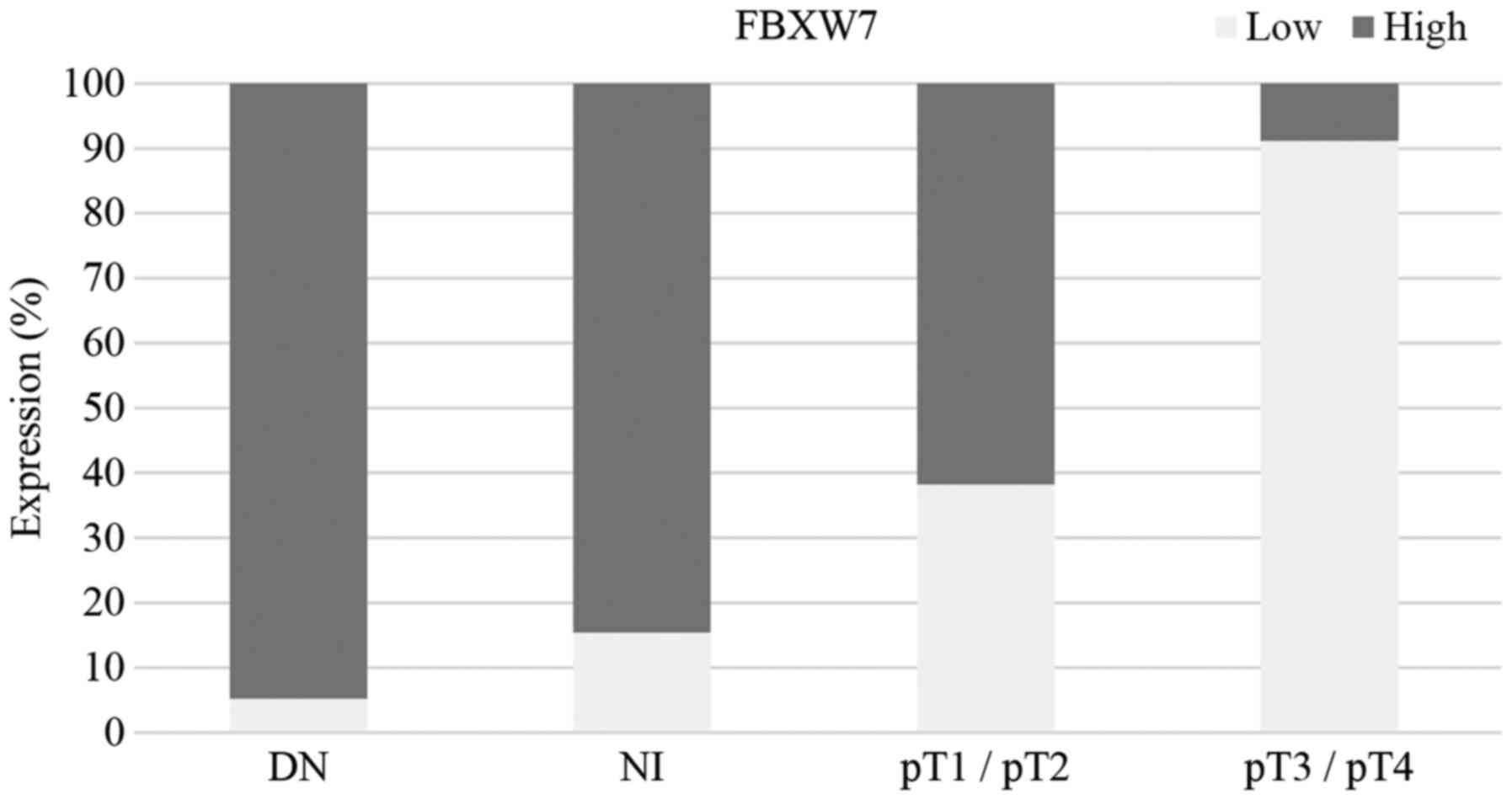
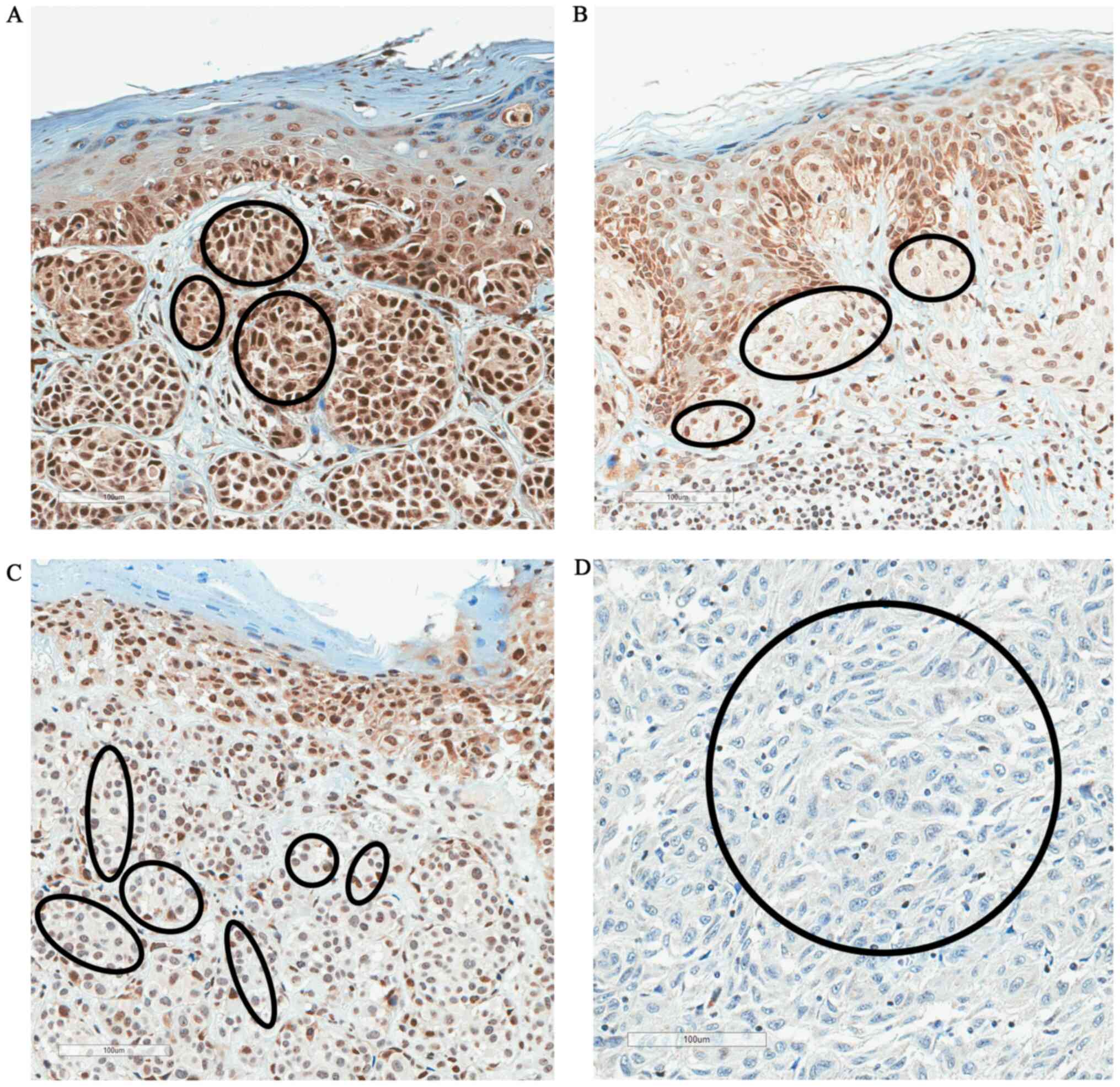
