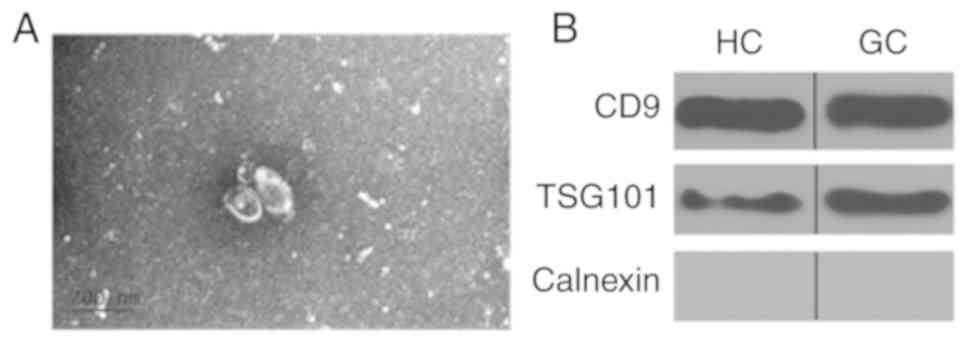
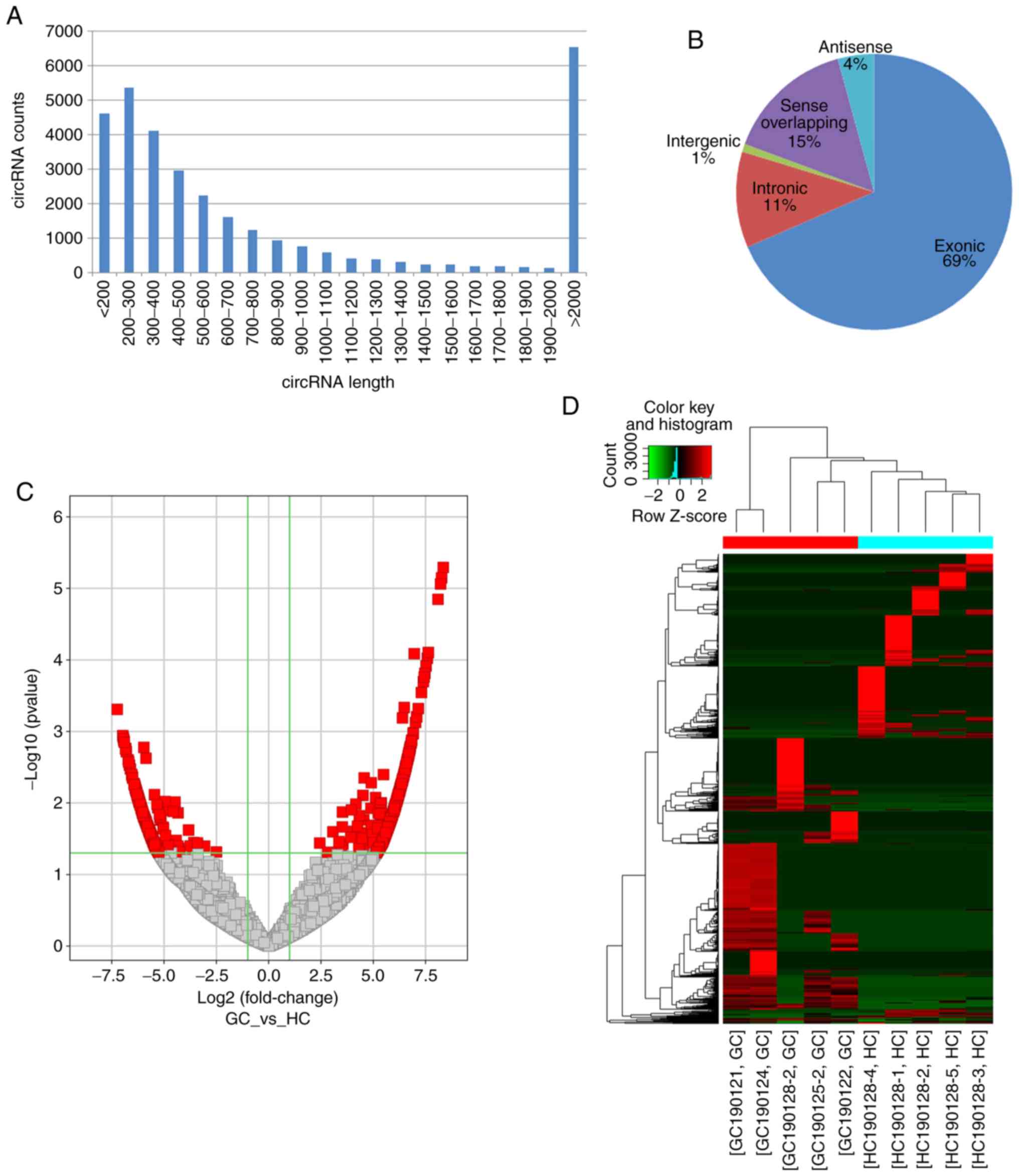
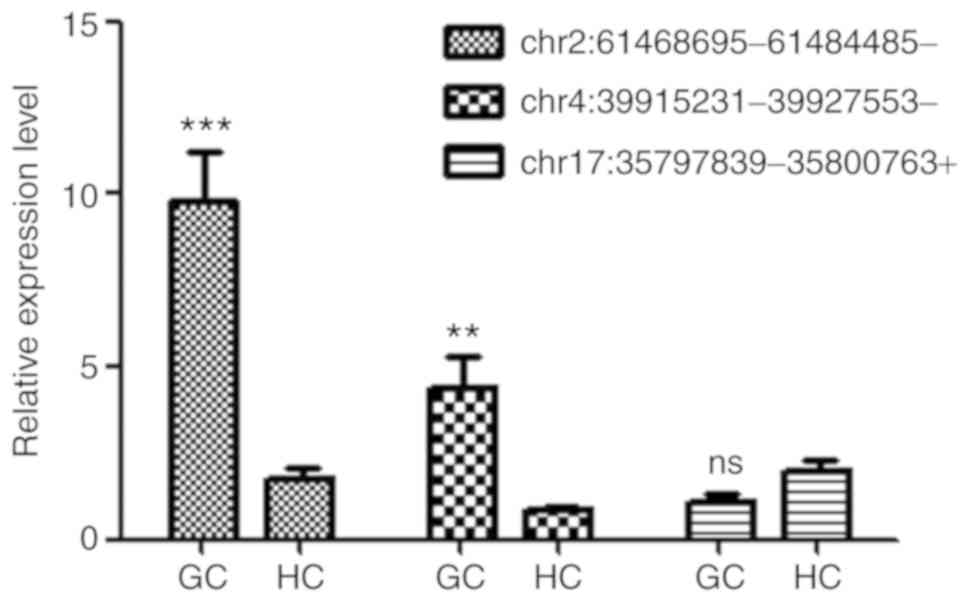
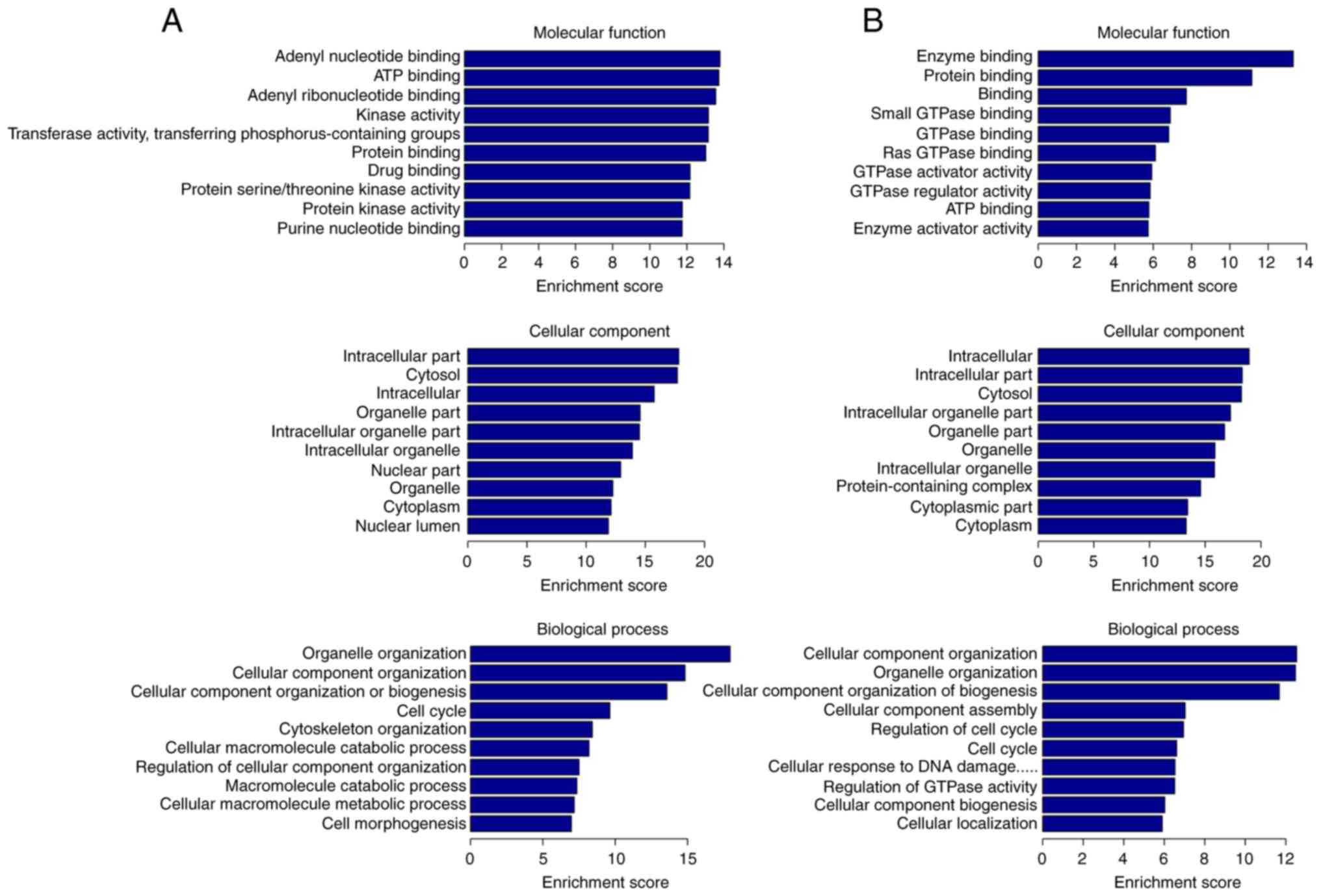
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang T, Liu C, Huang S, Ma Y, Fang J and
Chen Y: A down modulated MicroRNA profiling in patients with
gastric cancer. Gastroenterol Res Pract. 2017:15269812017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yan Y, Fu G, Ye Y and Ming L: Exosomes
participate in the carcinogenesis and the malignant behavior of
gastric cancer. Scand J Gastroenterol. 52:499–504. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang J, Liu Y, Sun W, Zhang Q, Gu T and Li
G: Plasma exosomes as novel biomarker for the early diagnosis of
gastric cancer. Cancer Biomark. 21:805–812. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hou J, Jiang, Zhu L, Zhong S, Zhang H, Li
