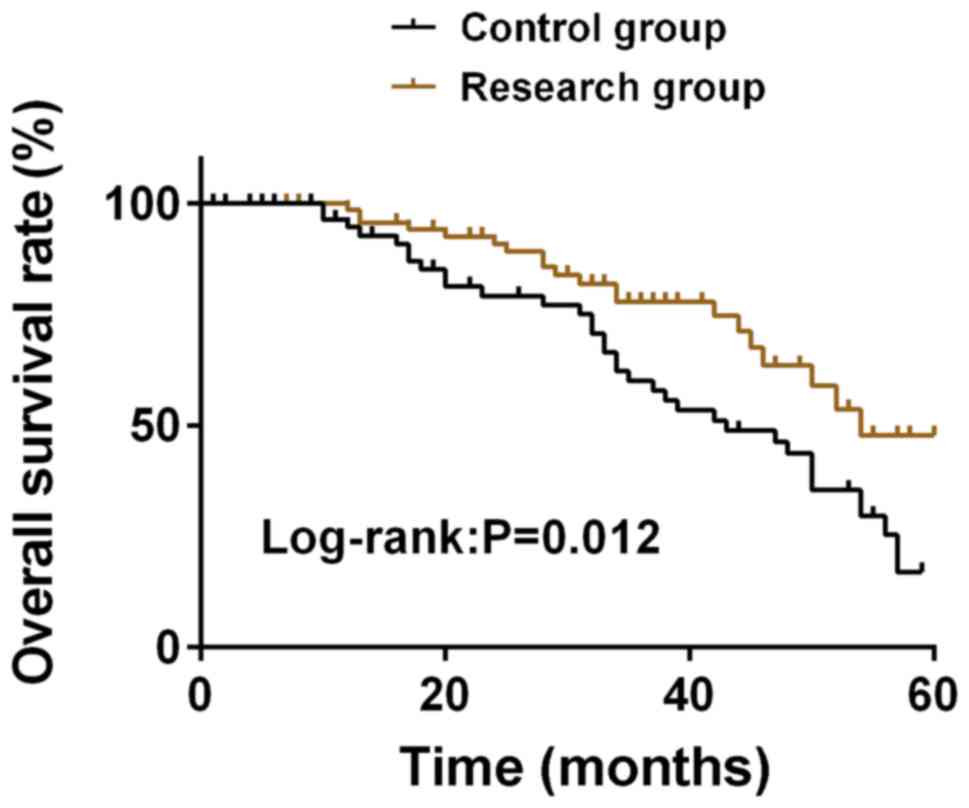
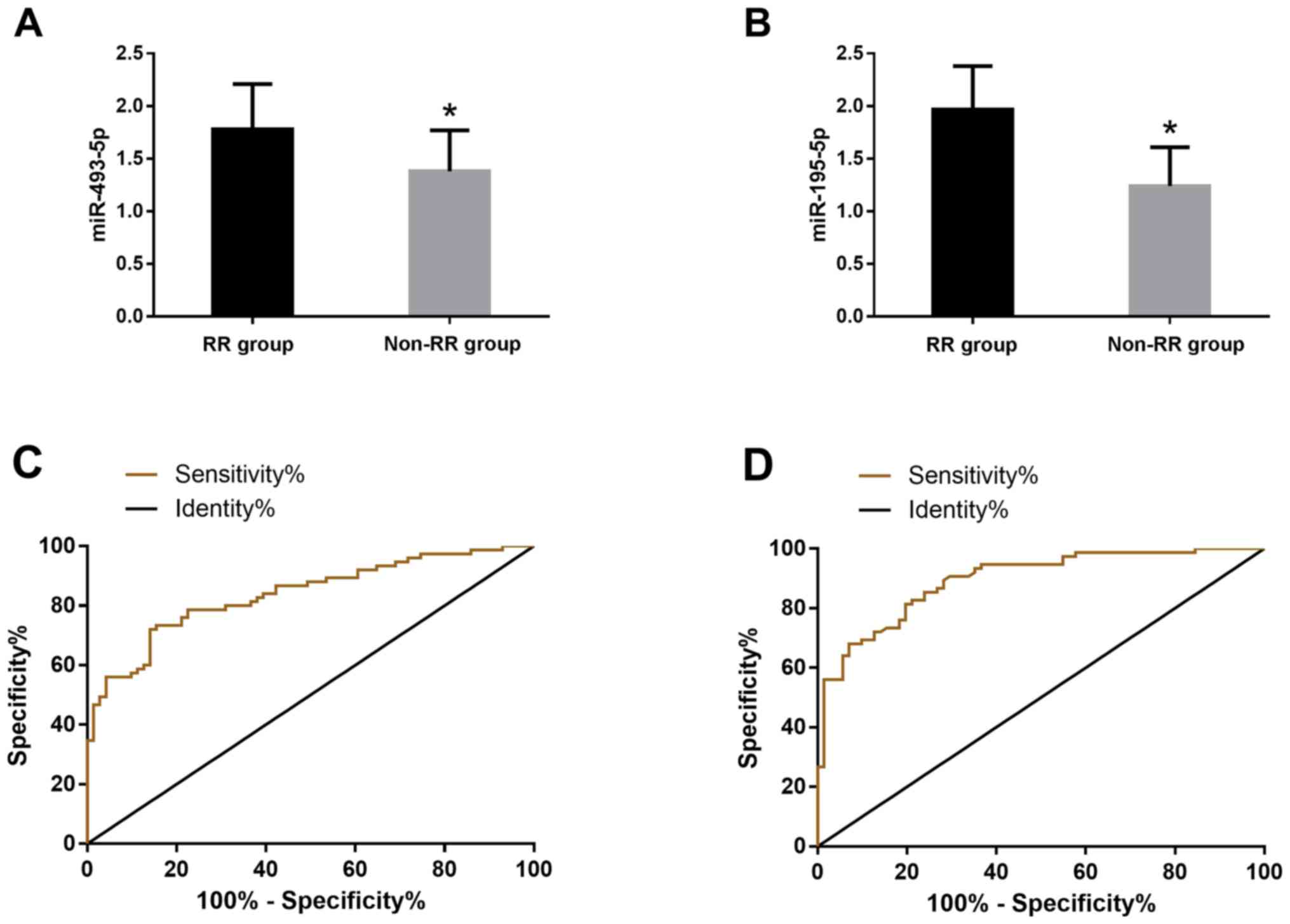
|
1
|
Afshar-Oromieh A, Holland-Letz T, Giesel
FL, Kratochwil C, Mier W, Haufe S, Debus N, Eder M, Eisenhut M,
Schäfer M, et al: Diagnostic performance of 68Ga-PSMA-11 (HBED-CC)
PET/CT in patients with recurrent prostate cancer: Evaluation in
1007 patients. Eur J Nucl Med Mol Imaging. 44:1258–1268. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Watson PA, Arora VK and Sawyers CL:
Emerging mechanisms of resistance to androgen receptor inhibitors
in prostate cancer. Nat Rev Cancer. 15:701–711. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yang Y, Chen R, Sun T, Zhao L, Liu F, Ren
S, Wang H, Lu X, Gao X, Xu C, et al: Efficacy and safety of
combined androgen blockade with antiandrogen for advanced prostate
cancer. Curr Oncol. 26:e39–e47. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kambale PR, Haldar D, Kabi BC and Kambale
KP: Study of vitamin D receptor gene polymorphism (FokI, TaqI and
ApaI) among prostate cancer patients in North India. J Clin Diagn
Res. 11:BC05–BC08. 2017.PubMed/NCBI
|
|
5
|
Benoist GE, Hendriks RJ, Mulders PF,
Gerritsen WR, Somford DM, Schalken JA, van Oort IM, Burger DM and
van Erp NP: Pharmacokinetic aspects of the two novel oral drugs
used for metastatic castration-resistant prostate cancer:
Abiraterone acetate and enzalutamide. Clin Pharmacokinet.
55:1369–1380. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yasui M, Uemura K, Yoneyama S, Kawahara T,
Hattori Y, Teranishi JI, Inoue M, Ohta JI, Yokomizo Y, Yao M, et
al: Predictors of poor response to secondary alternative
antiandrogen therapy with flutamide in metastatic
castration-resistant prostate cancer. Jpn J Clin Oncol.
