Introduction
Pancreatic ductal adenocarcinoma (PDAC), a malignant
tumor, is associated with poor prognosis, which is mainly due to
the difficulties in early diagnosis and the low efficacy of
adjuvant therapy. There is a need to develop methods for early
diagnosis and multimodal treatment involving surgical resection and
chemo-radiotherapy to improve the clinical outcomes of patients
with PDAC. The development of mass spectrometry (MS)-based
proteomics technology has facilitated the detection of disease
biomarkers (1). This technology,
which involves the analysis of peptides and proteins in biological
samples, has enabled the identification of many biomarker
candidates in the human plasma or serum and can provide
cancer-specific diagnostic information (2,3).
A comprehensive analysis of glycoproteins in the
serum samples of patients with PDAC using Tandem Mass Tag™ with MS
identified C4b-binding protein α-chain (C4BPA) as a novel serum
PDAC biomarker (4). C4BPA is
reported to be markedly upregulated in patients with clear-cell
ovarian cancer (5) and
non-small-cell lung cancer (6). This
indicated the diagnostic value of serum C4BPA in different cancers.
However, the objective comparison of serum C4BPA levels is
difficult because of the use of different measurement methods. High
reproducibility and specificity are the key parameters for
establishing useful clinical biomarkers, large-scale validation of
these markers, and high-throughput screening for such markers.
Fucosylation and sialylation are major glycosylation
events in post-translational protein modifications, which are
involved in the pathogenesis of inflammation and cancer (7). In fucosylation, the transfer of fucose
from GDP-fucose to a molecule is catalyzed by fucosyltransferases.
Sialylation involves the addition of a sialic acid unit to the end
of an oligosaccharide chain in a glycoprotein. The glycoproteins,
alpha-fetoprotein-L3 (AFP-L3; fucosylated AFP) and carbohydrate
antigen 19-9 (CA19-9; sialyl Lewis A antigen) are used as tumor
markers in hepatocellular carcinoma (8) and PDAC (9), respectively. Thus, glycoproteins are
potential serum diagnostic biomarkers for cancer. Previous studies
have reported that the fully-sialylated C4BPA levels are
upregulated in the serum of patients with ovarian cancer (10). However, there are no studies on the
importance of serum fucosylated (Fuc-) C4BPA in patients with
cancer. Therefore, we hypothesized that the serum Fuc-C4BPA level
has diagnostic and clinical significance in patients with PDAC.
In this study, we established a novel hybrid
enzyme-linked immunosorbent assay (ELISA) for measuring Fuc-C4BPA
using lens culinaris agglutinin (LCA)-lectin, which specifically
binds to fucose. We demonstrated that serum Fuc-C4BPA is a
potential diagnostic biomarker for PDAC with clinical
significance.
Materials and methods
Serum samples of participants
The inclusion criterion for the study was patients
aged 20–85 who have been histologically diagnosed with PDAC or
chronic pancreatitis (CP). Patients with other malignancies in the
active phase were excluded from the study. For the training set, we
measured the serum Fuc-C4BPA level in 19 patients with PDAC, 10
patients with CP, and 40 age and gender-matched healthy volunteers
(HVs). For the validation set 1, the Fuc-C4BPA levels were
comparatively analyzed in nine pairs of pre- and post-operative
(3-4 weeks post-surgery when the serum levels of C-reactive protein
returned to the normal range) sera obtained from patients with PDAC
who underwent curative surgery. Additionally, the diagnostic values
of conventional tumor markers, such as CA19-9, carcinoembryonic
antigen (CEA), total C4BPA, and Fuc-C4BPA were investigated in the
validation set 2. In the validation set 3, the serum Fuc-C4BPA
levels were comparatively analyzed among 50 HVs, 20 patients with
CP, and 45 patients with PDAC to examine the correlation between
the serum C4BPA levels and the clinicopathological features of
patients with PDAC (Table I). The
blood samples from patients with PDAC or CP were collected at the
Department of General Surgery, Chiba University Hospital between
May 2011 and March 2019. The samples from HVs were collected at the
Kashiwado Hospital. The procedures for sample collection and
processing were performed as previously reported (11). The ethics committee of each institute
approved the protocol (approval number: 2155 for Chiba University,
and 007 for Kashiwado Hospital). Written informed consent was
obtained from all the patients and HVs. All data from the
participants were fully anonymized, and the study was performed
according to the guidelines of the Declaration of Helsinki
1975.
 | Table I.Clinical characteristics of
participants. |
Table I.
Clinical characteristics of
participants.
|
|
| Sex |
|---|
|
|
|
|
|---|
| Patient set | Age, mean ± SD | M | F |
|---|
| Training set |
|
|
|
| HVs
(n=40) | 65±5 | 25 | 15 |
| CP
(n=10) | 51±14 | 9 | 1 |
| PDAC
(n=19) | 68±10 | 11 | 8 |
| Validation set 1 |
|
|
|
|
Pre-/post-operative sera of
PDAC (n=9) | 69±13 | 5 | 4 |
| Validation set 2 |
|
|
|
| HVs
(n=10) | 66±5 | 5 | 5 |
| CP
(n=10) | 58±12 | 9 | 1 |
| PDAC (n=17) | 63±11 | 9 | 8 |
| Validation set 3 |
|
|
|
| HVs
(n=50; training set + validation set 2) | 67±5 | 30 | 20 |
| CP (n=20;
training set + validation set 2) | 55±13 | 18 | 2 |
| PDAC
(n=45; training set + validation set 1+2) | 66±11 | 25 | 20 |
Western blot analysis
To examine the specificity of the anti-C4BPA
antibody, recombinant human C4BPA protein (Abnova) and serum sample
of a patient with PDAC were subjected to sodium dodecyl
sulfate-polyacrylamide gel electrophoresis using a 10–20% gradient
gel (DRC, Tokyo, Japan) in the absence of β-mercaptoethanol. The
resolved proteins were transferred to a polyvinyl difluoride
membrane. To minimize nonspecific binding, the membrane was
incubated with Blocking One (Nacalai Tesque). The membrane was
washed thrice with PBST (phosphate-buffered saline (PBS) containing
0.1% (v/v) Tween-20) and incubated with anti-C4BPA polyclonal
antibodies (LifeSpan BioSciences, Inc.) for 1 h at room
temperature. Next, the membrane was washed thrice with PBST and
incubated with rabbit horseradish peroxidase (HRP)-conjugated
anti-mouse immunoglobulin secondary antibody (Dako Japan) for 1 h
at room temperature. The immunoreactive bands were visualized using
Pierce Western Blotting substrate (Thermo Fisher Scientific).
Precision plus protein dual-color standards (Bio-Rad Laboratories,
Inc.) were used as internal references.