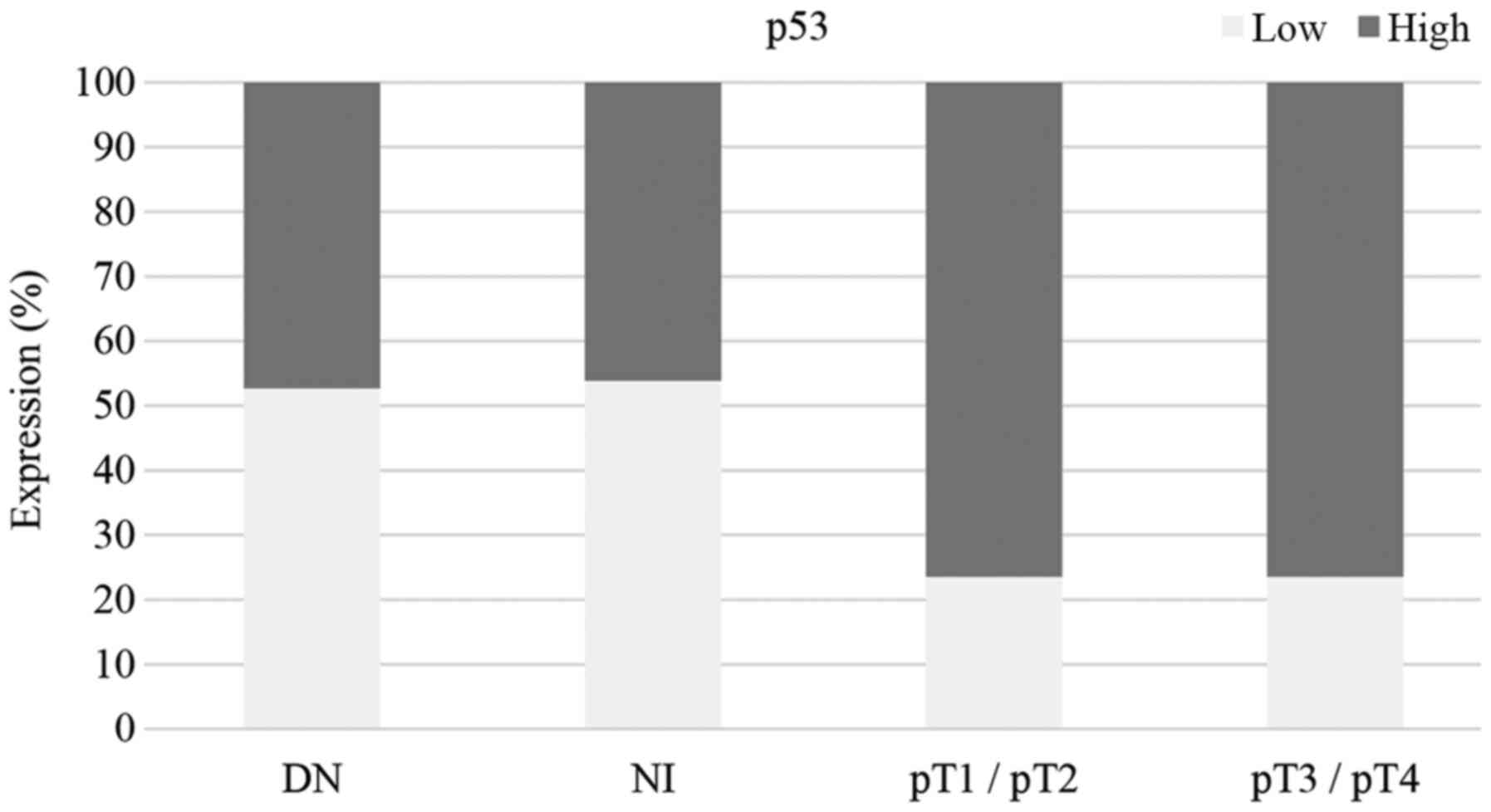
