Introduction
Lung cancer is the leading cause of
cancer-associated mortality worldwide (1). Despite advances in surgery,
chemotherapy, radiotherapy and molecular targeted therapy, the
prognosis of lung cancer remains poor, with a 5-year survival rate
ranging from 4–17% (2). Therefore,
there is a need to develop new therapeutic strategies for the
treatment of lung carcinoma.
Dendritic cells (DCs) are the most potent
antigen-presenting cells with a critical role in both the primary
and secondary immune response against tumor-associated antigens
(TAAs) (3). Vaccinations using DCs
loaded with tumor lysates or specific tumor-associated peptides can
trigger cytotoxic T cell responses against TAAs (4). However, the antitumor activity of DC
vaccines remains unsatisfactory in animal models and clinical
trials (5). DCs comprise of
heterogeneous cell populations, the two main populations being
plasmacytoid (p)DCs and myeloid (m)DCs (6). mDCs and pDCs are functionally distinct,
with different toll-like receptors (TLRs; pDCs express TLR7 and TLR
9, whereas mDC express all TLRs except TLR7 and TLR9) (7) and cytokines secretion (for example,
mDCs secret INF-λ, while pDCs secrete INF-α) following stimulation
(8,9). It has been shown that mDCs and pDCs can
act synergistically, resulting in improved antitumor efficacy of
the vaccine. For example, a mouse model showed that a pDCs and mDCs
combined vaccine has a synergistic effect and results in improved
antigen-specific antitumor efficacy against thymoma compared with a
pDC or mDC alone vaccine (10).
However, the antitumor efficacy of a pDC and mDC combined vaccine
against lung cancer remains to be investigated.
Antigens loading serves an important role in the
antitumor efficacy of DCs vaccines (4). Antigens used for vaccination include
peptides, DNA and recombinant tumor proteins, tumor lysates, heat
shock proteins, whole tumor cells and whole tumor RNAs (11,12). A
vaccine containing a single TAA is limited and elicits
tumor-associated major histocompatibility complex (MHC) class I
responses but not MHC class II and CD4+ T cell helper
responses (13). Meanwhile, tumor
lysate or whole tumor cells contain a full complement of TAAs,
including both MHC class I and class II-restricted epitopes, thus
decreasing the risk of immune escape (11,12).
Compared with a single TAA, loading with tumor lysates in DCs
induces a stronger and more extensive immunological response
against tumors (14–16).
The purpose of the present study was to evaluate the
therapeutic efficacy of the mDC and pDC combined vaccine loaded
with lung cancer cell lysates for the treatment of lung cancer.
Materials and methods
Materials and reagents
Mouse Lewis lung cancer cells were purchased from
the American Type Culture collection (https://www.lgcstandards-atcc.org/products/all/CRL-1642.aspx?geo_country=nl).
Mouse anti-CD11b magnetic beads and mouse anti-mPDCA magnetic beads
were purchased from Miltenyi Biotec GmbH. Mouse IL-6 (cat. no.
555240), IL-12 (cat. no. 555256) and TNF-α (cat. no. 555268) ELISA
kits were purchased from BD Bioscience. The TUNEL assay kit was
purchased from Roche Diagnostics. Unmethylated CpG oligonucleotide
(CpG ODN 1826) and the lactate dehydrogenase (LDH) cytotoxicity
assay kit were purchased from Sangon Biotech Co., Ltd. The Cell
Counting Kit (CCK)-8 was purchased from Dojindo Molecular
Technologies, Inc. Recombinant Fms-like tyrosine kinase 3 receptor
ligand (rmFlt3-Ligand), CD11c-PE mAb, anti-CD11b-APC mAb,
anti-B220-FITC mAb, anti-MHC-II FITC mAb, anti-CD40 PE mAb, anti-
CD80 PE mAb and anti-CD86 PE mAb were all purchased from
eBioscience; Thermo Fisher Scientific, Inc.
Animals
In total, 35 C57BL/6 male mice (6–8 weeks old,
weighing 20–25 g) and 12 NU/NU male nude mice (3–4 weeks old,
weighing 9–11 g) were purchased from the Laboratory animal center
of Sun Yat-sen University (http://zssom.sysu.edu.cn/cmc/). All animals were
housed under standardized conditions with a 12-h light/dark cycle,
humidity (50-60%) and temperature (20–27°C) with free access to
food and water. All animals were monitored daily for signs of
obvious behavioral changes and physical stress, and would be
euthanized if found in distress. No animals were found dead or were
euthanized during the experiments. All protocols used in the
present study were approved by The Institutional Animal Care and
Use Committee of the Third Affiliated Hospital, Sun Yat-sen
University (Guangzhou, China; approval no. 00155986). The
experiment was conducted between September 2016 and March 2017.
Preparation of mDCs and pDCs
After 3–5 days of breeding, the mice were sacrificed
by cervical dislocation, bone marrow cells of the femur and tibia
of C57BL/6 mice were collected and lysed using red blood cell lysis
buffer (Sigma-Aldrich; Merck KGaA) to obtain bone marrow
mononuclear cells. The cells were resuspended with RMPI-1640
complete medium (2 mM glutamine, 50 µM 2-ME, 7 mM HEPES, 100 U/ml
penicillin, 100 µg/ml streptomycin, rmFlt3-L 100 ng/ml and 10% FBS)
(Invitrogen; Thermo Fisher Scientific, Inc.) and seeded onto 6-well
plates (2×106/ml, 1 ml/well) at 37°C in a 5%
CO2 incubator.
On day 4 of culture, half of the medium was
replaced, and fresh rmFlt3-L was added into the medium. On day 8,
the mDC and pDC were sorted using mouse anti-CD11b and mouse
anti-mPDCA magnetic beads according to manufacturer's protocol, and
the cell purity was >99% according to flow cytometry analysis
with a FACScaliber™ cytometer (BD Biosciences) using
fluorescently-labeled monoclonal antibodies (anti-CD11c-PE mAb
(cat. no. 12-0401-81), anti-CD11b-APC mAb (cat. no. 17-0012-81),
anti-B220-FITC mAb (cat. no. 11-0452-81)). The analysis software
used was FlowJo version 7.6.1 (FlowJo LLC). An equal amount of pDC
and mDC (1:1 ratio) was used for the combination vaccine (10). The sorted DC subsets were divided
into three groups for subsequent experiments: mDCs, pDCs And mDCs:
pDC (1:1). The number of the total cells was equal in the three
groups.
DCs loaded with heat-treated with
Lewis lung cancer cell lysates
Lewis lung cancer cells were cultured in high-sugar
DMEM medium containing 100 U/ml penicillin, 100 µg/ml streptomycin
and 10% FBS at a concentration of 1.5–3×106 cell/ml in a
37°C and 5% CO2 incubator. Lewis lung cancer cells in
the logarithmic growth phase were placed in an incubator at 42°C
for 1 h, followed by placing in an incubator at 37°C, 5%
CO2 for 2 h. The cells were digested with trypsin,
washed twice with PBS and then resuspended in PBS (1×107
cells/ml). The cells were repeatedly frozen and thawed in liquid
nitrogen and 37°C water bath five times and centrifuged at 13,000 ×
g at 4°C for 10 min. The resulting supernatant was collected as
cell lysates.
