Introduction
Upper tract urothelial carcinoma (UTUC), derived
from the urothelium of the renal pelvis and ureter, is relatively
uncommon and comprises only 5–10% of all urothelial carcinoma cases
in Western countries (1). However,
in some regions of China there is a higher prevalence rate of UTUC,
accounting for up to 20–30% of all urothelial carcinoma cases
(2). Furthermore, compared with the
USA, patients in China appear to be younger and exhibit worse
prognostic features, including high histological grade, muscle
invasion and nodal metastasis (2),
suggesting that there may be specific genetic or environmental
factors that influence UTUC carcinogenesis in the Chinese
population. Aristolochic acid (AA), a constituent in numerous
traditional Chinese herbs, may contribute to the higher incidence
rate of UTUC in the Chinese population (2). This is corroborated by a histone
demethylase gene, lysine demethylase 6A (KDM6A), which has
been identified as the most frequently mutated gene in patients
with UTUC in China (3).
KDM6A, also known as UTX, was the
first mutated histone demethylase gene described in human cancer.
The gene was initially demonstrated to be essential in the
differentiation of the normal urothelium (4). Subsequently, somatic mutations and
deletions targeting the KDM6A gene were identified in a
variety of different types of human cancer, including multiple
myeloma, bladder cancer and prostate cancer (5–7).
KDM6A is the second most significantly altered
cancer-associated gene in bladder cancer, and bladder cancer is the
most frequent malignancy in tumors with the KDM6A mutation
(5,8), suggesting an important role for KDM6A
in the tumorigenesis of urothelial carcinoma. However, the
prognostic value of KDM6A expression in patients with UTUC has not
yet been investigated. Previous studies have indicated that
KDM6A functions, in part, by antagonizing histone-lysine
N-methyltransferase EZH2 (EZH2), which provides instructions
for synthesizing histone methyltransferase through the removal of
methyl groups from H3K27me3, which provides an epigenetic
modification of histone H3 (9,10).
Bladder cancer with inactivating KDM6A mutations is
potentially targetable through the inhibition of EZH2
(4). To the best of our knowledge,
this is the first study to analyze the expression levels of KDM6A,
EZH2 and H3K27me3, and the interactions among these proteins in
UTUC.
Materials and methods
Patients
Tissues were retrieved from 108 patients with UTUC,
who received radical nephroureterectomy between January 2007 and
March 2017 at Peking University Shougang Hospital and Peking
University Third Hospital (Beijing, China). Written informed
consent was obtained from all patients. None of the patients
received preoperative chemotherapy. All cases were reviewed, and
pathological diagnoses were confirmed independently by two
pathologists. The pathological grade was assigned according to the
2016 World Health Organization histological criteria (11), and the tumor stage was assigned using
the American Joint Committee on Cancer TNM Staging System for Renal
Pelvis and Ureter Cancer (8th edition, 2018) (12).
Patient follow-up data were collected until June
2017. However, 14 patients were lost to follow-up after surgery,
leaving 94 cases for final survival analysis, with a median
follow-up time of 28 months (range, 1–101 months). Bladder
recurrence was defined as the identification of a subsequent
bladder tumor during cystoscopy, with confirmation using a
pathological evaluation. Cancer-specific survival (CSS) was defined
as the interval between surgery and death from UTUC. Death was
scored as an event, and patients who were still alive were
contacted twice a year to assess the state of their health.
Disease-free survival (DFS) was calculated from the date of surgery
to the date of the first documented evidence of disease recurrence,
or the last follow-up visit, while the patient was alive. Patient
characteristics are shown in Table
I.
 | Table I.Summary of the clinicopathological
characteristics of patients with upper tract urothelial carcinoma
(n=108). |
Table I.
Summary of the clinicopathological
characteristics of patients with upper tract urothelial carcinoma
(n=108).
| Characteristics | n | % |
|---|
| Patient age,
years |
|
|
|
<65 | 40 | 37.0 |
| ≥65 | 68 | 63.0 |
| Sex |
|
|
| Male | 62 | 57.4 |
|
Female | 46 | 42.6 |
| Laterality |
|
|
| Left | 49 | 45.4 |
|
Right | 58 | 53.7 |
| Both | 1 | 0.9 |
| Tumor site |
|
|
| Renal
pelvis | 42 | 38.9 |
|
Ureter | 55 | 50.9 |
|
Transitional zone | 10 | 9.3 |
| Renal
pelvis and ureter | 1 | 0.9 |
| Tumor size, cm |
|
|
|
<3.0 | 60 | 55.6 |
| ≥3.0 | 48 | 44.4 |
| Tumor grade |
|
|
| Low | 31 | 28.7 |
| High | 77 | 71.3 |
| Pathological
stage |
|
|
| NMI (pTa
+ pT1) | 43 | 39.8 |
| pTa | 33 | 30.6 |
| pT1 | 10 | 9.3 |
| MI (pT2
+ pT3 + pT4) | 65 | 60.2 |
|
pT2 | 26 | 24.1 |
|
pT3 | 31 | 28.7 |
|
pT4 | 8 | 7.4 |
| LVI |
|
|
| No | 87 | 80.6 |
|
Yes | 21 | 19.4 |
| Neural
invasion |
|
|
| No | 99 | 91.7 |
|
Yes | 9 | 8.3 |
| Lymph node
metastasis |
|
|
| No | 98 | 90.7 |
|
