Introduction
Colorectal cancer (CRC) is a leading cause of
mortality worldwide (1), and it has
been estimated that the morbidity and mortality of CRC are
increasing rapidly, with nearly 1.2 million new patients and
600,000 mortalities annually (2).
CRC treatment including surgery, chemotherapy and radiation are
commonly used; however, patients suffer severe side effects
following these therapies, such as toxicity, hepatic injury and
drug resistance (3,4). Thus, it remains urgent to identify
novel therapies for CRC, with high efficacy and fewer adverse
effects.
Curcumin (CUR), a natural product extracted from the
rhizome of Curcuma longa (5), is a drug with strong pharmacological
effects and limited side effects (6,7).
Increasing evidence have confirmed that CUR exerts antioxidant,
antibacterial, anti-inflammatory, antiproliferation and anticancer
effects (8–11). Furthermore, curcuminoids have been
approved by the US Food and Drug Administration (FDA) as ‘Generally
Recognized As Safe’ (GRAS) (12),
and clinical trials have demonstrated good tolerability and safety
profiles, at doses between 4,000–8,000 mg/day (12,13). In
another phase I clinical trial, CUR (at doses 0.45–3.6 g) was
administered to 15 patients with advanced CRC who were resistant to
chemotherapy for 4 months. The results demonstrated that treatment
with CUR was well tolerated and there was no toxicity at any doses
(14).
Previous studies have reported several molecular
mechanisms for the anticancer effects of CUR. For example, CUR
inhibits Axin2 expression in the colorectal cancer line, HCT116,
and modulates the Wnt/β-catenin signaling pathway (15). Yan et al (16) demonstrated that Axin2 is upregulated
and the Wnt/β-catenin signaling pathway is activated in human colon
tumor samples. It has also been reported that CUR can target
colorectal stem cells via the Wnt pathway to inhibit their
proliferation and drug resistance to chemotherapy (14), indicating the important role of the
Wnt/β-catenin signaling pathway in the tumorigenesis of CRC.
Another study demonstrated that CUR inhibits the proliferation and
induces apoptosis of human non-small cell lung cancer cells via the
PI3K/Akt signaling pathway (17).
Furthermore, the results of a squamous cell carcinoma study
reported that CUR inhibits cancer cell proliferation via the
epidermal growth factor receptor signaling pathway (18). In addition to the effects of CUR on
CRC progression through the NBR2/AMPK/mTOR pathway (19), CUR regulates the NF-κB and Src
protein kinase signaling pathways by inhibiting IκBα kinase
activation and IκBα phosphorylation (20). Collectively, these results indicate
several molecular mechanisms of the anticancer effects of CUR in
vitro. However, the in vivo mechanism remains to be
elucidated. Thus, the present study aimed to investigate the
anticancer effects of CUR and determine its underlying mechanisms
in CRC, in vivo.
The results of the present study demonstrated that
CUR abrogated CRC induced by azoxymethane (AOM)-dextran sodium
sulfate (DSS). In addition, reverse transcription-quantitative
(RT-q)PCR, western blot and immunohistochemistry (IHC) analysis
demonstrated that CUR attenuated CRC by downregulating Axin2
expression, which is a key downstream target in the Wnt/β-catenin
signaling pathway (14), and
decreasing the expression of pro-inflammatory cytokines. Taken
together, these results demonstrate the mechanisms of the
anticancer effects of CUR in vivo, and suggest that Axin2
may act as a potential therapeutic target for CRC.
Materials and methods
Reagents and chemicals
AOM (cat. no. SLBX6213) was purchased from
Sigma-Aldrich; Merck KGaA, while DSS (cat. no. S0798) was purchased
from MP Biomedicals, LCC., and CUR (cat. no. LH80S20) was purchased
from J&K Scientific, Ltd. (https://www.jkchemical.com). PEG 400 (cat. no.
C1925030) was purchased from Aladdin Industrial Corporation
(https://www.linkedin.com). The Easy Pure RNA kit
(cat. no. N10312), TransScript ALL-in-One First-Strand cDNA
Synthesis Super Mix (cat. no. N10117) and TransStart Tip Green qPCR
Super Mix (cat. no. N10710) were all purchased from Beijing
Transgen Biotech Co., Ltd. The Rabbit primary antibody against
Axin2 (1:2,000; cat. no. GR3203132-3; Abcam) and HRP-conjugated
secondary antibody (goat anti-rabbit IgG; 1:10,000; cat. no.
ab6722; Abcam) were purchased from Xi'an Zhuangzhi Biotechnology
Co., Ltd. (https://www.chemicalbook.com/ContactUs_186262.htm).
All other reagents in the experiment were pure analytical pure
grade.
Animals and drug administration
A total of 60 C57BL/6 male mice (20±2 g; 6–8 weeks
old) were obtained from the Laboratory Animal Center of Xi'an
Jiaotong University. Mice were randomly divided into three groups
(20 mice/group) as follows: Control group, AOM-DSS group and mice
treated with AOM-DSS and 500 mg/kg of CUR per day through oral
gavage. Mice were fed in an air-conditioned room at 22±2°C and
relative humidity of 55±5%, received standard water and chow
freely, with a 12 h light/dark cycle for 1 week prior to treatment.
All experimental animal protocols were approved by the Northwest
University Animal Ethics Committee (Xi'an, China, approval no.
NWU-AWC-20190202M), and all procedures were in line with relevant
ethical norms (euthanasia was used to treat the animals) (21). Sevoflurane (8%; Xi'an Zhuangzhi
Biotechnology Co., Ltd., http://www.chemicalbook.com/ContactUs_186262.htm)
was used for anesthesia (21).
Mice in the AOM-DSS group were treated with 10 mg/kg
AOM via intraperitoneal injection. After 7 days, mice were given 2%
DSS drinking water for 1 week plus regular drinking water for 2
weeks in one of three rounds to induce colorectal tumors. Mice
treated with CUR were given 500 mg/kg CUR suspension, while mice in
the other groups were given the same volume of vehicle control PEG
400 via oral gavage until the end of the experiment. The behavior
of mice, changes in body weight, hematochezia and the number of
dead mice were recorded daily. At week 10, the mice were sacrificed
using inhaled 8% sevoflurane to harvest plasma and colorectal
tissues for analysis. Mortality was verified by checking for a
heartbeat and consciousness. Colorectal tissues were cut open
longitudinally, washed in PBS and inspected under a dissection
microscope (magnification, ×20). Tumor size was determined as
follows: π × d2/4, where d is the diameter of each
tumor.
RT-qPCR
RT-qPCR analysis was performed according to the
manufacturer's instructions. Total RNA was extracted from
colorectal tumor tissues using the Easy Pure RNA kit. Each colonic
tissue was ground with a pestle, with continuous addition of liquid
nitrogen. Different amounts of lysates were added according to the
weight of colorectal tissue (1:1) and incubated in a water bath at
56°C for 20 min. The mixture was centrifuged at 9,710 × g for 5 min
at room temperature to collect the supernatant. Equal volumes of
70% ethanol was subsequently added, mixed with the supernatant and
centrifuged 9,710 × g for 30 sec at 4°C. Finally, the mixture was
centrifuged at 9,710 × g for 2 min at room temperature to
completely remove the residual ethanol. A total of 30 µl RNase-free
water was added and the mixture was left to stand for 1 min, prior
to centrifugation 9,710 × g for 2 min at room temperature to elute
the RNA. The quality of extracted RNA was measured using Nano-Drop
2000 (Shanghai Danding Trading Co., Ltd., http://www.china.cn/fenguangguangduji/4679745796.html)
and subsequently reverse transcribed into cDNA using a cDNA
synthesis kit for 15 min at room temperature.