Equal amounts of heat-treated tumor cell lysates
were added to the mDC (1×105), pDC (1×105)
and mDC: pDC (1:1; 1×105) groups. The amount of antigen
was based on the number of tumor cells before freeze-thaw treatment
with DCs: Tumor cells =1:1. The control group consisted of an equal
amount of RMPI-1640 complete medium, and then CpG ODN 1826 was
added to each group at a final concentration of 2 µg/ml. The cell
was cultured for 24 h to induce DC maturation for subsequent
experiments.
Mixed lymphocyte proliferation
assay
C57BL/6 mouse spleens were taken and cut with
ophthalmology scissors, placed on a 70-µm filter screen, and ground
with a 1-ml syringe plunger and rinsed with PBS while grinding. The
spleen cells suspension was collected in a centrifuge tube and
centrifuged at 300 × g for 5 min, and the supernatant was
discarded. The red blood cell lysis buffer was added and incubated
for 1 min, and the RPMI-1640 medium was immediately added to stop
the reaction, then centrifuged at 300 × g for 5 min. After washing
twice with PBS, the resulting pellet was collected as spleen cells.
These experiments were performed at room temperature ~25°C.
DCs loaded with heat-treated tumor cell lysates from
the three groups of mDC, pDC and mDC+pDC were added to 96-well
culture plates at 1×104 cells/well. C57BL/6-derived
spleen cells were added into each well (1×105 cells
/well; DCs: Spleen cells =1:10) and cultured for 3 days. The DCs of
each group without tumor cell lysate loading were used as control
groups. There were three duplicated wells in each group. The
proliferation of spleen cells was detected using the CCK-8 assay
according to the manufacturer's protocol.
CTL cytotoxicity assay
DC cells in three groups loaded with heat-treated
tumor cell lysate were co-cultured in vitro by DCs: Spleen
cells =1:10. There were three duplicated wells in each group.
Recombinant mouse IL-2 was added into each well at a final
concentration of 20 U/ml, and the solution was replaced every 3
days and cultured at 37°C, 5% CO2. After culture for a
week, the cells were trypsinized and collected as the CTL
cells.
Lewis lung cancer cells in the logarithmic growth
phase were used as target cells, and CTL cells were used as
effector cells. The effector and target cells were mixed at the
effector: Target cell ratios (E:T) of 40:1 (CTL:tumor cells,
4×105:1×104), 20:1 (CTL:tumor cells,
2×105:1×104) and 10:1 (CTL:tumor cells,
1×105:1×104) in 100 µl medium, and plated
onto a 96-well flat-bottom plate and co-cultured for 12 h. There
were three duplicated wells in each group. At 1 h before the end of
co-culture, 10 µl of LDH releasing agent (Beyotime Institute of
Biotechnology) was added into each well and incubated at 37°C, 5%
CO2 for 1 h. At the end of the culture, the absorbance
at 490 nm (OD value) was measured using a microplate reader.
Determining surface markers on DCs and
cytokines secretion
For surface marker labeling, the cell suspension was
incubated with anti-CD11c-PE mAb (cat. no. 12-0401-81),
anti-CD11b-APC mAb (cat. no. 17-0012-81), anti-B220-FITC mAb (cat.
no. 11-0452-81), anti-mouse CD80 PE-Cy7 (cat. no. 15-0801-81),
anti-mouse CD86 APC (cat. no. 17-0862-81), anti-mouse CD40 PE (cat.
no. 12-0401-81) and anti-mouse MHC Class II FITC (cat. no.
11-5322-81) fluorescent antibodies at 20°C for 20 min in the dark.
All the antibodies were diluted by 1:100. The cells were washed
twice with PBS, resuspended in 300 µl PBS containing 1% FBS
(Invitrogen; Thermo Fisher Scientific, Inc.), and subjected to flow
cytometry as aforementioned. The experiment was repeated three
times. The analysis software used was Flowjo version 7.6.1 (FlowJo,
LLC). CD11c positive cells were gated, CD11b-B220+ was identified
as pDC subpopulation and CD11b+B220- was defined as mDC
subpopulation. The secretion level of IL-6, IL-12 and TNF-α in the
aforementioned cell supernatant was detected using the
corresponding ELISA kit according to the manufacturer's
protocols.
Establishment of tumor-bearing nude
mice model
Lewis lung cancer cells in the logarithmic growth
phase were resuspended at a cell density of 1×107/ml.
The cell suspension (0.1 ml; 1×106 cells) was injected
subcutaneously into the thigh roots of 12 NU/NU male nude mice to
establish tumor-bearing nude mice. The tumor-bearing mice were
randomly divided into three groups: Control, mDC group and the
mDC+pDC group, with four mice per group.
Preparation of CTLs
The tumor cell lysate-sensitized mDCs and pDCs were
added to the 6-well plate culture well (1×106/ml per
well). The ratio of DCs: Antigen was 1:1, and the amount of antigen
was based on the number of tumor cells before freeze-thaw
treatment. DC maturation was induced by the addition of CpG ODN1826
at a final concentration of 2 µg/ml overnight. The cells in the
wells were collected, and the cell suspension was prepared at the
cell density of 3×106/ml. Mouse spleen cells were
collected as aforementioned.
The DCs and spleen cells were mixed at a ratio of
1:10 and then added to a 6-well plate (3×106 cells/ml, 2
ml per well with 6×106 cells/well). rmIL-2 was added at
a final concentration of 20 U/ml The culture medium was replaced
with complete RMPI-1640 medium containing IL-2 once every 3 days
and then incubated for 2 weeks in a 37°C in a 5% CO2
incubator. The resulting cells were CTLs.
In vivo antitumor study of DC
vaccine
Tumor-bearing mice were randomly divided into three
groups: Control, mDC group and the mDC+pDC group. At day 7 post the
inoculation of Lewis lung cancer cells, PBS, mDC-induced CTL cells
and mDC+pDC induced CTL cells (1×107 cells/mouse) were
intravenously injected into mice in the three groups through the
tail vein. The injection of CTL cells was repeated 7 days after the
first injection.
The length (L) and width (D) of the tumor tissue
were measured with a vernier caliper, and the tumor volume was
calculated every 2 days. The tumor volume was calculated according
to the formula V=1/2LxD2.
At 25 days after the inoculation of Lewis lung
cancer cells, nude mice were sacrificed by cervical dislocation,
and then tumor tissues were collected. The necrosis of tumor
tissues was observed by hematoxylin and eosin (H&E) staining.
For H&E staining, tissue samples were fixed with 10% formalin
for 24 h, then embedded in paraffin and sectioned into slices with
a thickness of 5 µm. After dewaxing with xylene and rehydration
with descending ethanol series [100 (twice), 90, 80 and 70%
respectively], the slice was stained with H for 5 min, 5% acetic
acid for 1 min, and eosin staining for 1 min at room temperature
~25°C. After dehydration with ascending ethanol series (70, 80, 90
and 100% respectively), the slide was dripped with neutral resin,
covered with a cover glass, and then mounted. TUNEL staining was
used to observe the apoptosis of tumor cells by using the
aforementioned TUNEL assay kit according to the manufacturer's
protocol. The apoptotic cells were observed under a fluorescence
microscope (excitation light wavelength, 450–500 nm; detection
wavelength, 515–565 nm).
