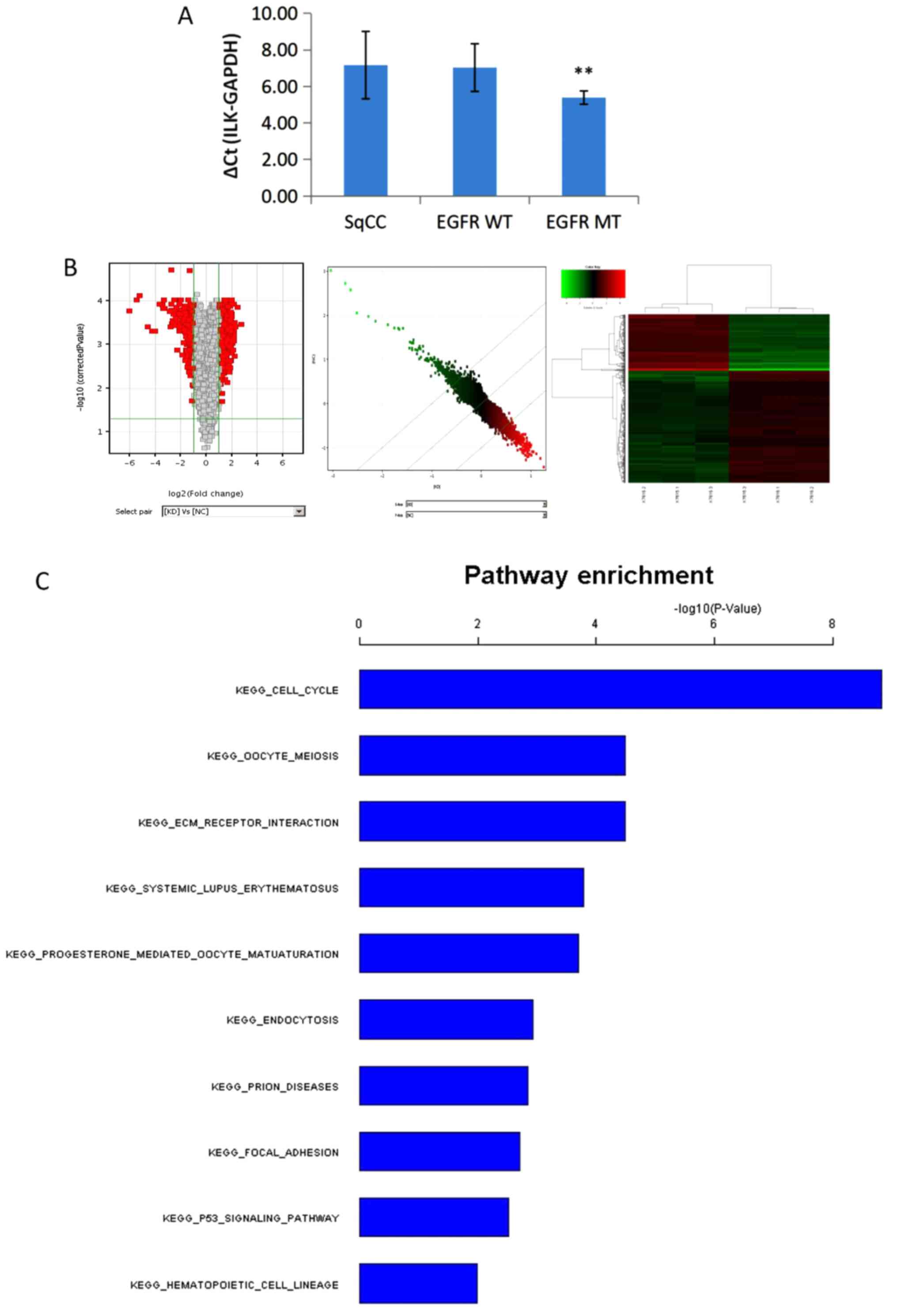
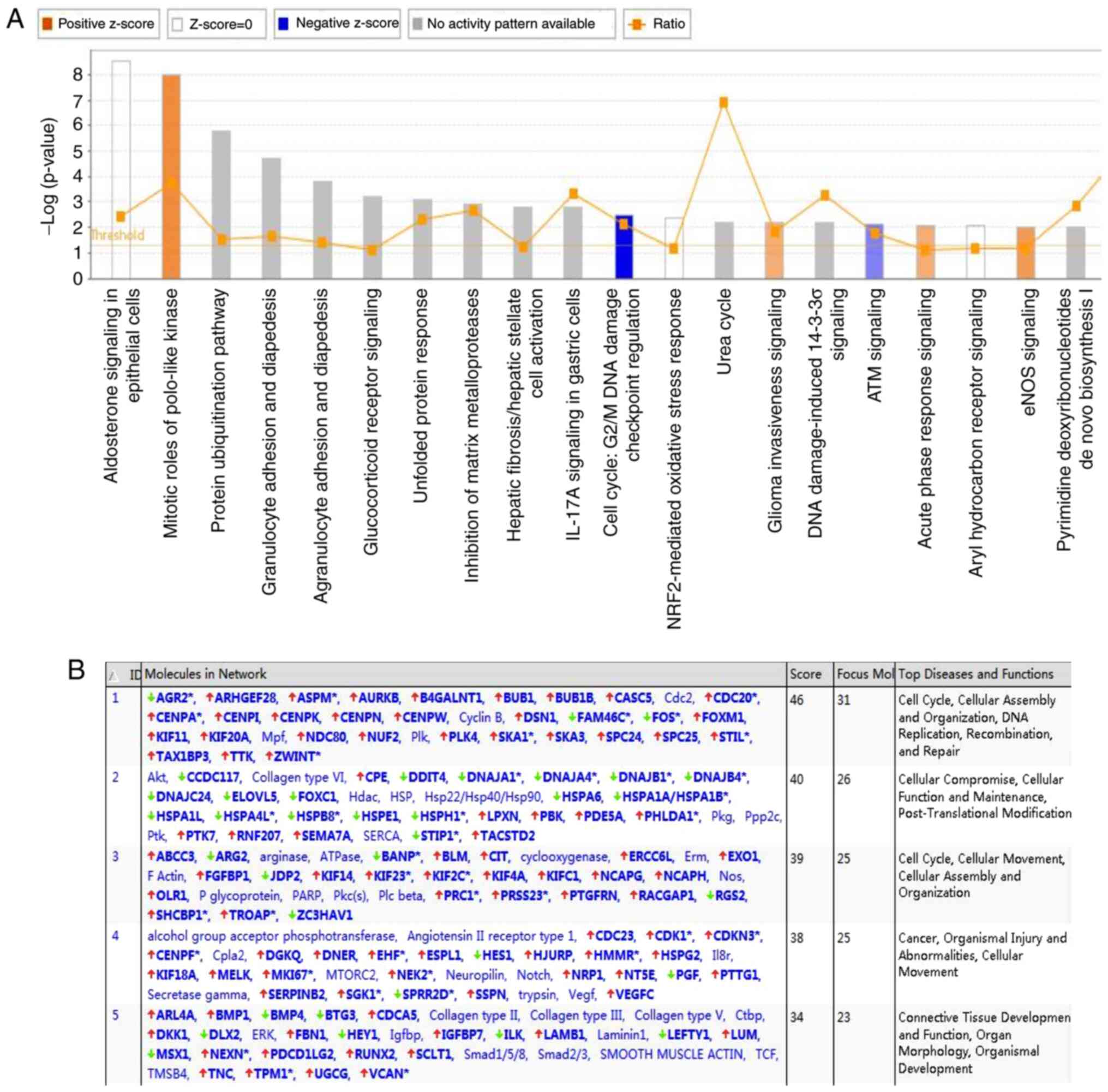
|
1
|
Wahbah M, Boroumand N, Castro C, El-Zeky F
and Eltorky M: Changing trends in the distribution of the
histologic types of lung cancer: A review of 4439 cases. Ann Diagn
Pathol. 11:89–96. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Camidge DR, Doebele RC and Kerr KM:
Comparing and contrasting predictive biomarkers for immunotherapy
and targeted therapy of NSCLC. Nat Rev Clin Oncol. 16:341–355.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sands JM, Nguyen T, Shivdasani P, Sacher
AG, Cheng ML, Alden RS, Jänne PA, Kuo FC, Oxnard GR and Sholl LM:
Next-generation sequencing informs diagnosis and identifies
unexpected therapeutic targets in lung squamous cell carcinomas.