46:1042–1046. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Reid AH, Attard G, Danila DC, Oommen NB,
Olmos D, Fong PC, Molife LR, Hunt J, Messiou C, Parker C, et al:
Significant and sustained antitumor activity in post-docetaxel,
castration-resistant prostate cancer with the CYP17 inhibitor
abiraterone acetate. J Clin Oncol. 28:1489–1495. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
de Bono JS, Logothetis CJ, Molina A,
Fizazi K, North S, Chu L, Chi KN, Jones RJ, Goodman OB Jr, Saad F,
et al COU-AA-301 Investigators, : Abiraterone and increased
survival in metastatic prostate cancer. N Engl J Med.
364:1995–2005. 2011. View Article : Google Scholar
|
|
9
|
Iguchi T, Tamada S, Kato M, Yasuda S,
Otoshi T, Hamada K, Yamasaki T and Nakatani T: Enzalutamide versus
flutamide for castration-resistant prostate cancer after combined
androgen blockade therapy with bicalutamide: A retrospective study.
Int J Clin Oncol. 24:848–856. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fukasawa S, Suzuki H, Kawaguchi K, Noguchi
H, Enjo K, Tran N, Todd M, Fizazi K and Matsubara N: Efficacy and
safety of abiraterone acetate plus prednisone in Japanese patients
with newly diagnosed, metastatic hormone-naïve prostate cancer: A
subgroup analysis of LATITUDE, a randomized, double-blind,
placebo-controlled, Phase 3 study. Jpn J Clin Oncol. 48:1012–1021.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nie W, Ge HJ, Yang XQ, Sun X, Huang H, Tao
X, Chen WS and Li B: LncRNA-UCA1 exerts oncogenic functions in
non-small cell lung cancer by targeting miR-193a-3p. Cancer Lett.
371:99–106. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fernandez S, Risolino M, Mandia N, Talotta
F, Soini Y, Incoronato M, Condorelli G, Banfi S and Verde P:
miR-340 inhibits tumor cell proliferation and induces apoptosis by
targeting multiple negative regulators of p27 in non-small cell
lung cancer. Oncogene. 34:3240–3250. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Beebe-Dimmer JL, Ruterbusch JJ, Bylsma LC,
Gillezeau C, Fryzek J, Schultz NM, Flanders SC, Barlev A, Heath E
and Quek RGW: Patterns of bicalutamide use in prostate cancer
treatment: A U.S. real-world analysis using the SEER-Medicare
database. Adv Ther. 35:1438–1451. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhao L, Feng X, Song X, Zhou H, Zhao Y,
Cheng L and Jia L: miR-493-5p attenuates the invasiveness and
tumorigenicity in human breast cancer by targeting FUT4. Oncol Rep.
36:1007–1015. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang G, Fang X, Han M, Wang X and Huang Q:
MicroRNA-493-5p promotes apoptosis and suppresses proliferation and
invasion in liver cancer cells by targeting VAMP2. Int J Mol Med.