The expression levels of β-catenin,
cyclooxygenase-2 (Cox-2), Axin2, interleukin (IL)-1β and
IL-6 were determined using the SYBR Green PCR kit (Beijing
Transgen Biotech Co., Ltd.) on an ABI7300 system (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The following
thermocycling conditions were used for qPCR: Initial denaturation
at 95°C for 3 min, followed by 39 cycles of 5 sec at 95°C and 1 min
at 58°C. Relative expression levels were quantified using the
2−∆∆Cq method (22) and
normalized to GAPDH. The primer sequences used for qPCR are listed
in Table I (https://pga.mgh.harvard.edu/primerbank).
 | Table I.Primer sequences used for
quantitative PCR. |
Table I.
Primer sequences used for
quantitative PCR.
| Gene | Forward
(5′-3′) | Reverse
(5′-3′) |
|---|
| GAPDH |
AGGTCGGTGTGAACGGATTTG |
TGTAGACCATGTAGTTGAGGTCA |
| β-catenin |
CAGCTTGAGTAGCCATTGTCC |
GAGCCGTCAGTGCAGGAG |
| Axin 2 |
TGCATCTCTCTCTGGAGCTG |
ACTGACCGACGATTCCATGT |
| IL-1β |
GCAACTGTTCCTGAACTCAACT |
ATCTTTTGGGGTCCGTCAACT |
| IL-6 |
TAGTCCTTCCTACCCCAATTTCC |
TTGGTCCTTAGCCACTCCTTC |
| Cox-2 |
TTCAACACACTCTATCACTGGC |
AGAAGCGTTTGCGGTACTCAT |
Western blotting
Total protein was extracted by centrifuging 1 ml
ice-cold RIPA buffer (cat. no. BC3710; Beijing Solarbio Science
& Technology Co., Ltd.) with 10 µl phosphatase inhibitor, 10 µl
protease inhibitor and 10 µl PMSF supplemented with 10–20 mg of
grounded colorectal tissues, at 9,710 × g for 30 min at 4°C.
Protein concentration was determined using a bicinchoninic acid kit
(cat. no. PC0020; Beijing Solarbio Science & Technology Co.,
Ltd.). Samples were mixed with 2X sample buffer supplemented with
0.5 mol/l Tris-HCl (pH=6.8), 10% SDS, 50% glycerol,
β-mercaptoethanol and 1% bromophenol blue. (Xi'an Zhuangzhi
Biotechnology Co., Ltd., http://www.chemicalbook.com/ContactUs_186262.htm). The
mixture was boiled at 100°C for 5–8 min. Proteins (50–100 µg) were
separated using SDS-PAGE with 10% concentrated glue and 15%
separated glue and transferred onto PVDF membranes (Pall
Corporation) using transfer buffer supplemented with 10 X electrode
buffer and methanol (Xi'an Zhuangzhi Biotechnology Co., Ltd.). The
membranes were blocked with 5% non-fat milk (Inner Mongolia Yili
Industrial Group Limited by Share Ltd., http://www.yili.com/cms/index#section-1) for 2 h at
room temperature and incubated with primary antibody against Axin2
for 12 h at 4°C (1:2,000). Following the primary incubation,
membranes were incubated with HRP-conjugated rabbit secondary
antibodies (1:10,000) for 2 h at room temperature. Protein bands
were visualized using an enhanced chemiluminescence kit (cat. no.
c6100460100, Xi'an Zhuangzhi Biotechnology Co., Ltd.). GAPDH was
used as the loading control (1:10,000; cat. no. 20536IAP;
ProteinTech Group).
Hematoxylin and eosin (H&E)
staining and IHC
H&E staining was performed as previously
described (23). Briefly, fresh
colorectal tissue was placed in a glass dish filled with
physiological saline (Xi'an Zhuangzhi Biotechnology Co., Ltd.) and
washed several times. One third of the colorectal tissue at the
anal end was placed in an embedding box and fixed with 4%
paraformaldehyde overnight at 4°C. Tissue sections were
subsequently cut into 4-µm-thick sections and dewaxed in a series
of xylene at about 60°C. Pathological changes of the colorectal
tissues were observed via H&E staining for 15–20 min at room
temperature.
For IHC analysis, samples were fixed with 40%
paraformaldehyde for 24–48 h at room temperature, embedded in
paraffin, and blocked with goat serum (Beijing Zhongshan Jinqiao
Biology Technology Co., Ltd., http://www.zsbio.com) for 30 min at room temperature.
Tissue sections were incubated with anti-Axin2 (1:150) overnight at
4°C, and subsequently incubated with a secondary antibody for 50
min at room temperature, followed by addition of HRP-labeled
streptavidin triple-antibody. Tissue sections were stained with
3,3′-diaminobenzidine for 50 sec at −20°C, and Axin2 expression was
observed under a light microscope (Eclipse C1) http://zh.medwow.com/med/confocal-microscope/nikon/eclipse-c1-plus/33643.model-spec.
The results were analyzed using ImageJ v1.51 software (National
Institutes of Health).
Statistical analysis
Statistical analysis was performed using GraphPad
Prism v8.0.1 software (GraphPad Software, Inc.). At least three
mice in each group were analyzed in the experiments. RT-qPCR and
western blot analyses were performed three times, while all other
experiments were performed once, and data are presented as the mean
± standard deviation. One-way ANOVA and Bonferroni's post hoc test
were used to compare differences in normally distributed data, and
singularity of variance and independence between three groups.
One-way ANOVA and Bonferroni's post hoc test were used to compare
differences between three independent groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
CUR suppresses AOM-DSS induced
colorectal tumorigenesis in mice
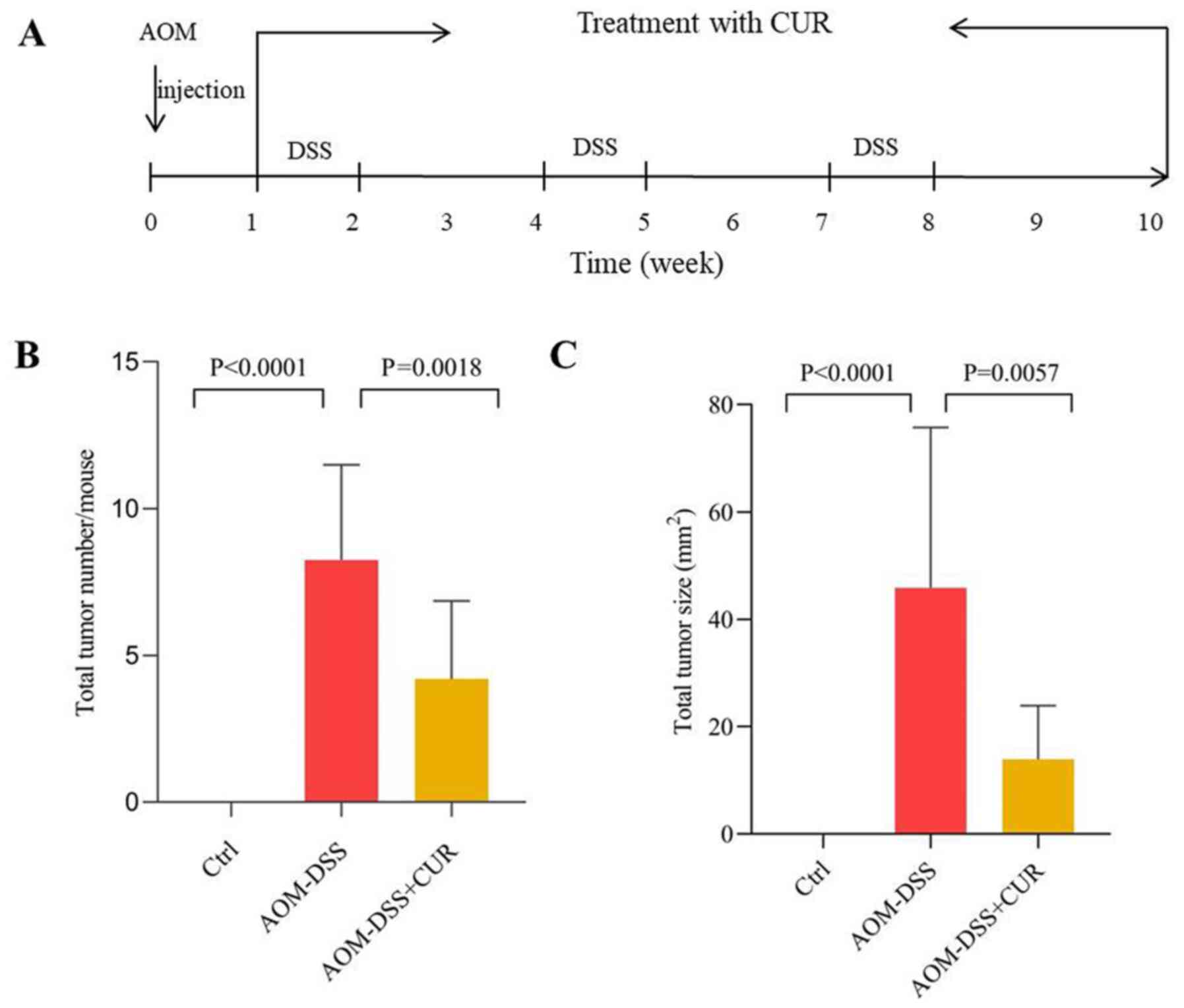
The animal experimental scheme is presented in
Fig. 1A. The effects of CUR on
AOM-DSS induced CRC was investigated simultaneously. The results
demonstrated that treatment with CUR significantly decreased the
colorectal tumor number and tumor size (The maximum tumor diameter
is about 11 mm) compared with the AOM-DSS treated group. The
average number of tumors in the AOM-DSS group was 8.25±3.24.