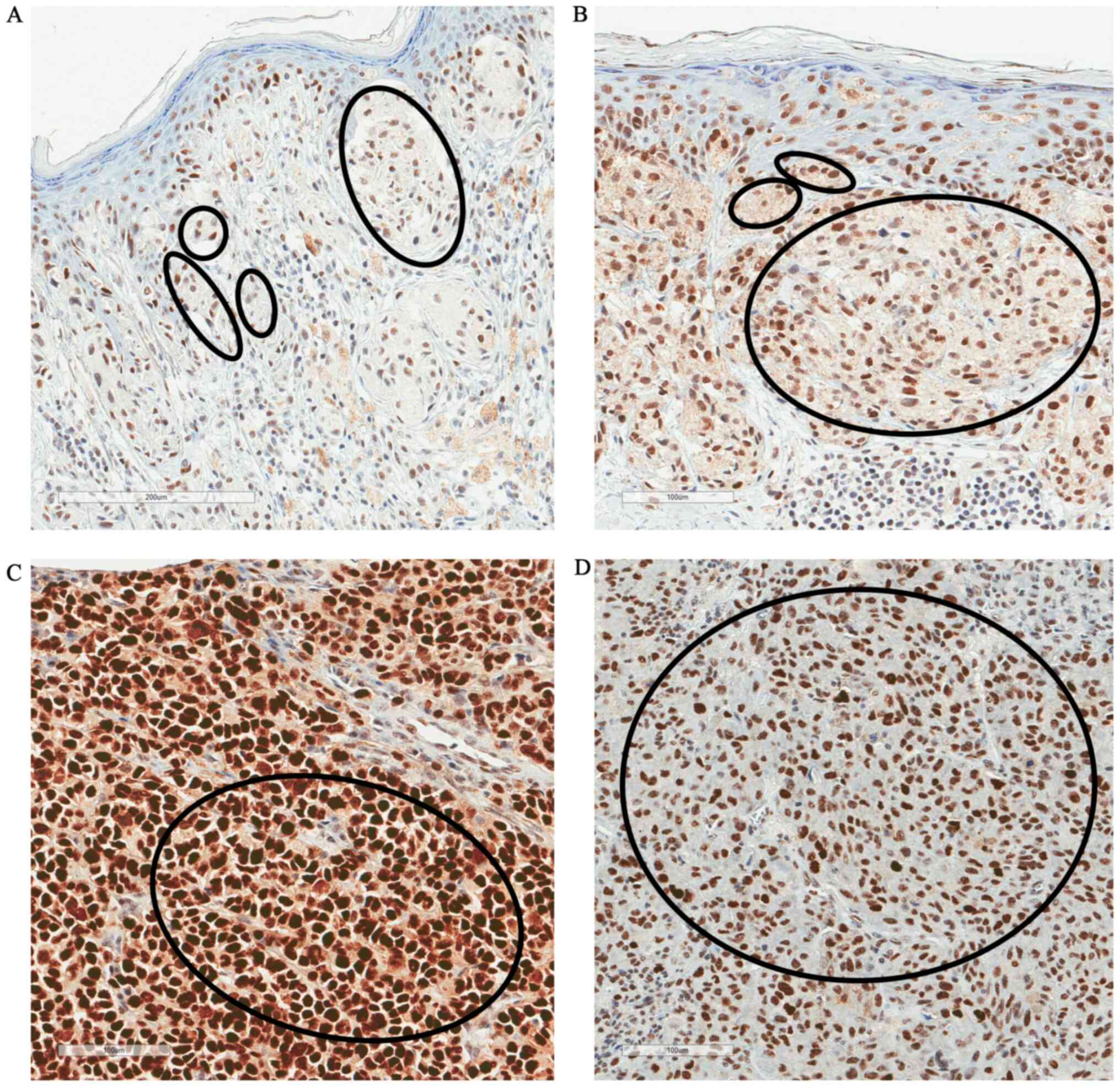
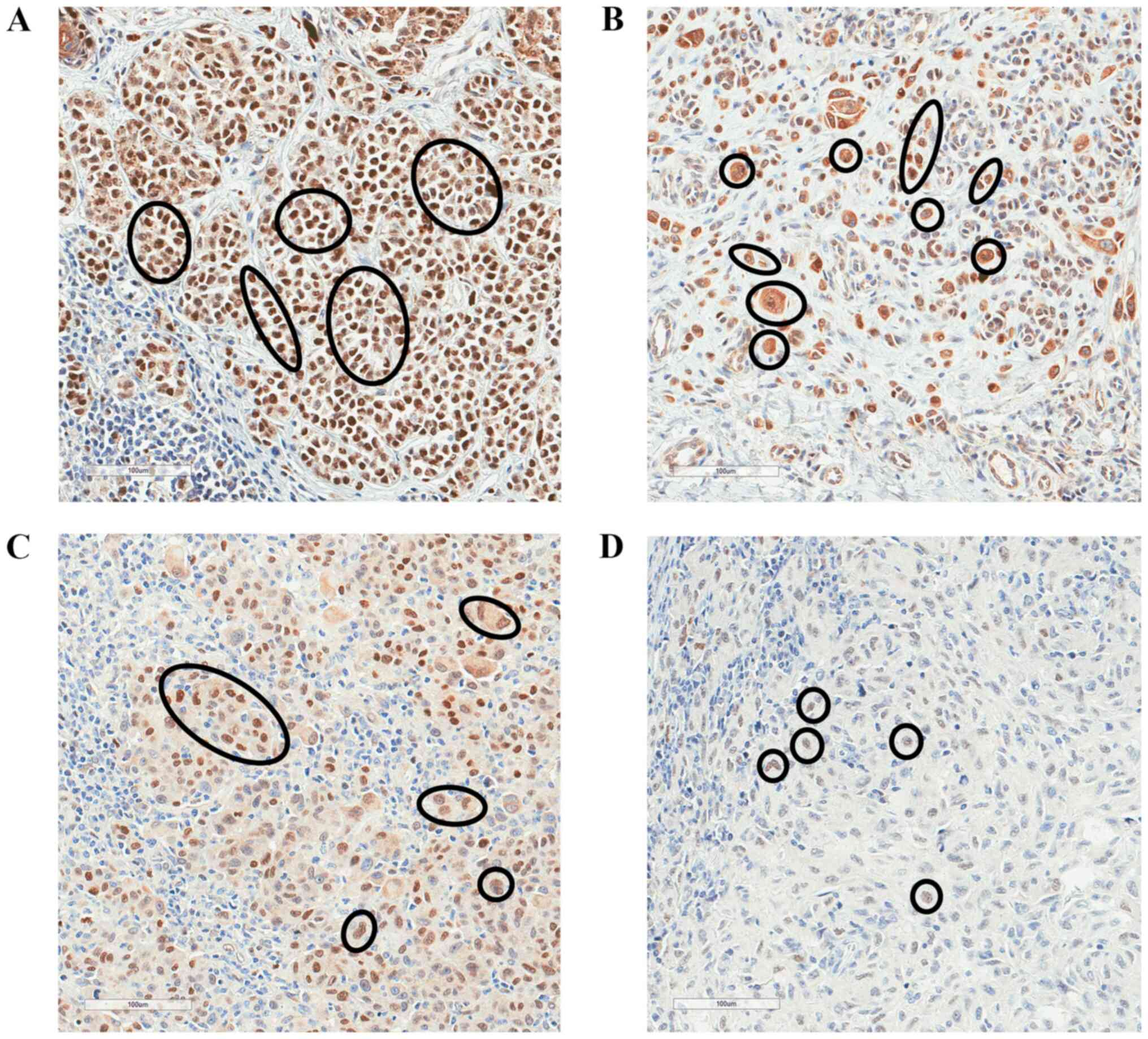
