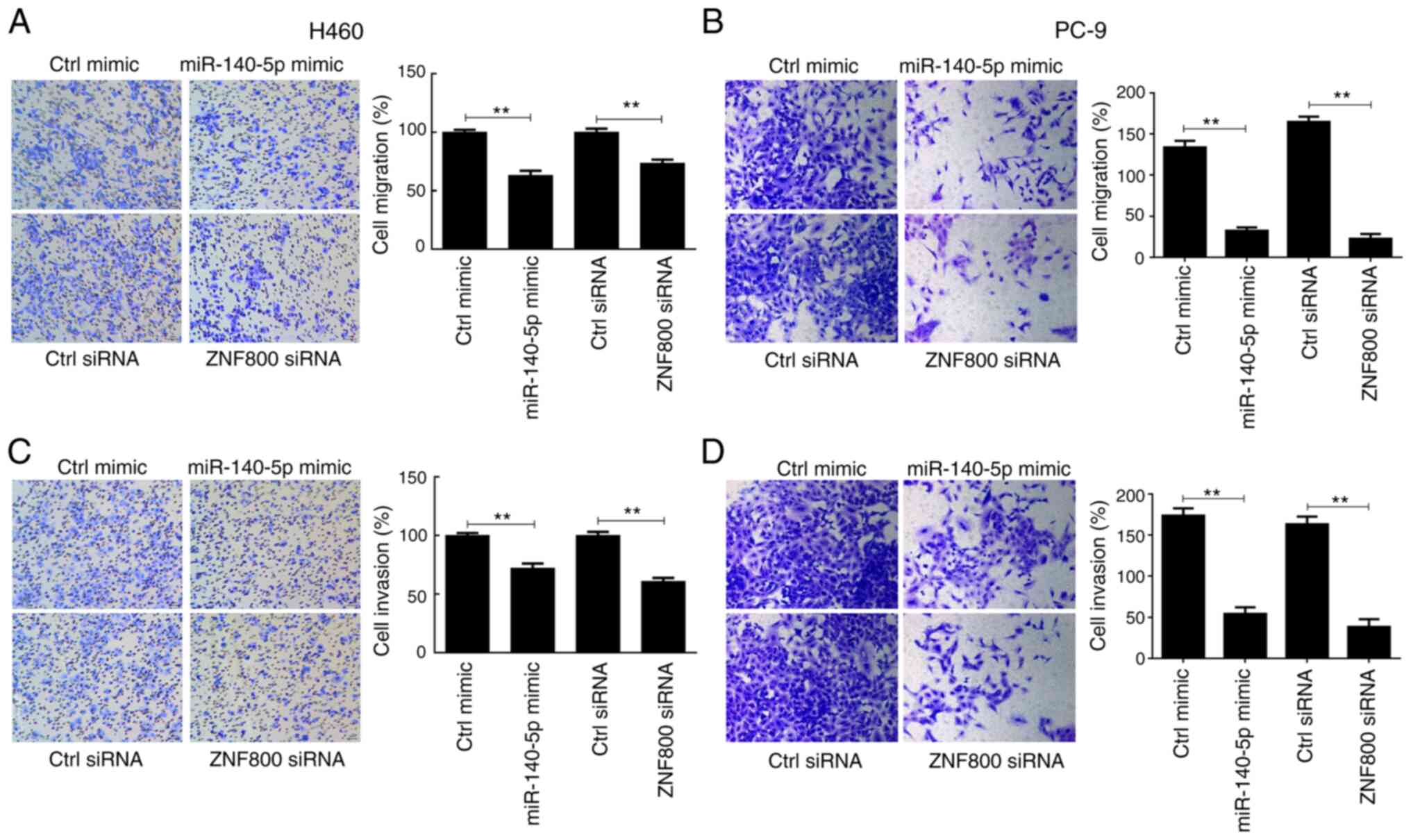
|
1
|
Nanavaty P, Alvarez MS and Alberts WM:
Lung cancer screening: advantages, controversies, and applications.
Cancer Control. 21:9–14. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olak J and Colson Y: Gender differences in
lung cancer: Have we really come a long way, baby? J Thorac
Cardiovasc Surg. 128:346–351. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chang YC, Chiou J, Yang YF, Su CY, Lin YF,
Yang CN, Lu PJ, Huang MS, Yang CJ and Hsiao M: Therapeutic
Targeting of Aldolase A Interactions Inhibits Lung Cancer
Metastasis and Prolongs Survival. Cancer Res. 79:4754–4766.