Following administration of CUR, the average total number of tumors
in mice decreased to 5.25±2.65 (P=0.0018), while the tumor size
decreased from 45.86±29.86 mm2 to 13.89±9.99
mm2 (P=0.0057; Fig. 1B and
C). The mice were not sacrificed in the middle of the
experiment in order to assess the tumor size at different time
points since the AOM-DSS induced CRC model is a long-term and
integral process. Thus, sacrificing the mice in the middle of the
experiment may have resulted in failure of the whole animal model
(no tumors in mice).
CUR exhibits slower weight loss and
has little effect on the survival rate of AOM-DSS induced CRC
mice
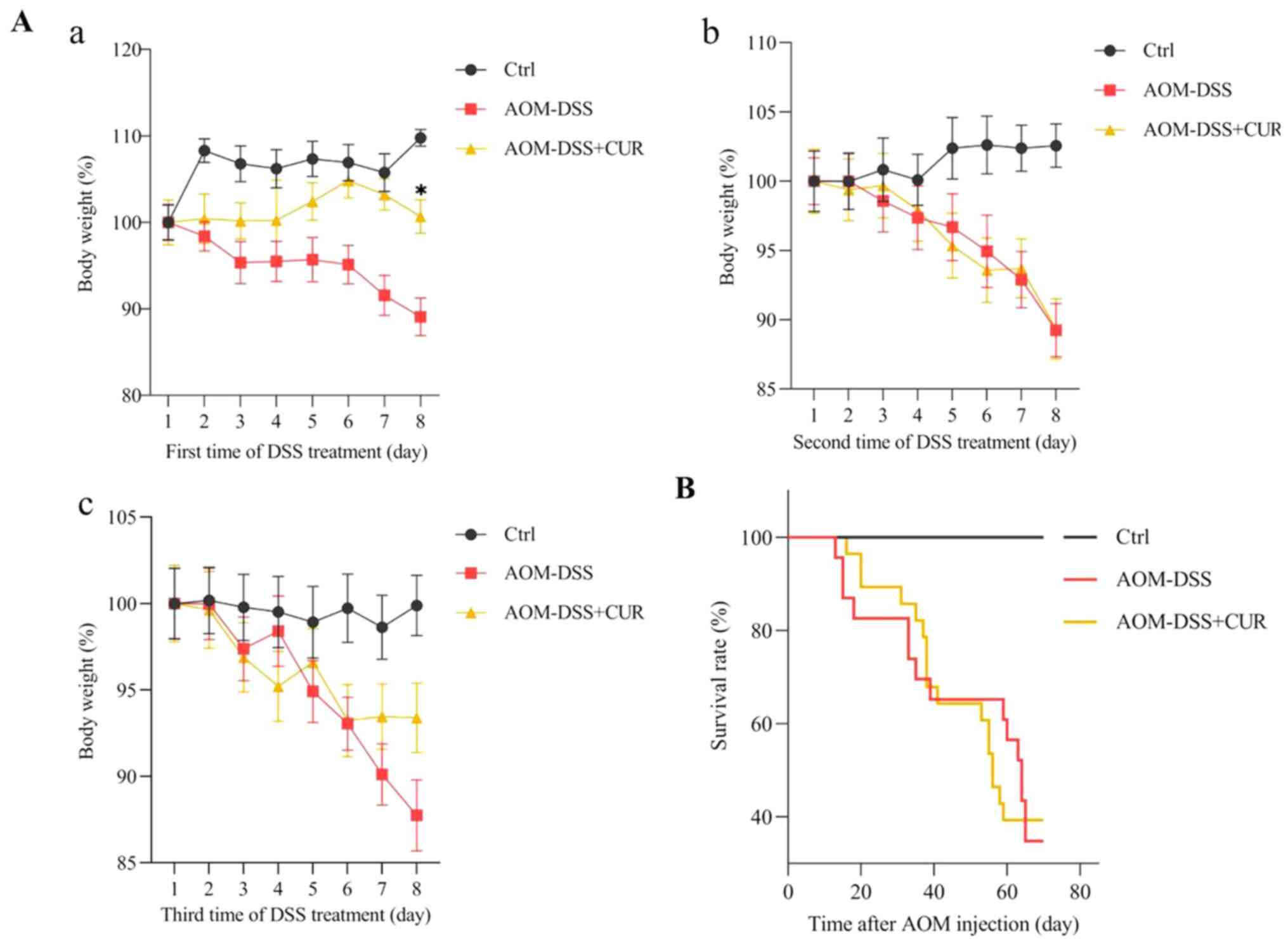
Mice were treated with 500 mg/kg CUR for 7 weeks,
and the body weight of the mice was recorded every day following
administration of 2% DSS water. During the three rounds of 7 days
of DSS treatment, little body weight was lost in the first 3–5
days. Notably, the body weight rapidly decreased on day 6, whereby
7–8 mice died due to excessive weight loss (the maximum percentage
weight loss was 12.26% during the full course of the experiment).
In addition to the rapid weight loss, the mental state of the mice
weakened following administration of 2% DSS water. Thus, for humane
endpoints, 8% sevoflurane was used to euthanize the mice.
The results demonstrated that CUR significantly
slowed the weight loss of mice on the last day of the first round
of DSS water compared with the AOM-DSS group (P<0.05; Fig. 2Aa). However, no significant
differences were observed in mortality between the AOM-DSS (65%)
and AOM-DSS+CUR (60%) groups (Fig.
2B). This may be due to the oral gavage treatment period of
vehicle control or CUR, and the small sample size. The oral gavage
is a common technique used in rodent experiments and the most
straight forward approach involving passing a feeding needle
through the mouth and into the esophagus (24). However, this technique is associated
with potential adverse effects, including (but not limited to)
esophageal trauma, aspiration pneumonia and weight loss (24), which may have led to no significant
differences in the mortality rates between the AOM-DSS and
AOM-DSS+CUR groups.