Statistical analysis
All data was presented as mean ± SD and were
compared using one-way ANOVA (for one independent variable) or
two-way ANOVA (for two independent variables) with Sidak's post hoc
test for multiple comparisons. P<0.05 was considered to indicate
a statistically significant difference. Statistical analyses were
performed using SPSS version 16 software (SPSS Inc.).
Results
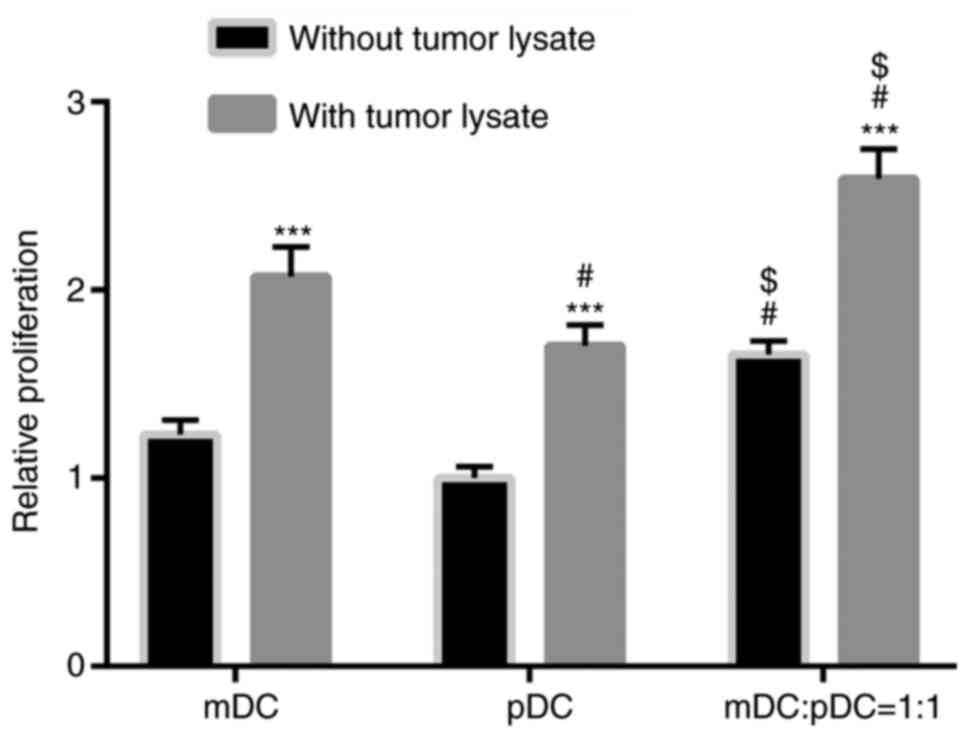
Combining tumor cell lysate-loaded mDC
and pDC promotes lymphocyte proliferation
The effect of mDC and pDC vaccines on lymphocyte
proliferation was determined using a CCK-8 assay. As shown in
Fig. 1, in both the absence or
presence of tumor cell lysates, the lymphocyte proliferation was
significantly higher in the mDC+pDC group compared with the mDC or
pDC group (all P<0.05). After loading with tumor cell lysate,
the promotive effect on lymphocytes proliferation was significantly
increased in the three groups (all P<0.001 without tumor lysate
vs. with tumor lysate; Fig. 1).
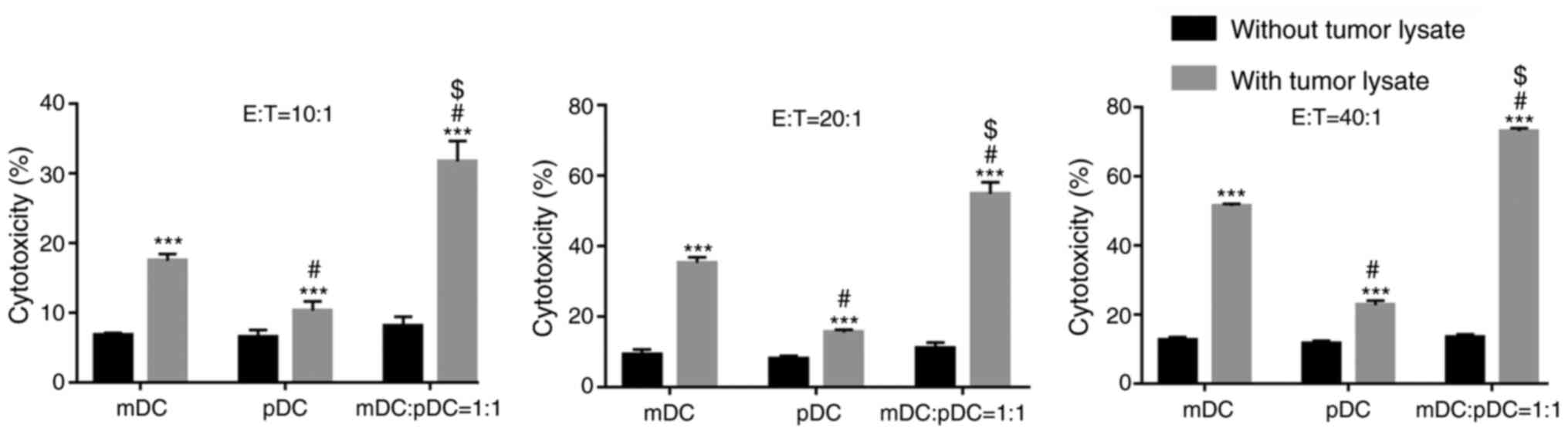
Combining tumor cell lysate-loaded mDC
and pDC enhances CTL-mediated cytotoxicity against tumor cells
The effect of mDC and pDC vaccines on CTL
cytotoxicity against tumor cells was investigated. In the absence
of tumor cell lysates, there was no difference in CTL cytotoxicity
among the three groups at all the effector: Target cell ratios
(E:T), (all P>0.05; Fig. 2).
Tumor cell lysate loading significantly enhanced the
CTL-mediated cytotoxicity in all the groups at all the E:T (all
P<0.001 without tumor lysate vs. with tumor lysate; Fig. 2). At all the three E:T, the mDC+pDC
group had significantly higher cytotoxicity compared with the mDC
or pDC group (all P<0.05; Fig.
2). The cytotoxicity of the mDC+pDC group was 31.68, 54.77 and
73.01% at the E:T of 1:10, 1:20 and 1:40 (Fig. 2), respectively.