Yes | 10 | 9.3 |
| Concurrent CIS |
|
|
| No | 93 | 86.1 |
|
Yes | 15 | 13.9 |
| Extensive
necrosis |
|
|
| No | 91 | 84.3 |
|
Yes | 17 | 15.7 |
| Glomerular
sclerosis |
|
|
| No | 73 | 67.6 |
|
Yes | 35 | 32.4 |
| Solitary or
multifocality |
|
|
|
Solitary | 93 | 86.1 |
|
Multifocality | 15 | 13.9 |
| Bladder
cancera |
|
|
| No | 61 | 72.6 |
|
Simultaneous | 5 | 6.0 |
|
Postoperative | 16 | 19.0 |
|
Preoperative | 2 | 2.4 |
| Statusb,c |
|
|
|
Survival | 66 | 70.2 |
|
Death | 28 | 29.8 |
| Cancer-specific
survival, yearsc |
|
|
|
<1 | 19 | 20.2 |
|
1-3 | 41 | 43.6 |
|
>3 | 34 | 36.2 |
| Disease-free
survival, yearsc |
|
|
|
<1 | 36 | 38.3 |
|
1-3 | 31 | 33.0 |
|
>3 | 27 | 28.7 |
Construction of tissue microarrays
(TMAs)
TMAs were constructed from 108 samples of UTUC, and
29 samples of adjacent normal urothelium. A power calculation was
performed to support the inclusion of the number of samples. TMAs
were constructed with the three representative areas of the tumor,
which were selected manually. The representative areas were defined
as the areas with enough tumor cells, less necrosis, no extrusion
and heterogeneous regions if they exist. A previous study found
that ≥3 cores from each sample provides an acceptable statistical
analysis in TMAs in various tumor types (13), and so three 1-mm tissue cores from
each 10% neutral buffered formalin-fixed (12–48 h at room
temperature) paraffin-embedded donor block were selected and
precisely arrayed into a new recipient paraffin block.
Immunohistochemistry (IHC) of TMA and
scoring
A total of four serial 4-mm sections were prepared
from each TMA section. The first slide was stained with hematoxylin
(5 min) and eosin (1–3 min) at room temperature for hematoxylin to
confirm the presence of tumor cells, and subsequent slides were
used to evaluate the reactivity of primary antibodies, including
rabbit polyclonal anti-KDM6A antibody (1:100; cat. no. ab36938;
Abcam), mouse monoclonal anti-EZH2 antibody (1:150; cat. no. 6A10;
Origene Technologies, Inc.) and rabbit polyclonal anti-H3K27me3
antibody (1:150; cat. no. A2363; ABclonal Biotech Co., Ltd.). The
Tris-buffered saline without antibodies were used as a negative
control. Hydrogen peroxide (3%) was used as blocking reagent at
room temperature for 10 min. Primary antibodies were all incubated
for 4°C overnight. Secondary antibody for Pika (neat; cat. no.
PV-6,000; Origene Technologies, Inc.) was used at room temperature
for 30 min. Immunostaining results were evaluated independently by
two pathologists. Discrepancies in analysis were reconciled
following evaluation by a third reviewer. Semi-quantitative scoring
criteria, including staining intensity and proportion of positive
cells, were used for KDM6A, EZH2 and H3K27me3 staining evaluation.
The normal urothelium showed strong immunoreactivity for KDM6A,
allowing its use as a positive internal control. Tonsil tissue and
glioma cells served as positive controls for EZH2 and H3K27me3,
respectively. These tissues and cells were obtained from Peking
University Shougang Hospital (Beijing, China) (approval no.
IRBK-2017-047-01). Written informed consent was obtained from all
patients. Thus, staining intensity that was equivalent to a
positive internal control was graded as 3 (strong positive), with
lower levels of expression graded as 2 (positive) or 1 (weak
positive), and no detectable expression graded as 0 (negative).
Proportions of positive cells were graded as 0 (0–5% positive), 1
(6–25% positive), 2 (26–50% positive) or 3 (>50% positive). The
scores for staining intensity and proportion of positive cells were
multiplied to produce a weighted score for each case. A mean value
was calculated when heterogeneity was present in the three separate
TMA cores. Different cut-off values were investigated in the study
and the best ones selected. For KDM6A, the complete absence of
nuclear staining within tumor cells was considered negative. For
EZH2 and H3K27me3, cases with weighted scores of ≤1 were defined as
negative. All other scores were considered positive.
Statistical analysis
All data processing and statistical analyses were
performed using SPSS software (version 16.0; SPSS, Inc.). Pearson's
χ2 test and Fisher's exact test were used to analyze the
associations between the IHC staining results and the
clinicopathological parameters. The 3-year survival rates were
calculated using the Kaplan-Meier method. CSS and DFS analyses were
performed using the Kaplan-Meier method and log-rank test.
Multivariate predictive modeling was performed using multivariate
Cox regression analysis. Qualifying criteria for inclusion in the
Cox regression analysis were a P-value of ≤0.1 or a risk ratio of
≤0.5 or >2 on univariate Cox analysis. For the aforementioned
statistical tests, P<0.05 was used to indicate a statistically
significant difference. McNemar's test and Bonferroni's correction
were used to analyze the association between the expression levels
of any two out of the three proteins; a P-value <0.0167 (the
P-value equals 0.05 divided by 3) represented a significant
difference.