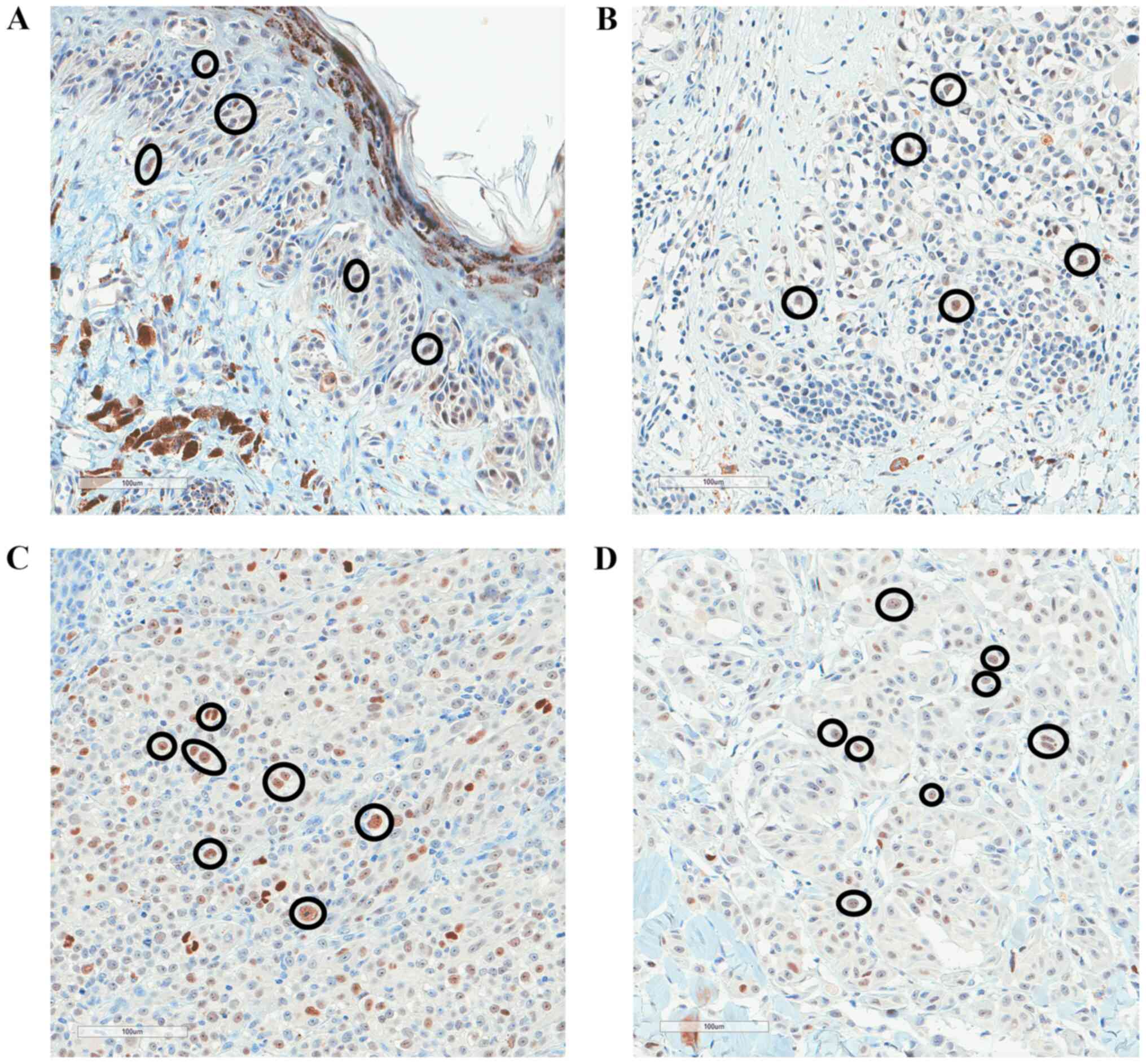
|
1
|
Morrow JK, Lin HK, Sun SC and Zhang S:
Targeting ubiquitination for cancer therapies. Future Med Chem.
