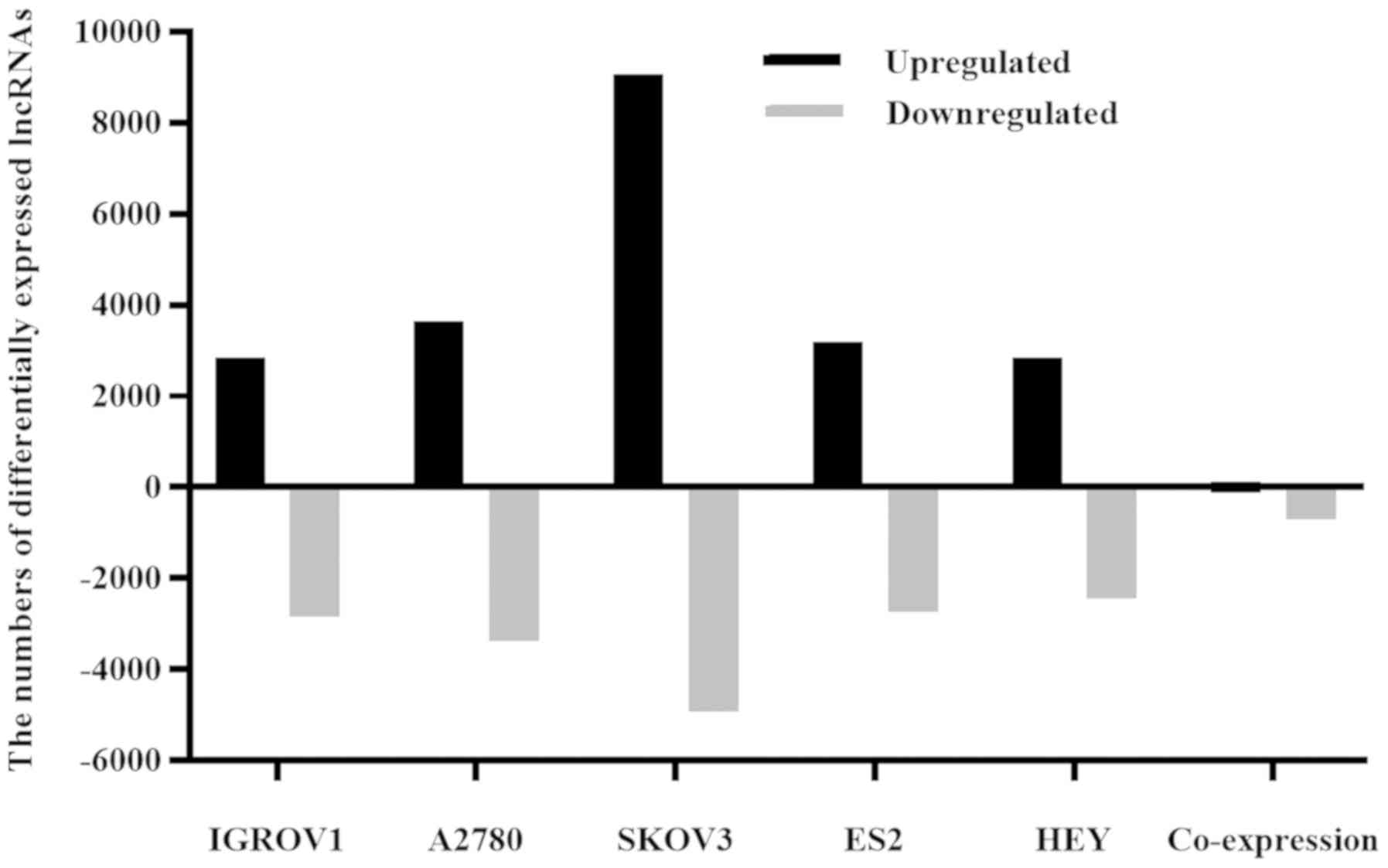
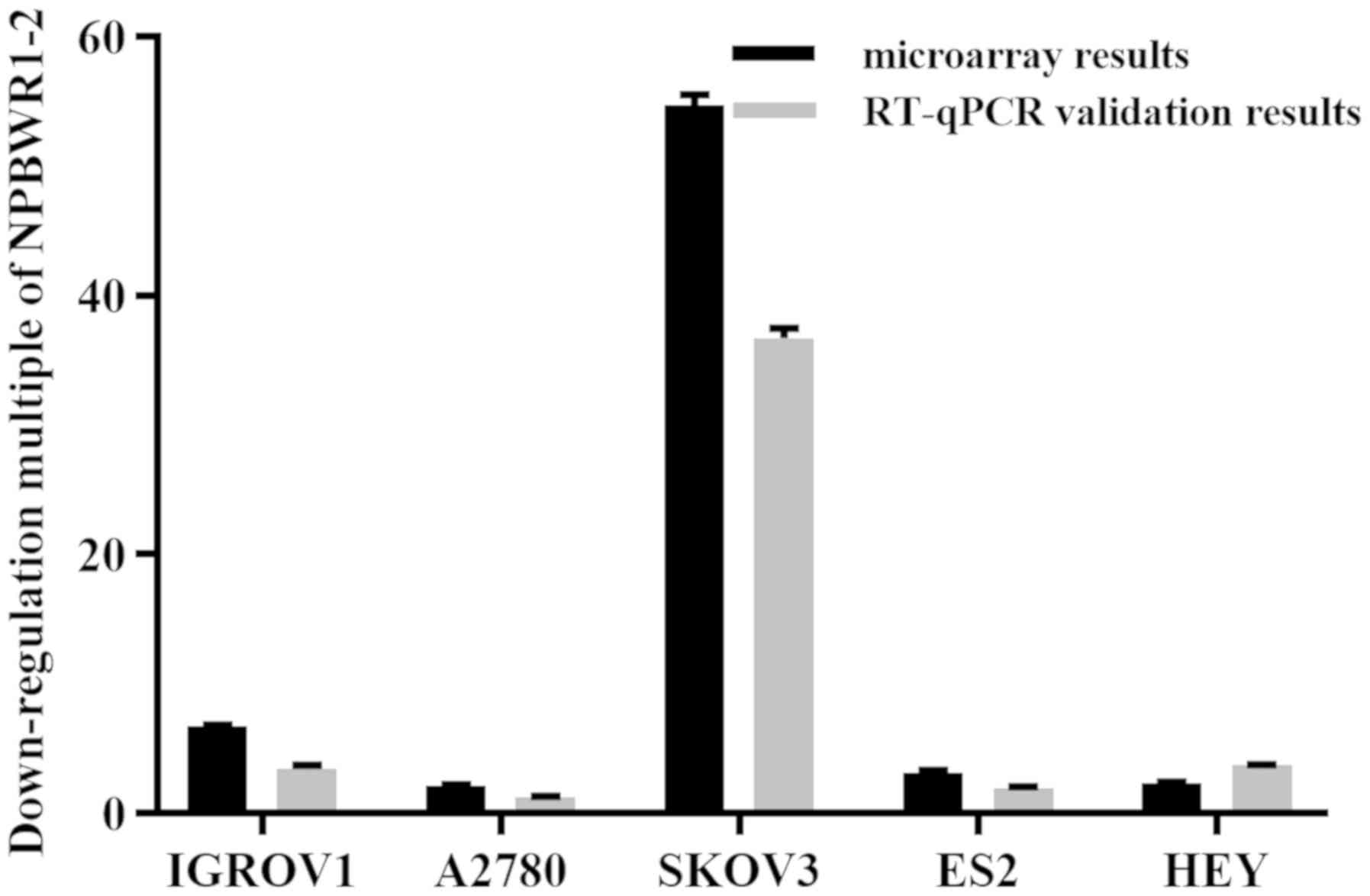
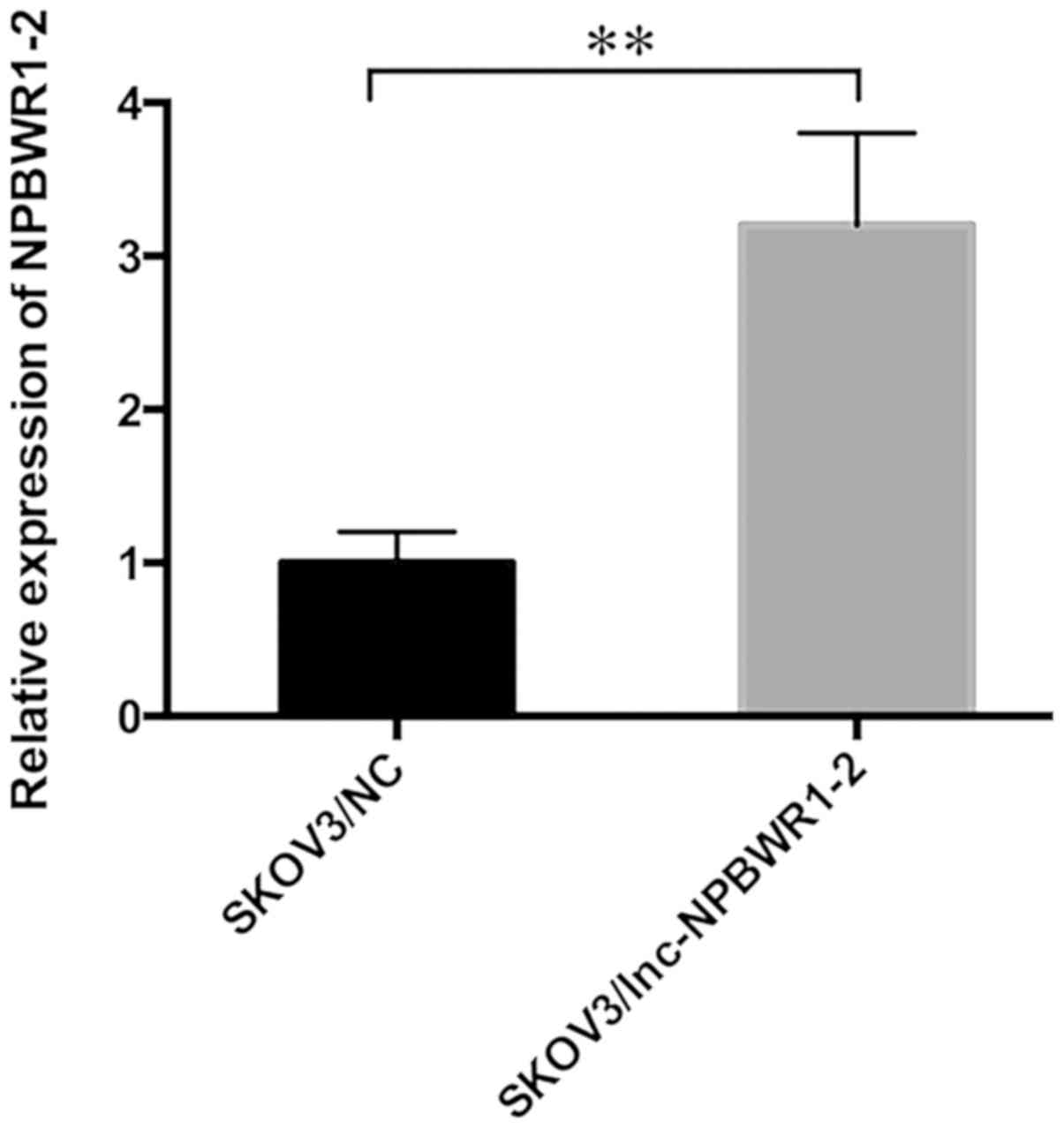
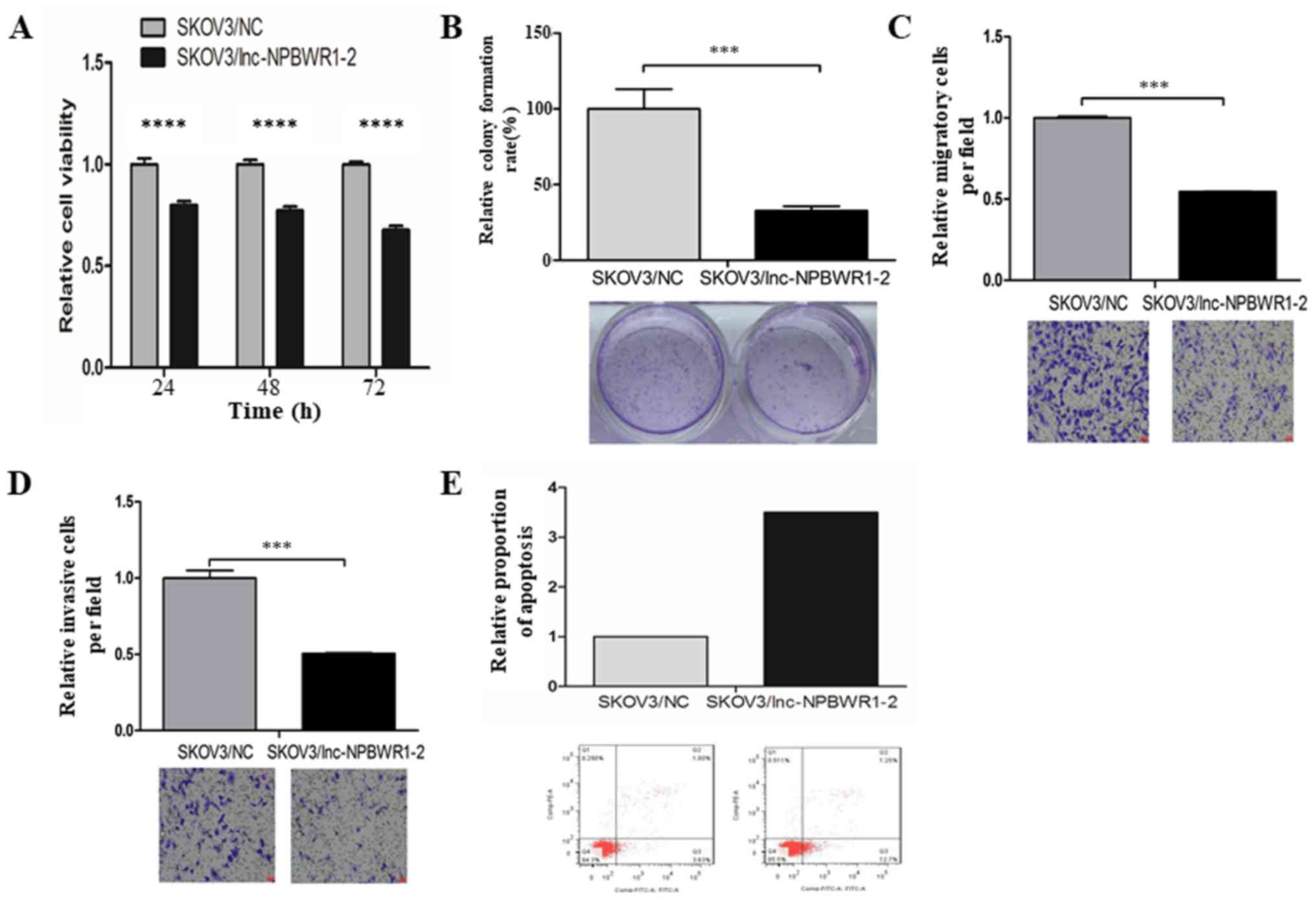
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Korkmaz T, Seber S and Basaran G: Review
of the current role of targeted therapies as maintenance therapies
in first and second line treatment of epithelial ovarian cancer; In
the light of completed trials. Crit Rev Oncol Hematol. 98:180–188.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li H, Xiao N, Li Z and Wang Q: Expression
of inorganic pyrophosphatase (PPA1) correlates with poor prognosis
of epithelial ovarian cancer. Tohoku J Exp Med. 241:165–173. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mercer TR, Dinger ME, Sunkin SM, Mehler MF
and Mattick JS: Specific expression of long noncoding RNAs in the
