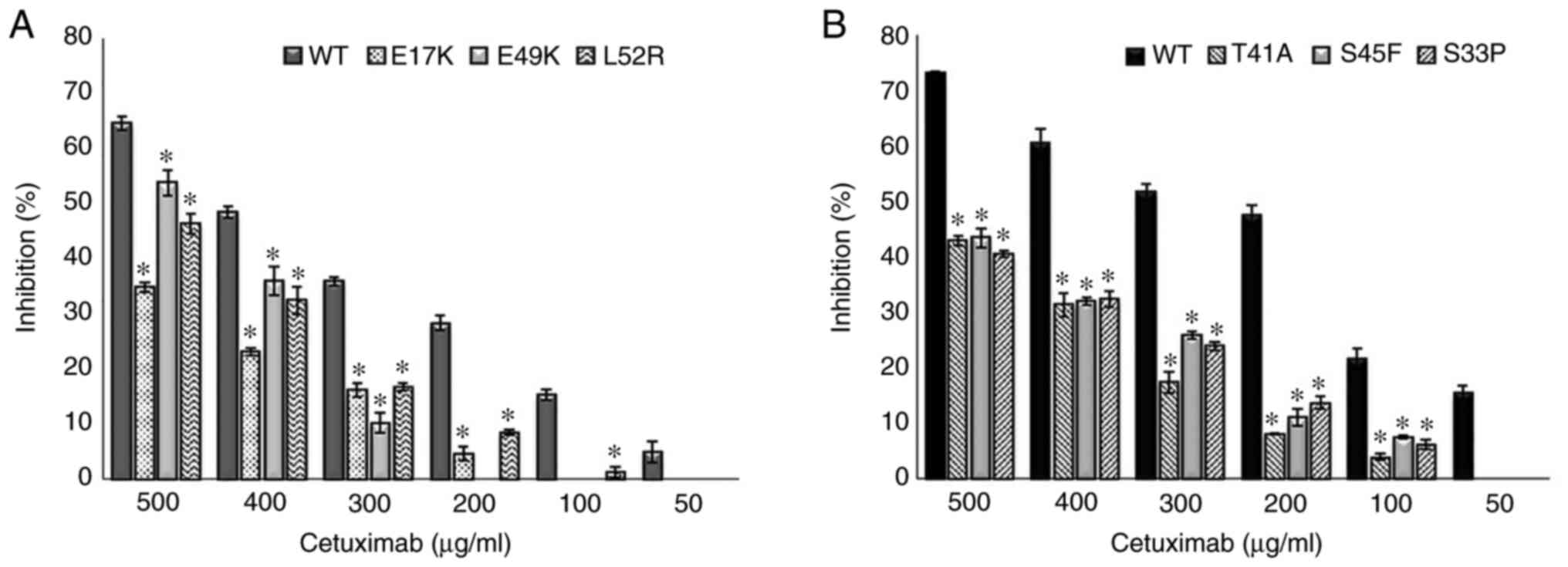
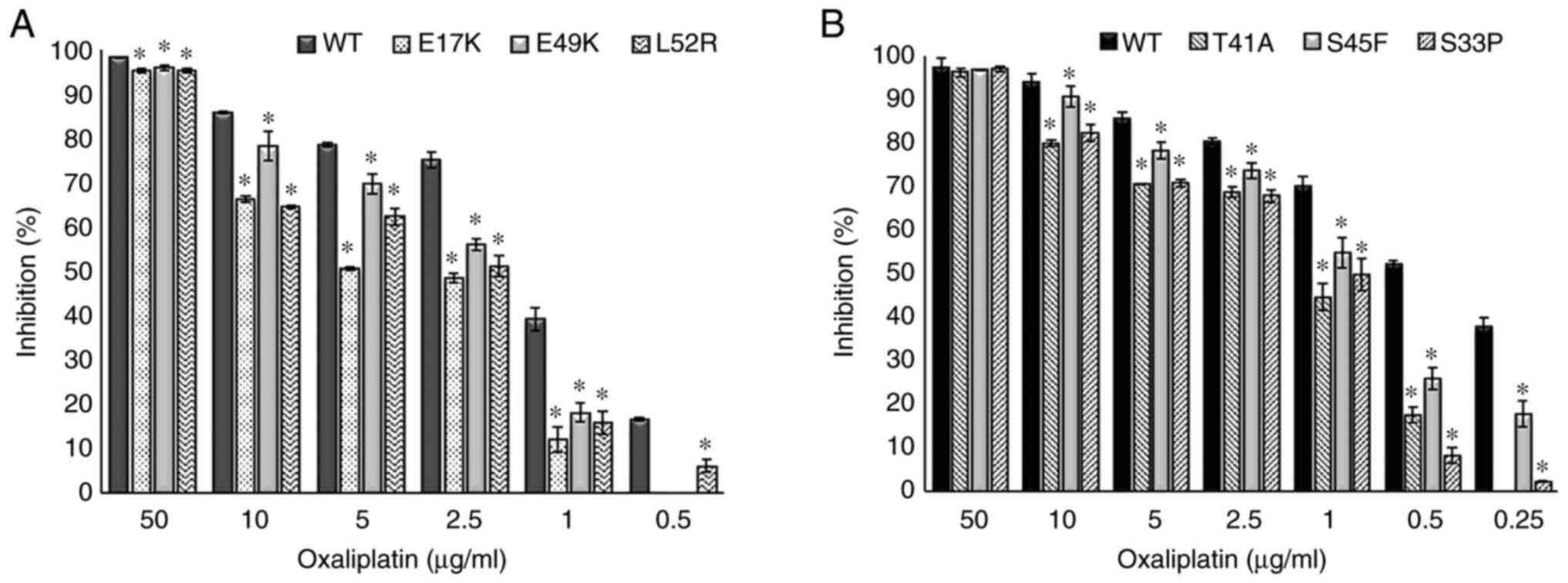