Results
Patient characteristics
UTUC specimens were obtained from 108 patients with
a male-to-female ratio of 1.35:1. The median age of the patients at
diagnosis was 70 (range, 41–86 years). Tumor grade was low in 31
patients and high in 77 patients. Low grade means the tumor is
composed of slender papillary structure with common papillary
branches and fusions and increased epithelial layers. The cell
abnormalities are not obvious under low magnification. While high
grade means the coated epithelium showed obvious disorder of
arrangement and cell atypia. Characteristically, there is striking
nuclear pleomorphism with variably sized and shaped hyperchromatic
nuclei. Mitoses are numerous and abnormal mitotic figures are
present. The prognosis of patients with muscle-invasive urothelial
carcinoma was worse than the patients with non-muscle invasive ones
and the clinical protocol was different for these two groups
(4,14). Thus, a total of 43 patients with
pathological stage pTa-pT1 were categorized as the non-muscle
invasive group, while 65 patients with stage pT2-pT4 were
categorized as the muscle invasive group. During the observation
period, complete follow-up information was obtained for 94 patients
and included 28 patients (29.8%) who died of tumor progression.
The data from all 108 patients were analyzed and
only those with follow-up data were included in the analysis of
prognosis. The clinicopathological characteristics of these
patients with UTUC are summarized in Table I.
Negative KDM6A expression status is
associated with aggressive clinical behavior in UTUC
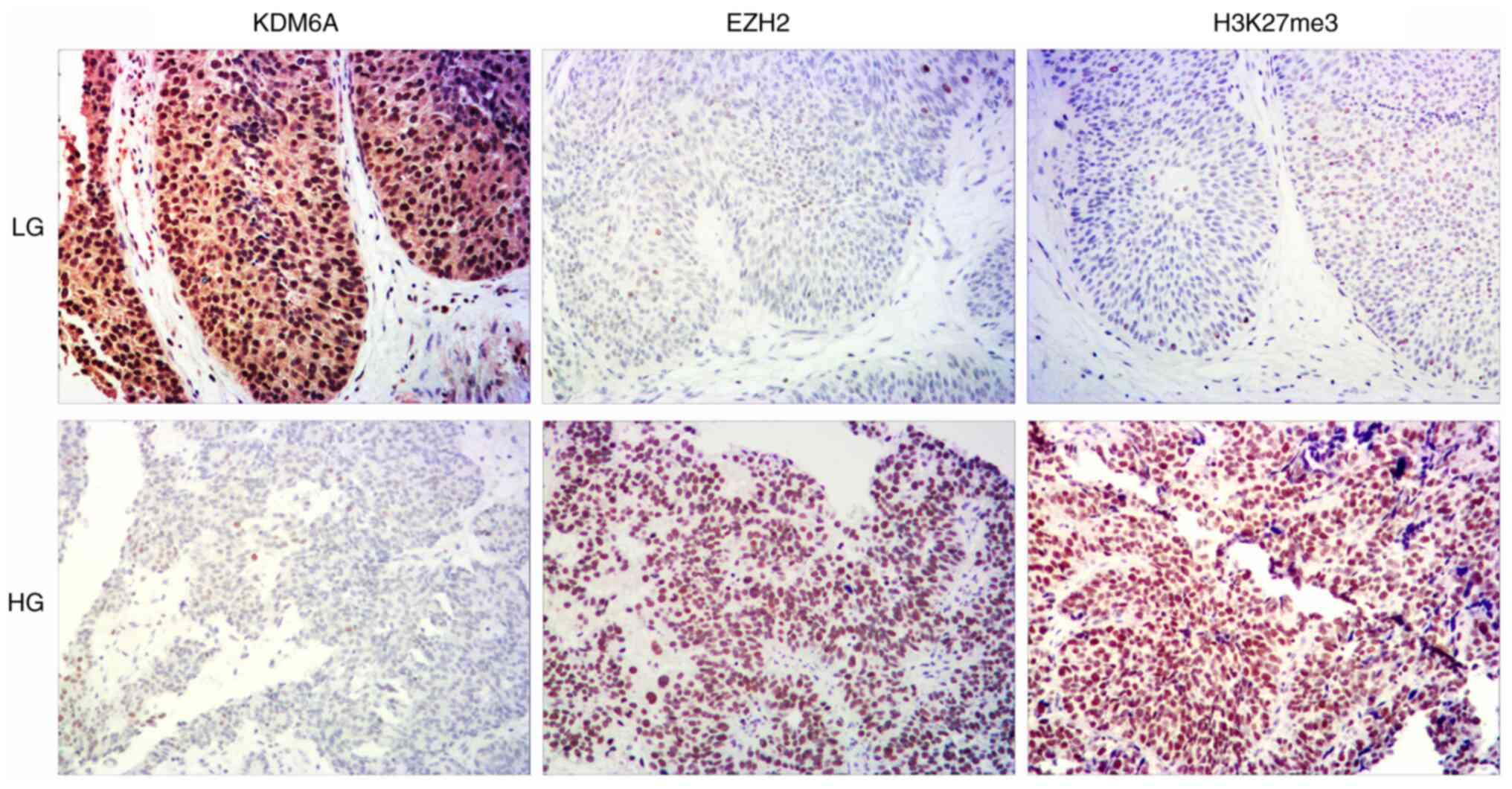
Immunohistochemical staining of KDM6A expression in
the TMAs is shown in Fig. 1 and
Fig. S1. Compared with that in
normal urothelial cells, the expression of KDM6A was significantly
lower in tumor cells (79.3 vs. 27.6%; P=0.033) (Table SI).