7:2333–2350. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ma J, Guo W and Li C: Ubiquitination in
melanoma pathogenesis and treatment. Cancer Med. 6:1362–1377. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Aydin IT, Melamed RD, Adams SJ,
Castillo-Martin M, Demir A, Bryk D, Brunner G, Cordon-Cardo C,
Osman I, Rabadan R, et al: FBXW7 mutations in melanoma and a new
therapeutic paradigm. J Natl Cancer Inst. 106:dju1072014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xie CM, Wei W and Sun Y: Role of
SKP1-CUL1-F-box-protein (SCF) E3 ubiquitin ligases in skin cancer.
J Genet Genomics. 40:97–106. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Welcker M and Clurman BE: FBW7 ubiquitin
ligase: A tumour suppressor at the crossroads of cell division,
growth and differentiation. Nat Rev Cancer. 8:83–93. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang Z, Inuzuka H, Zhong J, Wan L,
Fukushima H, Sarkar FH and Wei W: Tumor suppressor functions of
FBW7 in cancer development and progression. FEBS Lett.
586:1409–1418. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang HL, Weng HY, Wang LQ, Yu CH, Huang
QJ, Zhao PP, Wen JZ, Zhou H and Qu LH: Triggering Fbw7-mediated
proteasomal degradation of c-Myc by oridonin induces cell growth
inhibition and apoptosis. Mol Cancer Ther. 11:1155–1165. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu H, Wang K, Fu H and Song J: Low
expression of the ubiquitin ligase FBXW7 correlates with poor
prognosis of patients with colorectal cancer. Int J Clin Exp
Pathol. 11:413–419. 2018.PubMed/NCBI
|
|
9
|
Cheng Y, Chen G, Martinka M, Ho V and Li
G: Prognostic significance of Fbw7 in human melanoma and its role
in cell migration. J Invest Dermatol. 133:1794–1802. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang L, Ye X, Liu Y, Wei W and Wang Z:
Aberrant regulation of FBW7 in cancer. Oncotarget. 5:2000–2015.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mao JH, Perez-Losada J, Wu D, Delrosario
R, Tsunematsu R, Nakayama KI, Brown K, Bryson S and Balmain A:
Fbxw7/Cdc4 is a p53-dependent, haploinsufficient tumour suppressor
gene. Nature. 432:775–779. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pei D, Zhang Y and Zheng J: Regulation of
p53: A collaboration between Mdm2 and Mdmx. Oncotarget. 3:228–235.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Madan E, Gogna R, Bhatt M, Pati U,
Kuppusamy P and Mahdi AA: Regulation of glucose metabolism by p53:
Emerging new roles for the tumor suppressor. Oncotarget. 2:948–957.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Muller PAJ and Vousden KH: p53 mutations
in cancer. Nat Cell Biol. 15:2–8. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wade M, Li Y and Wahl G: MDM2, MDMX and
p53 in oncogenesis and cancer therapy. Nat Rev Cancer. 13:83–96.
2013. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Olivier M, Goldgar DE, Sodha N, Ohgaki H,
Kleihues P, Hainaut P and Eeles RA: Li-Fraumeni and related
syndromes: Correlation between tumor type, family structure, and
TP53 genotype. Cancer Res. 63:6643–6650. 2003.PubMed/NCBI
|
|
17
|
Birch JM, Alston RD, McNally RJ, Evans DG,
Kelsey AM, Harris M, Eden OB and Varley JM: Relative frequency and
morphology of cancers in carriers of germline TP53 mutations.
Oncogene. 20:4621–4628. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Felsher DW and Bishop JM: Reversible
tumorigenesis by MYC in hematopoietic lineages. Mol Cell.
4:199–207. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Marinkovic D, Marinkovic T, Mahr B, Hess J
and Wirth T: Reversible lymphomagenesis in conditionally c-MYC
expressing mice. Int J Cancer. 110:336–342. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jain M, Arvanitis C, Chu K, Dewey W,
Leonhardt E, Trinh M, Sundberg CD, Bishop JM and Felsher DW:
Sustained loss of a neoplastic phenotype by brief inactivation of
MYC. Science. 297:102–104. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gabay M, Li Y and Felsher DW: MYC
activation is a hallmark of cancer initiation and maintenance. Cold
Spring Harb Perspect Med. 4:1–14. 2014. View Article : Google Scholar
|
|
22
|
Shachaf CM, Kopelman AM, Arvanitis C,
Karlsson A, Beer S, Mandl S, Bachmann MH, Borowsky AD, Ruebner B,
Cardiff RD, et al: MYC inactivation uncovers pluripotent
differentiation and tumour dormancy in hepatocellular cancer.