2019.PubMed/NCBI
|
|
4
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Miranda-Filho A, Piñeros M and Bray F: The
descriptive epidemiology of lung cancer and tobacco control: A
global overview 2018. Salud Publica Mex. 61:219–229. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thawani R, McLane M, Beig N, Ghose S,
Prasanna P, Velcheti V and Madabhushi A: Radiomics and
radiogenomics in lung cancer: A review for the clinician. Lung
Cancer. 115:34–41. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kambayashi Y, Fujimura T, Hidaka T and
Aiba S: Biomarkers for predicting efficacies of anti-PD1
antibodies. Front Med (Lausanne). 6:1742019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hjelmborg J, Korhonen T, Holst K, Skytthe
A, Pukkala E, Kutschke J, Harris JR, Mucci LA, Christensen K, Czene
K, et al Nordic Twin Study of Cancer (NorTwinCan) collaboration, :
Lung cancer, genetic predisposition and smoking: The Nordic Twin
Study of Cancer. Thorax. 72:1021–1027. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chudgar NP, Bucciarelli PR, Jeffries EM,
Rizk NP, Park BJ, Adusumilli PS and Jones DR: Results of the
national lung cancer screening trial: Where are we now? Thorac Surg
Clin. 25:145–153. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cong L, Zhao Y, Pogue AI and Lukiw WJ:
Role of microRNA (miRNA) and viroids in lethal diseases of plants
and animals. potential contribution to human neurodegenerative
disorders. Biochemistry (Mosc). 83:1018–1029. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bahreini F, Rayzan E and Rezaei N:
microRNA-related single-nucleotide polymorphisms and breast cancer.
J Cell Physiol. Jul 27–2020.(Epub ahead of print). doi:
10.1002/jcp.29966. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang Y, He R, An J, Deng P, Huang L and
Yang W: lncRNA XIST interacts with miR-140 to modulate lung cancer
growth by targeting iASPP. Oncol Rep. 38:941–948. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu M, Wang G, Tian W, Deng Y and Xu Y:
MiRNA-based therapeutics for lung cancer. Curr Pharm Des.
23:5989–5996. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhao JP, Jiang XL, Zhang BY and Su XH:
Involvement of microRNA-mediated gene expression regulation in the
pathological development of stem canker disease in Populus
trichocarpa. PLoS One. 7:e449682012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ling B, Wang GX, Long G, Qiu JH and Hu ZL:
Tumor suppressor miR-22 suppresses lung cancer cell progression
through post-transcriptional regulation of ErbB3. J Cancer Res Clin
Oncol. 138:1355–1361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Turchinovich A, Weiz L, Langheinz A and
Burwinkel B: Characterization of extracellular circulating
microRNA. Nucleic Acids Res. 39:7223–7233. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kropp J, Salih SM and Khatib H: Expression
of microRNAs in bovine and human pre-implantation embryo culture
media. Front Genet. 5:912014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang X, Wang T, Chen C, Wu Z, Bai P, Li S,
Chen B, Liu R, Zhang K, Li W, et al: Serum exosomal miR-210 as a
potential biomarker for clear cell renal cell carcinoma. J Cell
Biochem. Oct 10–2018.(Epub ahead of print). doi:
1002/jcb.27347.
|
|
20
|
Yang F, Ning Z, Ma L, Liu W, Shao C, Shu Y
and Shen H: Exosomal miRNAs and miRNA dysregulation in
cancer-associated fibroblasts. Mol Cancer. 16:1482017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sueta A, Yamamoto Y, Tomiguchi M,
Takeshita T, Yamamoto-Ibusuki M and Iwase H: Differential
expression of exosomal miRNAs between breast cancer patients with
and without recurrence. Oncotarget. 8:69934–69944. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dong F, Xu T, Shen Y, Zhong S, Chen S,
Ding Q and Shen Z: Dysregulation of miRNAs in bladder cancer:
Altered expression with aberrant biogenesis procedure. Oncotarget.
8:27547–27568. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu L, Lai X, Yuan C, Lv X, Yu T, He W,
Liu J and Zhang H: Aberrant expression of miR-153 is associated
with the poor prognosis of cervical cancer. Oncol Lett.
15:9183–9187. 2018.PubMed/NCBI
|
|
24
|
Zhang W, Zou C, Pan L, Xu Y, Qi W, Ma G,
Hou Y and Jiang P: MicroRNA-140-5p inhibits the progression of
colorectal cancer by targeting VEGFA. Cell Physiol Biochem.
37:1123–1133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hu Y, Li Y, Wu C, Zhou L, Han X, Wang Q,
Xie X, Zhou Y and Du Z: MicroRNA-140-5p inhibits cell proliferation
and invasion by regulating VEGFA/MMP2 signaling in glioma. Tumour
Biol. 39:10104283176975582017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li W and He F: Monocyte to macrophage
differentiation-associated (MMD) targeted by miR-140-5p regulates
tumor growth in non-small cell lung cancer. Biochem Biophys Res
Commun. 450:844–850. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang P, Xiong J, Zuo L, Liu K and Zhang H:
miR 140 5p regulates cell migration and invasion of non small cell
lung cancer cells through targeting VEGFA. Mol Med Rep.