CUR downregulates the expression of
pro-inflammatory cytokines and protumorigenic proteins
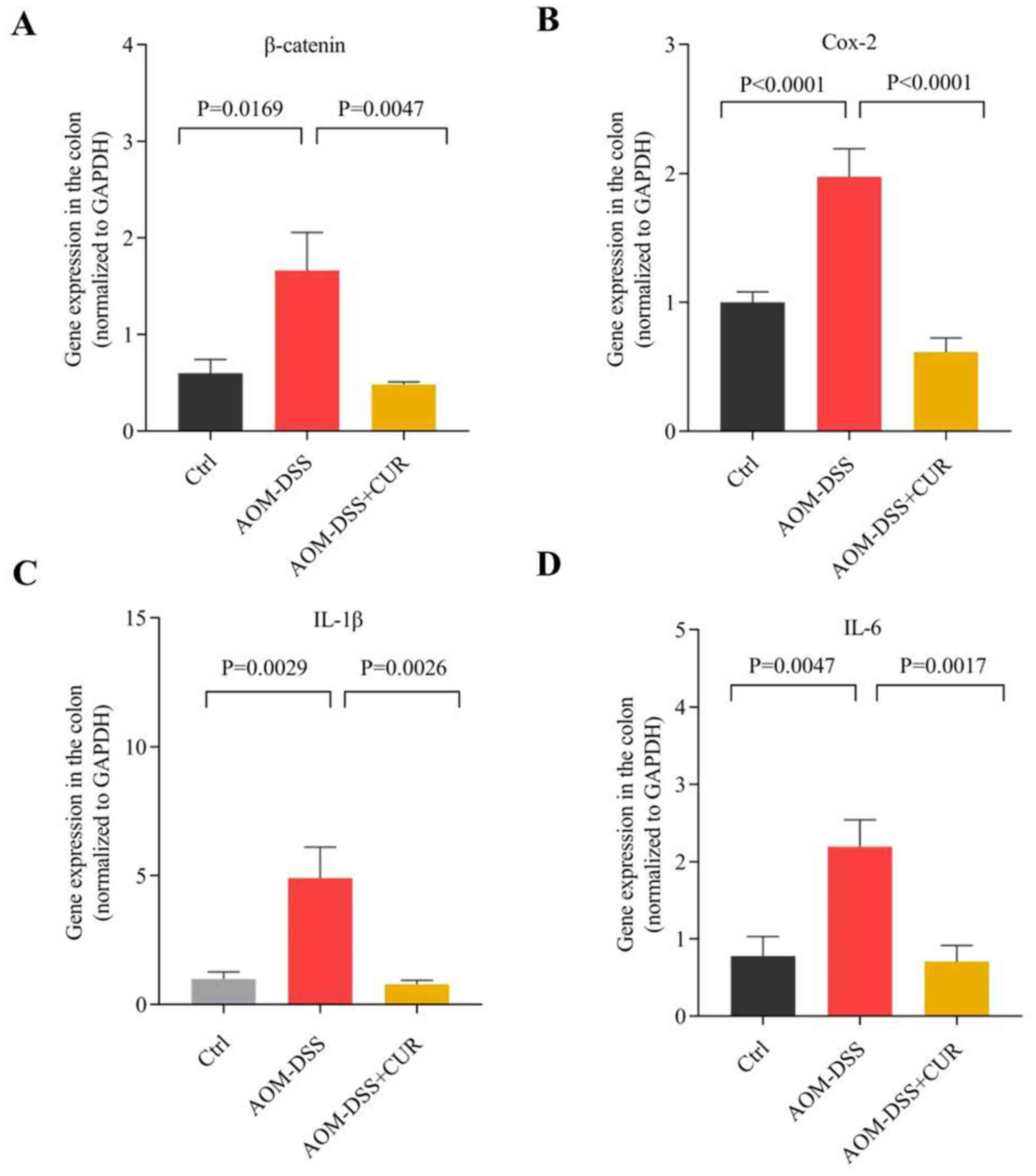
RT-qPCR analysis demonstrated that the expression
levels of IL-1β, IL-6, Cox-2 (P<0.0001) and
β-catenin (P<0.01) significantly decreased in the
CUR-treated group compared with the AOM-DSS group (Fig. 3), suggesting that CUR suppresses
AOM-DSS induced tumorigenesis by downregulating cytokines
expression.
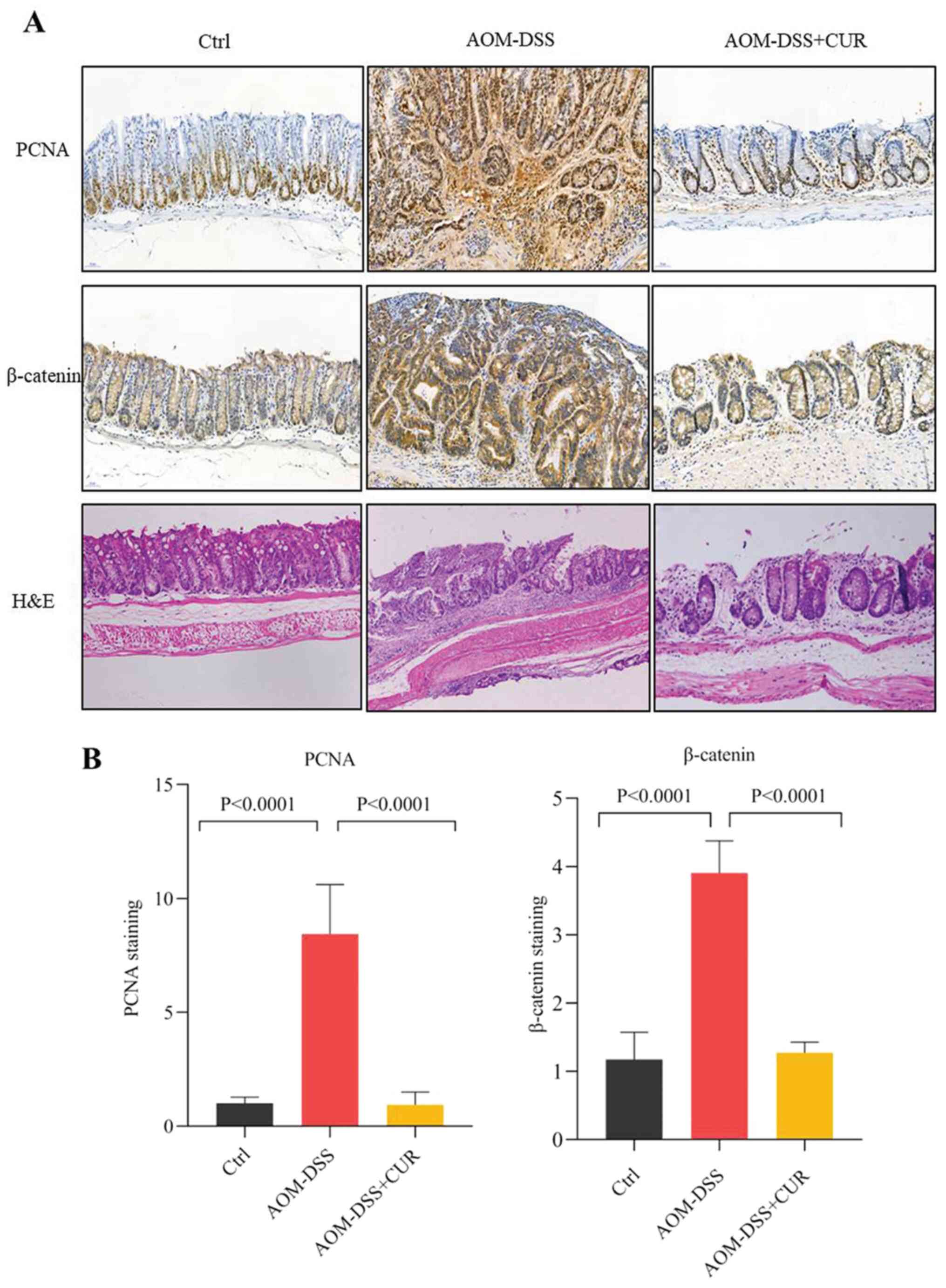
The H&E staining results demonstrated that
treatment with AOM-DSS disordered cell arrangement, enlarged the
nucleus and imbalanced the ratio of nuclear-cytoplasmic cell
morphology compared with the control group (Fig. 4A).
