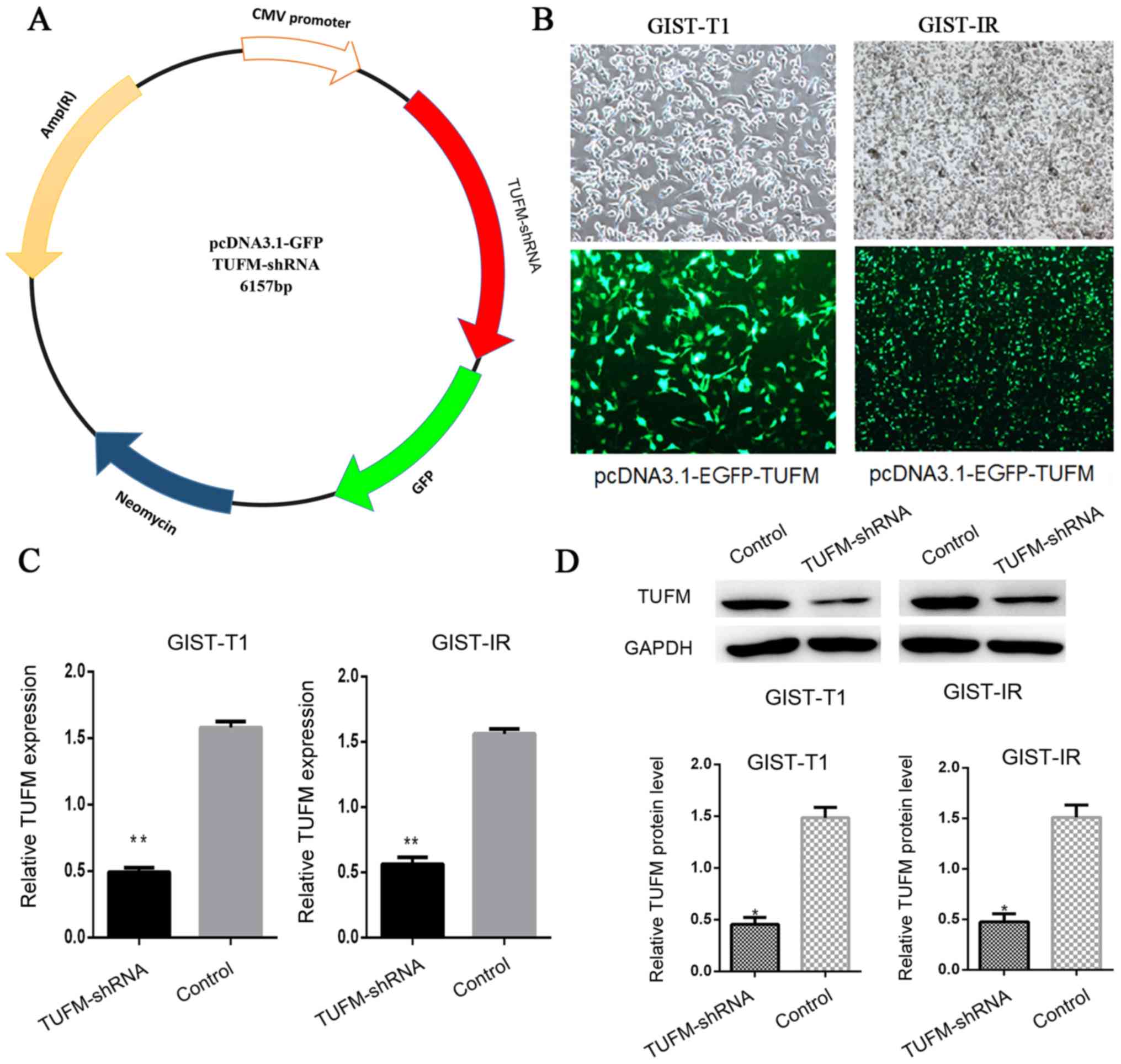
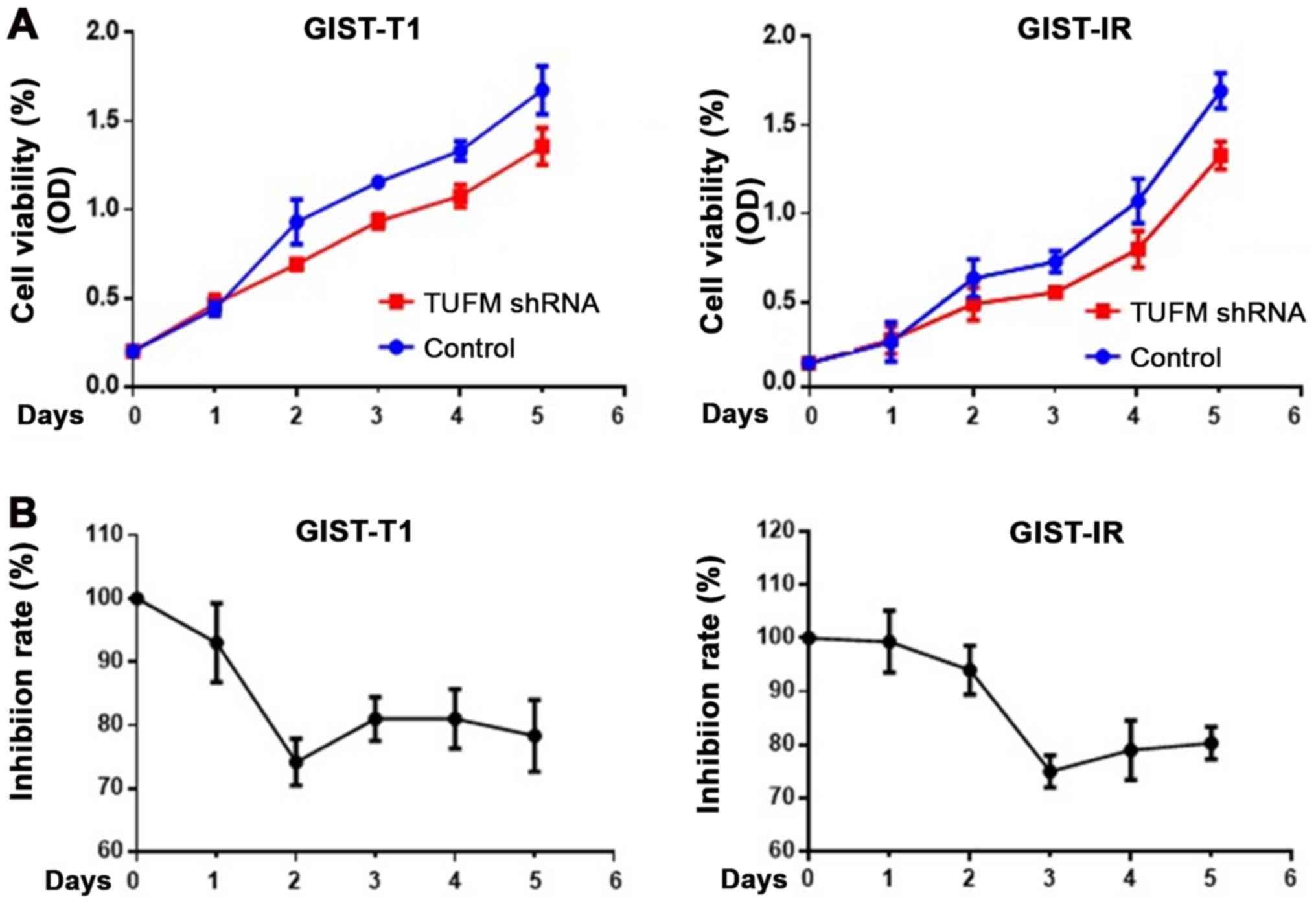
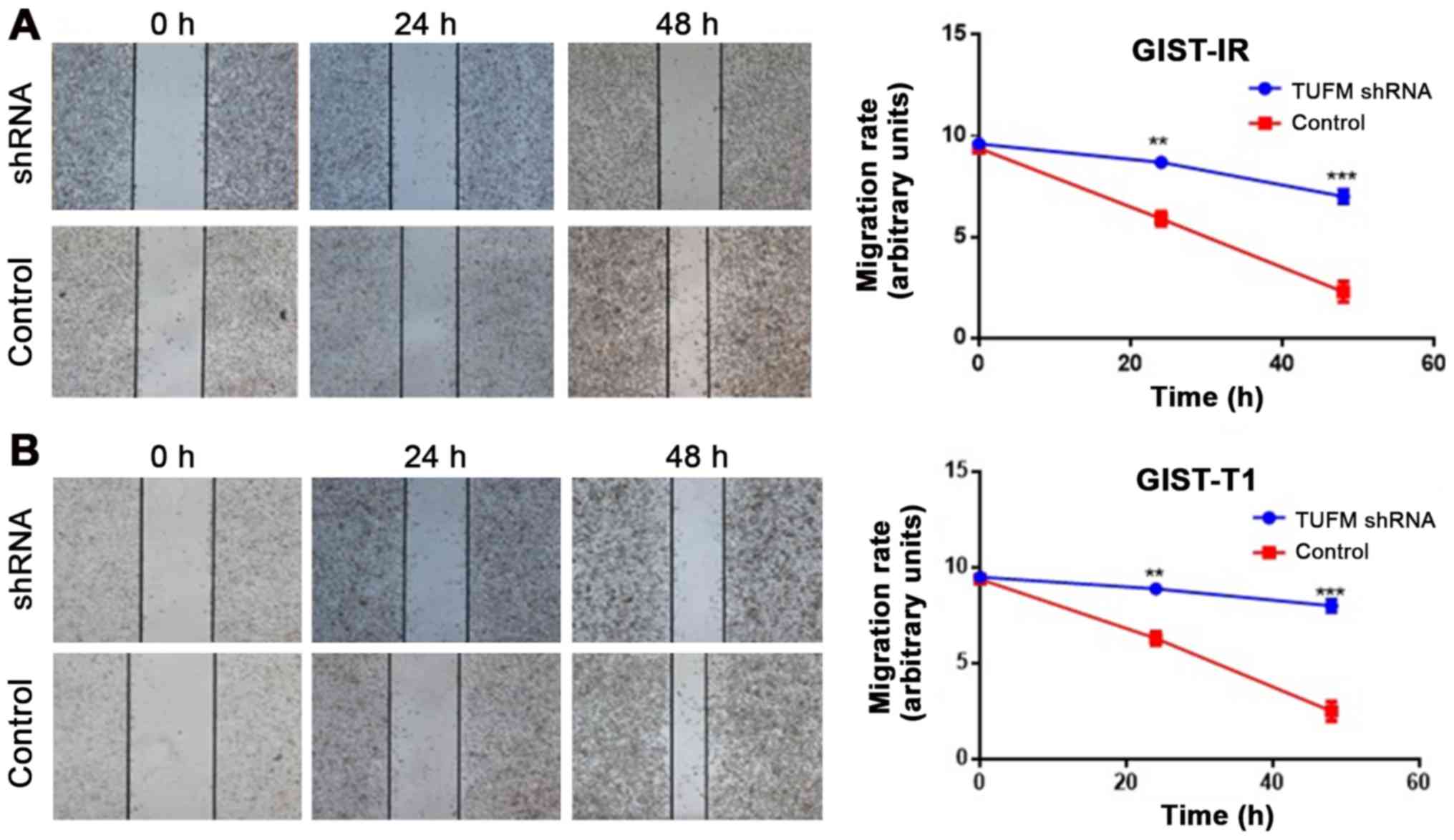
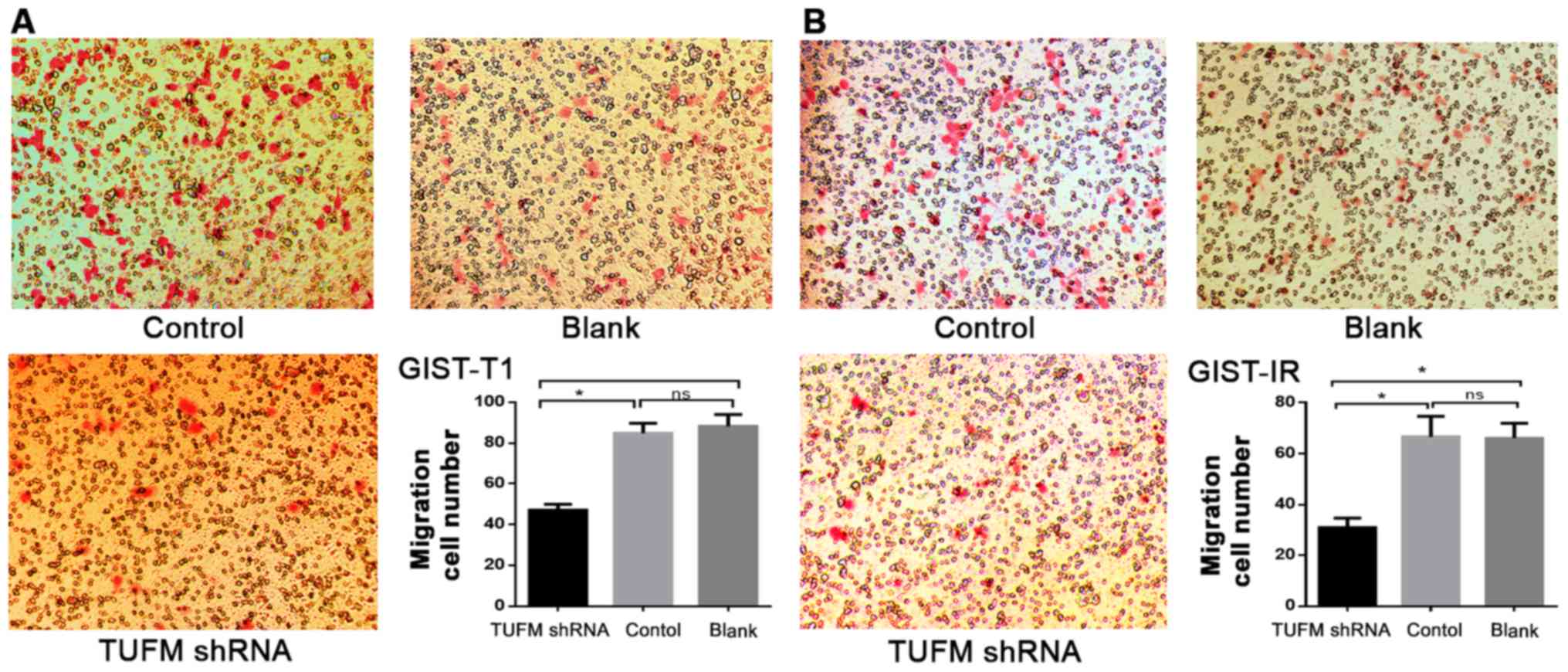
|
1
|
Mazur MT and Clark HB: Gastric stromal
tumors. Reappraisal of histogenesis. Am J Surg Pathol. 7:507–519.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roggin KK and Posner MC: Modern treatment
of gastric gastrointestinal stromal tumors. World J Gastroenterol.
18:6720–6728. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Søreide K, Sandvik OM, Søreide JA, Giljaca