Immobilization of antibody to a
polystyrene microtiter plate
The anti-C4BPA polyclonal antibodies in PBS were
dispensed into a 96-well polystyrene microtiter plate (Thermo
Fisher Scientific) at a concentration of 0.5 mg/well and incubated
for 1 day at 4°C. The plate was washed thrice with PBS containing
0.05% Tween-20. Next, the wells of the microtiter plate were coated
with 20% NOF102 containing 10% sucrose for one day at 4°C, followed
by drying for seven days at 4°C. The microtiter plate was
maintained at 4°C until use.
LCA-lectin ELISA conditions
First, 100-µl aliquots of 100-fold diluted serum
samples were added in duplicates to each well of a microtiter plate
washed with PBS. The samples were incubated at room temperature for
1 h and washed thrice. HRP-conjugated LCA-lectin (J-Chemical, Inc.)
in PBS containing 0.05% Tween-20 (100 µl) was added to each well
and the samples were incubated at room temperature for 30 min. The
plate was washed thrice and 100 µl of TMB solution (Fujifilm Wako
Pure Chemical Corporation) was added to each well. After incubation
at room temperature for 10 min, 100 µl of stop solution was added.
The absorbance of the mixtures was measured at 450 nm.
Statistical analysis
Numerical data are presented as the means ± standard
deviations. All statistical analyses, including linear regression
analysis and Pearson's correlation coefficient calculation were
performed, and receiver operator characteristic (ROC) and the area
under the ROC curves (AUC) were calculated using SPSS v.19.0
(SPSS). For non-parametric data, the means of the two groups were
compared using Mann-Whitney U test, Welch's t-test. The
means of the two groups among three groups were compared using
Dunn's nonparametric comparison for post hoc testing after a
Kruskal-Wallis test. The means of four groups were compared the
differences using ANOVA test and the Tukey-Kramer post hoc test.
The differences were considered significant at
P<0.05.
Results
Establishment and characterization of
a hybrid LCA-lectin ELISA for measuring Fuc-C4BPA
We first developed a sandwich ELISA system, which
has a higher sensitivity than a previously reported ELISA system
(4), for measuring total C4BPA
levels. The anti-C4BPA antibody was used to detect recombinant
human C4BPA protein and serum C4BPA in a patient with PDAC
(Fig. 1A). This antibody was used as
a capture antibody to establish a novel hybrid LCA-lectin ELISA for
measuring the Fuc-C4BPA levels (Fig.
1B). A standard curve was generated based on the absorbance
value of the diluted C4BPA in the HEK293T cell lysate (OriGene
Technologies, Inc.) to evaluate the correlation between the
absorbance values and the C4BPA concentrations. The working
concentration range of Fuc-C4BPA was 0.0–2.0 AU/ml (Fig. 1C). The assay results revealed a
linear correlation between the absorbance values and the C4BPA
levels at a concentration range of 0.0–2.0 AU/ml (y=1.0009×,
r2=0.9988, P<0.0001; Pearson's correlation) (Fig. 1D). The detection limit was estimated
by assaying the zero concentration eight times and was defined as
the C4BPA ‘zero’ concentration + 3 SD. The limit of Fuc-C4BPA
detection was 0.13 AU/ml. To examine the within-run and between-run
reproducibility, the precision of the assay was examined at
Fuc-C4BPA concentrations of 0.23 and 1.39 AU/ml. Within-assay
coefficient of variations (CVs) were determined using eight
replicates of each sample. The assay was repeated on five different
days to determine the between-assay CVs (two replicates of each
sample per day). The within-run and between-run CVs were 2.6–6.7
and 1.8–3.6%, respectively.
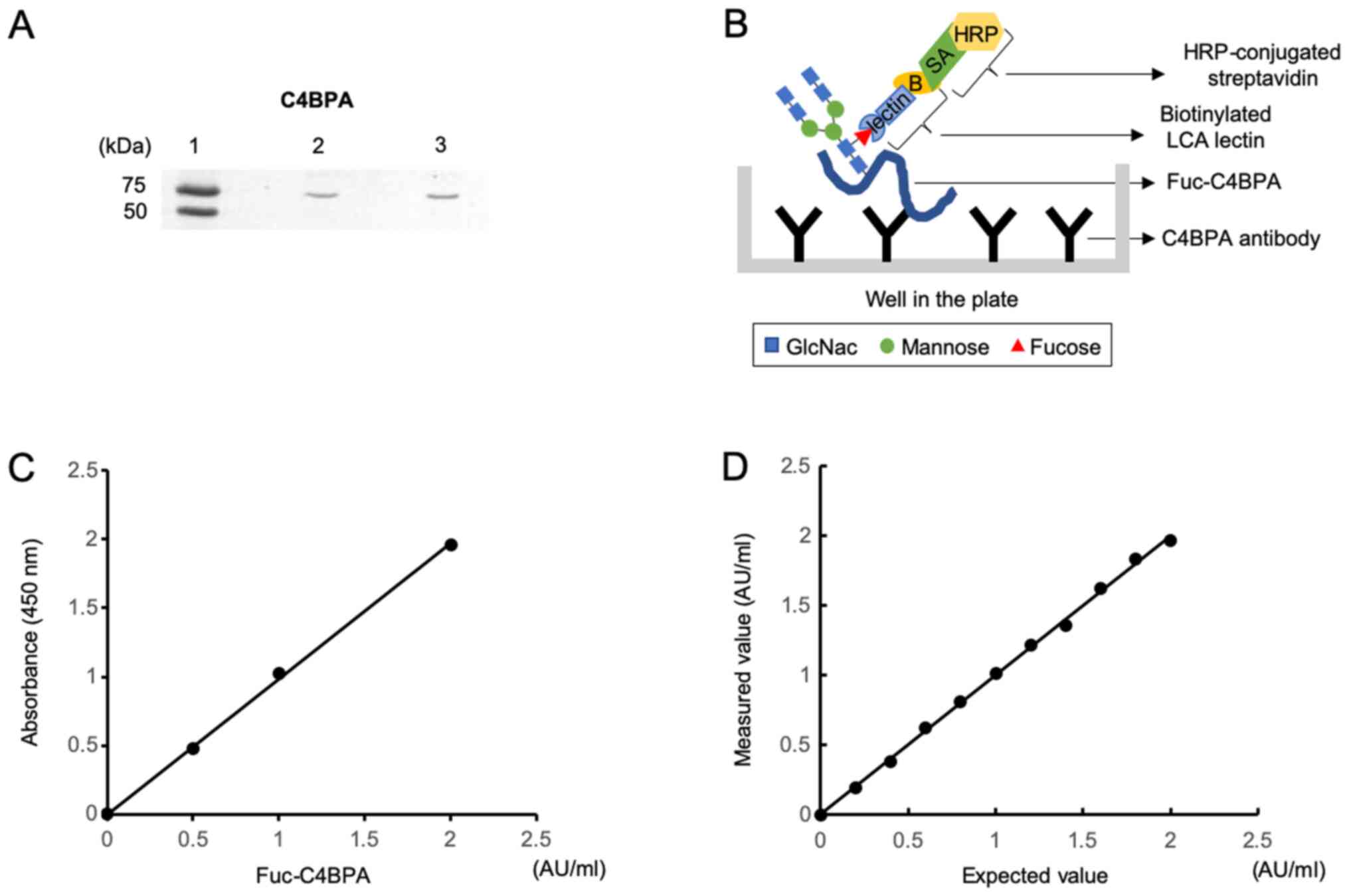 | Figure 1.Establishment of lens culinaris
agglutinin-lectin ELISA for the detection of fucosylated C4BPA. (A)
Western blot analysis. Immunoreactive bands were observed at the
predicted molecular weight of C4BPA (67 kDa) upon incubation of
recombinant human C4BPA protein and the serum sample from a patient
with PDAC with anti-C4BPA polyclonal antibodies. Lane 1, internal
references. The molecular weights of the upper and lower bands are
75 and 50 kDa, respectively. Lane 2, recombinant human C4BPA
protein. Lane 3, serum sample from a patient with PDAC. (B)
Schematic diagram of the antibody-lectin sandwich assay.