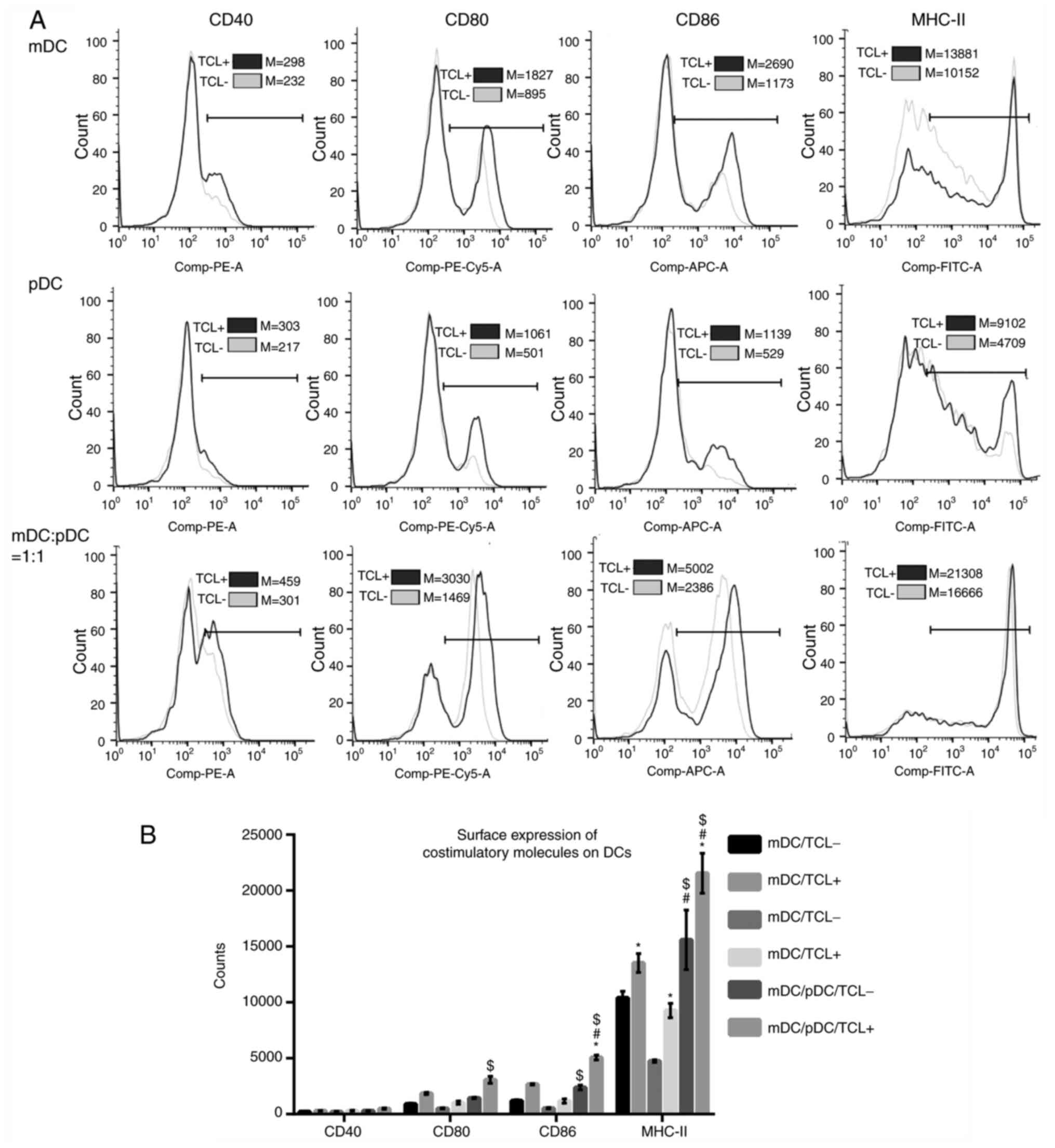
Heat-treated tumor cell lysate loading
upregulates the DCs surface expression of costimulatory
molecules
Surface expressions of costimulatory molecules,
including CD80, CD86, CD40 and MHC-II, were assessed using flow
cytometry (Fig. 3A). The results
showed that in the absence of tumor cell lysate, the surface
expressions of MHC-II were significantly higher in the
mDC+pDC/TCL− group compared with the mDC/TCL−
or pDC/TCL− group (all P<0.05; Fig. 3B). After loading with the tumor cell
lysate, the surface expressions of CD86 and MHC-II were
significantly higher in the mDC+pDC/TCL+ group compared
with those in the mDC/TCL+ or pDC group/TCL+
(all P<0.05; Fig. 3B). In
addition, after loading with the tumor cell lysate, surface
expression of MHC-II was significantly increased in all cell types
(all P<0.05; Fig. 3B).
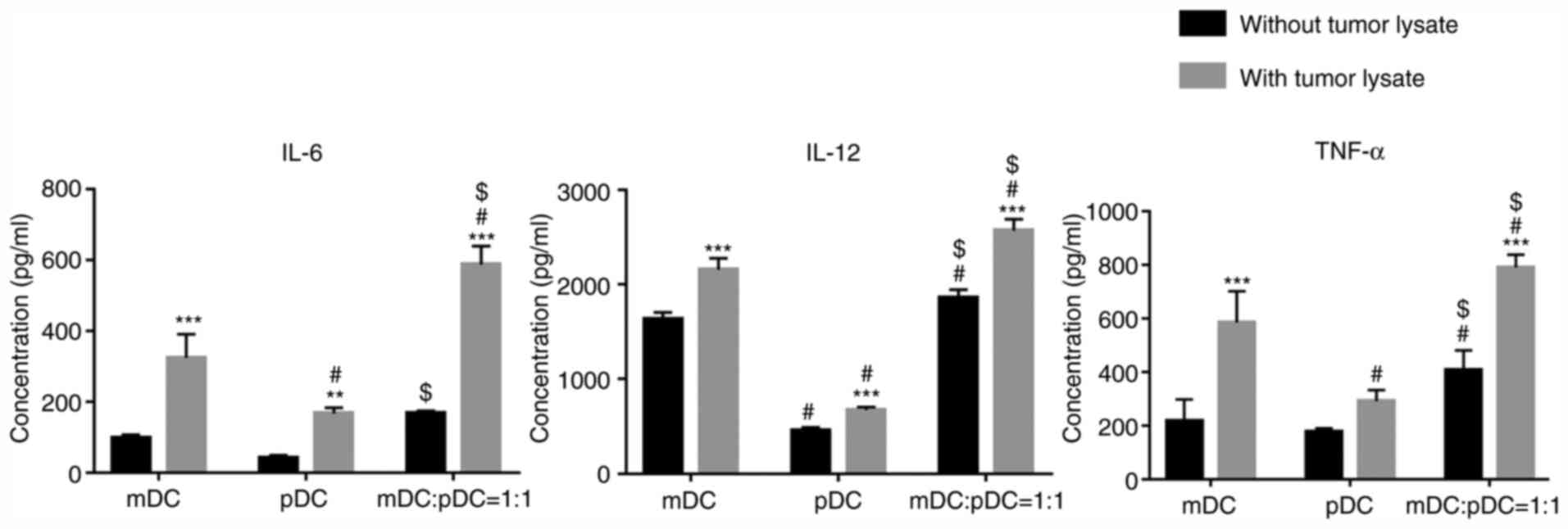
Tumor cell lysate loading stimulates
cytokines secretion in DCs
The secretion of cytokines in DCs was determined
using ELISA kits. As shown in Fig.