V, Jureckova A and Bulusu VS: Global epidemiology of
gastrointestinal stromal tumors (GIST): A systematic review of
population-based cohort studies. Cancer Epidemiol. 40:39–46. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hirota S, Isozaki K, Moriyama Y, Hashimoto
K, Nishida T, Ishiguro S, Kawano K, Hanada M, Kurata A, Takeda M,
et al: Gain-of-function mutations of KIT in human gastrointestinal
stromal tumors. Science. 279:577–580. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hayashi Y, Bardsley MR, Toyomasu Y,
Taguchi T, Rubin BP, Carter M, Ramachandran A and Ordog T: Abstract
1035: Crenolanib, a highly selective platelet-derived growth factor
receptor/(PDGFRA/B) tyrosine kinase inhibitor, destabilizes ETV1
protein in KIT-mutant gastrointestinal stromal tumor (GIST) cells.
Cancer Res. 73 (Suppl 8):S10352013.
|
|
6
|
Gill AJ: Succinate dehydrogenase (SDH) and
mitochondrial driven neoplasia. Pathology. 44:285–292. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang WL, Addona T, Nair DG, Qi L and
Ravikumar TS: Apoptosis induced by cryo-injury in human colorectal
cancer cells is associated with mitochondrial dysfunction. Int J
Cancer. 103:360–369. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ma L, Wang R, Duan H, Nan Y, Wang Q and
Jin F: Mitochondrial dysfunction rather than mtDNA sequence
mutation is responsible for the multi-drug resistance of small cell
lung cancer. Oncol Rep. 34:3238–3246. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shah ZH, Migliosi V, Miller SCM, Wang A,
Friedman TB and Jacobs HT: Chromosomal locations of three human
nuclear genes (RPSM12, TUFM, and AFG3L1) specifying putative
components of the mitochondrial gene expression apparatus.