Immobilized anti-C4BPA antibody captures C4BPA in the serum.
Fucosylation of C4BPA is detected using a biotinylated lectin,
followed by incubation with horseradish peroxidase-conjugated
streptavidin. (C) Standard curves of Fuc-C4BPA following ELISA.
Correlation between absorbance values and concentration of
Fuc-C4BPA. Four concentrations of Fuc-C4BPA were examined using
ELISA. (D) Linearity of the ELISA results, which fitted into the
following linear equation: y=1.0009× (r2=0.9988;
P<0.0001). C4BPA, C4b-binding protein α-chain; PDAC, pancreatic
ductal adenocarcinoma; HRP, horseradish peroxidase; GlcNac,
N-Acetylglucosamine; Fuc, fucosylated; LCA, lens culinaris
agglutinin. |
Finally, interference was assessed in the samples
containing 0.23 AU/ml of Fuc-C4BPA. Potential interference
materials were added to the sera at various concentrations. There
was no substantial interference from hemoglobin (up to 5,000 mg/l),
free bilirubin (up to 207 mg/l), ditaurobilirubin (up to 204 mg/l),
chyle (up to 1,400 formazine turbidity units, equal to 1,176 mg/l
triglycerides), ascorbic acid (up to 500 mg/l), and rheumatoid
factor (up to 500 U/l).
Fucosylated C4BPA levels are
upregulated in the serum of patients with PDAC
The established LCA-lectin ELISA was used to examine
the serum Fuc-C4BPA levels in 69 serum samples (40 age-matched HVs,
10 patients with CP, and 19 patients with PDAC) of the training set
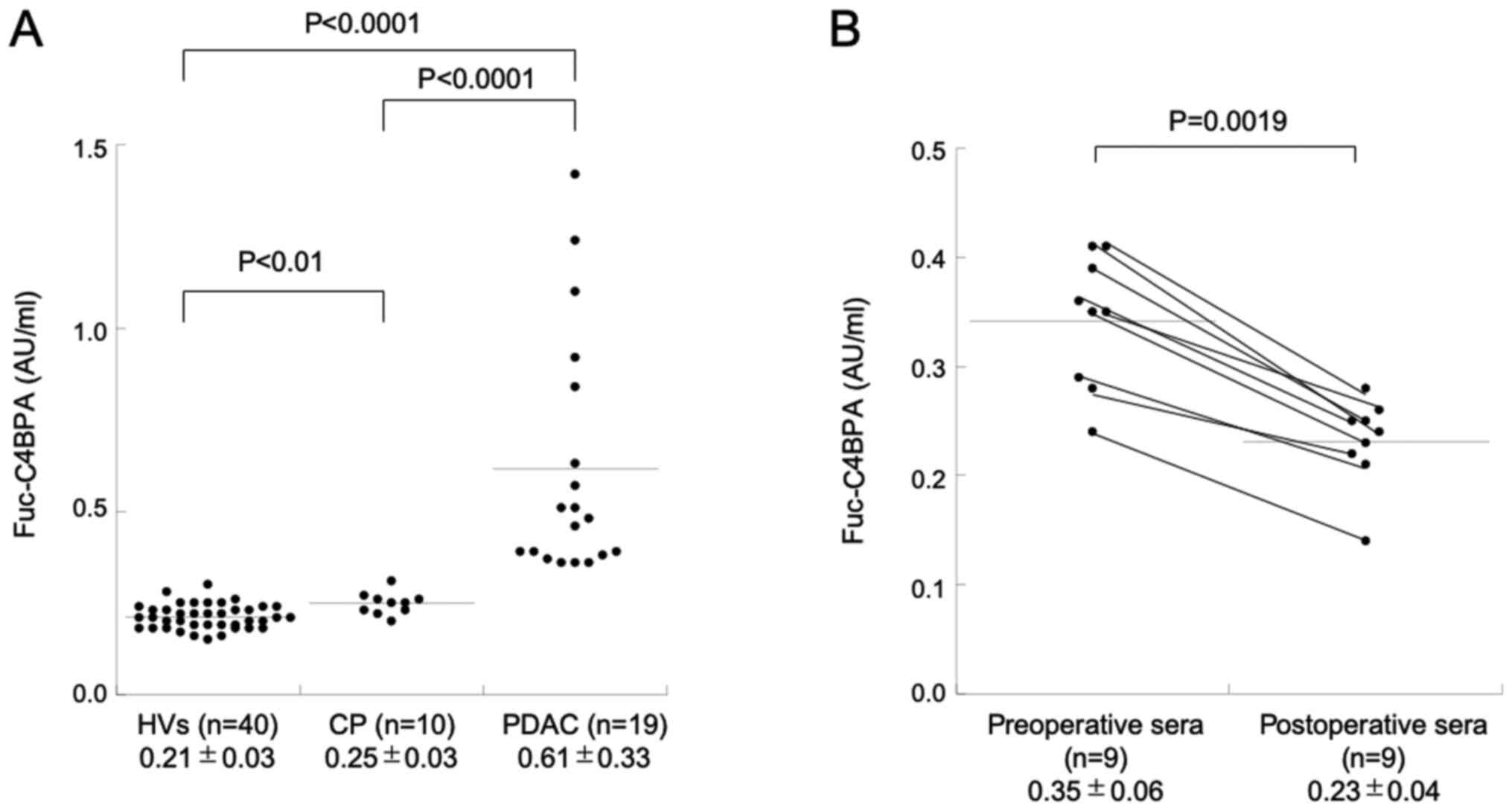
(Table I). As shown in Fig. 2A, the serum level of Fuc-C4BPA in
patients with PDAC (0.61±0.33 AU/ml) was significantly higher than
that in HVs (0.21±0.06 AU/ml; P<0.0001) and patients with CP
(0.25±0.03 AU/ml; P<0.0001). Additionally, the serum level of
Fuc-C4BPA in patients with CP was higher than that in HVs
(P<0.01). Next, the Fuc-C4BPA levels were measured in nine pairs
of pre- and post-operative sera in the validation set 1.