J, Zhou S, Yang S, He Y, Wang D, et al: Circular RNAs and exosomes
in cancer: A mysterious connection. Clin Transl Oncol.
20:1109–1116. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kharaziha P, Ceder S, Li Q and Panaretakis
T: Tumor cell-derived exosomes: A message in a bottle. Biochim
Biophys Acta. 1826:103–111. 2012.PubMed/NCBI
|
|
7
|
Gao Y, Wang J, Zheng Y, Zhang J, Chen S
and Zhao F: Comprehensive identification of internal structure and
alternative splicing events in circular RNAs. Nat Commun.
7:120602016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang Y and Wang Z: Efficient back splicing
produces translatable circular mRNAs. RNA. 21:172–179. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xia L, Song M, Sun M, Wang F and Yang C:
Circular RNAs as biomarkers for cancer. Adv Exp Med Biol.
1087:171–187. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sui W, Shi Z, Xue W, Ou M, Zhu Y, Chen J,
Liu H, Liu F and Dai L: Circular RNA and gene expression profiles
in gastric cancer based on microarray chip technology. Oncol Rep.
37:1804–1814. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang H, Zhu L, Bai M, Liu Y, Zhan Y, Deng
T, Yang H, Sun W, Wang X, Zhu K, et al: Exosomal circRNA derived
from gastric tumor promotes white adipose browning by targeting the
miR-133/PRDM16 pathway. Int J Cancer. 144:2501–2515. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kumar D, Gupta D, Shankar S and Srivastava
RK: Biomolecular characterization of exosomes released from cancer
stem cells: Possible implications for biomarker and treatment of
cancer. Oncotarget. 6:3280–3291. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Martin M: Cutadapt removes adapter
sequences from high-throughput sequencing reads. EMBnet J. 17:2011.