mouse brain. Proc Natl Acad Sci USA. 105:716–721. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dinger ME, Pang KC, Mercer TR and Mattick
JS: Differentiating protein-coding and noncoding RNA: Challenges
and ambiguities. PLOS Comput Biol. 4:e10001762008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bonasio R and Shiekhattar R: Regulation of
transcription by long noncoding RNAs. Annu Rev Genet. 48:433–455.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mercer TR and Mattick JS: Structure and
function of long noncoding RNAs in epigenetic regulation. Nat
Struct Mol Biol. 20:300–307. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yoon JH, Abdelmohsen K and Gorospe M:
Posttranscriptional gene regulation by long noncoding RNA. J Mol
Biol. 425:3723–3730. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Avgeris M, Tsilimantou A, Levis PK,
Rampias T, Papadimitriou MA, Panoutsopoulou K, Stravodimos K and
Scorilas A: Unraveling UCA1 lncRNA prognostic utility in urothelial
bladder cancer. Carcinogenesis. 40:965–974. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang Y, Liu YT, Tang H, Xie WQ, Yao H, Gu
WT, Zheng YZ, Shang HB, Wang Y, Wei YX, et al: Exosome-transmitted
lncRNA H19 inhibits the growth of pituitary adenoma. J Clin
Endocrinol Metab. 104:126345–6356. 2019. View Article : Google Scholar
|
|
11
|
Li Y, Zhu G, Ma Y and Qu H: lncRNA CCAT1
contributes to the growth and invasion of gastric cancer via
targeting miR-219-1. J Cell Biochem. Dec 12–2017.(Epub ahead of
print).
|
|
12
|
Liu S, Zhang M and Qu P: Expression level
and clinical significance of HOX transcript antisense intergenic
RNA in cervical cancer: A meta-analysis. Sci Rep. 6:380472016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jun GJ, Zhong GG and Ming ZS: miR-218