Interestingly, the serum Fuc-C4BPA level significantly decreased in
all nine patients after the curative operation (Fig. 2B) (P=0.0019; Wilcoxon signed rank
test).
Fucosylated C4BPA levels have a higher
PDAC detection accuracy than total C4BPA levels
To examine the discriminatory power of serum
Fuc-C4BPA and total C4BPA levels, we compared the levels of
Fuc-C4BPA and total C4BPA among 17 patients with PDAC and
age-matched benign controls comprising 10 HVs and 10 patients with
CP in the validation set 2. The serum levels of total C4BPA in
patients with PDAC (87.7±41.7 µg/ml) were significantly higher than
those in HVs (38.9±8.4 µg/ml) (P<0.001) and patients with CP
(43.4±11.5 µg/ml) (P<0.01). The HVs and patients with CP did not
exhibit significant differences in the serum levels of total C4BPA
(P=0.35) (Fig. 3A). Meanwhile, the
serum levels of Fuc-C4BPA in patients with PDAC (0.54±0.22 AU/ml)
were significantly higher than those in HVs (0.20±0.04 AU/ml;
P<0.0001) and patients with CP (0.25±0.03 AU/ml; P<0.0001) in
the validation set 2 (Fig. 3B). The
serum levels of total C4BPA and Fuc-C4BPA were not significantly
correlated in this sample set (P=0.61, r=−0.13; Pearson's
correlation) (Fig. 3C).
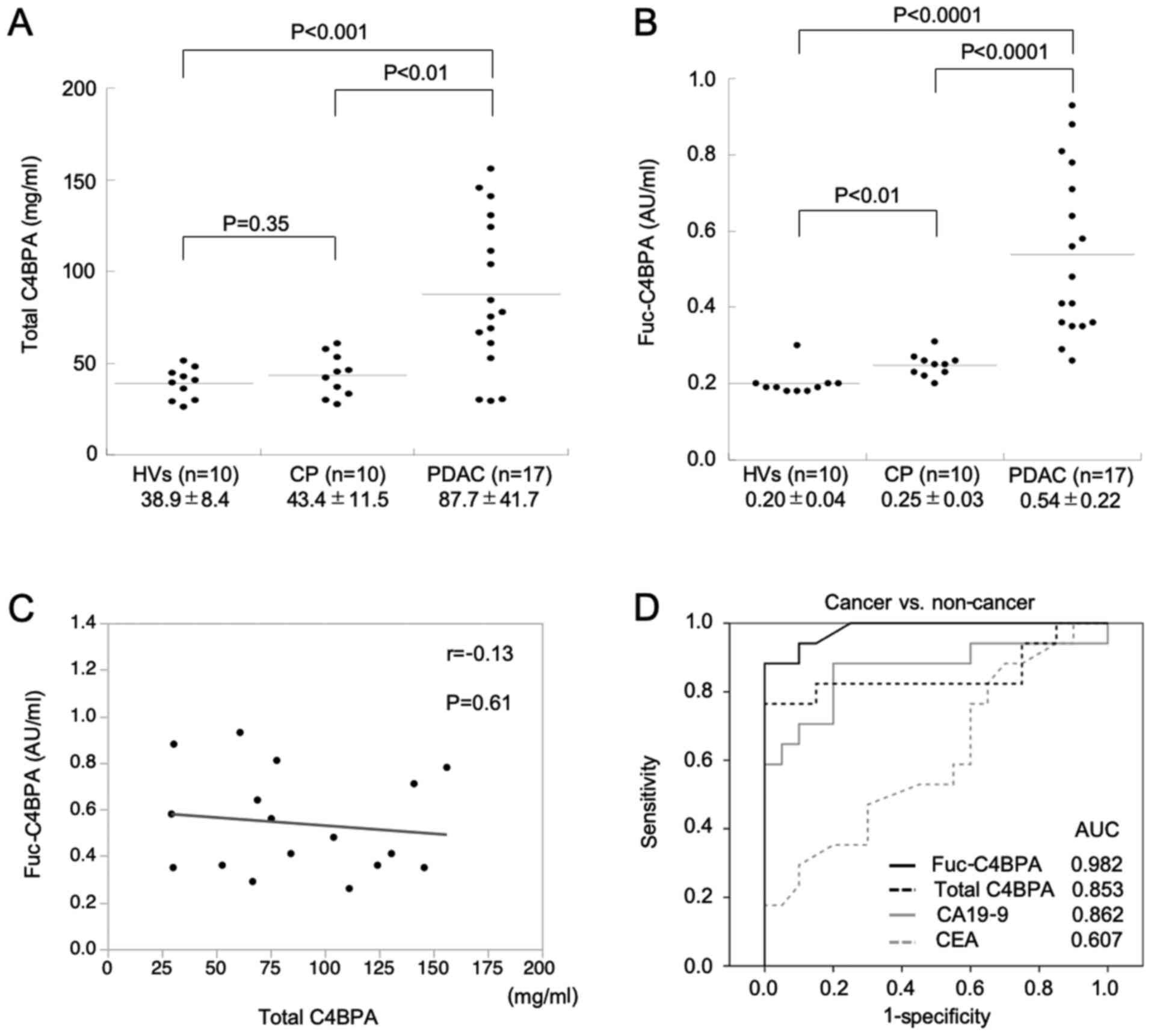 | Figure 3.Serum levels of total C4BPA and
Fuc-C4BPA are upregulated in patients with PDAC. (A) Comparison of
total serum C4BPA levels among HVs, patients with CP and patients
with PDAC using ELISA. Total serum C4BPA levels in patients with
PDAC were significantly higher than those in HVs and patients with
CP. HVs and patients with CP did not exhibit significant
differences in total serum C4BPA levels. (B) Comparison of
Fuc-C4BPA serum levels between HVs, patients with CP and patients
with PDAC using ELISA. The serum Fuc-C4BPA levels in patients with
PDAC were significantly higher than those in HVs and patients with
CP. (C) Comparison of serum levels of total C4BPA and Fuc-C4BPA.