Nature. 431:1112–1117. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sato M, Rodriguez-Barrueco R, Yu J, Do C,
Silva JM and Gautier J: MYC is a critical target of FBXW7.
Oncotarget. 6:3292–3305. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Meyer N and Penn LZ: Reflecting on 25
years with MYC. Nat Rev Cancer. 8:976–990. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ross DA and Wilson GD: Expression of c-myc
oncoprotein represents a new prognostic marker in cutaneous
melanoma. Br J Surg. 85:46–51. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Greulich KM, Utikal J, Peter RU and Krähn
G: c-MYC and nodular malignant melanoma. A case report. Cancer.
89:97–103. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kraehn GM, Utikal J, Udart M, Greulich KM,
Bezold G, Kaskel P, Leiter U and Peter RU: Extra c-myc oncogene
copies in high risk cutaneous malignant melanoma and melanoma
metastases. Br J Cancer. 84:72–79. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kanzler MH and Mraz-Gernhard S: Primary
cutaneous malignant melanoma and its precursor lesions: Diagnostic
and therapeutic overview. J Am Acad Dermatol. 45:260–276. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lin X, Sun R, Zhao X, Zhu D, Zhao X, Gu Q,
Dong X, Zhang D, Zhang Y, Li Y, et al: C-myc overexpression drives
melanoma metastasis by promoting vasculogenic mimicry via
c-myc/snail/Bax signaling. J Mol Med (Berl). 95:53–67. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nag S, Zhang X, Srivenugopal KS, Wang MH,
Wang W and Zhang R: Targeting MDM2-p53 interaction for cancer
therapy: Are we there yet? Curr Med Chem. 21:553–574. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Freedman DA, Wu L and Levine AJ: Functions
of the MDM2 oncoprotein. Cell Mol Life Sci. 55:96–107. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Momand J, Wu HH and Dasgupta G: MDM2 -
master regulator of the p53 tumor suppressor protein. Gene.
242:15–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Oliner JD, Pietenpol JA, Thiagalingam S,
Gyuris J, Kinzler KW and Vogelstein B: Oncoprotein MDM2 conceals
the activation domain of tumour suppressor p53. Nature.
362:857–860. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Momand J, Zambetti GP, Olson DC, George D
and Levine AJ: The mdm-2 oncogene product forms a complex with the
p53 protein and inhibits p53-mediated transactivation. Cell.
69:1237–1245. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li Q and Lozano G: Molecular pathways:
Targeting Mdm2 and Mdm4 in cancer therapy. Clin Cancer Res.
19:34–41. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vassilev LT: MDM2 inhibitors for cancer
therapy. Trends Mol Med. 13:23–31. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Amin MB, Edge S, Greene F, Byrd DR,
Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR,
Sullivan DC, et al: AJCC Cancer Staging Manual, 8th edition.
Springer; 2017
|
|
38
|
Cheng Y and Li G: Role of the ubiquitin
ligase Fbw7 in cancer progression. Cancer Metastasis Rev. 31:75–87.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Busino L, Millman SE, Scotto L, Kyratsous
CA, Basrur V, OConnor O, Hoffmann A, Elenitoba-Johnson KS and
Pagano M: Fbxw7α- and GSK3-mediated degradation of p100 is a
pro-survival mechanism in multiple myeloma. Nat Cell Biol.
14:375–385. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Min SH, Lau AW, Lee TH, Inuzuka H, Wei S,
Huang P, Shaik S, Lee DY, Finn G, Balastik M, et al: Negative
regulation of the stability and tumor suppressor function of Fbw7
by the Pin1 prolyl isomerase. Mol Cell. 46:771–783. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yada M, Hatakeyama S, Kamura T, Nishiyama
M, Tsunematsu R, Imaki H, Ishida N, Okumura F, Nakayama K and
Nakayama KI: Phosphorylation-dependent degradation of c-Myc is
mediated by the F-box protein Fbw7. EMBO J. 23:2116–2125. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Davis RJ, Welcker M and Clurman BE: Tumor
suppression by the Fbw7 ubiquitin ligase: Mechanisms and
opportunities. Cancer Cell. 26:455–464. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Bonetti P, Davoli T, Sironi C, Amati B,
Pelicci PG and Colombo E: Nucleophosmin and its AML-associated
mutant regulate c-Myc turnover through Fbw7γ. J Cell Biol.