41:1740–1748. 2018.PubMed/NCBI
|
|
16
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tamada S, Iguchi T, Kato M, Asakawa J,
Kita K, Yasuda S, Yamasaki T, Matsuoka Y, Yamaguchi K, Matsumura K,
et al: Time to progression to castration-resistant prostate cancer
after commencing combined androgen blockade for advanced
hormone-sensitive prostate cancer. Oncotarget. 9:36966–36974. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Teoh JY, Ng CF and Poon DM: Chemohormonal
therapy for metastatic hormone-sensitive prostate cancer: An Asian
perspective. Asia Pac J Clin Oncol. 14 (Suppl 5):5–8. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Robinson D, Van Allen EM, Wu YM, Schultz
N, Lonigro RJ, Mosquera JM, Montgomery B, Taplin ME, Pritchard CC,
Attard G, et al: Integrative clinical genomics of advanced prostate
cancer. Cell. 161:1215–1228. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mittal RD, Mishra DK, Srivastava P,
Manchanda P, Bid HK and Kapoor R: Polymorphisms in the vitamin D
receptor and the androgen receptor gene associated with the risk of
urolithiasis. Indian J Clin Biochem. 25:119–126. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dong L, Zieren RC, Xue W, de Reijke TM and
Pienta KJ: Metastatic prostate cancer remains incurable, why? Asian
J Urol. 6:26–41. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shariat SF, Semjonow A, Lilja H, Savage C,
Vickers AJ and Bjartell A: Tumor markers in prostate cancer I:
Blood-based markers. Acta Oncol. 50 (Suppl 1):61–75. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rescigno P, Lorente D, Bianchini D,
Ferraldeschi R, Kolinsky MP, Sideris S, Zafeiriou Z, Sumanasuriya
S, Smith AD, Mehra N, et al: Prostate-specific antigen decline
after 4 weeks of treatment with abiraterone acetate and overall
survival in patients with metastatic castration-resistant prostate
cancer. Eur Urol. 70:724–731. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Szmulewitz RZ, Peer CJ, Ibraheem A,
Martinez E, Kozloff MF, Carthon B, Harvey RD, Fishkin P, Yong WP,
Chiong E, et al: Prospective international randomized phase II
study of low-dose abiraterone with food versus standard dose
abiraterone in castration-resistant prostate cancer. J Clin Oncol.
36:1389–1395. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nagai T, Naiki T, Iida K, Etani T, Ando R,
Hamamoto S, Sugiyama Y, Akita H, Kubota H, Hashimoto Y, et al:
Early abiraterone acetate treatment is beneficial in Japanese
castration-resistant prostate cancer after failure of primary
combined androgen blockade. Prostate Int. 6:18–23. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xu L, Chen J, Liu W, Liang C, Hu H and
Huang J: Targeting androgen receptor-independent pathways in
therapy-resistant prostate cancer. Asian J Urol. 6:91–98. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ito Y and Sadar MD: Enzalutamide and
blocking androgen receptor in advanced prostate cancer: Lessons
learnt from the history of drug development of antiandrogens. Res
Rep Urol. 10:23–32. 2018.PubMed/NCBI
|
|
28
|
Basch E, Autio K, Ryan CJ, Mulders P,
Shore N, Kheoh T, Fizazi K, Logothetis CJ, Rathkopf D, Smith MR, et
al: Abiraterone acetate plus prednisone versus prednisone alone in
chemotherapy-naive men with metastatic castration-resistant
prostate cancer: Patient-reported outcome results of a randomised
phase 3 trial. Lancet Oncol. 14:1193–1199. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Fizazi K, Tran N, Fein L, Matsubara N,
Rodriguez-Antolin A, Alekseev BY, Özgüroğlu M, Ye D, Feyerabend S,
Protheroe A, et al LATITUDE investigators, : Abiraterone plus
prednisone in metastatic, castration-sensitive prostate cancer. N
Engl J Med. 377:352–360. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhao JG, Liu JD, Shen PF, Tang X, Sun GX,
Zhang XM, Chen JR, Shu KP, Shi M and Zeng H: Prior switching to a
second-line nonsteroidal antiandrogen does not impact the
therapeutic efficacy of abiraterone acetate in patients with
metastatic castration-resistant prostate cancer: A real-world
retrospective study. Asian J Androl. 20:545–550. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang S, Wang X, Li J, Meng S, Liang Z, Xu
X, Zhu Y, Li S, Wu J, Xu M, et al: c-Met, CREB1 and EGFR are
involved in miR-493-5p inhibition of EMT via AKT/GSK-3β/Snail
signaling in prostate cancer. Oncotarget. 8:82303–82313. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cai C, Chen QB, Han ZD, Zhang YQ, He HC,
Chen JH, Chen YR, Yang SB, Wu YD, Zeng YR, et al: miR-195-5
inhibits tumor progression by targeting RPS6KB1 in human prostate
cancer. Clin Cancer Res. 21:4922–4934. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Linder S, van der Poel HG, Bergman AM,
Zwart W and Prekovic S: Enzalutamide therapy for advanced prostate
cancer: Efficacy, resistance and beyond. Endocr Relat Cancer.
26:R31–R52. 2018.PubMed/NCBI
|