Serum Fuc-C4BPA levels were not correlated with total serum C4BPA
levels in patients with PDAC. (D) Fuc-C4BPA has higher PDAC
diagnostic accuracy than total C4BPA, CA19-9 and carcinoembryonic
antigen (CEA). The receiver operating characteristic curve analyses
of the serum levels of Fuc-C4BPA, total C4BPA, CA19-9 and CEA
between patients with PDAC, HVs and patients with CP. PDAC,
pancreatic ductal adenocarcinoma; C4BPA, C4b-binding protein
α-chain; Fuc, fucosylated; HVs, healthy volunteers; CP, chronic
pancreatitis; CA19-9, carbohydrate antigen 19-9; CEA,
carcinoembryonic antigen; AUC, area under the curve. |
Next, the receiver operating characteristic (ROC)
curves were generated to evaluate the ability of Fuc-C4BPA, total
C4BPA, CA19-9, and CEA levels to distinguish patients with PDAC
from non-cancer (HVs and CP) participants. The cutoff values of
these four markers were set at levels (mean + 2 SD) that yielded a
specificity of 97.7% when compared with those of the participants
without cancer (HVs and CP patients); 61.3 µg/ml, 0.297 AU/ml, 34.2
U/ml, and 7.3 ng/ml for total C4BPA, Fuc-C4BPA, CA19-9, and CEA,
respectively. The area under the ROC curve (AUC) of Fuc-C4BPA,
total C4BPA, CA19-9, and CEA was 0.982, 0.853, 0.862, and 0.607,
respectively, in the validation set 2 (Fig. 3D). These results suggest that serum
Fuc-C4BPA expression is a potential novel diagnostic biomarker for
PDAC.
Fuc-C4BPA can identify the CA19-9
false-negative cases among patients with PDAC
To validate the diagnostic value of serum Fuc-C4BPA
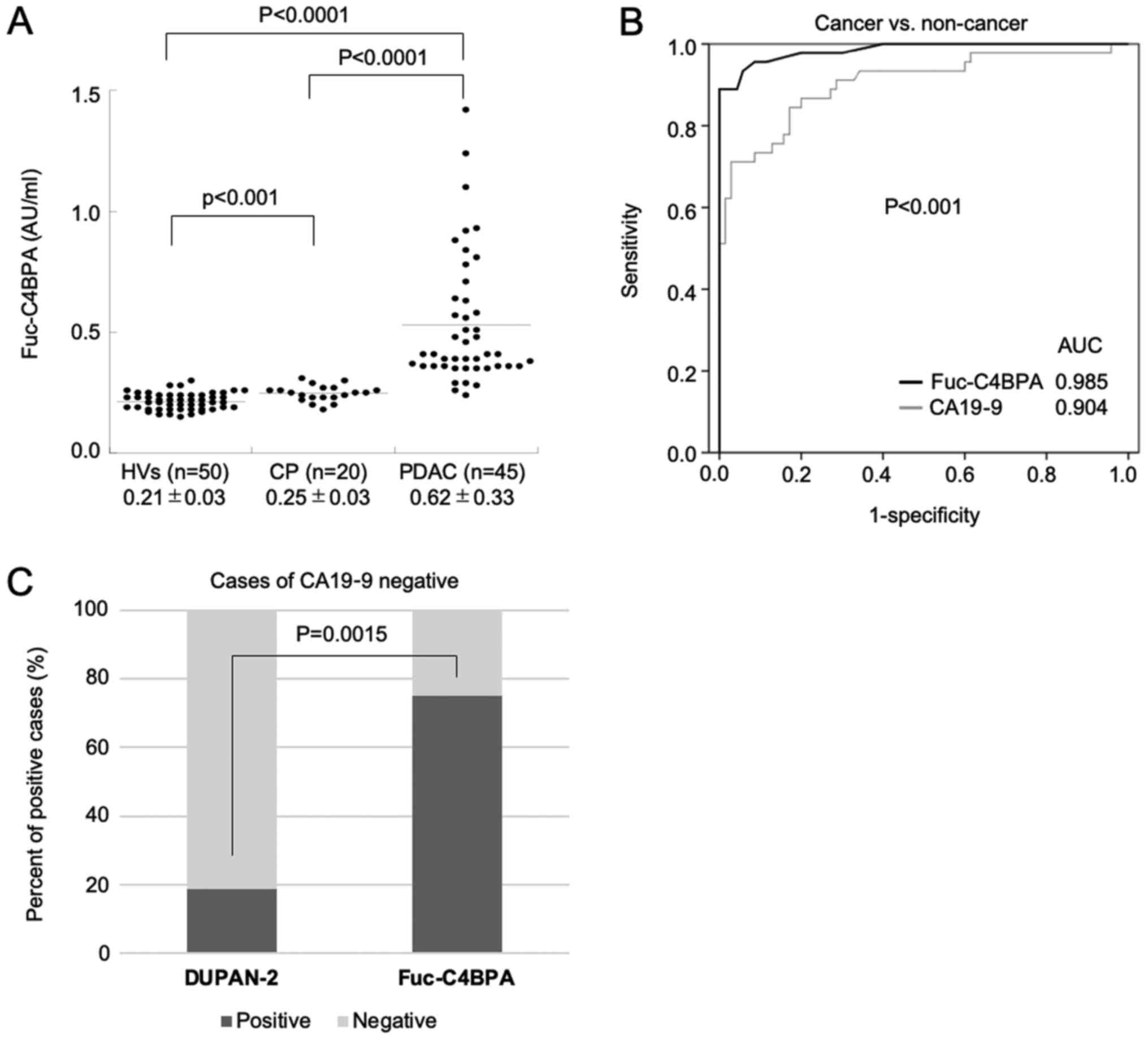
levels in PDAC, the PDAC sample size in the validation set 3 was
increased to 45 from the training set and the validation sets 1 and
2. The Fuc-C4BPA levels in patients with PDAC (0.62±0.33 AU/ml)
were significantly higher than those in HVs (0.21±0.03 AU/ml;
P<0.0001) and patients with CP (0.25±0.03 AU/ml; P<0.0001)
(Fig. 4A). The AUC of Fuc-C4BPA and
CA19-9 was 0.985 and 0.904, respectively. This indicated that serum
Fuc-C4BPA is a better diagnostic PDAC biomarker than serum CA19-9
(Fig. 4B; P<0.001).
The accuracy of a single serum biomarker is limited
in cancer detection. To increase the detection rate, additional
biomarkers are often tested in a clinical setting. CA19-9 reacts
with a monoclonal antibody directed against sialyl Lewis A antigen.