The association between KDM6A protein expression
level and patient clinicopathological parameters was also analyzed
(Table II). Low KDM6A expression
was associated with larger tumor size (36/48 vs. 34/60; P=0.047),
higher tumor grade (56/77 vs. 14/31; P=0.007) and advanced
pathological stage (48/65 vs. 22/43; P=0.016). There was no
association between KDM6A expression level and the other
parameters.
 | Table II.Clinicopathological characteristics
associated with KDM6A, EZH2 and H3K27me3 expression. |
Table II.
Clinicopathological characteristics
associated with KDM6A, EZH2 and H3K27me3 expression.
|
| KDM6A | EZH2 | H3K27me3 |
|---|
|
|
|
|
|
|---|
| Characteristic | -, n | +, n | P-value | -, n | +, n | P-value | -, n | +, n | P-value |
|---|
| Patient age,
years |
|
| 0.654 |
|
| 0.516 |
|
| 0.883 |
|
<65 | 27 | 13 |
| 18 | 22 |
| 20 | 20 |
|
|
≥65 | 43 | 25 |
| 35 | 33 |
| 35 | 33 |
|
| Sex |
|
| 1.000 |
|
| 0.316 |
|
| 0.868 |
|
Male | 40 | 22 |
| 33 | 29 |
| 32 | 30 |
|
|
Female | 30 | 16 |
| 20 | 26 |
| 23 | 23 |
|
| Laterality |
|
| 0.122 |
|
| 0.053 |
|
| 0.122 |
|
Left | 36 | 13 |
| 29 | 20 |
| 21 | 28 |
|
|
Right | 33 | 25 |
| 23 | 35 |
| 34 | 24 |
|
|
Both | 1 | 0 |
| 1 | 0 |
| 0 | 1 |
|
| Tumor site |
|
| 0.082 |
|
| 0.319 |
|
|
|
| Renal
pelvis | 32 | 10 |
| 24 | 18 |
| 24 | 18 | 0.518 |
|
Ureter | 31 | 24 |
| 23 | 32 |
| 26 | 29 |
|
|
Transitional zone | 7 | 3 |
| 5 | 5 |
| 4 | 6 |
|
| Renal
pelvis and ureter | 0 | 1 |
| 1 | 0 |
| 1 | 0 |
|
| Tumor size, cm |
|
| 0.047 |
|
| 0.863 |
|
| 0.344 |
|
<3.0 | 34 | 26 |
| 29 | 31 |
| 33 | 27 |
|
|
≥3.0 | 36 | 12 |
| 24 | 24 |
| 22 | 26 |
|
| Tumor grade |
|
| 0.007 |
|
| 0.001 |
|
| 0.073 |
|
Low | 14 | 17 |
| 23 | 8 |
| 20 | 11 |
|
|
High | 56 | 21 |
| 30 | 47 |
| 35 | 42 |
|
| Pathological
stage |
|
| 0.016 |
|
| 0.002 |
|
| 0.223 |
| NMI
(pTa-pT1) | 22 | 21 |
| 29 | 14 |
| 25 | 18 |
|
| MI
(pT2-pT4) | 48 | 17 |
| 24 | 41 |
| 30 | 35 |
|
| LVI |
|
| 0.126 |
|
| 0.882 |
|
| 0.736 |
| No | 53 | 34 |
| 43 | 44 |
| 45 | 42 |
|
|
Yes | 17 | 4 |
| 10 | 11 |
| 10 | 11 |
|
| Neural
invasion |
|
| 1.000 |
|
| 0.162 |
|
| 0.316 |
| No | 64 | 35 |
| 51 | 48 |
| 52 | 47 |
|
|
Yes | 6 | 3 |
| 2 | 7 |
| 3 | 6 |
|
| Lymph node
metastasis |
|
| 0.739 |
|
| 0.951 |
|
| 0.742 |
| No | 64 | 34 |
| 48 | 50 |
| 49 | 49 |
|
|
Yes | 6 | 4 |
| 5 | 5 |
| 6 | 4 |
|
| Concurrent CIS |
|
| 0.674 |
|
| 0.094 |
|
| 0.362 |
| No | 61 | 32 |
| 49 | 44 |
| 49 | 44 |
|
|
Yes | 9 | 6 |
| 4 | 11 |
| 6 | 9 |
|
| Extensive
necrosis |
|
| 0.408 |
|
| 0.033 |
|
| 0.856 |
| No | 57 | 34 |
| 49 | 42 |
| 46 | 45 |
|
|
Yes | 13 | 4 |