4, tumor cell lysate loading significantly upregulated the
secretions of IL-6, IL-12 and TNF-α in the mDC+pDC=1:1 group (all
P<0.001). In addition, in both the absence or presence of tumor
cell lysates, secretions of IL-6, IL-12 and TNF-α were
significantly higher in the mDC+pDC group compared with those in
the mDC or pDC group (P<0.05; Fig.
4), except for mDC:pDC without tumor lysate vs. mDC without
tumor lysate for IL-6.
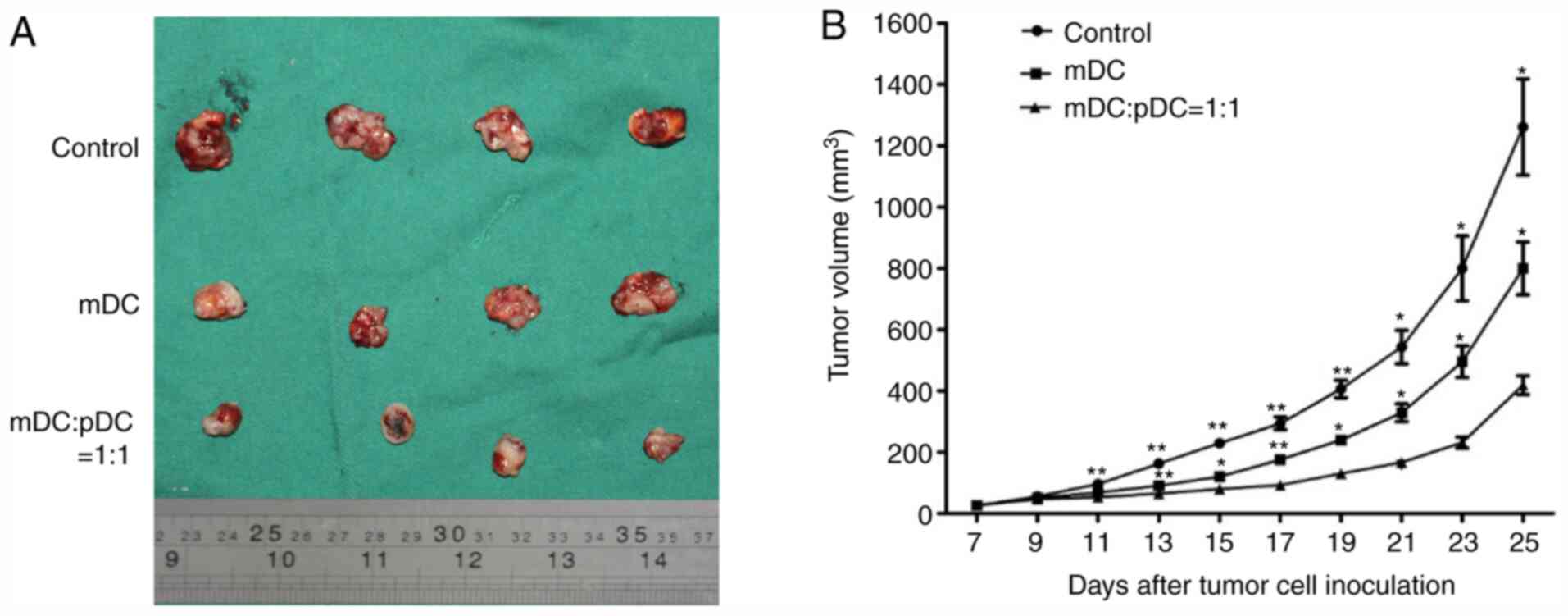
mDC and pDC combined vaccine inhibits
tumor growth in nude mice
The in vitro results indicated that DCs
vaccines loaded with tumor cell lysates induced a strong antitumor
effect, and the mDC and pDC combined vaccine loaded with tumor cell
lysates exhibited a synergistic antitumor effect in vitro.
To further evaluate the antitumor effect of the mDC and pDC
combined vaccine in vivo, a tumor-bearing nude mice model
was established. The pDC alone group was not included in the animal
study because this group exhibited the lowest in vitro
antitumor effect. After 7 days post subcutaneous inoculation of
Lewis lung cancer cells in nude mice, all mice developed a
subcutaneous mass and were administrated with the first DC vaccine.
At 14 days after inoculation, a second DC vaccine was
administrated. It was observed that between 11 and 25 days after
inoculation, the tumor volume was significantly lower in the