inhibits the proliferation of glioma U87 cells through the
inactivation of the CDK6/cyclin D1/p21Cip1/Waf1 pathway. Oncol
Lett. 9:2743–2749. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cesana M, Cacchiarelli D, Legnini I,
Santini T, Sthandier O, Chinappi M, Tramontano A and Bozzoni I: A
long noncoding RNA controls muscle differentiation by functioning
as a competing endogenous RNA. Cell. 147:358–369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou RS, Zhang EX, Sun QF, Ye ZJ, Liu JW,
Zhou DH and Tang Y: Integrated analysis of lncRNA-miRNA-mRNA ceRNA
network in squamous cell carcinoma of tongue. BMC Cancer.
19:7792019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fan CN, Ma L and Liu N: Systematic
analysis of lncRNA-miRNA-mRNA competing endogenous RNA network
identifies four-lncRNA signature as a prognostic biomarker for
breast cancer. J Transl Med. 16:2642018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lin C, Yuan G, Hu Z, Zeng Y, Qiu X, Yu H
and He S: Bioinformatics analysis of the interactions among lncRNA,
miRNA and mRNA expression, genetic mutations and epigenetic
modifications in hepatocellular carcinoma. Mol Med Rep.
19:1356–1364. 2019.PubMed/NCBI
|
|
18
|
Niu H, Zhou W, Xu Y, Yin Z, Shen W, Ye Z,
Liu Y, Chen Y, Yang S, Xiang R, et al: Silencing PPA1 inhibits
human epithelial ovarian cancer metastasis by suppressing the
Wnt/β-catenin signaling pathway. Oncotarget. 8:76266–76278. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Luo D, Wang G, Shen W, Zhao S, Zhou W, Wan
L, Yuan L, Yang S and Xiang R: Clinical significance and functional
validation of PPA1 in various tumors. Cancer Med. 5:2800–2812.
