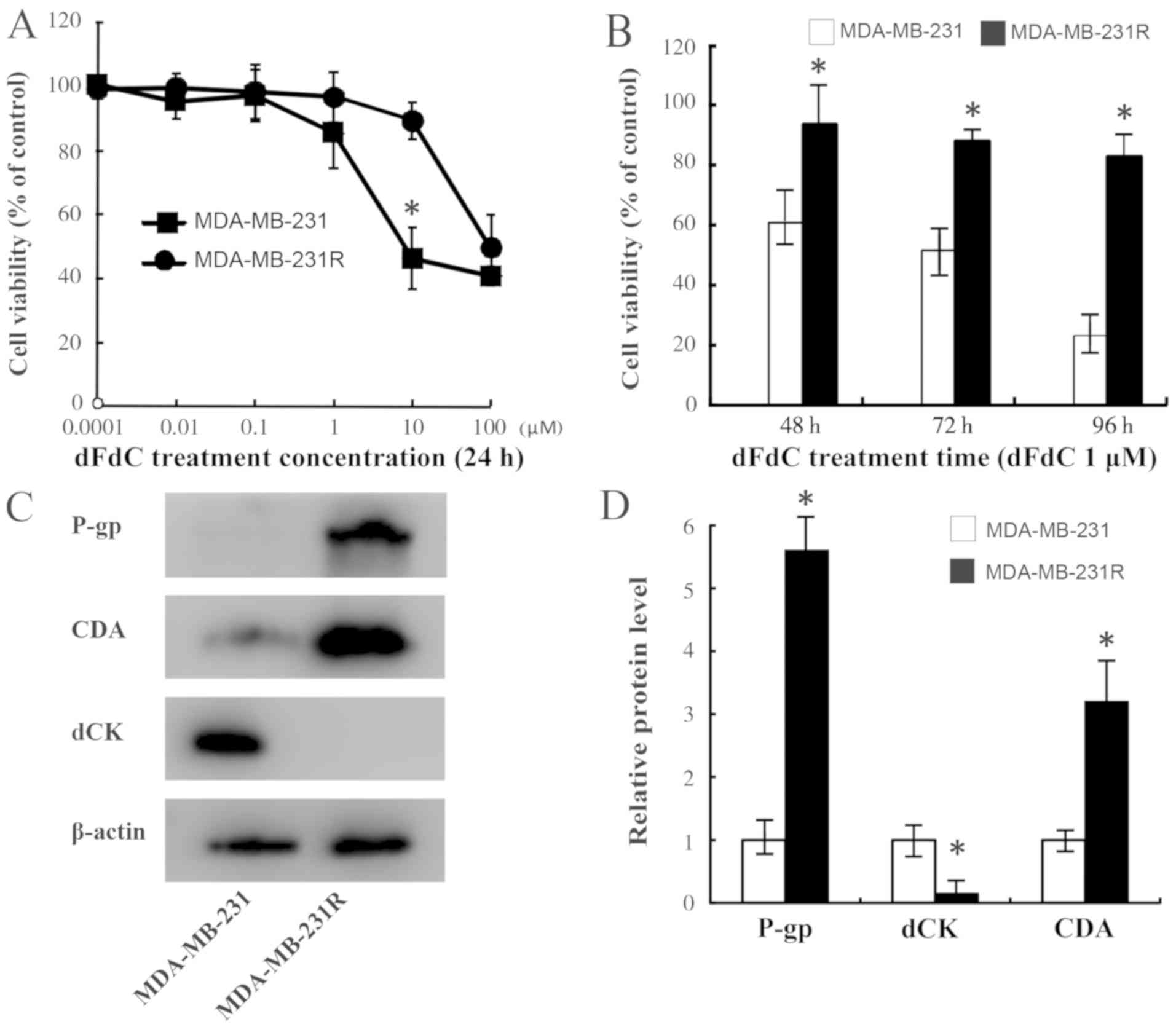
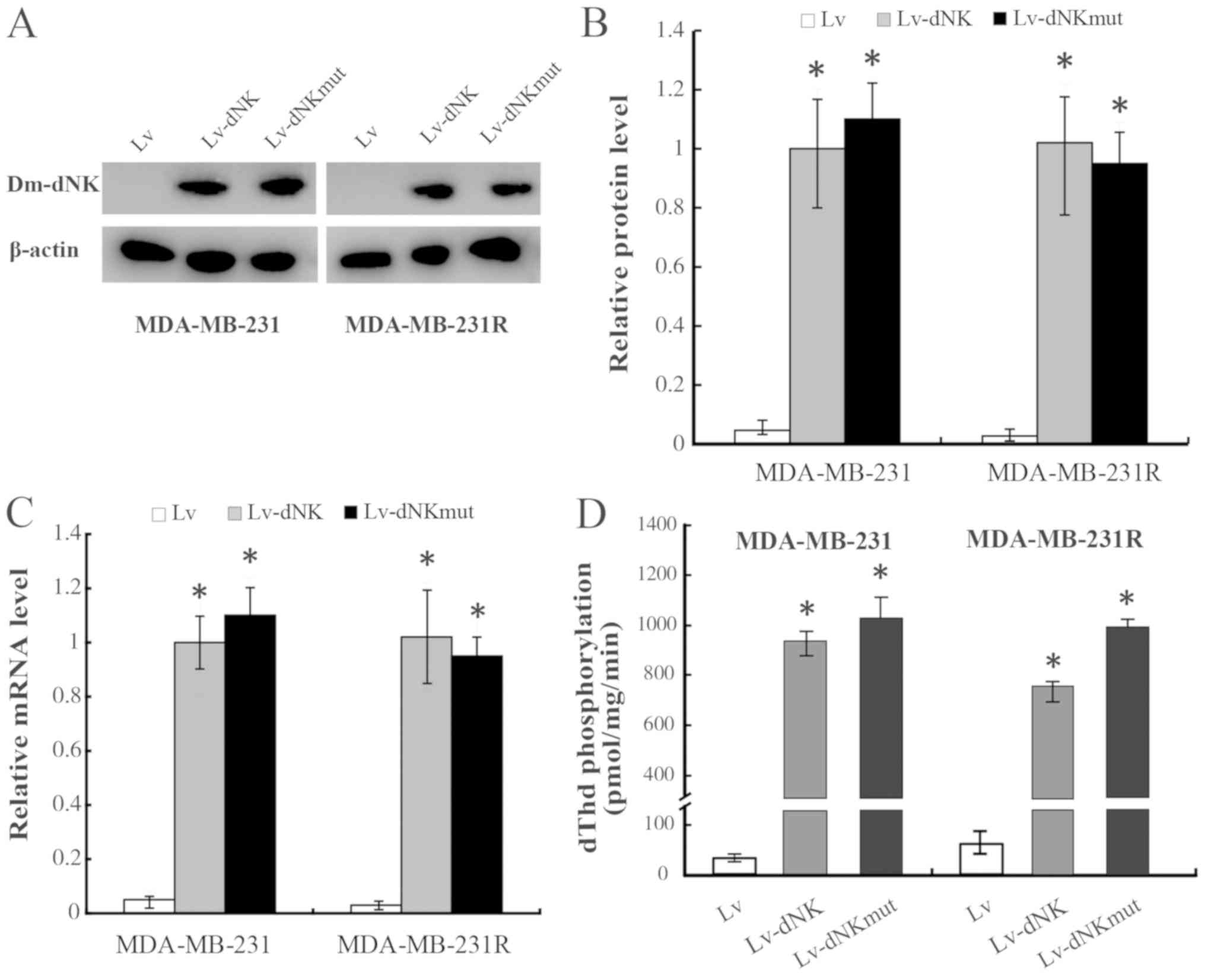
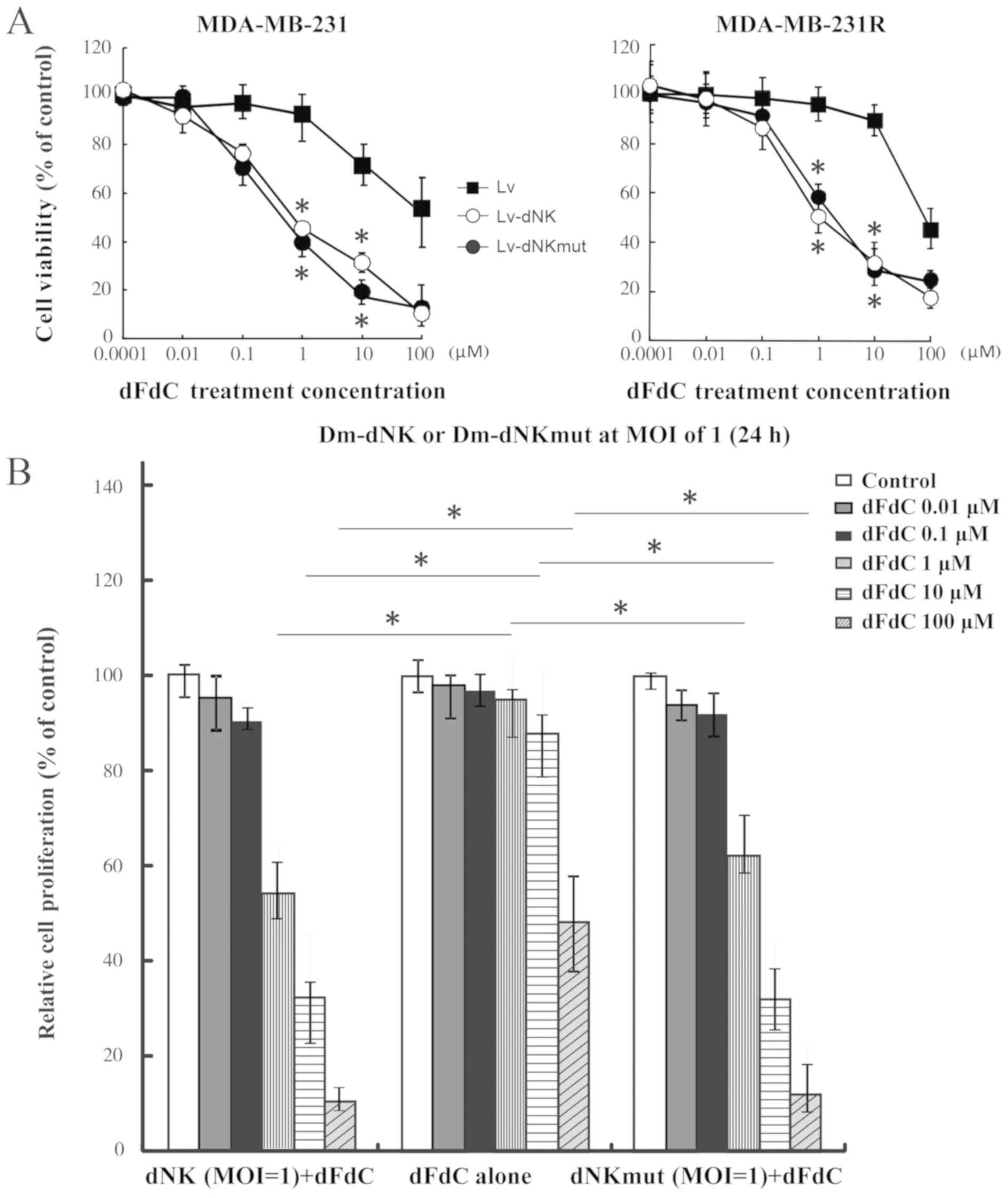
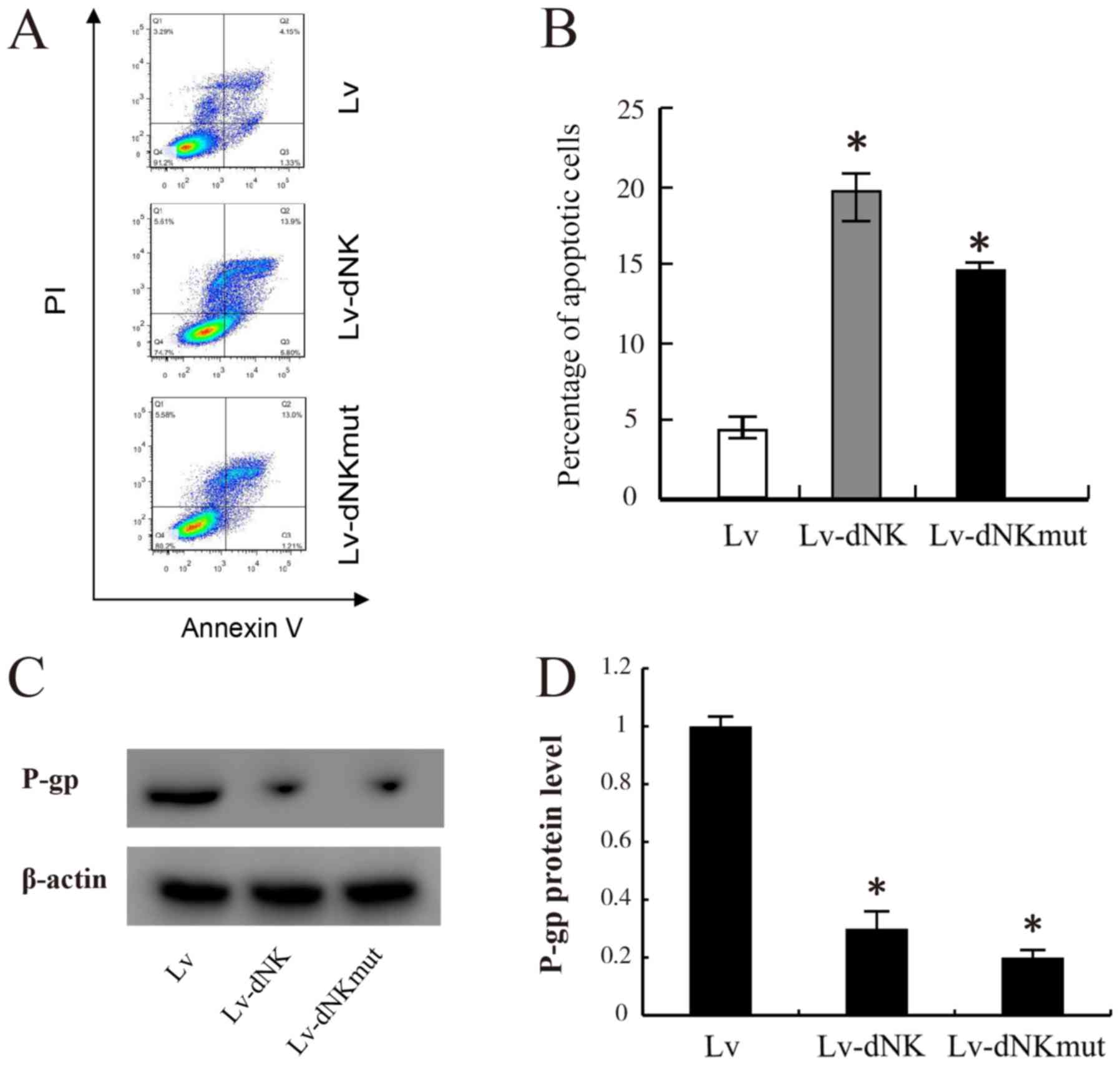
|
1
|
Peddi PF, Ellis MJ and Ma C: Molecular
basis of triple negative breast cancer and implications for
therapy. Int J Breast Cancer. 2012:2171852012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thomas H and Coley HM: Overcoming
multidrug resistance in cancer: An update on the clinical strategy
of inhibiting p-glycoprotein. Cancer Control. 10:159–165. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gonzalez-Angulo AM, Morales-Vasquez F and
Hortobagyi GN: Overview of resistance to systemic therapy in
patients with breast cancer. Adv Exp Med Biol. 608:1–22. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cleator S, Heller W and Coombes RC:
Triple-negative breast cancer: Therapeutic options. Lancet Oncol.
8:235–244. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Garrido-Castro AC, Lin NU and Polyak K:
Insights into molecular classifications of triple-negative breast
cancer: Improving patient selection for treatment. Cancer Discov.