IHC analysis demonstrated that AOM-DSS increased the
expression levels of the tumorigenic markers, proliferating cell
nuclear antigen (PCNA) and β-catenin, while treatment with CUR
significantly decreased PCNA and β-catenin expression (both
P<0.0001; Fig. 4B). Taken
together, these results suggest that CUR attenuates tumor
occurrence by downregulating the expression of β-catenin and
pro-inflammatory cytokines, and decreasing tumor cell
proliferation, which further validates the anticancer effect of CUR
in vivo.
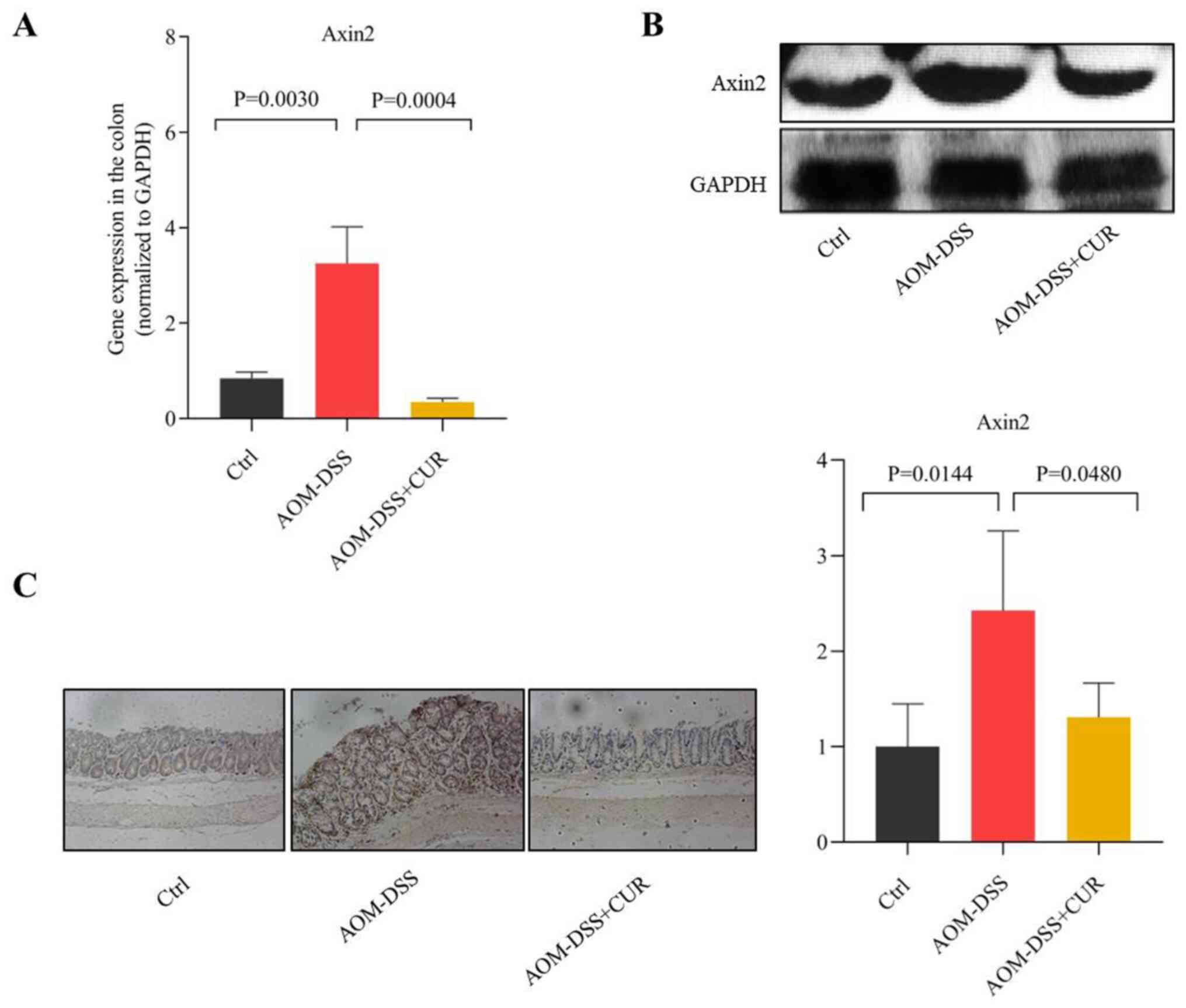
CUR attenuates AOM-DSS induced CRC by
downregulating Axin2
Axin2 is one of the important regulatory downstream
genes of the Wnt/β-catenin signaling pathway, which is involved in
several biological functions, including proliferation, mutation,
migration and apoptosis (25–28). The
results of the present study demonstrated that treatment with CUR
suppressed Axin2 expression in the colorectal tissue of mice
compared with the AOM-DSS group (P=0.0004; Fig. 5A). These results were consistent with
those of western blotting (Fig. 5B).
Furthermore, IHC analysis demonstrated that CUR decreased Axin2
expression induced by AOM-DSS (P=0.0480; Fig. 5C). Collectively, these results
suggest that CUR attenuates AOM-DSS induced CRC by downregulating
Axin2, which is as important downstream target gene involved in the
Wnt/β-catenin signaling pathway (14), thus indicating the potential
anticancer target of CUR.
Discussion
CUR, a spice widely used in cooking condiments for
hundreds of years, is a natural extract from rhizome of
Curcuma longa (5,29). Studies have confirmed that CUR exerts
antioxidant, antibacterial, anti-inflammatory, antiproliferation
and anticancer effects (8–11). CUR is unstable and easily degrades
(30–32). In vitro studies have
demonstrated that the anticancer effects are exerted by CUR rather
than its degradation products (30–32).
However, the in vivo molecular mechanism for the anticancer
effect of CUR remains to be elucidated. One of the mechanism
studies in vitro revealed that CUR can inhibit Axin2
expression in the CRC cell line, HCT116, and regulate the
Wnt/β-catenin signaling pathway (15), while the effect and mechanisms in
vivo remain largely unknown. Thus, the present study chose the
high reproducibility and potency AOM-DSS induced CRC model instead
of the xenograft model, as the AOM-DSS induced CRC model
recapitulates the aberrant crypt foci-adenoma-carcinoma sequence
that occurs in human CRC well (33),
while the xenograft model occurs by injecting colon cancer cells to
the subcutaneous, which cannot fully mimic the pathology of CRC in
humans. The results of the present study highlighted the effect of
CUR against CRC in vivo, and demonstrated that the in
vivo mechanism occurs via regulation of the Wnt/β-catenin
signaling pathway by decreasing Axin2 expression in AOM-DSS induced
CRC.