| 4 | 13 |
| 9 | 8 |
|
| Glomerular
sclerosis |
|
| 0.892 |
|
| 0.735 |
|
| 0.192 |
| No | 47 | 26 |
| 35 | 38 |
| 34 | 39 |
|
|
Yes | 23 | 12 |
| 18 | 17 |
| 21 | 14 |
|
| Solitary or
multifocality |
|
| 0.249 |
|
| 0.094 |
|
| 0.142 |
|
Solitary | 58 | 35 |
| 49 | 44 |
| 50 | 43 |
|
|
Multifocality | 12 | 3 |
| 4 | 11 |
| 5 | 10 |
|
| Bladder cancer |
|
| 0.842 |
|
| 0.116 |
|
| 0.701 |
| No | 35 | 26 |
| 29 | 32 |
| 32 | 29 |
|
|
Simultaneous | 3 | 2 |
| 5 | 0 |
| 3 | 2 |
|
|
Postoperative | 10 | 6 |
| 9 | 7 |
| 11 | 5 |
|
|
Preoperative | 2 | 0 |
| 1 | 1 |
| 1 | 1 |
|
Lower KDM6A expression level is a
useful prognostic factor for poor prognosis in patients with
UTUC
Of the 94 patients with complete follow-up
information, 66 patients (70.2%) were alive at a median follow-up
time of 35.5 months (range, 3–101 months). A total of 28 (29.8%)
patients died of the disease following a median period of 15 months
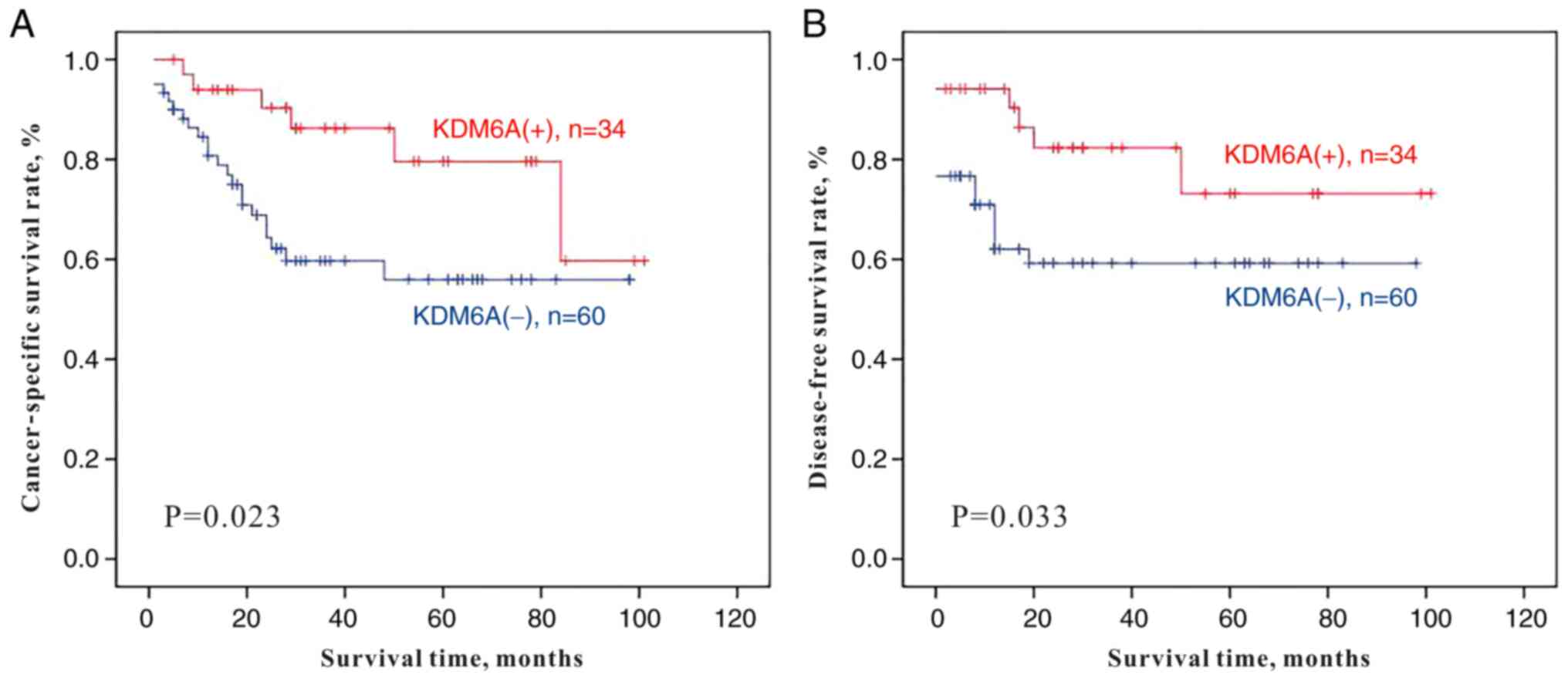
(range, 1–84 months). Kaplan-Meier single-factor analysis and the
log-rank test demonstrated a statistically significant decrease in
CSS (P=0.023) and DFS (P=0.033) times in patients with negative
staining for KDM6A (Table III;
Fig. 2). However, KDM6A was not an
independent risk factor for either CSS or DFS according to
multivariate Cox regression analysis. As expected, other adverse
clinicopathological parameters, such as high tumor grade, advanced
stage, presence of lymphovascular invasion and lymph node
metastasis were associated with both shorter CSS (P=0.036,
P<0.001, P=0.030 and P=0.002, respectively) and DFS (P=0.041,
P<0.001, P=0.031 and P=0.008, respectively) times. Furthermore,
only advanced stage was an independent prognostic factor for both
CSS and DFS times (P=0.013 and P=0.015, respectively).
 | Table III.Univariate and multivariate analyses
of CSS and DFS in patients with upper tract urothelial carcinoma
(n=108). |
Table III.
Univariate and multivariate analyses
of CSS and DFS in patients with upper tract urothelial carcinoma
(n=108).
|
| CSS | DFS |
|---|
|
|
|
|
|---|
|
| Univariate | Multivariate | Univariate | Multivariate |
|---|
|
|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| KDM6A (positive vs.
negative) | 0.364 | 0.147–0.904 | 0.023 | – | – | 0.272 | 0.404 | 0.163–1.000 | 0.033 | – | – | 0.272 |
| EZH2 (positive vs.
negative) | 4.083 | 1.653–10.087 | 0.002 | 0.205 | 0.074–0.567 | 0.022 | 3.608 | 1.462–8.904 | 0.005 | – | – | 0.058 |
| H3K27me3 (positive
vs. negative) | 1.508 | 0.708–3.209 | 0.287 | – | – | – | 1.420 | 0.672–3.002 | 0.359 | – | – | – |
| Grade (LG vs.