9:176–198. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li X, Lewis MT, Huang J, Gutierrez C,
Osborne CK, Wu MF, Hilsenbeck SG, Pavlick A, Zhang X, Chamness GC,
et al: Intrinsic resistance of tumorigenic breast cancer cells to
chemotherapy. J Natl Cancer Inst. 100:672–679. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rong D, Wang C, Zhang X, Wei Y, Zhang M,
Liu D, Farhan H, Ali SA, Liu Y, Taouil A, et al: A novel taxane,
difluorovinyl-ortataxel, effectively overcomes
paclitaxel-resistance in breast cancer cells. Cancer Lett.
491:36–39. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yuan D, Zhou H, Sun H, Tian R, Xia M, Sun
L and Liu Y: Identification of key genes for guiding
chemotherapeutic management in ovarian cancer using translational
bioinformatics. Oncol Lett. 20:1345–1359. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Niero EL, Rocha-Sales B, Lauand C, Cortez
BA, de Souza MM, Rezende-Teixeira P, Urabayashi MS, Martens AA,
Neves JH and Machado-Santelli GM: The multiple facets of drug
resistance: One history, different approaches. J Exp Clin Cancer
Res. 33:372014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhou M, Li L, Li L, Lin X, Wang F, Li Q
and Huang Y: Overcoming chemotherapy resistance via simultaneous
drug-efflux circumvention and mitochondrial targeting. Acta Pharm
Sin B. 9:615–625. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Agarwal R and Kaye SB: Ovarian cancer:
Strategies for overcoming resistance to chemotherapy. Nat Rev
Cancer. 3:502–516. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Asleh K, Lyck Carstensen S, Tykjaer
Jørgensen CL, Burugu S, Gao D, Won JR, Jensen MB, Balslev E,
Laenkholm AV, Nielsen DL, et al: Basal biomarkers nestin and INPP4B
predict gemcitabine benefit in metastatic breast cancer: Samples
from the phase III SBG0102 clinical trial. Int J Cancer.
144:2578–2586. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Diéras V, Bonnefoi H, Alba E, Awada A,
Coudert B, Pivot X, Gligorov J, Jager A, Zambelli S, Lindeman GJ,
et al: Iniparib administered weekly or twice-weekly in combination
with gemcitabine/carboplatin in patients with metastatic
triple-negative breast cancer: A phase II randomized open-label
study with pharmacokinetics. Breast Cancer Res Treat. 177:383–393.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Spratlin JL and Mackey JR: Human
equilibrative nucleoside transporter 1 (hENT1) in pancreatic
adenocarcinoma: Towards individualized treatment decisions. Cancers
(Basel). 2:2044–2054. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hung SW, Marrache S, Cummins S, Bhutia YD,
Mody H, Hooks SB, Dhar S and Govindarajan R: Defective hCNT1
transport contributes to gemcitabine chemoresistance in ovarian
cancer subtypes: Overcoming transport defects using a nanoparticle
approach. Cancer Lett. 359:233–240. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Honeywell RJ, Ruiz van Haperen VW, Veerman
G, Smid K and Peters GJ: Inhibition of thymidylate synthase by
2′,2′-difluoro-2′-deoxycytidine (Gemcitabine) and its metabolite
2′,2′-difluoro-2′-deoxyuridine. Int J Biochem Cell Biol. 60:73–81.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mitsuno M, Kitajima Y, Ohtaka K, Kai K,
Hashiguchi K, Nakamura J, Hiraki M, Noshiro H and Miyazaki K:
Tranilast strongly sensitizes pancreatic cancer cells to
gemcitabine via decreasing protein expression of ribonucleotide
reductase 1. Int J Oncol. 36:341–349. 2010.PubMed/NCBI
|
|
18
|
Bergman AM, Pinedo HM and Peters GJ:
Determinants of resistance to 2′,2′-difluorodeoxycytidine
(gemcitabine). Drug Resist Updat. 5:19–33. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Baker JA, Wickremsinhe ER, Li CH, Oluyedun
OA, Dantzig AH, Hall SD, Qian YW, Ring BJ, Wrighton SA and Guo Y:
Pharmacogenomics of gemcitabine metabolism: Functional analysis of
genetic variants in cytidine deaminase and deoxycytidine kinase.