View Article : Google Scholar
|
|
14
|
Langmead B and Salzberg SL: Fast
gapped-read alignment with bowtie 2. Nat Methods. 9:357–359. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Glažar P, Papavasileiou P and Rajewsky N:
circBase: A database for circular RNAs. RNA. 20:1666–1670. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
John B, Enright AJ, Aravin A, Tuschl T,
Sander C and Marks DS: Human MicroRNA targets. PLoS Biol.
2:e3632004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shi Y, Yang F, Wei S and Xu G:
Identification of key genes affecting results of hyperthermia in
osteosarcoma based on integrative ChIP-Seq/TargetScan analysis. Med
Sci Monit. 23:2042–2048. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hansen TB, Jensen TI, Clausen BH, Bramsen
JB, Finsen B, Damgaard CK and Kjems J: Natural RNA circles function
as efficient microRNA sponges. Nature. 495:384–388. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vishnoi A and Rani S: MiRNA biogenesis and
regulation of diseases: An overview. Methods Mol Biol. 1509:1–10.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Boukouris S and Mathivanan S: Exosomes in
bodily fluids are a highly stable resource of disease biomarkers.
Proteomics Clin Appl. 9:358–367. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nam S, Kim JH and Lee DH: RHOA in gastric
cancer: Functional roles and therapeutic potential. Front Genet.
10:4382019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu N, Zhang G, Bi F, Pan Y, Xue Y, Shi Y,
Yao L, Zhao L, Zheng Y and Fan D: RhoC is essential for the
metastasis of gastric cancer. J Mol Med (Berl). 85:1149–1156. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pan Y, Bi F, Liu N, Xue Y, Yao X, Zheng Y
and Fan D: Expression of seven main Rho family members in gastric
carcinoma. Biochem Biophys Res Commun. 315:686–691. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hu M, Zhu S, Xiong S, Xue X and Zhou X:
MicroRNAs and the PTEN/PI3K/Akt pathway in gastric cancer (Review).
Oncol Rep. 41:1439–1454. 2019.PubMed/NCBI
|
|
30
|
Klempner SJ, Myers AP and Cantley LC: What
a tangled web we weave: Emerging resistance mechanisms to
inhibition of the phosphoinositide 3-kinase pathway. Cancer Discov.
3:1345–1354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tran P, Nguyen C and Klempner SJ:
Targeting the phosphatidylinositol-3-kinase pathway in gastric
cancer: Can omics improve outcomes? Int Neurourol J. 20 (Supp
2):S131–S140. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yang M and Huang CZ: Mitogen-activated
protein kinase signaling pathway and invasion and metastasis of
gastric cancer. World J Gastroenterol. 21:11673–11679. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brown AR, Simmen RC and Simmen FA: The
role of thyroid hormone signaling in the prevention of digestive
system cancers. Int J Mol Sci. 14:16240–16257. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kandemir EG, Yonem A and Narin Y: Gastric
carcinoma and thyroid status. J Int Med Res. 33:222–227. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Aldinucci D and Casagrande N: Inhibition
of the CCL5/CCR5 axis against the progression of gastric cancer.
Int J Mol Sci. 19:14772018. View Article : Google Scholar
|
|
36
|
Xue LJ, Mao XB, Ren LL and Chu XY:
Inhibition of CXCL12/CXCR4 axis as a potential targeted therapy of
advanced gastric carcinoma. Cancer Med. 6:1424–1436. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ooi CH, Ivanova T, Wu J, Lee M, Tan IB,
Tao J, Ward L, Koo JH, Gopalakrishnan V, Zhu Y, et al: Oncogenic
pathway combinations predict clinical prognosis in gastric cancer.
PLoS Genet. 5:e10006762009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Korourian A, Roudi R, Shariftabrizi A,
Kalantari E, Sotoodeh K and Madjd Z: Differential role of wnt
signaling and base excision repair pathways in gastric
adenocarcinoma aggressiveness. Clin Exp Med. 17:505–517. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yousefi B, Mohammadlou M, Abdollahi M,
Salek Farrokhi A, Karbalaei M, Keikha M, Kokhaei P, Valizadeh S,
Rezaiemanesh A, Arabkari V and Eslami M: Epigenetic changes in
gastric cancer induction by Helicobacter pylori. J Cell
Physiol. 234:21770–21784. 2019. View Article : Google Scholar : PubMed/NCBI
|