OVPA8. Int J Mol Sci. 19(pii): E20802018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hadjicharalambous MR and Lindsay MA: Long
Non-coding RNAs and the innate immune response. Noncoding RNA.
5(pii): E432019.PubMed/NCBI
|
|
29
|
Chen J, Wang Y, Wang C, Hu JF and Li W:
LncRNA Functions as a new emerging epigenetic factor in determining
the fate of stem cells. Front Genet. 11:2772020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zampetaki A, Albrecht A and Steinhofel K:
Long Non-coding RNA structure and function: Is There a Link? Front
Physiol. 9:12012018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bhan A, Soleimani M and Mandal SS: Long
Noncoding RNA and cancer: A New paradigm. Cancer Res. 77:3965–3981.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu X, Yuan Y, Ma R, Xu B and Zhang R:
lncRNA SNHG7 affects malignant tumor behaviors through
downregulation of EZH2 in uveal melanoma cell lines. Oncol Lett.
19:1505–1515. 2020.PubMed/NCBI
|
|
33
|
Qiu MT, Hu JW, Yin R and Xu L: Long
noncoding RNA: An emerging paradigm of cancer research. Tumour
Biol. 34:613–620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Malek E, Jagannathan S and Driscoll JJ:
Correlation of long non-coding RNA expression with metastasis, drug
resistance and clinical outcome in cancer. Oncotarget. 5:8027–8038.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Ding W, Xu Y, Tao E, Mo M, Xu W,
Cai X, Chen X, Yuan J and Wu X: Long non-coding RNA RHPN1-AS1
promotes tumorigenesis and metastasis of ovarian cancer by acting
as a ceRNA against miR-596 and upregulating LETM1. Aging (Albany
NY). 12:4558–4572. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Watanabe N, Wada M, Irukayama-Tomobe Y,
Ogata Y, Tsujino N, Suzuki M, Furutani N, Sakurai T and Yamamoto M:
A single nucleotide polymorphism of the neuropeptide B/W receptor-1
gene influences the evaluation of facial expressions. PLoS One.
7:e353902012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim J, Kim WH, Byeon SJ, Lee BL and Kim
MA: Epigenetic downregulation and growth inhibition of IGFBP7 in
gastric cancer. Asian Pac J Cancer Prev. 19:667–675.
2018.PubMed/NCBI
|
|
39
|
Chen D, Yoo BK, Santhekadur PK, Gredler R,
Bhutia SK, Das SK, Fuller C, Su ZZ, Fisher PB and Sarkar D:
Insulin-like growth factor-binding protein-7 functions as a
potential tumor suppressor in hepatocellular carcinoma. Clin Cancer
Res. 17:6693–6701. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hiyama T, Yoshihara M, Tanaka S and
Chayama K: Genetic polymorphisms and esophageal cancer risk. Int J
Cancer. 121:1643–1658. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen J, Huang X, Wang W, Xie H, Li J, Hu
Z, Zheng Z, Li H and Teng L: LncRNA CDKN2BAS predicts poor
prognosis in patients with hepatocellular carcinoma and promotes
metastasis via the miR-153-5p/ARHGAP18 signaling axis. Aging
(Albany NY). 10:3371–3381. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chen Y, Feng F, Gao X, Wang C, Sun H,
Zhang C, Zeng Z, Lu Y, An L, Qu J, et al: MiRNA153 reduces effects
of chemotherapeutic agents or small molecular kinase inhibitor in
HCC cells. Curr Cancer Drug Targets. 15:176–187. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Niu G, Li B, Sun L and An C: MicroRNA-153
inhibits osteosarcoma cells proliferation and invasion by targeting
TGF-β2. PLoS One. 10:e01192252015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhou J, Xie M, Shi Y, Luo B, Gong G, Li J,
Wang J, Zhao W, Zi Y, Wu X and Wen J: MicroRNA-153 functions as a
tumor suppressor by targeting SET7 and ZEB2 in ovarian cancer
cells. Oncol Rep. 34:111–120. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rane JK, Scaravilli M, Ylipää A, Pellacani
D, Mann VM, Simms MS, Nykter M, Collins AT, Visakorpi T and
Maitland NJ: MicroRNA expression profile of primary prostate cancer
stem cells as a source of biomarkers and therapeutic targets. Eur
Urol. 67:7–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chang H, Kim N, Park JH, Nam RH, Choi YJ,
Lee HS, Yoon H, Shin CM, Park YS, Kim JM and Lee DH: Different
microRNA expression levels in gastric cancer depending on
Helicobacter pylori infection. Gut Liver. 9:188–196. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Stephens PJ, Tarpey PS, Davies H, Van Loo
P, Greenman C, Wedge DC, Nik-Zainal S, Martin S, Varela I, Bignell
GR, et al: Oslo Breast Cancer Consortium (OSBREAC): The landscape
of cancer genes and mutational processes in breast cancer. Nature.