2016. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim JA, Lee S, Kim DE, Kim M, Kwon BM and
Han DC: Fisetin, a dietary flavonoid, induces apoptosis of cancer
cells by inhibiting HSF1 activity through blocking its binding to
the hsp70 promoter. Carcinogenesis. 36:696–706. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bodnar M, Luczak M, Bednarek K, Szylberg
L, Marszalek A, Grenman R, Szyfter K, Jarmuz-Szymczak M and Giefing
M: Proteomic profiling identifies the inorganic pyrophosphatase
(PPA1) protein as a potential biomarker of metastasis in laryngeal
squamous cell carcinoma. Amino Acids. 48:1469–1476. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lei B, Chai W, Wang Z and Liu R: Highly
expressed UNC119 promotes hepatocellular carcinoma cell
proliferation through Wnt/β-catenin signaling and predicts a poor
prognosis. Am J Cancer Res. 5:3123–3134. 2015.PubMed/NCBI
|
|
23
|
Hwang KW, Won TJ, Kim H, Chun HJ, Chun T
and Park Y: Erratum to ‘Characterization of the regulatory roles of
the SUMO’. Diabetes Metab Res Rev. 28:196–202. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rivière L, Gerossier L, Ducroux A, Dion S,
Deng Q, Michel ML, Buendia MA, Hantz O and Neuveut C: HBx relieves
chromatin-mediated transcriptional repression of hepatitis B viral
cccDNA involving SETDB1 histone methyltransferase. J Hepatol.
63:1093–1102. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou M, Ye Z, Gu Y, Tian B, Wu B and Li J:
Genomic analysis of drug resistant pancreatic cancer cell line by
combining long non-coding RNA and mRNA expression profiling. Int J
Clin Exp Pathol. 8:38–52. 2015.PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tudrej P, Olbryt M, Zembala-Nożyńska E,
Kujawa KA, Cortez AJ, Fiszer-Kierzkowska A, Pigłowski W, Nikiel B,
Głowala-Kosińska M, Bartkowska-Chrobok A, et al: Establishment and
characterization of the novel High-grade serous ovarian cancer cell
line OVPA8. Int J Mol Sci. 19(pii): E20802018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hadjicharalambous MR and Lindsay MA: Long
Non-coding RNAs and the innate immune response. Noncoding RNA.
5(pii): E432019.PubMed/NCBI
|
|
29
|
Chen J, Wang Y, Wang C, Hu JF and Li W:
LncRNA Functions as a new emerging epigenetic factor in determining
the fate of stem cells. Front Genet. 11:2772020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zampetaki A, Albrecht A and Steinhofel K:
Long Non-coding RNA structure and function: Is There a Link? Front
Physiol. 9:12012018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bhan A, Soleimani M and Mandal SS: Long
Noncoding RNA and cancer: A New paradigm. Cancer Res. 77:3965–3981.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu X, Yuan Y, Ma R, Xu B and Zhang R:
lncRNA SNHG7 affects malignant tumor behaviors through
downregulation of EZH2 in uveal melanoma cell lines. Oncol Lett.
19:1505–1515. 2020.PubMed/NCBI
|
|
33
|
Qiu MT, Hu JW, Yin R and Xu L: Long
noncoding RNA: An emerging paradigm of cancer research. Tumour
Biol. 34:613–620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Malek E, Jagannathan S and Driscoll JJ:
Correlation of long non-coding RNA expression with metastasis, drug
resistance and clinical outcome in cancer. Oncotarget. 5:8027–8038.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Ding W, Xu Y, Tao E, Mo M, Xu W,
Cai X, Chen X, Yuan J and Wu X: Long non-coding RNA RHPN1-AS1
promotes tumorigenesis and metastasis of ovarian cancer by acting
as a ceRNA against miR-596 and upregulating LETM1. Aging (Albany
NY). 12:4558–4572. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Watanabe N, Wada M, Irukayama-Tomobe Y,
Ogata Y, Tsujino N, Suzuki M, Furutani N, Sakurai T and Yamamoto M:
A single nucleotide polymorphism of the neuropeptide B/W receptor-1
gene influences the evaluation of facial expressions. PLoS One.