Drug Metab Dispos. 41:541–545. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Garcia-Diaz M, Murray MS, Kunkel TA and
Chou KM: Interaction between DNA polymerase lambda and anticancer
nucleoside analogs. J Biol Chem. 285:16874–16879. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Galmarini CM, Clarke ML, Santos CL,
Jordheim L, Perigaud C, Gosselin G, Cros E, Mackey JR and Dumontet
C: Sensitization of ara-C-resistant lymphoma cells by a
pronucleotide analogue. Int J Cancer. 107:149–154. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Réjiba S, Bigand C, Parmentier C and Hajri
A: Gemcitabine-based chemogene therapy for pancreatic cancer using
Ad-dCK::UMK GDEPT and TS/RR siRNA strategies. Neoplasia.
11:637–650. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Saiki Y, Yoshino Y, Fujimura H, Manabe T,
Kudo Y, Shimada M, Mano N, Nakano T, Lee Y, Shimizu S, et al: DCK
is frequently inactivated in acquired gemcitabine-resistant human
cancer cells. Biochem Biophys Res Commun. 421:98–104. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang Q, Yang M, Zhang Y, Zhong L and Zheng
X: Novel combination oncolytic adenoviral gene therapy armed with
Dm-dNK and CD40L for breast cancer. Curr Gene Ther. 19:54–65. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dong X, Qu W, Ma S, Zhu Z, Zheng C, He A,
Karlsson A, Xu K and Zheng X: Potent antitumoral effects of
targeted promoter-driven oncolytic adenovirus armed with Dm-dNK for
breast cancer in vitro and in vivo. Cancer Lett. 328:95–103. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jiang H, Zhao L, Dong X, He A, Zheng C,
Johansson M, Karlsson A and Zheng X: Tanshinone IIA enhances
bystander cell killing of cancer cells expressing Drosophila
melanogaster deoxyribonucleoside kinase in nuclei and
mitochondria. Oncol Rep. 34:1487–1493. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma S, Qu W, Mao L, Zhu Z, Jia L, Zhao L
and Zheng X: Antitumor effects of oncolytic adenovirus armed with
Drosophila melanogaster deoxyribonucleoside kinase in
colorectal cancer. Oncol Rep. 27:1443–1450. 2012.PubMed/NCBI
|
|
28
|
Qu W, Zhu Z, Zhao L, He A and Zheng X:
Conditionally replicating adenovirus SG500-expressed mutant Dm-dNK
gene for breast cancer therapy. Int J Oncol. 41:2175–2183. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tang M, Zu C, He A, Wang W, Chen B and
Zheng X: Synergistic antitumor effect of adenovirus armed with
Drosophila melanogaster deoxyribonucleoside kinase and
nucleoside analogs for human breast carcinoma in vitro and in vivo.
Drug Des Devel Ther. 9:3301–3312. 2015.PubMed/NCBI
|
|
30
|
Zhang N, Dong X, Sun Y, Cai X, Zheng C, He
A, Xu K and Zheng X: Cytotoxic effects of adenovirus- and
lentivirus-mediated expression of Drosophila melanogaster
deoxyribonucleoside kinase on Bcap37 breast cancer cells. Oncol
Rep. 29:960–966. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang N, Zhao L, Ma S, Gu M and Zheng X:
Lentivirus-mediated expression of Drosophila melanogaster
deoxyribonucleoside kinase driven by the hTERT promoter combined
with gemcitabine: A potential strategy for cancer therapy. Int J
Mol Med. 30:659–665. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zheng X, Lundberg M, Karlsson A and
Johansson M: Lipid-mediated protein delivery of suicide nucleoside
kinases. Cancer Res. 63:6909–6913. 2003.PubMed/NCBI
|
|
33
|
Zhu Z, Ma S, Zhao L, Sun Z, He A, Xu H and
Zheng X: Adenovirus-mediated Drosophila melanogaster
deoxyribonucleoside kinase mutants combined with gemcitabine harbor
a safe cancer treatment profile. Int J Oncol. 38:745–753.