mDC+pDC group compared with the control group (all P<0.05 or
P<0.01; Fig. 5B). In addition,
the tumor volume was all significantly lower in the mDC+pDC group
compared with the mDC group (Fig.
5B). This result suggested that the mDC and pDC combined
vaccine had a good in vivo antitumor effect.
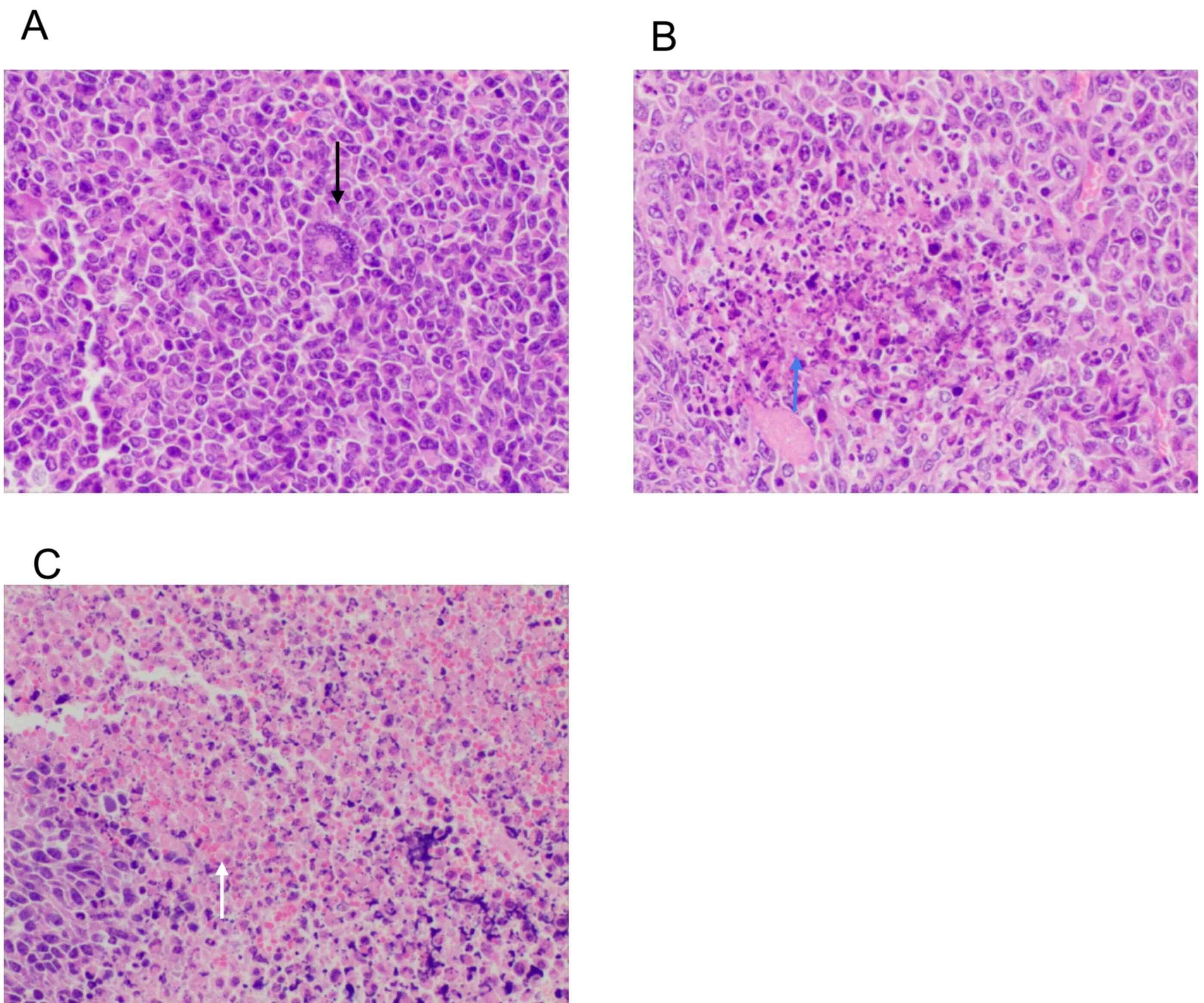
Histological changes of tumor cells
after mDC and pDC combined vaccine treatment
Histological changes in tumor cells following DC
vaccine treatment were evaluated. H&E staining showed that in
the control group, the tumor cells had different sizes and obvious
atypia (Fig. 6A, black arrow). The
cancer cells were round, elliptical or fusiform in shape. There was
no tumor giant cell or necrosis. Tumor cells in the mDC group
showed focal necrosis and hemorrhage (Fig. 6B, blue arrow), while those in the
mDC+pDC group exhibited massive necrosis and hemorrhage (Fig. 6C, white arrow).
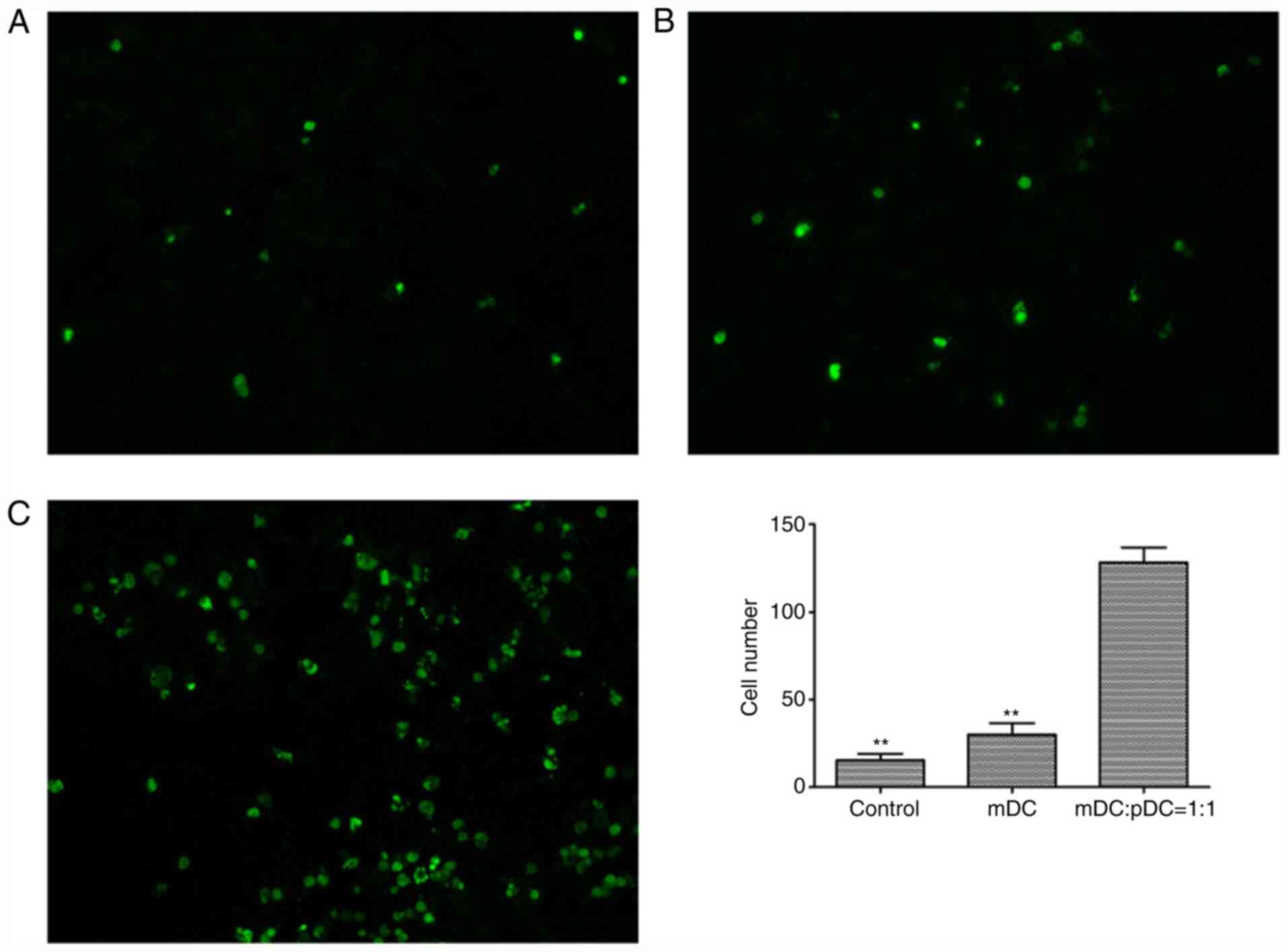
mDC and pDC combined vaccine treatment
induces apoptosis in tumor cells
A TUNEL assay was performed to assess the number of
apoptotic tumor cells after DC vaccine treatment (Fig. 7A-C). The results showed that the
number of apoptotic tumor cells was significantly higher in the
mDC+pDC=1:1 group compared with the mDC group and the control group
(both P<0.01; Fig. 7D),
indicating that the mDC and pDC combined vaccine treatment
decreased apoptosis within the tumor.