Hence, Lewis A antigen-negative patients do not exhibit increased
levels of CA19-9 (12). To overcome
this limitation, the level of duke pancreatic monoclonal antigen
type 2 (DUPAN-2) is measured because it is recognized by a
monoclonal antibody directed against sialyl Lewis C antigen, which
is the precursor of sialyl Lewis A antigen (13). We examined the ability of Fuc-C4BPA
to identify the CA19-9false-negative cases among patients with
PDAC. Among the 16 CA19-9-negative patients, the percentage of the
Fuc-C4BPA-positive cases (12/16; 75%) was significantly higher than
that of DUPAN-2-positive cases (3/16; 18.8%) (P=0.0015; Chi-square
test) (Fig. 4C). These results
indicated that Fuc-C4BPA can identify the CA19-9 false-negative
cases among patients with PDAC.
Fucosylated C4BPA predicts
pathological lymph node (LN) metastasis in patients with PDAC
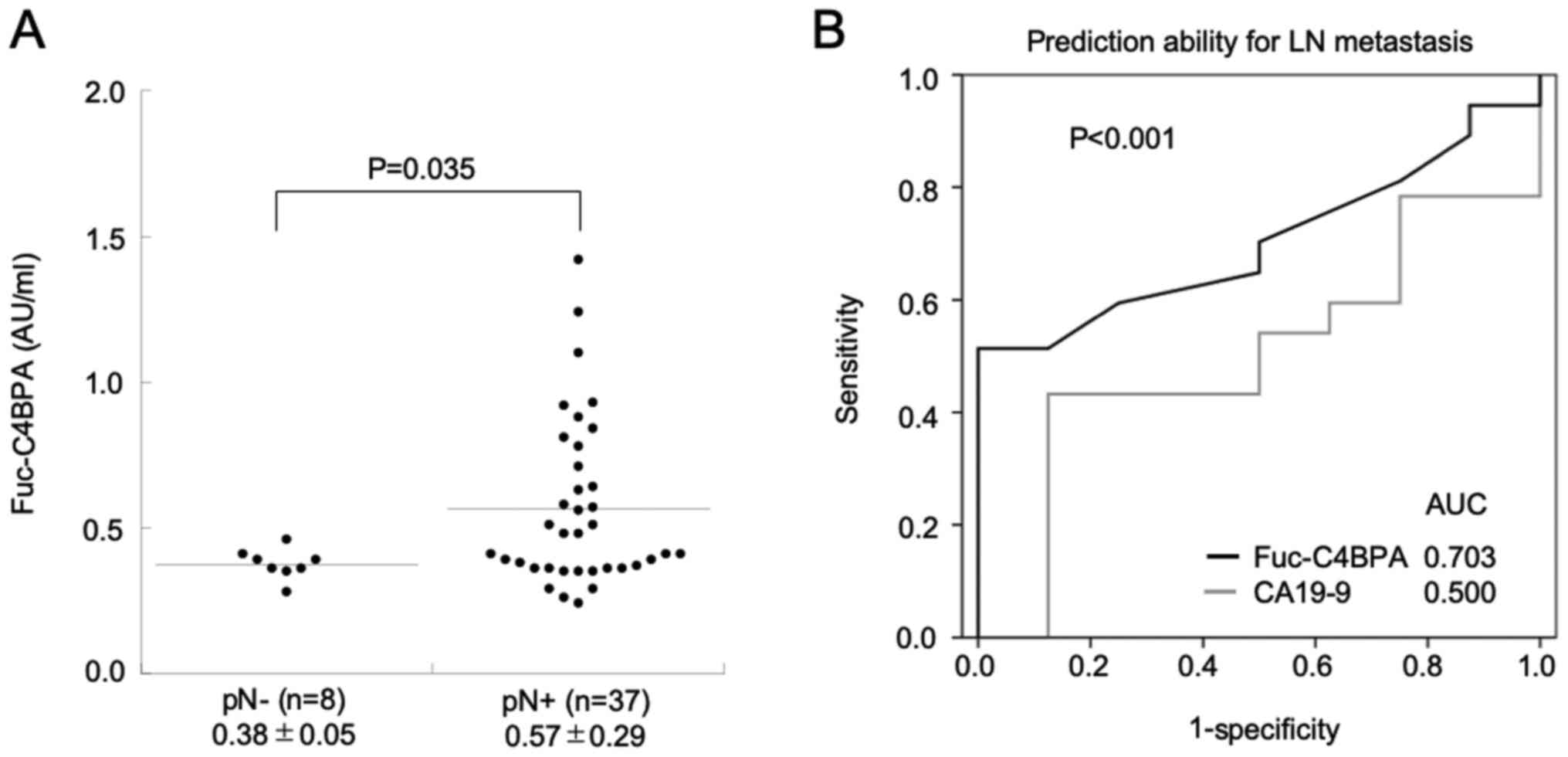
Next, we investigated the correlation between the
levels of Fuc-C4BPA and the clinicopathological features of
patients with PDAC. Among the major clinical parameters, such as
factors contributing to the TNM classification system of malignant
tumors, the tumor stage was not correlated with the levels of
Fuc-C4BPA and CA19-9 (Table II).
Pathological LN metastasis (pN+) was positively correlated with
high serum Fuc-C4BPA levels in the validation set 3 (P=0.035;
Welch's t-test) (Fig. 5A).
Furthermore, the ability of Fuc-C4BPA and CA19-9 levels to predict
LN metastasis was comparatively evaluated. The AUC of serum
Fuc-C4BPA levels (0.703) was significantly higher than that of
serum CA19-9 levels (0.500) (P<0.001) (Fig. 5B). These findings suggested that
Fuc-C4BPA is a better predictor of LN metastasis than CA19-9 in
patients with PDAC.
 | Table II.Fuc-C4BPA, CA19-9 and CEA levels in
the sera of participants. |
Table II.
Fuc-C4BPA, CA19-9 and CEA levels in
the sera of participants.
|
| Non-cancer
(n=70) | PDAC (validation set
3; n=45) |
|---|
|
|
|
|
|---|
| Serum markers | HVs (n=50) | CP (n=20) | Stage I (n=8) | Stage II (n=14) | Stage III (n=19) | Stage IV (n=4) | P-value |
|---|
| Fuc-C4BPA
(AU/ml) | 0.21±0.03 | 0.25±0.03 | 0.38±0.05 | 0.57±0.29 | 0.58±0.29 | 0.48±0.17 | 0.13 |
| CA19-9 (U/ml) | 11.2±8.1 | 16.7±14.5 | 1,697.5±4,301.4 | 208.8±398.8 | 365.2±467.5 | 347.6±317.7 | 0.65 |
| CEA (ng/ml) | 3.1±1.9 | 3.5±2.4 | 10.8±18.6 | 4.6±3.6 | 3.1±2.5 | 3.0±1.7 | 0.47 |
Discussion
Although several novel biomarkers have been
identified using proteomic analysis, most of them are not available
for clinical diagnosis. Generally, biomarkers are identified
through comparative analysis of the disease and control groups
based on semiquantitative methods (discovery phase) and
identification of candidates using MS (validation phase). Multiple
reaction monitoring (MRM) in MS, which does not involve the use of
an antibody, and selective reaction monitoring are used to detect
and quantify the target proteins (11). The serum Fuc-C4BPA level cannot be
measured using MRM as its molecular weight exceeds the detectable
range of MRM. Clinically, steroid hormones (14), vitamins (15), and thyroid hormones (16) can be measured using MS. However, the
clinical applications of MS for diagnosis are limited due to
various limitations, including complex sample preparation, lack of
high-throughput methods, and the need for automation, interface to
the laboratory information system, and device standardization.
