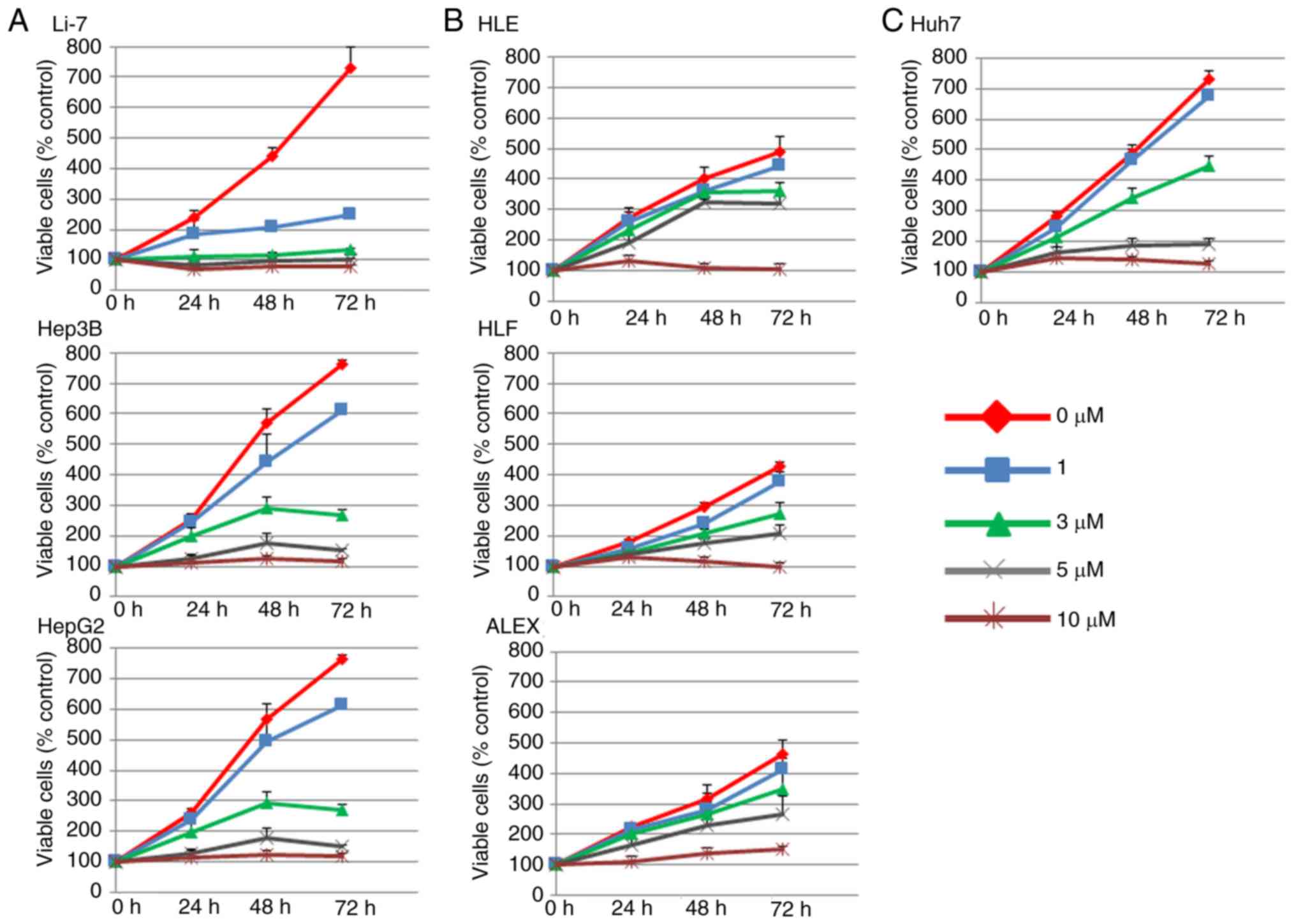
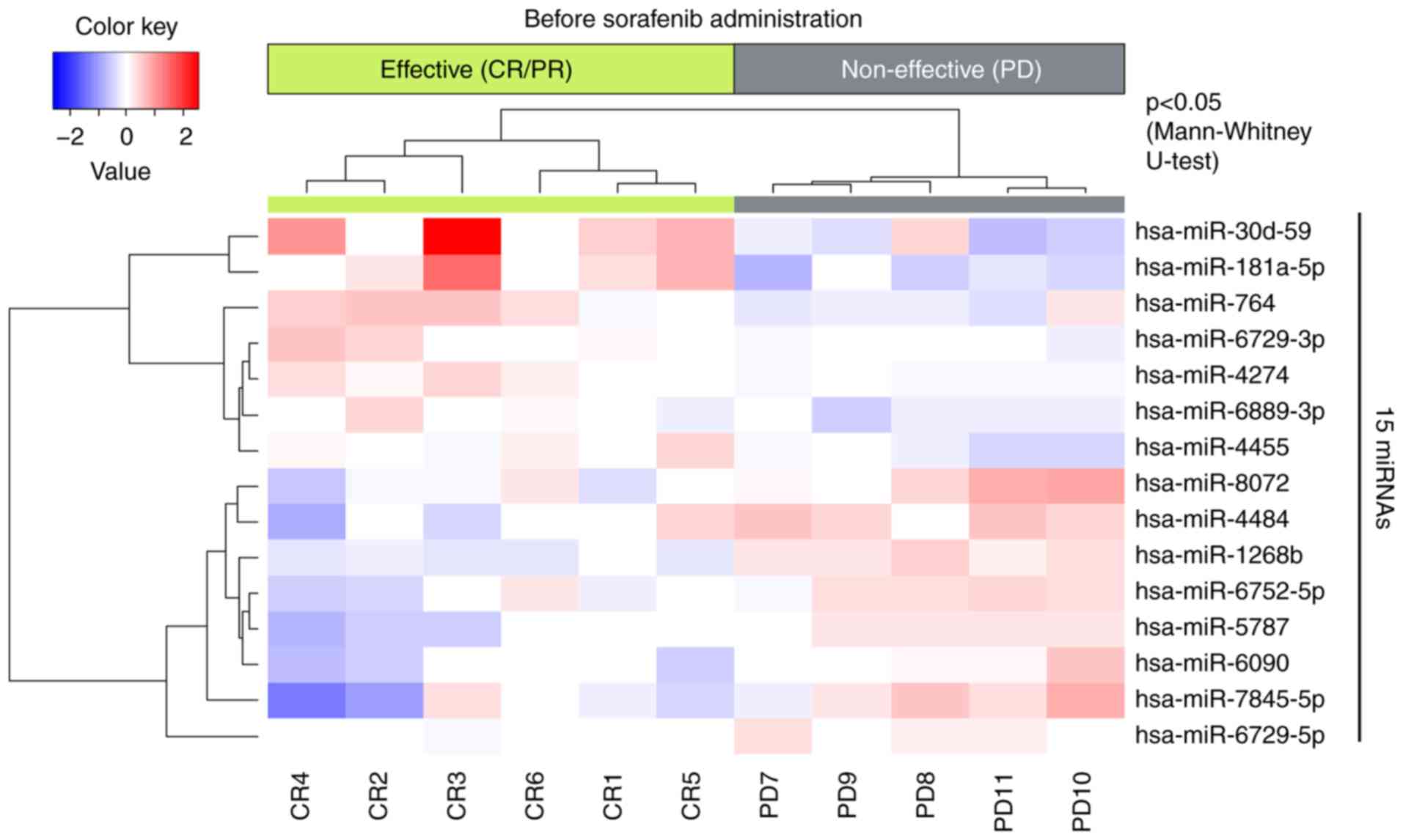
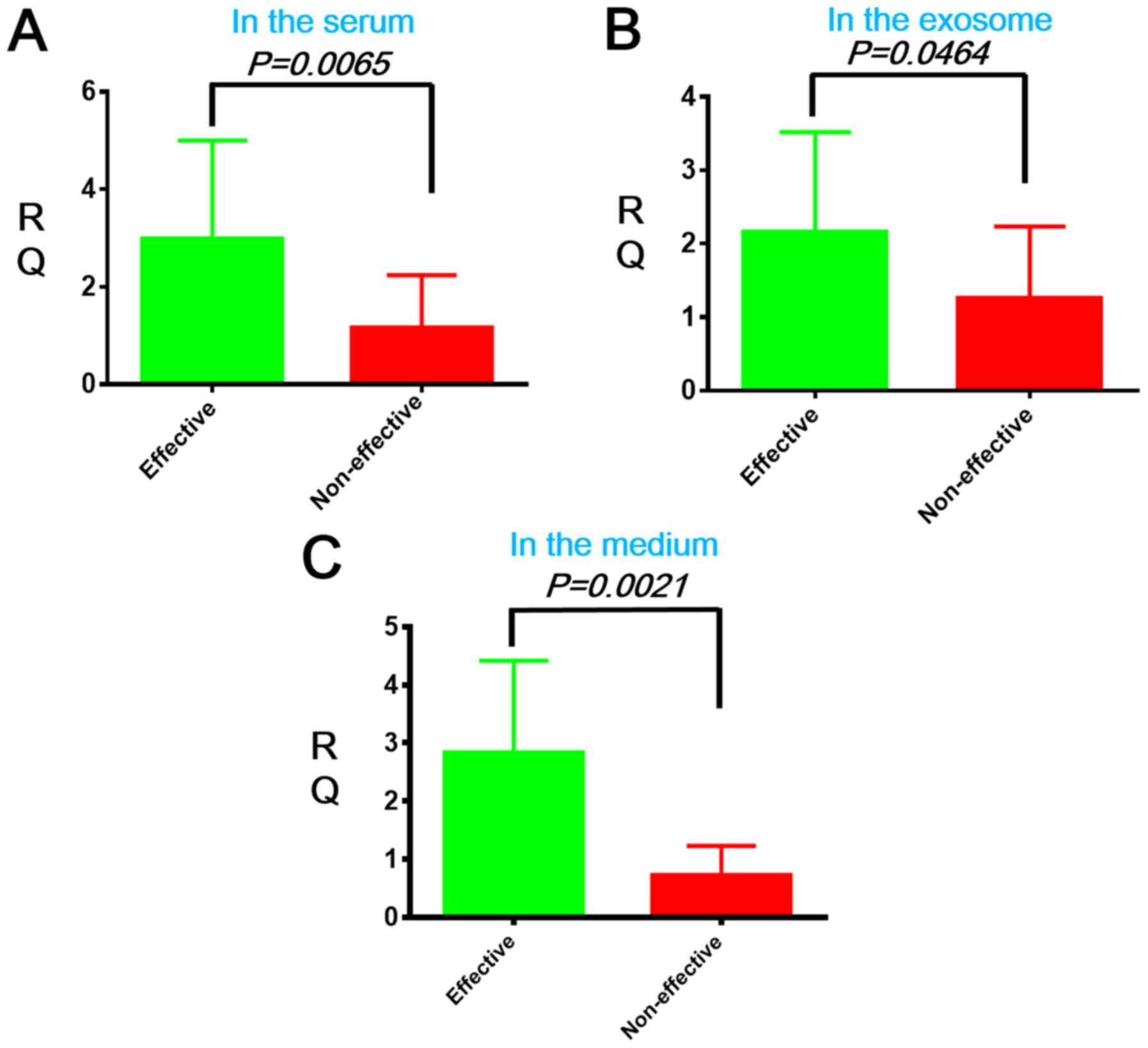
|
1
|
Morishita A and Masaki T: miRNA in
hepatocellular carcinoma. Hepatol Res. 45:128–141. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lacaze L and Scotté M: Surgical treatment
of intra hepatic recurrence of hepatocellular carcinoma. World J
Hepatol. 7:1755–1760. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Imamura H, Matsuyama Y, Tanaka E, Ohkubo
T, Hasegawa K, Miyagawa S, Sugawara Y, Minagawa M, Takayama T,
Kawasaki S and Makuuchi M: Risk factors contributing to early and
late phase intrahepatic recurrence of hepatocellular carcinoma