7:e353902012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim J, Kim WH, Byeon SJ, Lee BL and Kim
MA: Epigenetic downregulation and growth inhibition of IGFBP7 in
gastric cancer. Asian Pac J Cancer Prev. 19:667–675.
2018.PubMed/NCBI
|
|
39
|
Chen D, Yoo BK, Santhekadur PK, Gredler R,
Bhutia SK, Das SK, Fuller C, Su ZZ, Fisher PB and Sarkar D:
Insulin-like growth factor-binding protein-7 functions as a
potential tumor suppressor in hepatocellular carcinoma. Clin Cancer
Res. 17:6693–6701. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hiyama T, Yoshihara M, Tanaka S and
Chayama K: Genetic polymorphisms and esophageal cancer risk. Int J
Cancer. 121:1643–1658. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen J, Huang X, Wang W, Xie H, Li J, Hu
Z, Zheng Z, Li H and Teng L: LncRNA CDKN2BAS predicts poor
prognosis in patients with hepatocellular carcinoma and promotes
metastasis via the miR-153-5p/ARHGAP18 signaling axis. Aging
(Albany NY). 10:3371–3381. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chen Y, Feng F, Gao X, Wang C, Sun H,
Zhang C, Zeng Z, Lu Y, An L, Qu J, et al: MiRNA153 reduces effects
of chemotherapeutic agents or small molecular kinase inhibitor in
HCC cells. Curr Cancer Drug Targets. 15:176–187. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Niu G, Li B, Sun L and An C: MicroRNA-153
inhibits osteosarcoma cells proliferation and invasion by targeting
TGF-β2. PLoS One. 10:e01192252015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhou J, Xie M, Shi Y, Luo B, Gong G, Li J,
Wang J, Zhao W, Zi Y, Wu X and Wen J: MicroRNA-153 functions as a
tumor suppressor by targeting SET7 and ZEB2 in ovarian cancer
cells. Oncol Rep. 34:111–120. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rane JK, Scaravilli M, Ylipää A, Pellacani
D, Mann VM, Simms MS, Nykter M, Collins AT, Visakorpi T and
Maitland NJ: MicroRNA expression profile of primary prostate cancer
stem cells as a source of biomarkers and therapeutic targets. Eur
Urol. 67:7–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chang H, Kim N, Park JH, Nam RH, Choi YJ,
Lee HS, Yoon H, Shin CM, Park YS, Kim JM and Lee DH: Different
microRNA expression levels in gastric cancer depending on
Helicobacter pylori infection. Gut Liver. 9:188–196. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Stephens PJ, Tarpey PS, Davies H, Van Loo
P, Greenman C, Wedge DC, Nik-Zainal S, Martin S, Varela I, Bignell
GR, et al: Oslo Breast Cancer Consortium (OSBREAC): The landscape
of cancer genes and mutational processes in breast cancer. Nature.