HG) | 36.575 | 1.262–1060 | 0.036 | – | – | 0.296 | 35.464 | 1.149–1094 | 0.041 | – | – | 0.301 |
| pT stage (pTa-T1
vs. pT2-T4) | 50.838 | 2.309–1119 | 0.001 | 0.020 | 0.001–0.433 | 0.013 | 48.004 | 2.107–1093 | 0.001 | 0.021 | 0.001–0.474 | 0.015 |
| LVI(yes vs.
no) | 2.379 | 1.089–5.200 | 0.030 | – | – | 0.404 | 2.345 | 1.082–5.086 | 0.031 | – | – | 0.328 |
| Neural invasion(yes
vs. no) | 1.994 | 0.687–5.787 | 0.204 | – | – | – | 1.785 | 0.617–5.165 | 0.285 | – | – | – |
| LN metastasis(yes
vs. no) | 4.734 | 1.751–12.802 | 0.002 | – | – | 0.047 | 3.793 | 1.422–10.123 | 0.008 | – | – | 0.099 |
| Concurrent CIS(yes
vs. no) | 0.883 | 0.305–2.554 | 0.818 | – | – | – | 0.882 | 0.306–2.545 | 0.816 | – | – | – |
Lower KDM6A expression level is
neither associated with a significant increase in H3K27me3
expression nor with EZH2 expression
To evaluate the association between KDM6A and EZH2,
and H3K27me3 expression levels in UTUC, the expression levels of
EZH2 and H3K27me3 in UTUC and the adjacent normal urothelium were
investigated. There was a significant increase in EZH2 expression
level in UTUC tumors compared with that in the normal urothelium
(P=0.035; Table SI). However,
H3K27me3 expression levels were similar between UTUC tumor and
normal urothelium samples (P=0.390; Table SI). In view of the fact that
KDM6A antagonizes EZH2 activity and functions as a
histone H3K27me3 demethylase, pairwise associations among the
expression levels of these three proteins were also analyzed.
However, there was no statistical association observed (Table IV).
 | Table IV.Pairwise association among the KDM6A,
EZH2 and H3K27me3 expression levels in upper tract urothelial
carcinoma. |
Table IV.
Pairwise association among the KDM6A,
EZH2 and H3K27me3 expression levels in upper tract urothelial
carcinoma.
|
|
| KDM6A |
|
|---|
|
|
|
|
|
|---|
| Expression | n | + (cutoff 0),
n |
P-valuea |
P-valueb |
|---|
|
EZH2− | 53 | 21 | 0.037 | 0.871 |
|
EZH2+ | 55 | 17 |
|
|
|
H3K27me3− | 55 | 27 | 0.092 |
|
|
H3K27me3+ | 53 | 11 |
|
|
The clinicopathological significance of EZH2 and
H3K27me3 expression levels was also analyzed (Table II). There was an association between
EZH2 expression levels and tumor grade (P=0.001), advanced
pathological stage (P=0.002) and extensive necrosis (P=0.033).
Using multivariate survival analysis, increased EZH2 expression
levels were a significant predictor for poor CSS time (P=0.022),
while there was no significance for DFS time (P=0.058) (Table III). Upon analyzing only H3K27me3
protein expression, its level was neither significantly associated
with any clinicopathological parameter nor was it of prognostic
value.
Discussion
A previous study has shown that KDM6A is the most
frequently mutated gene in AA-associated UTUCs, and this type of
cancer was found to be the most common UTUC in a Chinese cohort in
one study (3). However, currently
there is a lack of relevant research with respect to KDM6A
expression in patients with UTUC in China. To the best of our
knowledge, this is the first study in which the association between
KDM6A expression levels and clinicopathologically relevant features
of UTUC has been evaluated.
KDM6A mutations have been reported to be involved in
both hematological and solid neoplasms, including multiple myeloma,
clear cell renal cell carcinoma, esophageal squamous cell carcinoma
and bladder cancer (5,15–18);
however, the expression levels of KDM6A vary in the different tumor
types. Inactivating mutations and downregulation of KDM6A have been
described in a wide spectrum of bladder urothelial carcinomas
(4). Conversely, in breast cancer
and clear cell renal cell carcinoma, KDM6A overexpression and
upregulation has been associated with a poor prognosis (19–21).
These varying results suggest that the epigenetic function of KDM6A
in human cancer may depend on a number of factors, including the
type of cell involved in the cancer. In the present study, KDM6A
expression was significantly lower in Chinese patients with UTUC
compared with that in adjacent normal tissues, with adverse
pathological features and an unfavorable prognosis, which suggested
that lower KDM6A expression may accelerate UTUC progression.
Patients with UTUC in China were associated with
specific epidemiological characteristics (2). An observed epidemiological difference
between patients with UTUC in China and Western countries is sex
distribution (2). In Western
countries, urothelial carcinoma is more common in men than in women
(2). A previous study revealed that
the X chromosome protects against bladder cancer among women, via a
KDM6A-dependent epigenetic mechanism, and that decreased expression
of KDM6A predicts a poor prognosis in women with bladder cancer
(22). This suggests that KDM6A
could be a prototypical sex-biasing tumor suppressor (22). By contrast, the incidence rate of
UTUC in China is higher in women compared with that in men
(2). However, in the present study,
it was found that KDM6A was neither significantly associated with
sex or with the survival time of female patients with UTUC.