after hepatectomy. J Hepatol. 38:200–207. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Venook AP, Papandreou C, Furuse J and de
Guevara LL: The incidence and epidemiology of hepatocellular
carcinoma: A global and regional perspective. Oncologist. 15 (Suppl
4):S5–S13. 2010. View Article : Google Scholar
|
|
6
|
Kudo M: Systemic therapy for
hepatocellular carcinoma: 2017 update. Oncology. 93 (Suppl
1):S135–S146. 2017. View Article : Google Scholar
|
|
7
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pelosof L, Lemery S, Casak S, Jiang X,
Rodriguez L, Pierre V, Bi Y, Liu J, Zirkelbach JF, Patel A, et al:
Benefit-risk summary of regorafenib for the treatment of patients
with advanced hepatocellular carcinoma that has progressed on
Sorafenib. Oncologist. 23:496–500. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Belghiti J and Kianmanesh R: Surgical
treatment of hepatocellular carcinoma. HPB (Oxford). 7:42–49. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Masaki T: MicroRNA and hepatocellular
carcinoma. Hepatol Res. 39:751–752. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Krek A, Grün D, Poy MN, Wolf R, Rosenberg
L, Epstein EJ, MacMenamin P, da Piedade I, Gunsalus KC, Stoffel M
and Rajewsky N: Combinatorial microRNA target predictions. Nat
Genet. 37:495–500. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Meng F, Henson R, Wehbe-Janek H, Ghoshal
K, Jacob ST and Patel T: MicroRNA-21 regulates expression of the
PTEN tumor suppressor gene in human hepatocellular cancer.