Previously, the serum level of fully-sialylated C4BPA was reported
to be upregulated in patients with clear-cell ovarian cancer.
However, the utility of the serum level of fully-sialylated C4BPA
for cancer diagnosis has been limited due to the absence of a
high-throughput method for measurement (10).
Cancer progression encompasses major alterations in
molecular glycosylation, such as incomplete synthesis of O-glycan
structures, increased expression of branched N-glycans, expression
of terminal sialylated glycans, and altered fucosylation. Capturing
lectin using Aleuria aurantia lectin, Pisum sativum
agglutinin, and LCA, which can recognize the core fucosylation
structure, is the established approach to identify the core
fucosylated glycoproteins. In this study, we developed an ELISA
using C4BPA polyclonal antibodies and LCA-lectin. The ELISA system
exhibited good basic performances, including day-to-day
reproducibility and repeatability. The easy-to-use ELISA system
developed in this study to detect serum Fuc-C4BPA is a useful
method for diagnosing PDAC.
Cancer-related immunocompetent cells or complement
factors in the tumor microenvironment are associated with cancer
progression (17). The contribution
of aberrant fucosylation to the interactions within the tumor
microenvironment remains poorly understood. In this study, we
demonstrated that the serum Fuc-C4BPA levels were significantly
upregulated in patients with PDAC. Additionally, the serum
Fuc-C4BPA levels in the post-operative patients were significantly
lower than those in the preoperative patients. Fucosylation is
reported to be upregulated at an early stage of colon
carcinogenesis (18). However, other
studies have reported that de-fucosylation through genetic mutation
promotes the escape from natural killer cell-mediated tumor
surveillance and the development of malignant characteristics in
certain types of advanced cancer (19). To understand the role of fucosylation
during cancer progression from early to late-stage, the mechanisms
underlying fucosylation or de-fucosylation induced by the
interaction between tumor cells and molecules in the tumor
microenvironments must be elucidated.
The upregulated CA 19-9 levels are reported to be an
independent prognostic factor for LN metastasis. Preoperative
chemotherapy can improve the prognosis of patients with PDAC
exhibiting LN metastasis (20,21).
Therefore, the prediction of LN metastasis is important to develop
an efficient therapeutic strategy for patients with PDAC.
Contrast-enhanced computed tomography (CECT) is commonly used for
the preoperative diagnosis of LN metastasis in patients with PDAC.
A previous prospective study has demonstrated that the diagnostic
accuracy of LN metastasis through CECT is low (73%) in PDAC
(22). The significance of enlarged
LNs in PDAC is not well defined because the LNs can enlarge due to
local inflammation or biliary obstruction. Additionally, LN
metastasis is not correlated with this enlargement (23). A recent study reported that a
six-microRNA risk prediction model could distinguish patients with
PDAC patients exhibiting LN metastases from those with PDAC not
exhibiting LN metastases (AUCof 0.84 and 0.73 in the training and
validation sets, respectively) (24). In this study, the upregulated levels
of Fuc-C4BPA were positively correlated with LN metastasis. The AUC
of Fuc-C4BPA (0.703) was significantly higher than that of CA19-9
(0.500). This indicated that a single serum biomarker, Fuc-C4BPA,
is a good indicator of LN metastasis and can aid in determining the
treatment strategy for patients with PDAC.
There are several limitations associated with the
clinical application of serum Fuc-C4BPA level as a diagnostic
marker for PDAC. The diagnostic window (e.g. the detection limit)
of the lectin ELISA for measuring the Fuc-C4BPA level is narrow.
Additionally, the serum Fuc-C4BPA level, but not the serum total
C4BPA level, in patients with CP was significantly higher than that
in HVs. This may be due to the correlation between inflammation and
carcinogenesis, which are critical for the activation of various
common molecules. The identification and measurement of specific
target sites of fucosylation, which are directly involved in PDAC
progression, may resolve these issues. Moreover, this is a
retrospective study involving a small sample size. Further
validation in an independent and prospective large cohort is needed
to establish Fuc-C4BPA as a promising serum diagnostic biomarker
for PDAC. In conclusion, we established a novel lectin ELISA for
the measurement of serum Fuc-C4BPA. Serum Fuc-C4BPA has a powerful
diagnostic ability with potential applications for the development
of therapeutic strategies for PDAC.
Acknowledgements
Not applicable.
Funding
This work was supported by a Grant-in-Aid for
Scientific Research (KAKENHI; grant nos. ‘KIBAN’ C:19K07947,
19K09113, 20K09073 and ‘KIBAN’ B:19H03725).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KaS, SY, ST, FN, and MO were involved in the study
design, data analysis and development of the study. KoS, YM, HT KF,
TT, and SK collected and analyzed the clinical data. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Chiba University, Graduate School of Medicine
(approval no. #2155) and Kashiwado Hospital (approval no. #007),
respectively. Informed consent was obtained from all participants
and patients for the acquisition of clinical and pathological
information and for the use of serum samples.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Takano S, Yoshitomi H, Togawa A, Sogawa K,
Shida T, Kimura F, Shimizu H, Tomonaga T, Nomura F and Miyazaki M:
Apolipoprotein C-1 maintains cell survival by preventing from
apoptosis in pancreatic cancer cells. Oncogene. 27:2810–2822. 2008.
View Article : Google Scholar
|
|
2
|
Moulder R, Bhosale SD, Goodlett DR and
Lahesmaa R: Analysis of the plasma proteome using iTRAQ and
TMT-based Isobaric labeling. Mass Spectrom Rev. 37:583–606. 2018.
View Article : Google Scholar
|
|
3
|
Bhawal R, Oberg AL, Zhang S and Kohli M:
Challenges and opportunities in clinical applications of
blood-based proteomics in cancer. Cancers (Basel). 12:24282020.