Lung Cancer. 140:35–41. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
National Comprehensive Cancer Network
(NCCN). Clinical Practice Guidelines in Oncology. Plymouth; PA:
2018
|
|
5
|
Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho
BC, Turna HZ, Castro G Jr, Srimuninnimit V, Laktionov KK,
Bondarenko I, et al: Pembrolizumab versus chemotherapy for
previously untreated, PD-L1-expressing, locally advanced or
metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised,
open-label, controlled, phase 3 trial. Lancet. 393:1819–1830. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hochmair MJ, Morabito A, Hao D, Yang CT,
Soo RA, Yang JC, Gucalp R, Halmos B, Wang L, Märten A and Cufer T:
Sequential afatinib and osimertinib in patients with EGFR
mutation-positive non-small-cell lung cancer: Updated analysis of
the observational GioTag study. Future Oncol. 15:2905–2914. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Brahmer J, Reckamp KL, Baas P, Crinò L,
Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE,
Holgado E, et al: Nivolumab versus Docetaxel in advanced
squamous-cell non-small-cell lung cancer. N Engl J Med.
373:123–135. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Herbst RS, Baas P, Kim DW, Felip E,
Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, et al:
Pembrolizumab versus docetaxel for previously treated,
PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010):
A randomised controlled trial. Lancet. 387:1540–1550. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Solassol I, Pinguet F and Quantin X: FDA-
and EMA-approved tyrosine kinase inhibitors in advanced
EGFR-mutated non-small cell lung cancer: Safety,
tolerability, plasma concentration monitoring, and management.
Biomolecules. 9:6682019. View Article : Google Scholar
|
|
10
|
Hirsch FR, Varella-Garcia M, Bunn PA Jr,
Di Maria MV, Veve R, Bremmes RM, Barón AE, Zeng C and Franklin WA:
Epidermal growth factor receptor in non-small-cell lung carcinomas:
Correlation between gene copy number and protein expression and
impact on prognosis. J Clin Oncol. 21:3798–3807. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cancer Genome Atlas Research Network, .
Comprehensive genomic characterization of squamous cell lung
cancers. Nature. 489:519–525. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang Z, Shen Z, Li Z, Duan J, Fu S, Liu Z,
Bai H, Zhang Z, Zhao J, Wang X and Wang J: Activation of the
BMP-BMPR pathway conferred resistance to EGFR-TKIs inlung squamous
cell carcinoma patients with EGFR mutations. Proc Natl Acad Sci
USA. 112:9990–9995. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hata A, Katakami N, Yoshioka H, Kunimasa
K, Fujita S, Kaji R, Notohara K, Imai Y, Tachikawa R, Tomii K, et
al: How sensitive are epidermal growth factor receptor-tyrosine
kinase inhibitors for squamous cell carcinoma of the lung harboring
EGFR gene-sensitive mutations? J Thorac Oncol. 8:89–95. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shukuya T, Takahashi T, Kaira R, Ono A,
Nakamura Y, Tsuya A, Kenmotsu H, Naito T, Kaira K, Murakami H, et
al: Efficacy of gefitinib for non-adenocarcinoma non-small-cell
lung cancer patients harboring epidermal growth factor receptor
mutations: A pooled analysis of published reports. Cancer Sci.
102:1032–1037. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Filipits M: New developments in the
treatment of squamous cell lung cancer. Curr Opin Oncol.
26:152–158. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Schwaederle M, Elkin SK, Tomson BN, Carter
JL and Kurzrock R: Squamousness: Next-generation sequencing reveals
shared molecular features across squamous tumor types. Cell Cycle.
14:2355–2361. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Goss GD and Spaans JN: Epidermal growth
factor receptor inhibition in the management of squamous cell
carcinoma of the lung. Oncologist. 21:205–213. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Memon AA, Zhang H, Gu Y, Luo Q, Shi J,
Deng Z, Ma J and Ma W: EGFR with TKI-sensitive mutations in exon 19
is highly expressed and frequently detected in Chinese patients
with lung squamous carcinoma. Onco Targets Ther. 10:4607–4613.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ray P, Raghunathan K, Ahsan A, Allam US,
Shukla S, Basrur V, Veatch S, Lawrence TS, Nyati MK and Ray D:
Ubiquitin ligase SMURF2 enhances epidermal growth factor receptor
stability and tyrosine-kinase inhibitor resistance. J Biol Chem.