Therefore, the mechanisms underlying sex differences of urothelial
carcinoma associated with KDM6A, and the extent to which KDM6A
contributes to sex differences in Chinese patients with UTUC
warrant further research.
Geographic differences in risk factors for UTUC,
particularly AA consumption, may explain some differences in the
epidemiological and clinicopathological characteristics of patients
with UTUC in China and Western countries (3). Notably, high-throughput genome-wide
screening identified KDM6A as the most frequently mutated gene in
AA-associated UTUC (3). However,
patient information regarding AA usage was not available in the
present retrospective study, as it was difficult to trace back the
patient history of medication with Chinese herbs, which take
decades to cause nephrotoxicity and UTUC. Therefore, prospective
studies will be required to investigate the role of KDM6A in
Chinese patients with UTUC, particularly in AA-associated UTUC.
Previous studies have shown that homeostasis of
H3K27me3 levels is maintained by coordination of the demethylase
KDM6A and PRC2 complex, containing methyltransferase EZH2 (4,23), and
that this balance is disrupted when KDM6A is mutated. The
association between KDM6A and EZH2, as investigated in different
studies on various types of carcinoma, remains unclear. For
example, in human lung cancer, KDM6A expression was shown to be
inversely correlated with EZH2 expression (24). However, a study of chronic
myelomonocytic leukemia identified loss-of-function mutations in
both KDM6A and EZH2 despite their opposing roles in H3K27me3
regulation (25). In bladder cancer,
the levels of EZH2 expression were found to be similar in different
KDM6A gene status (4).
Consistent with the findings in bladder cancer, no statistical
correlation between KDM6A and EZH2 expression levels was found. A
possible explanation is that KDM6A may antagonize
EZH2 activity at the genetic level, but is not associated
with EZH2 at the protein expression level. Aside from the enzymatic
subunit of EZH2, the PRC2 complex also contains other cofactors,
such as SUZ12, EZH1 and EED (26).
It would therefore be of interest to investigate the expression
level of all the other core PRC components independent of EZH2
status in future studies.
The present study has some limitations. The study is
retrospective in nature, and the materials used were
paraffin-embedded tissues, which were not suitable for western
blotting or reverse transcription quantitative-PCR analysis.
Further investigations of the mechanism of KDM6A in UTUC
progression will therefore be analyzed using fresh UTUC tumor
tissues and cell lines in the next project.
In conclusion, a lower KDM6A expression level was
associated with adverse pathological features, such as a high grade
and advanced stage of UTUC, and was a significant predictor of a
poor prognosis in patients with UTUC. These data suggested that
immunohistochemical analysis of KDM6A provides an easier and more
efficient method, compared with complex genome screening procedures
used in other studies (3,4), for predicting the prognosis in patients
with UTUC in a clinical setting. KDM6A expression level was not
associated with EZH2 or H3K27me3 expression levels. Therefore,
lower KDM6A expression may not confer sensitivity to either EZH2
inhibition or EZH2 inhibitor-based therapy and remains to be
further investigated.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Dr Lei Wang
(Department of Urology, Peking University Wu Jieping Urology
Center, Peking University Shougang Hospital, Beijing, China) for
providing suggestions on the study design.
Funding
The present study was funded by the Leading Academic
Discipline Project of Beijing Education Bureau (grant no.
BMU20110254) and the Peking University Shougang Hospital Youth
Foundation (grant no. SGYYQ201701).
Availability of data and materials
The datasets analyzed during the present study are
available from the corresponding author on reasonable request.
Authors' contributions
YW and HYH designed the study and performed the
experiments, SFW and YZ collected and interpreted the
immunohistochemistry data, YW and JXZ analyzed the data, and YW and
HYH prepared the manuscript. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was performed according to the
principles of the Helsinki declaration. Experimental protocols were
reviewed and approved by the Ethics Committee of the Peking
University Shougang Hospital (approval no. IRBK-2017-047-01).
Written informed consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen XP, Xiong GY, Li XS, Matin SF, Garcia
M, Fang D, Wang TY, Yu W, Gong K, Song Y, et al: Predictive factors
for worse pathological outcomes of upper tract urothelial
carcinoma: Experience from a nationwide high-volume centre in
China. BJU Int. 112:917–924. 2013.PubMed/NCBI
|
|
2
|
Singla N, Fang D, Su X, Bao Z, Cao Z,
Jafri SM, Xiong G, Zhang L, Hutchinson R, Sagalowsky A, et al: A
multi-institutional comparison of clinicopathologic characteristics
and oncologic outcomes of upper tract urothelial carcinoma in China
and the United States. J Urol. 197:1208–1213. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Poon SL, Pang ST, McPherson JR, Yu W,
Huang KK, Guan P, Weng WH, Siew EY, Liu Y, Heng HL, et al:
Genome-wide mutational signatures of aristolochic acid and its
application as a screening tool. Sci Transl Med. 5:197ra1012013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ler LD, Ghosh S, Chai X, Thike AA, Heng
HL, Siew EY, Dey S, Koh LK, Lim JQ, Lim WK, et al: Loss of tumor
suppressor KDM6A amplifies PRC2-regulated transcriptional
repression in bladder cancer and can be targeted through inhibition
of EZH2. Sci Transl Med. 9:eaai83122017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Van der Meulen J, Speleman F and Van
Vlierberghe P: The H3K27me3 demethylase UTX in normal development
and disease. Epigenetics. 9:658–668. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nickerson ML, Dancik GM, Im KM, Edwards
MG, Turan S, Brown J, Ruiz-Rodriguez C, Owens C, Costello JC, Guo
G, et al: Concurrent alterations in TERT, KDM6A, and the BRCA
pathway in bladder cancer. Clin Cancer Res. 20:4935–4948. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Grasso CS, Wu YM, Robinson DR, Cao X,
Dhanasekaran SM, Khan AP, Quist MJ, Jing X, Lonigro RJ, Brenner JC,
et al: The mutational landscape of lethal castrate resistant
prostate cancer. Nature. 487:239–243. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kandoth C, McLellan MD, Vandin F, Ye K,
Niu B, Lu C, Xie M, Zhang Q, McMichael JF, Wyczalkowski MA, et al:
Mutational landscape and significance across 12 major cancer types.