Discussion
The use of the DC vaccine for tumor immunotherapy
has not yet achieved satisfactory results (5). Therefore, how to enhance the antigen
presentation ability of the DC vaccine in vivo and how
improve the clinical efficacy of the vaccine have been focuses of
research. The present study evaluated the therapeutic efficacy of
the mDC and pDC combined vaccine loaded with heat-treated cancer
cell lysates for the treatment of lung cancer and the underlying
mechanism. The results showed that tumor cell lysate loading
upregulated surface expression of costimulatory molecules on DCs
and enhanced secretions of IL-6, IL-12 and TNF-α in the mDC+pDC=1:1
group. In addition, combining tumor cell lysate-loaded mDCs and
pDCs significantly promoted lymphocyte proliferation and enhanced
CTL-mediated cytotoxicity against Lewis lung cancer cells compared
with mDC or pDC alone treatment. Furthermore, intravenous injection
of the mDC and pDC combined vaccine into tumor-bearing nude mice
significantly inhibited subcutaneous tumor growth and induced
necrosis and apoptosis within the tumor tissue. Taken together,
these results suggested that the combined pDC and mDC vaccine
loaded with heat-treated Lewis lung cancer cell lysate had a
synergistic effect on the induction of T lymphocyte proliferation
and antitumor efficacy. This synergistic antitumor effect may be
associated with the upregulation of co-stimulatory molecules and
cytokine secretions.
The method for preparing tumor lysates affects the
efficacy of the DCs vaccine. It has been shown that freeze-thaw
tumor cell lysates do not effectively mimic in vivo
immunogenic necrosis and inhibit the maturation and function of DCs
(17–20). Hatfield et al (20) reported that heat-treated tumor
lysates (≥42°C) compared with tumor lysates are more effective TAAs
for DC vaccination. DCs loaded with heat-treated tumor cell lysates
can induce strong and broad T cell responses against pancreatic
cancer, thereby enhancing antitumor effects (21). Heat treatment increases the synthesis
of heat shock protein (HSP) in tumor cells, and HSP may promote the
expression of TAAs and enhance their presentation to
antigen-presenting cells (22).
Stress-induced HSP can also induce the expression of MHC-II
molecules, and cytokine and chemokine secretions in DCs (23). Therefore, the present study adopted
heat treatment to prepare tumor cells lysates for DC
vaccination.
The present study used nude mice to generate a tumor
model. In our preliminary study, C57BL rats were used to generate a
tumor model, but the rate of tumor formation was low, with a small
tumor volume and relatively long tumor formation time (unpublished
data). In addition, DC-activated lymphocytes, rather than the DC
vaccine, were transplanted into the animals. The C57BL rats
inoculated with subcutaneous tumor would produce specific T cells
against tumor that can affect the antitumor activity of the
injected DC-activated lymphocytes. Since nude mice lack
lymphocytes, this problem can be overcome. In the present study,
the experimental animals were the NU/NU nude mice that lack thymus
and T lymphocytes (24). The
activation of T lymphocytes requires antigen and accessory
molecules to effectively induce CTL formation (25). Therefore, the DC vaccines could not
be directly injected into tumor-bearing nude mice. T lymphocytes
should be activated in vitro and then the CTL effector cells
infused into the animal. Previous studies have shown that
antigen-loaded DCs induce T lymphocytes in vitro, and
infusion of CTL effector cells exhibits significant antitumor
effects in mice (26–28), which are in line with the present
results.
Lou et al (10) demonstrated that pDCs and mDCs have a
synergistic effect on the induction of antigen-specific antitumor
immune responses. Piccioli et al reported that human mDC and
pDC mixed culture can upregulate the expression of DC activation
markers CD40, CD80 and CD83, which can enhance the proliferation of
allogeneic T cells and secrete more IFN-γ compared with single pDC
or mDC alone (29). Consistent with
these observations, the present results demonstrated that compared
with mDC or pDC alone treatment, combining tumor lysate-loaded mDC
and pDC enhanced lymphocyte proliferation and CTL-mediated
cytotoxicity in vitro. The in vivo experiments showed
that the mDC and pDC combined vaccine inhibited subcutaneous tumor
growth, and induced extensive necrosis, hemorrhage and apoptosis
within the tumor tissue. The efficacy of the combined vaccines was
improved compared with that of the mDC group. These results
suggested that the mDC and pDC combined vaccine exhibited a
synergistic antitumor effect against lung cancer cells. To the best
of our knowledge, the present study is the first to demonstrate the
efficacy of mDC and pDC vaccine combined against lung cancer.
Our results demonstrated that the combined mDCs and
pDCs induced increased secretion of IL-12, IL-6 and TNF-α compared
with mDC or pDC treatment alone, and upregulated surface
costimulatory molecules and MHC-II expression. Immature DCs undergo
a complex maturation process after antigen capture, during which
DCs upregulate cell surface costimulatory molecules, such as CD80,
CD86 and CD40, that enhances the ability of DCs to activate T cells
(30). IL-12 is the most important
cytokine secreted by DCs, regulating the balance between the T
helper (h)1 and Th2 response, and further promoting the maturation
of DC (31). IL-6 can promote T and
B cell proliferation and differentiation, antibody production, DC
maturation and enhance DC antigen presentation (32). TNF-α can promote DC maturation and
increase the viability of DCs in an autocrine or paracrine manner,
and indirectly promote DC antigen presentation (32). Therefore, the synergistic effect of
the mDC and pDC combined vaccine observed in the present study may
be associated with upregulated surface costimulatory molecules
expression and cytokine secretions. However, the detailed molecular
mechanisms underpinning this need to be further elucidated.
There are still some limitations to the present
study. First, the number of mDCs and pDCs after magnetic bead
sorting was limited, which were insufficient for large-scale animal
experiments. In addition, although an equal amount of mDC and pDC
exhibited a synergistic antitumor effect, the optimized ratio of
mDC and pDC remains to be investigated. Moreover, to minimize the
number of animals used, the pDC alone group was not included in the
animal study because this group exhibited the lowest in
vitro antitumor effect. All these limitations should be
addressed in the future study.
In summary, the present study suggested that the
mixed culture of mDC and pDC produced a synergistic effect, which
enhanced the cytotoxic effect of T lymphocytes against Lewis lung
cancer cells in vitro and in vivo. These results may
help improve the design and clinical efficacy of DC vaccines.
Acknowledgements
Not applicable.
Funding
The study was supported by The Science and
Technology Planning Project of Guangdong Province (grant no.
20130319c).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
LG and JZ designed the study. HC and JT conducted
the experiments, collected and analyzed the data and drafted the
manuscript. XL guided and performed the experiments and revised the
manuscript. HL, WW and YW cultured the cells and determined the
level of cytokines. All authors read and approved the final version
of the manuscript.
Ethics approval and consent to
participate
All protocols used in the present study were
approved by The Institutional Animal Care and Use Committee of the
Third Affiliated Hospital (Guangzhou, China; approval no.
00155986).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mellstedt H, Vansteenkiste J and Thatcher
N: Vaccines for the treatment of non-small cell lung cancer:
Investigational approaches and clinical experience. Lung Cancer.