486:400–404. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ni XF, Zhao LH, Li G, Hou M, Su M, Zou CL
and Deng X: MicroRNA-548-3p and MicroRNA-576-5p enhance the
migration and invasion of esophageal squamous cell carcinoma cells
via NRIP1 down-regulation. Neoplasma. 65:881–887. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shi Y, Qiu M, Wu Y and Hai L: MiR-548-3p
functions as an anti-oncogenic regulator in breast cancer. Biomed
Pharmacother. 75:111–116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lu J, Zhang M, Yang X, Cui T and Dai J:
MicroRNA-548c-3p inhibits T98G glioma cell proliferation and
migration by downregulating c-Myb. Oncol Lett. 13:3866–3872. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ding Z, Jian S, Peng X, Liu Y, Wang J,
Zheng L, Ou C, Wang Y, Zeng W and Zhou M: Loss of MiR-664
expression enhances cutaneous malignant melanoma proliferation by
upregulating PLP2. Medicine (Baltimore). 94:e13272015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yang H, Cho ME, Li TW, Peng H, Ko KS, Mato
JM and Lu SC: MicroRNAs regulate methionine adenosyltransferase 1A
expression in hepatocellular carcinoma. J Clin Invest. 123:285–298.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bao Y, Chen B, Wu Q, Hu K, Xi X, Zhu W,
Zhong X and Chen J: Overexpression of miR-664 is associated with
enhanced osteosarcoma cell migration and invasion ability via
targeting SOX7. Clin Exp Med. 17:51–58. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sahin Y, Altan Z, Arman K, Bozgeyik E,
Koruk Ozer M and Arslan A: Inhibition of miR-664a interferes with
the migration of osteosarcoma cells via modulation of MEG3. Biochem
Biophys Res Commun. 490:1100–1105. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Liu Z, He W, Gao J, Luo J, Huang X and Gao
C: Computational prediction and experimental validation of a novel
synthesized pan-PIM inhibitor PI003 and its apoptosis-inducing
mechanisms in cervical cancer. Oncotarget. 6:8019–8035. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Liu Y, Chen SH and Li YM: Differential
expression profiles of genes and miRNAs in alcoholic hepatitis.
Zhonghua gan zang bing za zhi. 20:883–887. 2012.(in Chinese).
PubMed/NCBI
|
|
58
|
Venkatesan N, Deepa PR, Khetan V and
Krishnakumar S: Computational and in vitro investigation of
miRNA-Gene regulations in retinoblastoma pathogenesis: miRNA mimics
strategy. Bioinform Biol Insights. 9:89–101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhu H, Wang G, Zhou X, Song X, Gao H, Ma
C, Chang H, Li H, Liu FF, Lu J and Ma J: miR-1299 suppresses cell
proliferation of hepatocellular carcinoma (HCC) by targeting CDK6.
Biomed Pharmacother. 83:792–797. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Cai Q, Zhang B, Sung H, Low SK, Kweon SS,
Lu W, Shi J, Long J, Wen W, Choi JY, et al: Genome-wide association
analysis in East Asians identifies breast cancer susceptibility
loci at 1q32.1, 5q14.3 and 15q26.1. Nat Genet. 46:886–890. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lin M, Chen W, Huang J, Gao H, Ye Y, Song
Z and Shen X: MicroRNA expression profiles in human colorectal
cancers with liver metastases. Oncol Rep. 25:739–747.
2011.PubMed/NCBI
|
|
62
|
Medimegh I, Troudi W, Stambouli N,
Khodjet-El-Khil H, Baroudi O, Ayari H, Omrane I, Uhrhammer N,
Privat M, Mezlini A, et al: Wild-type genotypes of BRCA1 gene SNPs
combined with micro-RNA over-expression in mammary tissue leading
to familial breast cancer with an increased risk of distant
metastases' occurrence. Med Oncol. 31:2552014. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Xu X, Cai N, Zhi T, Bao Z, Wang D, Liu Y,
Jiang K, Fan L, Ji J and Liu N: MicroRNA-1179 inhibits glioblastoma
cell proliferation and cell cycle progression via directly
targeting E2F transcription factor 5. Am J Cancer Res. 7:1680–1692.
2017.PubMed/NCBI
|
|
64
|
Mancikova V, Castelblanco E, Pineiro-Yanez
E, Perales-Paton J, de Cubas AA, Inglada-Perez L, Matias-Guiu X,
Capel I, Bella M, Lerma E, et al: MicroRNA deep-sequencing reveals
master regulators of follicular and papillary thyroid tumors. Mod
Pathol. 28:748–757. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lin C, Hu Z, Yuan G, Su H, Zeng Y, Guo Z,
Zhong F, Jiang K and He S: MicroRNA-1179 inhibits the
proliferation, migration and invasion of human pancreatic cancer
cells by targeting E2F5. Chem Biol Interact. 291:65–71. 2018.
View Article : Google Scholar : PubMed/NCBI
|