Nature. 502:333–339. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lan F, Bayliss PE, Rinn JL, Whetstine JR,
Wang JK, Chen S, Iwase S, Alpatov R, Issaeva I, Canaani E, et al: A
histone H3 lysine 27 demethylase regulates animal posterior
development. Nature. 449:689–694. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hong S, Cho YW, Yu LR, Yu H, Veenstra TD
and Ge K: Identification of JmjC domain-containing UTX and JMJD3 as
histone H3 lysine 27 demethylases. Proc Natl Acad Sci USA.
104:18439–18444. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Moch H, Humphrey PA, Ulbright TM, Cubilla
AL and Reuter VE: WHO Classification of Tumours of the Urinary
System and Male Genital Organ. International Agency for Research on
Cancer (IARC); Lyon: pp. 1312016
|
|
12
|
Paner GP, Stadler WM, Hansel DE, Montironi
R, Lin DW and Amin MB: Updates in the eighth edition of the
Tumor-Node-Metastasis staging classification for urologic cancers.
Eur Uro. l73:560–569. 2018. View Article : Google Scholar
|
|
13
|
Rubin MA, Dunn R, Strawderman M and Pienta
KJ: Tissue microarray sampling strategy for prostate cancer
biomarker analysis. Am J Surg Pathol. 26:312–319. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li YR, Yu KJ, Chang YH, Lin PH, Shao IH,
Kan HC, Chu YC, Chuang CK, Pang ST and Liu CY: Predictors of
intravesical recurrence after radical nephroureterectomy and
prognosis in patients with upper tract urothelial carcinoma. Cancer
Manag Res. 12:7439–7450. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gossage L, Murtaza M, Slatter AF,
Lichtenstein CP, Warren A, Haynes B, Marass F, Roberts I, Shanahan
SJ, Claas A, et al: Clinical and pathological impact of VHL, PBRMI,
BAPI, SETD2, KDM6A, and JARIDIc in clear cell renal cell carcinoma.
Genes Chromosomes Cancer. 53:38–51. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ntziachiristos P, Tsirigos A, Welstead GG,
Trimarchi T, Bakogianni S, Xu L, Loizou E, Holmfeldt L, Strikoudis
A, King B, et al: Contrasting roles for histone 3 lysine 27
demethylases in acute lymphoblastic leukemia. Nature. 514:513–517.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bailey P, Chang DK, Nones K, Johns AL,
Patch AM, Gingras MC, Miller DK, Christ AN, Bruxner TJC, Quinn MC,
et al: Genomic analyses identify molecular subtypes if pancreatic
cancer. Nature. 531:47–52. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li SH, Lu HI, Huang WT, Tien WY, Lan YC,
Lin WC, Tsai HT and Chen CH: The prognostic significance of histone
demethylase UTX in esophageal squamous cell carcinoma. Int J Mol
Sci. 19:2972018. View Article : Google Scholar
|
|
19
|
Paolicchi E, Crea F, Farrar WL, Green JE
and Danesi R: Histone lysine demethylases in breast cancer. Crit
Rev Oncol Hematol. 86:97–103. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Curtis C, Shah SP, Chin SF, Turashvili G,
Rueda OM, Dunning MJ, Speed D, Lynch AG, Samarajiwa S, Yuan Y, et
al: The genomic and transcriptomic architecture of 2,000 breast
tumours reveals novel subgroups. Nature. 486:346–352. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang J, Liu L, Xi W, Long Q, Wang Y, Bai
Q, Xia Y, Xu J and Guo J: Prognostic value of UTX expression in
patients with clear cell renal cell carcinoma. Urol Oncol.
34:338.e19–27. 2016. View Article : Google Scholar
|
|
22
|
Kaneko S and Li X: X chromosome protects
against bladder cancer in females via a KDM6A-dependent epigenetic
mechanism. Sci Adv. 4:eaar55982018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Martinez-Garcia E and Licht JD:
Deregulation of H3K27 methylation in cancer. Nat Genet. 42:100–101.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wu Q, Tian Y, Zhang J, Tong X, Huang H, Li
S, Zhao H, Tang Y, Yuan C, Wang K, et al: In vivo CRISPR screening
unveils histone demethylase UTX as an important epigenetic
regulator in lung tumorigenesis. Proc Natl Acad Sci USA.
115:E3978–E3986. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jankowska AM, Makishima H, Tiu RV, Szpurka
H, Huang Y, Traina F, Visconte V, Sugimoto Y, Prince C, O'Keefe C,
et al: Mutational spectrum analysis of chronic myelomonocytic
leukemia includes genes associated with epigenetic regulation: UTX,
EZH2, and DNMT3A. Blood. 118:3932–3941. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim KH and Roberts CW: Targeting EZH2 in
cancer. Nat Med. 22:128–134. 2016. View
Article : Google Scholar : PubMed/NCBI
|