295:12661–12673. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lei T, Zhang L, Song Y, Wang B, Shen Y,
Zhang N and Yang M: miR-1262 Transcriptionally modulated by
an enhancer genetic variant improves efficiency of epidermal growth
factor receptor-tyrosine kinase inhibitors in advanced lung
adenocarcinoma. DNA Cell Biol. 39:1111–1118. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nilsson MB, Sun H, Robichaux J, Pfeifer M,
McDermott U, Travers J, Diao L, Xi Y, Tong P, Shen L, et al: A
YAP/FOXM1 axis mediates EMT-associated EGFR inhibitor resistance
and increased expression of spindle assembly checkpoint components.
Sci Transl Med. 12:eaaz45892020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rong X, Liang Y, Han Q, Zhao Y, Jiang G,
Zhang X, Lin X, Liu Y, Zhang Y, Han X, et al: Molecular mechanisms
of tyrosine kinase inhibitor resistance induced by
membranous/cytoplasmic/nuclear translocation of epidermal growth
factor receptor. J Thorac Oncol. 14:1766–1783. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pinheiro FD, Teixeira AF, de Brito BB, da
Silva FAF, Santos MLC and de Melo FF: Immunotherapy-new perspective
in lung cancer. World J Clin Oncol. 11:250–259. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Assi K, Bergstrom K, Vallance B, Owen D
and Salh B: Requirement of epithelial integrin-linked kinase for
facilitation of Citrobacter rodentium-induced colitis. BMC
Gastroenterol. 13:1372013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chen D, Zhang Y, Zhang X, Li J, Han B, Liu
S, Wang L, Ling Y, Mao S and Wang X: Overexpression of
integrin-linked kinase correlates with malignant phenotype in
non-small cell lung cancer and promotes lung cancer cell invasion
and migration via regulating epithelial-mesenchymal transition
(EMT)-related genes. Acta Histochem. 115:128–136. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Espinoza I and Miele L: Deadly crosstalk:
Notch signaling at the intersection of EMT and cancer stem cells.
Cancer Lett. 341:41–45. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yu J, Shi R, Zhang D, Wang E and Qiu X:
Expression of integrin-linked kinase in lung squamous cell
carcinoma and adenocarcinoma: Correlation with E-cadherin
expression, tumor microvessel density and clinical outcome.
Virchows Arch. 458:99–107. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Posch F, Setinek U, Flores RM, Bernhard D,
Hannigan GE, Mueller MR and Watzka SB: Serum integrin-linked kinase
(sILK) concentration and survival in non-small cell lung cancer: A
pilot study. Clin Transl Oncol. 16:455–462. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jia Z: Role of integrin-linked kinase in
drug resistance of lung cancer. Onco Targets Ther. 8:1561–1565.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Augustin A, Lamerz J, Meistermann H,
Golling S, Scheiblich S, Hermann JC, Duchateau-Nguyen G, Tzouros M,
Avila DW, Langen H, et al: Quantitative chemical proteomics
profiling differentiates erlotinib from gefitinib in EGFR wild-type
non-small cell lung carcinoma cell lines. Mol Cancer Ther.
12:520–529. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu M, Xu S, Wang Y, Li Y, Li Y, Zhang H,
Liu H and Chen J: PD 0332991, a selective cyclin D kinase 4/6
inhibitor, sensitizes lung cancer cells to treatment with epidermal
growth factor receptor tyrosine kinase inhibitors. Oncotarget.
7:84951–84964. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dermawan JK, Gurova K, Pink J, Dowlati A,
De S, Narla G, Sharma N and Stark GR: Quinacrine overcomes
resistance to erlotinib by inhibiting FACT, NF-κB, and cell-cycle
progression in non-small cell lung cancer. Mol Cancer Ther.
13:2203–2214. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kaylan KB, Gentile SD, Milling LE, Bhinge
KN, Kosari F and Underhill GH: Mapping lung tumor cell drug
responses as a function of matrix context and genotype using cell
microarrays. Integr Biol (Camb). 8:1221–1231. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Wang B, Chu H and Yao Y: Intrinsic
resistance to EGFR tyrosine kinase inhibitors in advanced
non-small-cell lung cancer with activating EGFR mutations. Onco
Targets Ther. 9:3711–3726. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Salmela AL and Kallio MJ: Mitosis as an
anti-cancer drug target. Chromosoma. 122:431–449. 2013. View Article : Google Scholar : PubMed/NCBI
|