Gastroenterology. 133:647–658. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gramantieri L, Ferracin M, Fornari F,
Veronese A, Sabbioni S, Liu CG, Calin GA, Giovannini C, Ferrazzi E,
Grazi GL, et al: Cyclin G1 is a target of miR-122a, a microRNA
frequently down-regulated in human hepatocellular carcinoma. Cancer
Res. 67:6092–6099. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wong QW, Lung RW, Law PT, Lai PB, Chan KY,
To KF and Wong N: MicroRNA-223 is commonly repressed in
hepatocellular carcinoma and potentiates expression of Stathmin1.
Gastroenterology. 135:257–269. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Varnholt H, Drebber U, Schulze F,
Wedemeyer I, Schirmacher P, Dienes HP and Odenthal M: MicroRNA gene
expression profile of hepatitis C virus-associated hepatocellular
carcinoma. Hepatology. 47:1223–1232. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Vaira V, Roncalli M, Carnaghi C, Faversani
A, Maggioni M, Augello C, Rimassa L, Pressiani T, Spagnuolo G, Di
Tommaso L, et al: MicroRNA-425-3p predicts response to sorafenib
therapy in patients with hepatocellular carcinoma. Liver Int.
35:1077–1086. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gyöngyösi B, Végh É, Járay B, Székely E,
Fassan M, Bodoky G, Schaff Z and Kiss A: Pretreatment MicroRNA
level and outcome in Sorafenib-treated hepatocellular carcinoma. J
Histochem Cytochem. 62:547–555. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
El-Serag HB: Hepatocellular carcinoma. N
Engl J Med. 365:1118–1127. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kane RC, Farrell AT, Madabushi R, Booth B,
Chattopadhyay S, Sridhara R, Justice R and Pazdur R: Sorafenib for
the treatment of unresectable hepatocellular carcinoma. Oncologist.
14:95–100. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nishida N, Arizumi T, Hagiwara S, Ida H,
Sakurai T and Kudo M: MicroRNAs for the prediction of early
response to sorafenib treatment in human hepatocellular carcinoma.
Liver Cancer. 6:113–125. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yoon EL, Yeon JE, Ko E, Lee HJ, Je JH, Yoo
YJ, Kang SH, Suh SJ, Kim JH, Seo YS, et al: An explorative analysis
for the role of serum miR-10b-3p levels in predicting response to
sorafenib in patients with advanced hepatocellular carcinoma. J
Korean Med Sci. 32:212–220. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Díez-Planelles C, Sánchez-Lozano P, Crespo
MC, Gil-Zamorano J, Ribacoba R, González N, Suárez E,
Martínez-Descals A, Martínez-Camblor P, Álvarez V, et al:
Circulating microRNAs in Huntington's disease: Emerging mediators
in metabolic impairment. Pharmacol Res. 108:102–110. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Miquelestorena-Standley E, Tallet A,
Collin C, Piver E, De Muret A, Salamé E, Bourlier P, Kervarrec T,
Guyétant S and Pagès JC: Interest of variations in microRNA-152 and
−122 in a series of hepatocellular carcinomas related to hepatitis
C virus infection. Hepatol Res. 48:566–573. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Coenen-Stass AM, Betts CA, Lee YF, Mäger
I, Turunen MP, El Andaloussi S, Morgan JE, Wood MJ and Roberts TC:
Selective release of muscle-specific, extracellular microRNAs
during myogenic differentiation. Hum Mol Genet. 25:3960–3974. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu AM, Yao TJ, Wang W, Wong KF, Lee NP,
Fan ST, Poon RT, Gao C and Luk JM: Circulating miR-15b and miR-130b
in serum as potential markers for detecting hepatocellular
carcinoma: A retrospective cohort study. BMJ Open. 2:e0008252012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nakao K, Miyaaki H and Ichikawa T:
Antitumor function of microRNA-122 against hepatocellular
carcinoma. J Gastroenterol. 49:589–593. 2014. View Article : Google Scholar : PubMed/NCBI
|