18:2866–2872. 2018.PubMed/NCBI
|
|
28
|
Jing P, Sa N, Liu X, Liu X and Xu W:
MicroR-140-5p suppresses tumor cell migration and invasion by
targeting ADAM10-mediated Notch1 signaling pathway in
hypopharyngeal squamous cell carcinoma. Exp Mol Pathol.
100:132–138. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lu Y, Qin T, Li J, Wang L, Zhang Q, Jiang
Z and Mao J: MicroRNA-140-5p inhibits invasion and angiogenesis
through targeting VEGF-A in breast cancer. Cancer Gene Ther.
24:386–392. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cha Y, He Y, Ouyang K, Xiong H, Li J and
Yuan X: MicroRNA-140-5p suppresses cell proliferation and invasion
in gastric cancer by targeting WNT1 in the WNT/β-catenin signaling
pathway. Oncol Lett. 16:6369–6376. 2018.PubMed/NCBI
|
|
31
|
Fang Z, Yin S, Sun R, Zhang S, Fu M, Wu Y,
Zhang T, Khaliq J and Li Y: miR-140-5p suppresses the
proliferation, migration and invasion of gastric cancer by
regulating YES1. Mol Cancer. 16:1392017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
He Y, Deng F, Zhao S, Zhong S, Zhao J,
Wang D, Chen X, Zhang J, Hou J, Zhang W, et al: Analysis of
miRNA-mRNA network reveals miR-140-5p as a suppressor of breast
cancer glycolysis via targeting GLUT1. Epigenomics. 11:1021–1036.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chang QQ, Chen CY, Chen Z and Chang S:
LncRNA PVT1 promotes proliferation and invasion through enhancing
Smad3 expression by sponging miR-140-5p in cervical cancer. Radiol
Oncol. 53:443–452. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cunningham F, Achuthan P, Akanni W, Allen
J, Amode MR, Armean IM, Bennett R, Bhai J, Billis K, Boddu S, et
al: Ensembl 2019. Nucleic Acids Res. 47:D745–D751. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang Y, Xu R, Li G, Xie X, Long J and
Wang H: Loss of expression of the differentially expressed in
adenocarcinoma of the lung (DAL-1) protein is associated with
metastasis of non-small cell lung carcinoma cells. Tumour Biol.
33:1915–1925. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sano Y, Hashimoto E, Nakatani N, Abe M,
Satoh Y, Sakata K, Fujii T, Fujimoto-Ouchi K, Sugimoto M, Nagahashi
S, et al: Combining onartuzumab with erlotinib inhibits growth of
non-small cell lung cancer with activating EGFR mutations and HGF
overexpression. Mol Cancer Ther. 14:533–541. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Crone M, Hallman K, Lloyd V, Szmyd M,
Badamo B, Morse M and Dinda S: The antiestrogenic effects of black
cohosh on BRCA1 and steroid receptors in breast cancer cells.
Breast Cancer (Dove Med Press). 11:99–110. 2019.PubMed/NCBI
|
|
39
|
Lee YS and Dutta A: MicroRNAs in cancer.
Annu Rev Pathol. 4:199–227. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Billeter AT, Barnett RE, Druen D, Polk HC
Jr and van Berkel VH: MicroRNA as a new factor in lung and
esophageal cancer. Semin Thorac Cardiovasc Surg. 24:155–165. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Tong X, Su P, Yang H, Chi F, Shen L, Feng
X, Jiang H, Zhang X and Wang Z: MicroRNA-598 inhibits the
proliferation and invasion of non-small cell lung cancer cells by
directly targeting ZEB2. Exp Ther Med. 16:5417–5423.
2018.PubMed/NCBI
|
|
42
|
Lamichhane SR, Thachil T, De Ieso P, Gee
H, Moss SA and Milic N: Prognostic role of MicroRNAs in human
non-small-cell lung cancer: A systematic review and meta-analysis.
Dis Markers. 2018:83090152018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Jeong HC, Kim EK, Lee JH, Lee JM, Yoo HN
and Kim JK: Aberrant expression of let-7a miRNA in the blood of
non-small cell lung cancer patients. Mol Med Rep. 4:383–387.
2011.PubMed/NCBI
|
|
44
|
Jia Z, Zhang Y, Xu Q, Guo W and Guo A:
miR-126 suppresses epithelial-to-mesenchymal transition and
metastasis by targeting PI3K/AKT/Snail signaling of lung cancer
cells. Oncol Lett. 15:7369–7375. 2018.PubMed/NCBI
|
|
45
|
Huang H, Wang Y, Li Q, Fei X, Ma H and Hu
R: miR-140-3p functions as a tumor suppressor in squamous cell lung
cancer by regulating BRD9. Cancer Lett. 446:81–89. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Flamini V, Dudley E, Jiang WG and Cui Y:
Distinct mechanisms by which two forms of miR-140 suppress the
malignant properties of lung cancer cells. Oncotarget.
9:36474–36491. 2018. View Article : Google Scholar : PubMed/NCBI
|