The results of the present study demonstrated that
CUR attenuated AOM-DSS induced CRC in mice, which is consistent
with previous studies that have reported the anticancer effects of
CUR (17,18,34–36). The
results of the present study demonstrated that administration of
CUR significantly decreased both the tumor number and tumor size
compared with the AOM-DSS treatment group. Dou et al
(36) reported that CUR also
suppresses tumor growth in a xenograft CRC model, where 200 mg/kg
CUR attenuates cell growth in the xenograft model via subcutaneous
injection of human colon cancer cells SW480 into the mice. Lv et
al (37) demonstrated similar
results in a breast cancer model, which indicated that CUR inhibits
tumor growth in a xenograft breast tumor model by subcutaneous
injection of MDA-MB-231 cells in mice. These results are consistent
with the present study, demonstrating the inhibitory effects of CUR
on tumor growth. In addition, the results of the present study
demonstrated that CUR downregulated the expression of
pro-inflammatory cytokines, which further supports the hypothesis
that inflammation plays an important role in the progress of
tumorigenesis. A previous study demonstrated that inflammation is
associated with at least 15% of malignant tumors, worldwide
(38). Patients with inflammatory
bowel disease (IBD) have a higher risk of developing CRC compared
with the general population (39).
In addition, CUR has been extensively reported for its efficacy in
the prevention and treatment of several inflammatory diseases, such
as IBD, rheumatoid arthritis, systemic lupus erythematosus (SLE),
atherosclerosis and multiple sclerosis (40). Chen et al (41) demonstrated that CUR prevents
cytokines production (IL-1β, IL-6 and TNF-α) induced
by hyperosmotic stimulation in corneal epithelial cells. β-catenin
is the downstream effector of the Wnt/adenomatous polyposis coli
protein (APC)/β-catenin signaling pathway, which controls the
proliferation of colon epithelial cells and is generally imbalanced
in colon aberrant crypt foci and tumors (42,43).
TNF-α is an important inflammatory cytokine in the early stages of
inflammation, which is mainly produced by monocytes and
macrophages. TNF-α can activate the cytokine cascade reaction,
resulting in a waterfall effect and triggering the synthesis of
secondary inflammatory mediators, including IL-1β and IL-6
(44,45). IL-1β and IL-6 are important
indicators for assessing inflammatory reactions and therapeutic
effects in clinical and basic research (44,45).
Taken together, the results of the present study suggest that the
antitumor effects of CUR may be partially exerted through its
anti-inflammatory effects.
The present study demonstrated that CUR suppressed
tumorigenesis by downregulating the Wnt/β-catenin signaling
pathway. The Wnt/β-catenin signaling pathway is important for
normal growth and development, and is one of the most frequently
dysregulated pathways in CRC (46).
Aberrant activation of this pathway is associated with cell
proliferation, invasive behavior and cell resistance, suggesting
its potential value as a therapeutic target in CRC treatment
(47). Axin2, which is one of the
main downstream target genes (48),
acts as a negative feedback regulator of the stability of the
Wnt/β-catenin signaling pathway in CRC (49). In the absence of stimulation,
β-catenin is phosphorylated by a multiprotein complex containing
adenomatous polyposis coli (APC), casein kinase 1, glycogen
synthase kinase 3β and either Axin1 or Axin2, subsequently
undergoing ubiquitin-dependent proteolysis (50). Under abnormal conditions, the
multiprotein complex is disintegrated, resulting in the
accumulation and nuclear translocation of β-catenin (51–53).
β-catenin is associated with the lymphoid enhancer factor/T-cell
factor family (TCF), converting them from transcription repressors
to activators, thereby activating several Wnt-responsive genes
(51–53). Compared with Axin1, Axin2 is more
likely to promote β-catenin degradation even in the presence of
upstream signaling, which is a characteristic of negative feedback
regulation (51–53). However, Wu et al (54) demonstrated that endogenous Axin2
promotes the epithelial-to-mesenchymal transition (EMT) initiated
by β-catenin/TCF in β-catenin-mutant and APC mutant CRC.
Upregulated Axin2 levels in CRC trigger a significant increase in
Snail1 activity and induce EMT, while silencing of Axin2 initiates
mesenchymal-like epithelioid-like conversion, which not only
downregulates Snail1 but also triggers extensive changes in the
classical Wnt signaling transcriptome (54). These studies have demonstrated that
Axin2 can function as an effective tumor promoter rather than a
tumor suppressor (54). Yan et
al (16) came to the same
conclusion by analyzing the gene expression profile produced by the
CRC cell line, SW620, in which antisense oligo deoxy nucleotide
transfection decreased β-catenin protein levels. In the present
study, Axin2 was demonstrated to be the target gene induced by
decreased β-catenin. Notably, the present study performed
experiments on human colon tumor samples, where Axin2 mRNA
expression was upregulated in colorectal tumors when the
Wnt/β-catenin pathway was activated. These results, along with the
aforementioned studies, indicate the importance of Axin2 in the
Wnt/β-catenin signaling pathway in CRC. The results of the present
study demonstrated that CUR inhibited CRC via Axin2 downregulation
in the Wnt/β-catenin pathway, further demonstrating the molecular
mechanism of the anticancer effects of CUR in vivo. These
results suggest that CUR may be a promising drug for the prevention
and treatment of CRC. However, further studies are required to
confirm that CUR regulates Axin2.
Previous studies have reported that the molecular
mechanisms of CUR anticancer effects include several other
signaling pathways, such as the PI3k/Akt-1/Mtor (55–57),
Akt/GSK-3β (58) and NF-κB signaling
pathways (59). The PI3K/Akt-1/mTOR
signaling pathway is important in cell proliferation, cell
survival, cell development and signal transduction during cellular
metabolism (60). A previous study
demonstrated that CUR induces G2/M arrest and autophagy
by suppressing the Akt/mTOR/p70S6K pathway (61). Zhao et al (56) validated that CUR inhibits cancer cell
proliferation and invasion by downregulating the AKT/mTOR signaling
pathway in human melanoma cells. CUR induces apoptotic cell death
through GSK-3β activation in NCCIT human embryonic carcinoma cells
(58). The NF-κB signaling pathway
abates the survival and proliferation of cancer cells (59). CUR inhibits NF-κB reporter activity,
which is associated with decreased levels of phosphorylated-IκBα
and expression of its downstream target genes, Cox-2 and cyclin D1,
to act as an anticancer effector (59).
In conclusion, a better understanding of CUR roles
and mechanisms involved in colorectal tumorigenesis may aid in the
development of novel therapeutic targets of CRC, promoting the
progress of effective, safe and mechanism-based strategies to
decrease the risks of CRC.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81702832).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author upon reasonable
request.
Authors' contributions
YW conceived and designed the present study. JH and
XD performed the experiments and analyzed the datasets. JH was a
major contributor in drafting the initial manuscript. JG, YL, ZH
and ZC helped perform the experiments. All authors have read and
approved the final manuscript and agree to be accountable for all
aspects of the research in ensuring that the accuracy or integrity
of any part of the work are appropriately investigated and
resolved.
Ethics approval and consent to
participate
All experimental animal protocols were approved by
the Northwest University Animal Ethics Committee (Xi'an, China;
approval no. NWU-AWC-20190202M), and all procedures were in line
with relevant ethical norms (21).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AOM-DSS
|
azoxymethane-dextran sodium
sulfate
|
|
CUR
|
curcumin
|
|
CRC
|
colorectal cancer
|
|
IL
|
interleukin
|
|
H&E
|
hematoxylin and eosin
|
References
|
1
|
Isnida IN, Iekhsan O, Faridah A, Nordin H
and Rakesh N: Mechanism of apoptosis induced by curcumin in
colorectal cancer. Int J Mol Sci. 20:24542019. View Article : Google Scholar
|
|
2
|
Bahrami A, Khazaei M, Hasanzadeh M,
ShahidSales S, Mashhad MJ, Farazestanian M, Sadeghnia HR, Rezayi M,
Maftouh M, Hassanian SM and Avan A: Therapeutic potential of
targeting PI3K/AKT pathway in treatment of colorectal cancer:
Rational and progress. J Cell Biochem. 119:2460–2469. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wong KE, Ngai SC, Chan KG, Lee LH, Goh BH
and Chuan LH: Curcumin nanoformulations for colorectal cancer: A
review. Front Pharmacol. 10:1522019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Molodecky NA, Soon IS, Rabi DM, Ghali WA,
Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema
HW and Kaplan GG: Increasing incidence and prevalence of the
inflammatory bowel diseases with time, based on systematic review.