Genomics. 48:384–388. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang S, Wei AC, Fu Z, Ruker J, Foster BD,
O'Rourke B and Van Eyk JE: Abstract 16664: Cyclophilin D binds to
tufm and suppresses mitochondrial translation. Circulation.
130:A166642014.
|
|
11
|
Zhang J, Huang JY, Chen YN, Yuan F, Zhang
H, Yan FH, Wang MJ, Wang G, Su M, Lu G, et al: Erratum: Whole
genome and transcriptome sequencing of matched primary and
peritoneal metastatic gastric carcinoma. Sci Rep. 5:137502015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hou J, Liao LD, Xie YM, Zeng FM, Ji X,
Chen B, Li LY, Zhu MX, Yang CX, Zhao Q, et al: DACT2 is a candidate
tumor suppressor and prognostic marker in esophageal squamous cell
carcinoma. Cancer Prev Res (Phila). 6:791–800. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen DB, Jiang WZ, Hong CY, Wang QL, Hong
ZP, Shi LP and Lin CP: Comparative proteomics study of KIT positive
gastrointestinal stromal tumors based on risk stratification
differences. Chinese J Exper Surg. 34:217–220. 2017.
|
|
14
|
Shi H, Hayes M, Kirana C, Miller R,
Keating J, Macartney-Coxson D and Stubbs R: TUFM is a potential new
prognostic indicator for colorectal carcinoma. Pathology.
44:506–512. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Staab CA, Ceder R, Jägerbrink T, Nilsson
JA, Roberg K, Jörnvall H, Höög JO and Grafström RC: Bioinformatics
processing of protein and transcript profiles of normal and
transformed cell lines indicates functional impairment of
transcriptional regulators in buccal carcinoma. J Proteome Res.
6:3705–3717. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bümming P, Andersson J, Meis-Kindblom JM,
Klingenstierna H, Engström K, Stierner U, Wängberg B, Jansson S,
Ahlman H, Kindblom LG and Nilsson B: Neoadjuvant, adjuvant and
palliative treatment of gastrointestinal stromal tumors (GIST) with
imatinib: A center-based study of 17 patients. Br J Cancer.
89:460–464. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Taguchi T, Sonobe H, Toyonaga SI, Yamasaki
I, Shuin T, Takano A, Araki K, Akimaru K and Yuri K: Conventional
and molecular cytogenetic characterization of a new human cell
line, GIST-T1, established from gastrointestinal stromal tumor. Lab
Invest. 82:663–665. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hiroyuki S, Kazumasa F, Yoshiro S, et al:
Establishment of imatinib-resistant GIST cell lines. PLoS One.
2014.
|
|
19
|
Isakov O, Perrone M and Shomron N: Exome
sequencing analysis: A guide to disease variant detection. Methods
Mol Biol. 1038:137–158. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sanger F and Coulson AR: The use of thin
acrylamide gels for DNA sequencing. FEBS Lett. 87:107–110. 1978.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pfaffl MW: A new mathematical model for
relative quantification in realtime RT-PCR. Nucleic Acids Res.
29:e452001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Smith PK, Krohn RI, Hermanson GT, Mallia
AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ and
Klenk DC: Measurment of protein using bicinchoninic acid. Anal
Biochem. 150:76–85. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lasota J and Miettinen M: Clinical
significance of oncogenic KIT and PDGFRA mutations in
gastrointestinal stromal tumors. Histopathology. 53:245–266. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Coe TM, Fero KE, Fanta PT, Mallory RJ,
Tang CM, Murphy JD and Sicklick JK: Population-based epidemiology
and mortality of small malignant gastrointestinal stromal tumors in
the USA. J Gastrointestinal Surg. 6:1132–1140. 2016. View Article : Google Scholar
|
|
25
|
Wilkening A, Rüb C, Sylvester M and Voos
W: Analysis of heat-induced protein aggregation in human
mitochondria. J Biol Chem. 293:11537–11552. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pavlin D, Tozon N, Sersa G, Pogacnik A and
Cemazar M: Efficient electrotransfection into canine muscle.
Technol Cancer Res Treat. 7:45–54. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jhas B, Sriskanthadevan S, Skrtic M,
Sukhai MA, Voisin V, Jitkova Y, Gronda M, Hurren R, Laister RC,
Bader GD, et al: Metabolic adaptation to chronic inhibition of
mitochondrial protein synthesis in acute myeloid leukemia cells.