View Article : Google Scholar
|
|
4
|
Sogawa K, Takano S, Iida F, Satoh M,
Tsuchida S, Kawashima Y, Yoshitomi H, Sanda A, Kodera Y, Takizawa
H, et al: Identification of a novel serum biomarker for pancreatic
cancer, C4b-binding protein α-chain (C4BPA) by quantitative
proteomic analysis using tandem mass tags. Br J Cancer.
115:949–956. 2016. View Article : Google Scholar
|
|
5
|
Mikami M, Tanabe K, Matsuo K, Miyazaki Y,
Miyazawa M, Hayashi M, Asai S, Ikeda M, Shida M, Hirasawa T, et al:
Fully-sialylated alpha-chain of complement 4-binding protein:
Diagnostic utility for ovarian clear cell carcinoma. Gynecol Oncol.
139:520–528. 2015. View Article : Google Scholar
|
|
6
|
Liu YS, Luo XY, Li QR, Li H, Li C, Ni H,
Li RX, Wang R, Hu HC, Pan YJ, et al: Shotgun and targeted
proteomics reveal that pre-surgery serum levels of LRG1, SAA, and
C4BP may refine prognosis of resected squamous cell lung cancer. J
Mol Cell Biol. 4:344–347. 2012. View Article : Google Scholar
|
|
7
|
Jia L, Zhang J, Ma T, Guo Y, Yu Y and Cui
J: The function of fucosylation in progression of lung cancer.
Front Oncol. 8:5652018. View Article : Google Scholar
|
|
8
|
Li D and Satomura S: Biomarkers for
hepatocellular carcinoma (HCC): An update. Adv Exp Med Biol.
867:179–193. 2015. View Article : Google Scholar
|
|
9
|
Jelski W and Mroczko B: Biochemical
diagnostics of pancreatic cancer-Present and future. Clin Chim
Acta. 498:47–51. 2019. View Article : Google Scholar
|
|
10
|
Tanabe K, Matsuo K, Miyazawa M, Hayashi M,
Ikeda M, Shida M, Hirasawa T, Sho R and Mikami M: UPLC-MS/MS based
diagnostics for epithelial ovarian cancer using fully sialylated
C4-binding protein. Biomed Chromatogr. 32:e41802018. View Article : Google Scholar
|
|
11
|
Umemura H, Nezu M, Kodera Y, Satoh M,
Kimura A, Tomonaga T and Nomura F: Effects of the time intervals
between venipuncture and serum preparation for serum peptidome
analysis by matrix-assisted laser desorption/ionization
time-of-flight mass spectrometry. Clin Chim Acta. 406:179–180.
2009. View Article : Google Scholar
|
|
12
|
Narimatsu H, Iwasaki H, Nakayama F,
Ikehara Y, Kudo T, Nishihara S, Sugano K, Okura H, Fujita S and
Hirohashi S: Lewis and secretor gene dosages affect CA19-9 and
DU-PAN-2 serum levels in normal individuals and colorectal cancer
patients. Cancer Res. 58:512–518. 1998.
|
|
13
|
Kawa S, Oguchi H, Kobayashi T, Tokoo M,
Furuta S, Kanai M and Homma T: Elevated serum levels of Dupan-2 in
pancreatic cancer patients negative for Lewis blood group
phenotype. Br J Cancer. 64:899–902. 1991. View Article : Google Scholar
|
|
14
|
Stanczyk FZ and Clarke NJ: Advantages and
challenges of mass spectrometry assays for steroid hormones. J
Steroid Biochem Mol Biol. 121:491–495. 2010. View Article : Google Scholar
|
|
15
|
Farrell CJ, Martin S, McWhinney B, Straub
I, Williams P and Herrmann M: State-of-the-art vitamin D assays: A
comparison of automated immunoassays with liquid
chromatography-tandem mass spectrometry methods. Clin Chem.
58:531–542. 2012. View Article : Google Scholar
|
|
16
|
Soldin SJ, Soukhova N, Janicic N, Jonklaas
J and Soldin OP: The measurement of free thyroxine by isotope
dilution tandem mass spectrometry. Clin Chim Acta. 358:113–118.
2005. View Article : Google Scholar
|
|
17
|
Reis ES, Mastellos DC, Ricklin D,
Mantovani A and Lambris JD: Complement in cancer: Untangling an
intricate relationship. Nat Rev Immunol. 18:15–18. 2018. View Article : Google Scholar
|
|
18
|
Muinelo-Romay L, Vazquez-Martin C,
Villar-Portela S, Cuevas E, Gil-Martín E and Fernández-Briera A:
Expression and enzyme activity of alpha (1,6) fucosyltransferase in
human colorectal cancer. Int J Cancer. 123:641–646. 2008.
View Article : Google Scholar
|
|
19
|
Moriwaki K, Noda K, Furukawa Y, Ohshima K,
Uchiyama A, Nakagawa T, Taniguchi N, Daigo Y, Nakamura Y, Hayashi N
and Miyoshi E: Deficiency of GMDS leads to escape from NK
cell-mediated tumor surveillance through modulation of TRAIL
signaling. Gastroenterology. 137:188–198. 2009. View Article : Google Scholar
|
|
20
|
Lambert A, Schwarz L, Borbath I, Henry A,
Van Laethem JL, Malka D, Ducreux M and Conroy T: An update on
treatment options for pancreatic adenocarcinoma. Ther Adv Med
Oncol. 11:17588359198755682019. View Article : Google Scholar
|
|
21
|
Tran Cao HS, Zhang Q, Sada YH, Silberfein
EJ, Hsu C, Van Buren G II, Chai C, Katz MHG, Fisher WE and
Massarweh NN: Value of lymph node positivity in treatment planning
for early stage pancreatic cancer. Surgery. 162:557–567. 2017.
View Article : Google Scholar
|
|
22
|
Roche CJ, Hughes ML, Garvey CJ, Campbell
F, White DA, Jones L and Neoptolemos JP: CT and pathologic
assessment of prospective nodal staging in patients with ductal
adenocarcinoma of the head of the pancreas. AJR Am J Roentgenol.
180:475–480. 2003. View Article : Google Scholar
|
|
23
|
Tseng DS, van Santvoort HC, Fegrachi S,
Besselink MG, Zuithoff NP, Borel Rinkes IH, van Leeuwen MS and
Molenaar IQ: Diagnostic accuracy of CT in assessing extra-regional
lymphadenopathy in pancreatic and peri-ampullary cancer: A
systematic review and meta-analysis. Surg Oncol. 23:229–235. 2014.
View Article : Google Scholar
|
|
24
|
Nishiwada S, Sho M, Banwait JK, Yamamura
K, Akahori T, Nakamura K, Baba H and Goel A: A microRNA signature
identifies pancreatic ductal adenocarcinoma patients at risk for
lymph node metastases. Gastroenterology. 159:562–574. 2020.
View Article : Google Scholar
|