Gastroenterology. 142:46–54. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goel A, Kunnumakkara AB and Aggarwal BB:
Curcumin as ‘Curecumin’: From kitchen to clinic. Biochem Pharmacol.
75:787–809. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ahmad A, Sakr WA and Rahman KM: Novel
targets for detection of cancer and their modulation by
chemopreventive natural compounds. Front Biosci (Elite ED).
4:410–425. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nobili S, Lippi D, Witort E, Donnini M,
Bausi L, Mini E and Capaccioli S: Natural compounds for cancer
treatment and prevention. Pharmacol Res. 59:365–378. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Srimal RC and Dhawan BN: Pharmacology of
diferuloyl methane (curcumin), a non-steroidal anti-inflammatory
agent. J Pharm Pharmacol. 25:447–452. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sugiyama Y, Kawakishi S and Osawa T:
Involvement of the beta-diketone moiety in the antioxidative
mechanism of tetrahydrocurcumin. Biochem Pharmacol. 52:519–525.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mahady GB, Pendland SL, Yun G and Lu ZZ:
Turmeric (Curcuma longa) and curcumin inhibit the growth of
helicobacter pylori, a group 1 carcinogen. Anticancer Res.
22:4179–4181. 2002.PubMed/NCBI
|
|
11
|
Kuttan R, Bhanumathy P, Nirmala K and
George MC: Potential anticancer activity of turmeric (curcuma
longa). Cancer Lett. 29:197–202. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gupta SC, Patchva S and Aggarwal BB:
Therapeutic roles of curcumin: Lessons learned from clinical
trials. AAPS J. 15:195–218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hewlings SJ and Kalman DS: Curcumin: A
review of its' effects on human health. Foods. 6:922017. View Article : Google Scholar
|
|
14
|
Moradi-Marjaneh R, Hassanian SM,
Shahidsalea S, Avan A and Khazaei M: Curcumin effects on the wnt
signaling pathway in colorectal cancer stem cells. Basic Clin
Cancer Res. 10:33–48. 2018.
|
|
15
|
Srivastava NS and Srivastava RA: Curcumin
and quercetin synergistically inhibit cancer cell proliferation in
multiple cancer cells and modulate Wnt/β-catenin signaling and
apoptotic pathways in A375 cells. Phytomedicine. 52:117–128. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yan D, Wiesmann M, Rohan M, Chan V,
Jefferson AB, Guo L, Sakamoto D, Caothien RH, Fuller JH, Reinhard
C, et al: Elevated expression of axin2 and hnkd mRNA provides
evidence that Wnt/beta-catenin signaling is activated in human
colon tumors. Proc Natl Acad Sci USA. 98:14973–14978. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hai J, Fan Q, Yan W, Xu Y and Yan S:
Curcumin inhibits cell proliferation and induces apoptosis of human
non-small cell lung cancer cells through the upregulation of
miR-192-5p and suppression of PI3K/Akt signaling pathway. Oncol
Rep. 34:2782–2789. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhen L, Fan D, Yi X, Cao X, Chen D and
Wang L: Curcumin inhibits oral squamous cell carcinoma
proliferation and invasion via EGFR signaling pathways. Int J Clin
Exp Pathol. 7:6438–6446. 2014.PubMed/NCBI
|
|
19
|
Yu H, Xie Y, Zhou Z, Wu Z, Dai X and Xu B:
Curcumin regulates the progression of colorectal cancer via LncRNA
NBR2/AMPK pathway. Technol Cancer Res Treat.
18:15330338198707812019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shakibaei M, Mobasheri A, Lueders C, Busch
F, Shayan P and Goel A: Curcumin enhances the effect of
chemotherapy against colorectal cancer cells by inhibition of NF-κB
and src protein kinase signaling pathways. PLoS One. 8:e572182013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hawkins P, Prescott MJ, Carbone L,
Dennison N, Johnson C, Makowska IJ, Marquardt N, Readman G, Weary
DM and Golledge HD: A good death? Report of the second newcastle
meeting on laboratory animal euthanasia. Animals (Basel). 9:502016.
View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guo Y, Liu Y, Zhang C, Su ZY, Li W, Huang
MT and Kong AN: The epigenetic effects of aspirin: The modification
of histone H3 lysine 27 acetylation in the prevention of colon
carcinogenesis in azoxymethane- and dextran sulfate sodium-treated
CF-1 mice. Carcinogenesis. 37:616–624. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jones CP, Boyd KL and Wallace JM:
Evaluation of mice undergoing serial oral gavage while awake or
anesthetized. J Am Assoc Lab Anim Sci. 6:805–810. 2016.
|
|
25
|
Salahshor S, Goncalves J, Chetty R,
Gallinger S and Woodgett JR: Differential gene expression profile
reveals deregulation of pregnancy specific beta1 glycoprotein 9
early during colorectal carcinogenesis. BMC Cancer. 5:662005.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gunes EG, Pinarbasi E and Pinarbasi H:
AXIN2 polymorphism and its association with astrocytoma in a
turkish population. Mol Med Rep. 3:705–709. 2010.PubMed/NCBI
|
|
27
|
Kanzaki H, Ouchida M, Hanafusa H, Yano M,
Suzuki H, Aoe M, Imai K, Shimizu N, Nakachi K and Shimizu K: Single
nucleotide polymorphism of the AXIN2 gene is preferentially
associated with human lung cancer risk in a Japanese population.
Int J Mol Med. 18:279–284. 2006.PubMed/NCBI
|
|
28
|
Aristizabal-Pachon AF, Carvalho TI,
Carrara HH, Andrade J and Takahashi CS: AXIN2 polymorphisms, the
β-Catenin destruction complex expression profile and breast cancer
susceptibility. Asian Pac J Cancer Prev. 16:7277–7284. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yallapu MM, Jaggi M and Chauhan SC:
Curcumin nanomedicine: A road to cancer therapeutics. Curr Pharm
Des. 19:1994–2010. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sanidad KZ, Zhu J, Wang W, Du Z and Zhang
G: Effects of stable degradation products of curcumin on cancer
cell proliferation and inflammation. J Agric Food Chem.
4:9189–9195. 2016. View Article : Google Scholar
|
|
31
|
Gordon ON, Luis PB, Sintim HO and
Schneider C: Unraveling curcumin degradation: Autoxidation proceeds
through spiroepoxide and vinylether intermediates enroute to the
main bicyclopentadione. J Biol Chem. 290:4817–4828. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Griesser M, Pistis V, Suzuki T, Tejera N,
Pratt DA and Schneider C: Autoxidative and cyclooxygenase-2
catalyzed transformation of the dietary chemopreventive agent
curcumin. J Biol Chem. 286:1114–1124. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
De Robertis M, Massi E, Poeta ML, Carotti
S, Morini S, Cecchetelli L, Signori E and Fazio VM: The AOM/DSS
murine model for the study of colon carcinogenesis: From pathways
to diagnosis and therapy studies. J Carcinog. 10:92011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guo H, Xu YM, Ye ZQ, Yu JH and Hu XY:
Curcumin induces cell cycle arrest and apoptosis of prostate cancer
cells by regulating the expression of IkappaBalpha, c-jun and
androgen receptor. Pharmazie. 68:431–434. 2013.PubMed/NCBI
|
|
35
|
Ma J, Fang B, Zeng F, Pang H, Zhang J, Shi
Y, Wu X, Cheng L, Ma C, Xia J and Wang Z: Curcumin inhibits cell
growth and invasion through up-regulation of miR-7 in pancreatic
cancer cells. Toxicol Lett. 231:82–91. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Dou H, Shen R, Tao J, Huang L, Shi H, Chen
H, Wang Y and Wang T: Curcumin suppresses the colon cancer
proliferation by inhibiting wnt/β-catenin pathways via miR-130a.