PLoS One. 8:e583672013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hershkovitz T, Kurolap A, Gonzaga-Jauregui
C, Paperna T, Mory A, Wolf SE; Regeneron Genetics Center, ; Overton
JD, Shuldiner AR, Saada A, et al: A novel TUFM homozygous variant
in a child with mitochondrial cardiomyopathy expands the phenotype
of combined oxidative phosphorylation deficiency 4. J Hum Genet.
64:589–595. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sajid A, Arora G, Gupta M, Singhal A,
Chakraborty K, Nandicoori VK and Singh Y: Interaction of
mycobacterium tuberculosis elongation factor Tu with GTP is
regulated by phosphorylation. J Bacteriol. 193:5347–5358. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ravn K, Schönewolf-Greulich B, Hansen RM,
Bohr AH, Duno M, Wibrand F and Ostergaard E: Neonatal mitochondrial
hepatoencephalopathy caused by novel GFM1 mutations. Mol Genet
Metab Rep. 3:5–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu J, Fang H, Chi Z, Wu Z, Wei D, Mo D,
Niu K, Balajee AS, Hei TK, Nie L and Zhao Y: XPD localizes in
mitochondria and protects the mitochondrial genome from oxidative
DNA damage. Nuclc Acids Res. 43:5476–5488. 2015. View Article : Google Scholar
|
|
32
|
Xi HQ, Zhang KC, Li JY, Cui JX, Zhao P and
Chen L: Expression and clinicopathologic significance of TUFM and
p53 for the normal-adenoma-carcinoma sequence in colorectal
epithelia. World J Surg Oncol. 15:902017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hashim YZHY, Worthington J, Allsopp P,
Ternan NG, Brown EM, McCann MJ, Rowland IR, Esposto S, Servili M
and Gill CIR: Virgin olive oil phenolics extract inhibit invasion
of ht115 human colon cancer cells in vitro and in vivo. Food Funct.
5:1513–1519. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang L, Xiao K, Zhao X, Sun X, Zhang J,
Wang X, Zhu Y and Zhang X: Quantitative proteomics reveals key
proteins regulated by eicosapentaenoic acid in endothelial
activation. Biochem Biophys Res Commun. 487:464–469. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
He K, Guo X, Liu Y, Li J, Hu Y, Wang D and
Song J: TUFM induces epithelial-mesenchymal transition and invasion
in lung cancer cells via a mechanism involving AMPK-GSK3β
signaling. Cell Mol Life Sci. 73:2105–2121. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Geido E, Giaretti W and Nüsse M: Detection
of M and early-G1 phase cells by scattering signals combined with
identification of G1, S, and G2 phase cells. Methods Cell Biol.
33:149–156. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lei Y, Wen H, Yu Y, Taxman DJ, Zhang L,
Widman DG, Swanson KV, Wen KW, Damania B, Moore CB, et al: The
mitochondrial proteins NLRX1 and TUFM form a complex that regulates
type 1 interferon and autophagy. Immunity. 36:933–946. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rakovic A, Grünewald A, Voges L, Hofmann
S, Orolicki S, Lohmann K and Klein C: PINK1-interacting proteins:
Proteomic analysis of overexpressed PINK1. Parkinsons Dis.
2011:1539792011.PubMed/NCBI
|
|
39
|
Lei Y, Kansy BA, Li J, Cong L, Liu Y,
Trivedi S, Wen H, Ting JPY, Ouyang H and Ferris RL: EGFR-targeted
mAb therapy modulates autophagy in head and neck squamous cell
carcinoma through NLRX1-TUFM protein complex. Oncogene.
35:4698–4707. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zottel A, Jovčevska I, Šamec N, Mlakar J,
Šribar J, Križaj I, Vidmar MS and Komel R: Anti-vimentin,
anti-TUFM, anti-NAP1L1 and anti-DPYSL2 nanobodies display cytotoxic
effect and reduce glioblastoma cell migration. Ther Adv Med Oncol.
12:17588359209153022020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Samec N, Jovcevska I, Stojan J, Zottel A,
Liovic M, Myers MP, Muyldermans S, Šribar J, Križaj I and Komel R:
Glioblastoma-specific anti-TUFM nanobody for in-vitro immunoimaging
and cancer stem cell targeting. Oncotarget. 9:172822018. View Article : Google Scholar : PubMed/NCBI
|