2011.PubMed/NCBI
|
|
34
|
Ma S, Zhao L, Zhu Z, Liu Q, Xu H,
Johansson M, Karlsson A and Zheng X: The multisubstrate
deoxyribonucleoside kinase of Drosophila melanogaster as a
therapeutic suicide gene of breast cancer cells. J Gene Med.
13:305–311. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sun X, Xu X, Chen Y, Guan R, Cheng T, Wang
Y, Jin R, Song M and Hang T: Danggui buxue decoction sensitizes the
response of non-small-cell lung cancer to gemcitabine via
regulating deoxycytidine kinase and P-glycoprotein. Molecules.
24:20112019. View Article : Google Scholar
|
|
36
|
Sierzega M, Pach R, Kulig P, Legutko J and
Kulig J: Prognostic implications of expression profiling for
gemcitabine-related genes (hENT1, dCK, RRM1, RRM2) in patients with
resectable pancreatic adenocarcinoma receiving adjuvant
chemotherapy. Pancreas. 46:684–689. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zheng X, Johansson M and Karlsson A:
Nucleoside analog cytotoxicity and bystander cell killing of cancer
cells expressing Drosophila melanogaster deoxyribonucleoside
kinase in the nucleus or cytosol. Biochem Biophys Res Commun.
289:229–233. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yang J, Wu Y, Wang X, Xu L, Zhao X and
Yang Y: Chemoresistance is associated with overexpression of HAX-1,
inhibition of which resensitizes drug-resistant breast cancer cells
to chemotherapy. Tumour Biol. 39:10104283176922282017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zheng X, Johansson M and Karlsson A:
Retroviral transduction of cancer cell lines with the gene encoding
Drosophila melanogaster multisubstrate deoxyribonucleoside
kinase. J Biol Chem. 275:39125–39129. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fraser SP, Salvador V, Manning EA, Mizal
J, Altun S, Raza M, Berridge RJ and Djamgoz MBA: Contribution of
functional voltage-gated Na+ channel expression to cell
behaviors involved in the metastatic cascade in rat prostate
cancer: I. Lateral motility. J Cell Physiol. 195:479–487. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hosokawa M, Saito M, Nakano A, Iwashita S,
Ishizaka A, Ueda K and Iwakawa S: Acquired resistance to decitabine
and cross-resistance to gemcitabine during the long-term treatment
of human HCT116 colorectal cancer cells with decitabine. Oncol
Lett. 10:761–767. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Erin N, Grahovac J, Brozovic A and Efferth
T: Tumor microenvironment and epithelial mesenchymal transition as
targets to overcome tumor multidrug resistance. Drug Resist Updat.
53:1007152020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang XL, Lin FJ, Guo YJ, Shao ZM and Ou
ZL: Gemcitabine resistance in breast cancer cells regulated by
PI3K/AKT-mediated cellular proliferation exerts negative feedback
via the MEK/MAPK and mTOR pathways. Onco Targets Ther. 7:1033–1042.
2014.PubMed/NCBI
|
|
45
|
Triller N, Korosec P, Kern I, Kosnik M and
Debeljak A: Multidrug resistance in small cell lung cancer:
Expression of P-glycoprotein, multidrug resistance protein 1 and
lung resistance protein in chemo-naive patients and in relapsed
disease. Lung Cancer. 54:235–240. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kroep JR, Loves WJ, van der Wilt CL,
Alvarez E, Talianidis I, Boven E, Braakhuis BJM, van Groeningen CJ,
Pinedo HM and Peters GJ: Pretreatment deoxycytidine kinase levels
predict in vivo gemcitabine sensitivity. Mol Cancer Ther.
1:371–376. 2002.PubMed/NCBI
|
|
47
|
Qin T, Castoro R, El Ahdab S, Jelinek J,
Wang X, Si J, Shu J, He R, Zhang N, Chung W, et al: Mechanisms of
resistance to decitabine in the myelodysplastic syndrome. PLoS One.
6:e233722011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Stordal B, Pavlakis N and Davey R:
Oxaliplatin for the treatment of cisplatin-resistant cancer: A
systematic review. Cancer Treat Rev. 33:347–357. 2007. View Article : Google Scholar : PubMed/NCBI
|