486:400–404. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ni XF, Zhao LH, Li G, Hou M, Su M, Zou CL
and Deng X: MicroRNA-548-3p and MicroRNA-576-5p enhance the
migration and invasion of esophageal squamous cell carcinoma cells
via NRIP1 down-regulation. Neoplasma. 65:881–887. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shi Y, Qiu M, Wu Y and Hai L: MiR-548-3p
functions as an anti-oncogenic regulator in breast cancer. Biomed
Pharmacother. 75:111–116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lu J, Zhang M, Yang X, Cui T and Dai J:
MicroRNA-548c-3p inhibits T98G glioma cell proliferation and
migration by downregulating c-Myb. Oncol Lett. 13:3866–3872. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ding Z, Jian S, Peng X, Liu Y, Wang J,
Zheng L, Ou C, Wang Y, Zeng W and Zhou M: Loss of MiR-664
expression enhances cutaneous malignant melanoma proliferation by
upregulating PLP2. Medicine (Baltimore). 94:e13272015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yang H, Cho ME, Li TW, Peng H, Ko KS, Mato
JM and Lu SC: MicroRNAs regulate methionine adenosyltransferase 1A
expression in hepatocellular carcinoma. J Clin Invest. 123:285–298.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bao Y, Chen B, Wu Q, Hu K, Xi X, Zhu W,
Zhong X and Chen J: Overexpression of miR-664 is associated with
enhanced osteosarcoma cell migration and invasion ability via
targeting SOX7. Clin Exp Med. 17:51–58. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sahin Y, Altan Z, Arman K, Bozgeyik E,
Koruk Ozer M and Arslan A: Inhibition of miR-664a interferes with
the migration of osteosarcoma cells via modulation of MEG3. Biochem
Biophys Res Commun. 490:1100–1105. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Liu Z, He W, Gao J, Luo J, Huang X and Gao
C: Computational prediction and experimental validation of a novel
synthesized pan-PIM inhibitor PI003 and its apoptosis-inducing
mechanisms in cervical cancer. Oncotarget. 6:8019–8035. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Liu Y, Chen SH and Li YM: Differential
expression profiles of genes and miRNAs in alcoholic hepatitis.
Zhonghua gan zang bing za zhi. 20:883–887. 2012.(in Chinese).
PubMed/NCBI
|
|
58
|
Venkatesan N, Deepa PR, Khetan V and
Krishnakumar S: Computational and in vitro investigation of
miRNA-Gene regulations in retinoblastoma pathogenesis: miRNA mimics
strategy. Bioinform Biol Insights. 9:89–101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zhu H, Wang G, Zhou X, Song X, Gao H, Ma
C, Chang H, Li H, Liu FF, Lu J and Ma J: miR-1299 suppresses cell
proliferation of hepatocellular carcinoma (HCC) by targeting CDK6.
Biomed Pharmacother. 83:792–797. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Cai Q, Zhang B, Sung H, Low SK, Kweon SS,
Lu W, Shi J, Long J, Wen W, Choi JY, et al: Genome-wide association
analysis in East Asians identifies breast cancer susceptibility
loci at 1q32.1, 5q14.3 and 15q26.1. Nat Genet. 46:886–890. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lin M, Chen W, Huang J, Gao H, Ye Y, Song
Z and Shen X: MicroRNA expression profiles in human colorectal
cancers with liver metastases. Oncol Rep. 25:739–747.
2011.PubMed/NCBI
|
|
62
|
Medimegh I, Troudi W, Stambouli N,
Khodjet-El-Khil H, Baroudi O, Ayari H, Omrane I, Uhrhammer N,
Privat M, Mezlini A, et al: Wild-type genotypes of BRCA1 gene SNPs
combined with micro-RNA over-expression in mammary tissue leading
to familial breast cancer with an increased risk of distant
metastases' occurrence. Med Oncol. 31:2552014. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Xu X, Cai N, Zhi T, Bao Z, Wang D, Liu Y,
Jiang K, Fan L, Ji J and Liu N: MicroRNA-1179 inhibits glioblastoma
cell proliferation and cell cycle progression via directly
targeting E2F transcription factor 5. Am J Cancer Res. 7:1680–1692.
2017.PubMed/NCBI
|
|
64
|
Mancikova V, Castelblanco E, Pineiro-Yanez
E, Perales-Paton J, de Cubas AA, Inglada-Perez L, Matias-Guiu X,
Capel I, Bella M, Lerma E, et al: MicroRNA deep-sequencing reveals
master regulators of follicular and papillary thyroid tumors. Mod
Pathol. 28:748–757. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lin C, Hu Z, Yuan G, Su H, Zeng Y, Guo Z,
Zhong F, Jiang K and He S: MicroRNA-1179 inhibits the
proliferation, migration and invasion of human pancreatic cancer
cells by targeting E2F5. Chem Biol Interact. 291:65–71. 2018.
View Article : Google Scholar : PubMed/NCBI
|