Front Pharmacol. 8:8772017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lv ZD, Liu XP, Zhao WJ, Dong Q, Li FN,
Wang HB and Kong B: Curcumin induces apoptosis in breast cancer
cells and inhibits tumor growth in vitro and in vivo. Int J Clin
Exp Pathol. 7:2818–2824. 2014.PubMed/NCBI
|
|
38
|
Hynes MJ, Huang KM and Huang EH: Review
paper: Implications of the ‘Cancer Stem Cell’ hypothesis on murine
models of colon cancer and colitis-associated cancer. Vet Pathol.
46:819–835. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ryan BM, Wolff RK, Valeri N, Khan M,
Robinson D, Paone A, Bowman ED, Lundgreen A, Caan B, Potter J, et
al: An analysis of genetic factors related to risk of inflammatory
bowel disease and colon cancer. Cancer Epidemiol. 38:583–590. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kahkhaie KR, Mirhosseini A, Aliabadi A,
Mohammadi A, Mousavi MJ, Haftcheshmeh SM, Sathyapalan T and
Sahebkar A: Curcumin: A modulator of inflammatory signaling
pathways in the immune system. Inflammopharmacology. 27:885–900.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen M, Hu DN, Pan Z, Lu CW, Xue CY and
Aass L: Curcumin protects against hyperosmoticity-induced Il-1beta
elevation in human corneal epithelial cell via MAPK pathways. Exp
Eye Res. 90:437–443. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Morin PJ, Sparks AB, Korinek V, Barker N,
Clevers H, Vogelstein B and Kinzler KW: Activation of
beta-catenin-tcf signaling in colon cancer by mutations in
beta-catenin or APC. Science. 275:1787–1790. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Korinek V, Barker N, Morin PJ, Wichen D,
de Weger R, Kinzler KW, Vogelstein B and Clevers H: Constitutive
transcriptional activation by a beta-catenin-tcf complex in
APC-/-colon carcinoma. Science. 275:1784–1787. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Park SH, Kang K, Giannopoulou E, Yu Q,
Kang K, Kim G, Park-Min KH and Ivashkiv LB: Type I interferons and
the cytokine TNF cooperatively reprogram the macrophage epigenome
to promote inflammatory activation. Nat Immunol. 10:1104–1116.
2017. View Article : Google Scholar
|
|
45
|
Wu Y, Liu Z, Wu W, Lin S, Zhang N, Wang H,
Tan S, Lin P, Chen X, Wu L and Xu J: Effects of FM0807, a novel
curcumin derivative, on lipopolysaccharide-induced inflammatory
factor release via the ROS/JNK/p53 pathway in RAW264.7 cells.
Biosci Rep. 5:BSR201808492018. View Article : Google Scholar
|
|
46
|
Mccubrey JA, Rakus D, Gizak A, Steelman
LS, Abrams SL, Lertpiriyapong K, Fitagerald TL, Yang LV, Montalto
G, Cervello M, et al: Effects of mutations in Wnt/β-catenin,
hedgehog, notch and PI3K pathways on GSK-3 activity-diverse effects
on cell growth, metabolism and cancer. Biochim Biophys Acta.
12:2942–2976. 2016. View Article : Google Scholar
|
|
47
|
Bahrami A, Amerizadeh F, Shahidsales S,
Khazaei M, Ghayour-Mobarhan M, Sadeghnia HR, Maftouh M, Hassanian
SM and Avan A: Therapeutic potential of targeting Wnt/β-catenin
pathway in treatment of colorectal cancer: Rational and progress. J
Cell Biochem. 118:1979–1983. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jho E, Zhang T, Domon C, Joo CK, Freund JN
and Costantini F: Wnt/beta-catenin/Tcf signaling induces the
transcription of axin2, a negative regulator of the signaling
pathway. Mol Cell Biol. 22:1172–1183. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen HY, Lang YD, Lin HN, Liu YR, Liao CC,
Nana AW, Yen Y and Chen RH: MiR-103/107 prolong wnt/β-catenin
signaling and colorectal cancer stemness by targeting axin2. Sci
Rep. 9:96872019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu C, Kato Y, Zhang Z, Do VM, Yankner BA
and He X: Beta-trcp couples beta-catenin
phosphorylation-degradation and regulates xenopus axis formation.
Proc Natl Acad Sci USA. 96:6273–6278. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
He TC, Sparks AB, Rago C, Hermeking H,
Zawel L, da Costa LT, Morin PJ, Vogelstein B and Kinzler KW:
Identifcation of c-myc as a target of the APC pathway. Science.
281:1509–1512. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jubb AM, Chalasani S, Frantz GD, Smits R,
Grabsch HI, Kavi V, Maughan NJ, Hillan KJ, Quirke P and Koeppen H:
Achaete-Scute like 2 (ascl2) is a target of wnt signalling and is
upregulated in intestinal neoplasia. Oncogene. 25:3445–3457. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lustig B, Jerchow B, Sachs M, Weiler S,
Pietsch T, Karsten U, van de Wetering M, Clevers H, Schlag PM,
Birchmeier W and Behrens J: Negative feedback loop of wnt signaling
through upregulation of conductin/axin2 in colorectal and liver
tumors. Mol Cell Biol. 22:1184–1193. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wu ZQ, Brabletz T, Fearon E, Willis AL, Hu
CY, Li XY and Weiss SJ: Canonical wnt suppressor, axin2, promotes
colon carcinoma oncogenic activity. Proc Natl Acad Sci USA.
109:11312–11317. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sokolosky ML, Stadelman KM, Chappell WH,
Abrams SL, Martelli AM, Stivala F, Libra M, Nicoletti F, Drobot LB,
Franklin RA, et al: Involvement of akt-1 and mTOR in sensitivity of
breast cancer to targeted therapy. Oncotarget. 2:538–550. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhao G, Han X, Zheng S, Li Z, Sha Y, Ni J,
Sun Z, Qiao S and Song Z: Curcumin induces autophagy, inhibits
proliferation and invasion by downregulating AKT/mTOR signaling
pathway in human melanoma cells. Oncol Rep. 35:1065–1074. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhu FQ, Chen MJ, Zhu M, Zhao RS, Qiu W, Xu
X, Liu H, Zhao HW, Yu RJ, Wu XF, et al: Curcumin suppresses
epithelial-mesenchymal transition of renal tubular epithelial cells
through the inhibition of Akt/mTOR pathway. Biol Pharm Bull.
40:17–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yun JH, Park YG, Lee KM, Kim J and Nho CW:
Curcumin induces apoptotic cell death via oct4 inhibition and
GSK-3β activation in NCCIT cells. Mol Nutr Food Res. 59:1053–1062.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Marin YE, Wall BA, Wang S, Namkoong J,
Martino JJ, Suh J, Lee HJ, Rabson AB, Yang CS, Chen S and Ryu JH:
Curcumin downregulates the constitutive activity of NF-κB and
induces apoptosis in novel mouse melanoma cells. Melanoma Res.
17:274–283. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Marjaneh RM, Rahmani F, Hassanian SM,
Rezaei N, Hashemzehi M, Bahrami A, Ariakia F, Fiuji H, Sahebkar A,
Avan A and Khazaei M: Phytosomal curcumin inhibits tumor growth in
colitis-associated colorectal cancer. J Cell Physiol.
233:6785–6798. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Aoki H, Takada Y, Kondo S, Sawaya R,
Aggarwal BB and Kondo Y: Evidence that curcumin suppresses the
growth of malignant gliomas in vitro and in vivo through induction
of autophagy: Role of Akt and extracellular signal-regulated kinase
signaling pathways. Mol Pharmacol. 72:29–39. 2007. View Article : Google Scholar : PubMed/NCBI
|