182:19–26. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Grim JE, Gustafson MP, Hirata RK, Hagar
AC, Swanger J, Welcker M, Hwang HC, Ericsson J, Russell DW and
Clurman BE: Isoform- and cell cycle-dependent substrate degradation
by the Fbw7 ubiquitin ligase. J Cell Biol. 181:913–920. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Welcker M, Orian A, Grim JE, Eisenman RN
and Clurman BE: A nucleolar isoform of the Fbw7 ubiquitin ligase
regulates c-Myc and cell size. Curr Biol. 14:1852–1857. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Welcker M, Orian A, Jin J, Grim JE, Harper
JW, Eisenman RN and Clurman BE: The Fbw7 tumor suppressor regulates
glycogen synthase kinase 3 phosphorylation-dependent c-Myc protein
degradation. Proc Natl Acad Sci USA. 101:9085–9090. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Reavie L, Buckley SM, Loizou E, Takeishi
S, Aranda-Orgilles B, Ndiaye-Lobry D, et al: Regulation of c-Myc
ubiquitination controls CML initiation and progression. Cancer
Cell. 23:362–375. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Niezgoda A, Niezgoda P and Czajkowski R:
Novel approaches to treatment of advanced melanoma: A review on
targeted therapy and immunotherapy. Biomed Res Int. 2015:851382015.
View Article : Google Scholar
|
|
49
|
Kalkat M, De Melo J, Hickman KA, Lourenco
C, Redel C, Resetca D, Tamachi A, Tu WB and Penn LZ: MYC
deregulation in primary human cancers. Genes (Basel). 8:2–30. 2017.
View Article : Google Scholar
|
|
50
|
Cancer Genome Atlas Network: Comprehensive
molecular portraits of human breast tumors. Nature. 490:61–70.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Witkiewicz AK, McMillan EA, Balaji U, Baek
G, Lin WC, Mansour J, Mollaee M, Wagner KU, Koduru P, Yopp A, et
al: Whole-exome sequencing of pancreatic cancer defines genetic
diversity and therapeutic targets. Nat Commun. 6:67442015.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Cancer Genome Atlas Research Network, .
Integrated genomic analyses of ovarian carcinoma. Nature.
474:609–615. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Houben R, Hesbacher S, Schmid CP, Kauczok
CS, Flohr U, Haferkamp S, Müller CS, Schrama D, Wischhusen J and
Becker JC: High-level expression of wild-type p53 in melanoma cells
is frequently associated with inactivity in p53 reporter gene
assays. PLoS One. 6:e220962011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Winder M and Virós A: Mechanisms of drug
resistance in melanoma. Handb Exp Pharmacol. 249:91–108. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Webster MR, Fane ME, Alicea GM, Basu S,
Kossenkov AV, Marino GE, Douglass SM, Kaur A, Ecker BL,
Gnanapradeepan K, et al: Paradoxical role for wild-type p53 in
driving therapy resistance in melanoma. Mol Cell. 77:633–644.e5.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Soengas MS, Capodieci P, Polsky D, Mora J,
Esteller M, Opitz-Araya X, McCombie R, Herman JG, Gerald WL,
Lazebnik YA, et al: Inactivation of the apoptosis effector Apaf-1
in malignant melanoma. Nature. 409:207–211. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Karst AM, Dai DL, Martinka M and Li G:
PUMA expression is significantly reduced in human cutaneous
melanomas. Oncogene. 24:1111–1116. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Onel K and Cordon-Cardo C: MDM2 and
prognosis. Mol Cancer Res. 2:1–8. 2004.PubMed/NCBI
|
|
59
|
Sun Y: E3 ubiquitin ligases as cancer
targets and biomarkers. Neoplasia. 8:645–654. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Polsky D, Bastian B, Hazan C, Melzer K,
Pack J, Houghton A, Busam K, Cordon-Cardo C and Osman I: HDM2
protein overexpression, but not gene amplification, is related to
tumorigenesis of cutaneous melanoma. Cancer Res. 61:7642–7646.
2001.PubMed/NCBI
|
|
61
|
Rajabi P, Karimian P and Heidarpour M: The
relationship between MDM2 expression and tumor thickness and
invasion in primary cutaneous malignant melanoma. J Res Med Sci.
17:452–455. 2012.PubMed/NCBI
|
|
62
|
Polsky D, Melzer K, Hazan C, Panageas KS,
Busam K, Drobnjak M, Kamino H, Spira JG, Kopf AW, Houghton A, et
al: HDM2 protein overexpression and prognosis in primary malignant
melanoma. J Natl Cancer Inst. 94:1803–1806. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kimura T, Gotoh M, Nakamura Y and Arakawa
H: hCDC4b, a regulator of cyclin E, as a direct transcriptional
target of p53. Cancer Sci. 94:431–436. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Muthusamy V, Hobbs C, Nogueira C,
Cordon-Cardo C, McKee PH, Chin L and Bosenberg MW: Amplification of
CDK4 and MDM2 in malignant melanoma. Genes Chromosomes Cancer.
45:447–454. 2006. View Article : Google Scholar : PubMed/NCBI
|