73:11–17. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hirsch FR, Scagliotti GV, Mulshine JL,
Kwon R, Curran WJ, Wu YL and Paz-Ares L: Lung cancer: Current
therapies and new targeted treatments. Lancet. 389:299–311. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gardner A and Ruffell B: Dendritic cells
and cancer immunity. Trends Immunol. 37:855–865. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
González FE, Gleisner A, Falcón-Beas F,
Osorio F, López MN and Salazar-Onfray F: Tumor cell lysates as
immunogenic sources for cancer vaccine design. Hum Vaccines
Immunother. 10:3261–3269. 2014. View Article : Google Scholar
|
|
5
|
Ilett E, Prestwich R and Melcher A: The
evolving role of dendritic cells in cancer therapy. Expert Opin
Biol Ther. 10:369–379. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bol KF, Schreibelt G, Gerritsen WR, De
Vries IJM and Figdor CG: Dendritic cell-based immunotherapy: State
of the art and beyond. Clin Cancer Res. 22:1897–1906. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kadowaki N, Ho S, Antonenko S, Malefyt R,
Kastelein R, Bazan F and Liu Y: Subsets of human dendritic cell
precursors express different toll-like receptors and respond to
different microbial antigens. J Exp Med. 194:863–869. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Swiecki M and Colonna M: The multifaceted
biology of plasmacytoid dendritic cells. Nat Rev Immunol.
15:471–485. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nizzoli G, Krietsch J, Weick A,
Steinfelder S, Facciotti F, Gruarin P, Bianco A, Steckel B, Moro M,
Crosti M, et al: Human CD1c+ dendritic cells secrete
high levels of IL-12 and potently prime cytotoxic T-cell responses.
Blood. 122:932–942. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lou Y, Liu C, Kim GJ, Liu YJ, Hwu P and
Wang G: Plasmacytoid dendritic cells synergize with myeloid
dendritic cells in the induction of antigen-specific antitumor
immune responses. J Immunol. 178:1534–1541. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mocellin S, Mandruzzato S, Bronte V, Lise
M and Nitti D: Part I: Vaccines for solid tumours. Lancet Oncol.
5:681–689. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schuler G, Schuler-Thurner B and Steinman
RM: The use of dendritic cells in cancer immunotherapy. Curr Opin
Immunol. 15:138–147. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tagliamonte M, Petrizzo A, Tornesello ML,
Buonaguro FM and Buonaguro L: Antigen-specific vaccines for cancer
treatment. Hum Vaccines Immunother. 10:3332–3346. 2014. View Article : Google Scholar
|
|
14
|
Ridolfi R, Petrini M, Fiammenghi L,
Stefanelli M, Ridolfi L, Ballardini M, Migliori G and Riccobon A:
Improved overall survival in dendritic cell vaccination-induced
immunoreactive subgroup of advanced melanoma patients. J Transl
Med. 4:362006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Trefzer U, Herberth G, Wohlan K, Milling
A, Thiemann M, Sharav T, Sparbier K, Sterry W and Walden P:
Tumour-dendritic hybrid cell vaccination for the treatment of
patients with malignant melanoma: Immunological effects and
clinical results. Vaccine. 23:2367–2373. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nagayama H, Sato K, Morishita M, Uchimaru
K, Oyaizu N, Inazawa T, Yamasaki T, Enomoto M, Nakaoka T, Nakamura
T, et al: Results of a phase I clinical study using autologous
tumour lysate-pulsed monocyte-derived mature dendritic cell
vaccinations for stage IV malignant melanoma patients combined with
low dose interleukin-2. Melanoma Res. 13:521–530. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hersey P, Menzies SW, Halliday GM, Nguyen
T, Farrelly ML, DeSilva C and Lett M: Phase I/II study of treatment
with dendritic cell vaccines in patients with disseminated
melanoma. Cancer Immunol Immunother. 53:125–134. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jouanneau E, Poujol D, Gulia S, Le Mercier
I, Blay JY, Belin MF and Puisieux I: Dendritic cells are essential
for priming but inefficient for boosting antitumour immune response
in an orthotopic murine glioma model. Cancer Immunol Immunother.
55:254–267. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang Y, Yoneyama H, Wang Y, Ishikawa S,
Hashimoto S, Gao JL, Murphy P and Matsushima K: Mobilization of
dendritic cell precursors into the circulation by administration of
MIP-1alpha in mice. J Natl Cancer Inst. 96:201–209. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hatfield P, Merrick AE, West E, ODonnell
D, Selby P, Vile R and Melcher AA: Optimization of dendritic cell
loading with tumor cell lysates for cancer immunotherapy. J
Immunother. 31:620–632. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kim HS, Choo YS, Koo T, Bang S, Oh TY, Wen
J and Song SY: Enhancement of antitumor immunity of dendritic cells
pulsed with heat-treated tumor lysate in murine pancreatic cancer.
Immunol Lett. 103:142–148. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Qiu J, Li GW, Sui YF, Song HP, Si SY and
Ge W: Heat-shocked tumor cell lysate-pulsed dendritic cells induce
effective anti-tumor immune response in vivo. World J
Gastroenterol. 12:473–478. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Castelli C, Rivoltini L, Rini F, Belli F,
Testori A, Maio M, Mazzaferro V, Coppa J, Srivastava PK and
Parmiani G: Heat shock proteins: Biological functions and clinical
application as personalized vaccines for human cancer. Cancer
Immunol Immunother. 53:227–233. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pelleitier M and Montplaisir S: The nude
mouse: A model of deficient T-cell function. Methods Achiev Exp
Pathol. 7:149–166. 1975.PubMed/NCBI
|
|
25
|
Alberts B, Johnson A and Lewis J: Helper T
cells and lymphocyte activation. Molecular Biology of the Cell.
1–8. 2002.PubMed/NCBI
|
|
26
|
Zhang Y, Zhang N, Zhao M and Hoffman RM:
Comparison of the selective targeting efficacy of Salmonella
typhimurium A1-R and VNP20009 on the Lewis lung carcinoma in
nude mice. Oncotarget. 6:14625–14631. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fu Q, Wu Y, Yan F, Wang N, Wang W, Cao X,
Wang Y and Wan T: Efficient induction of a Her2-specific anti-tumor
response by dendritic cells pulsed with a Hsp70L1-Her2 341–456
fusion protein. Cell Mol Immunol. 8:424–432. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Song S, Zhang K, You H, Wang J, Wang Z,
Yan C and Liu F: Significant anti-tumour activity of adoptively
transferred T cells elicited by intratumoral dendritic cell vaccine
injection through enhancing the ratio of CD8+ T
cell/regulatory T cells in tumour. Clin Exp Immunol. 162:75–83.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Piccioli D, Tavarini S, Borgogni E, Steri
V, Nuti S, Sammicheli C, Bardelli M, Montagna D, Locatelli F and
Wack A: Functional specialization of human circulating CD16 and
CD1c myeloid dendritic-cell subsets. Blood. 109:5371–5379. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Audiger C, Rahman MJ, Yun TJ, Tarbell KV
and Lesage S: The importance of dendritic cells in maintaining
immune tolerance. J Immunol. 198:2223–2231. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu J, Cao S, Kim S, Chung EY, Homma Y,
Guan X, Jimenez V and Ma X: Interleukin-12: An update on its
immunological activities, signaling and regulation of gene
expression. Curr Immunol Rev. 1:119–137. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Blanco P, Palucka AK, Pascual V and
Banchereau J: Dendritic cells and cytokines in human inflammatory
and autoimmune diseases. Cytokine Growth Factor Rev. 19:41–52.
2008. View Article : Google Scholar : PubMed/NCBI
|