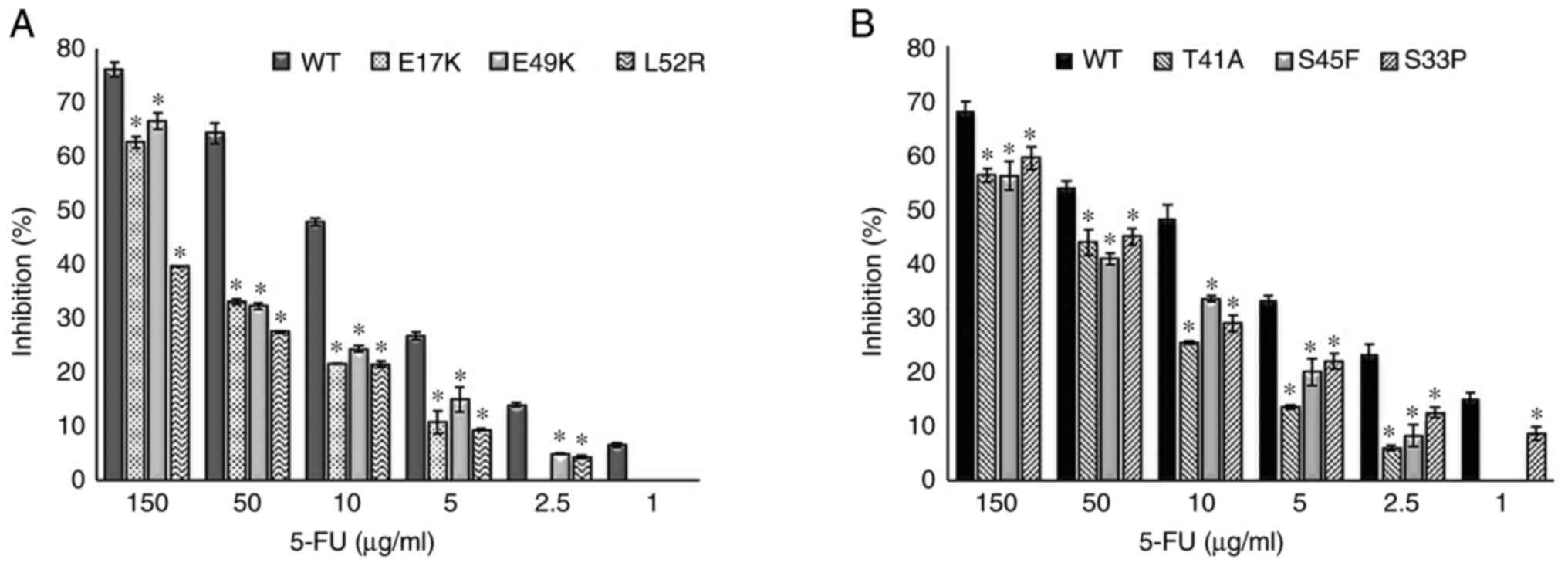
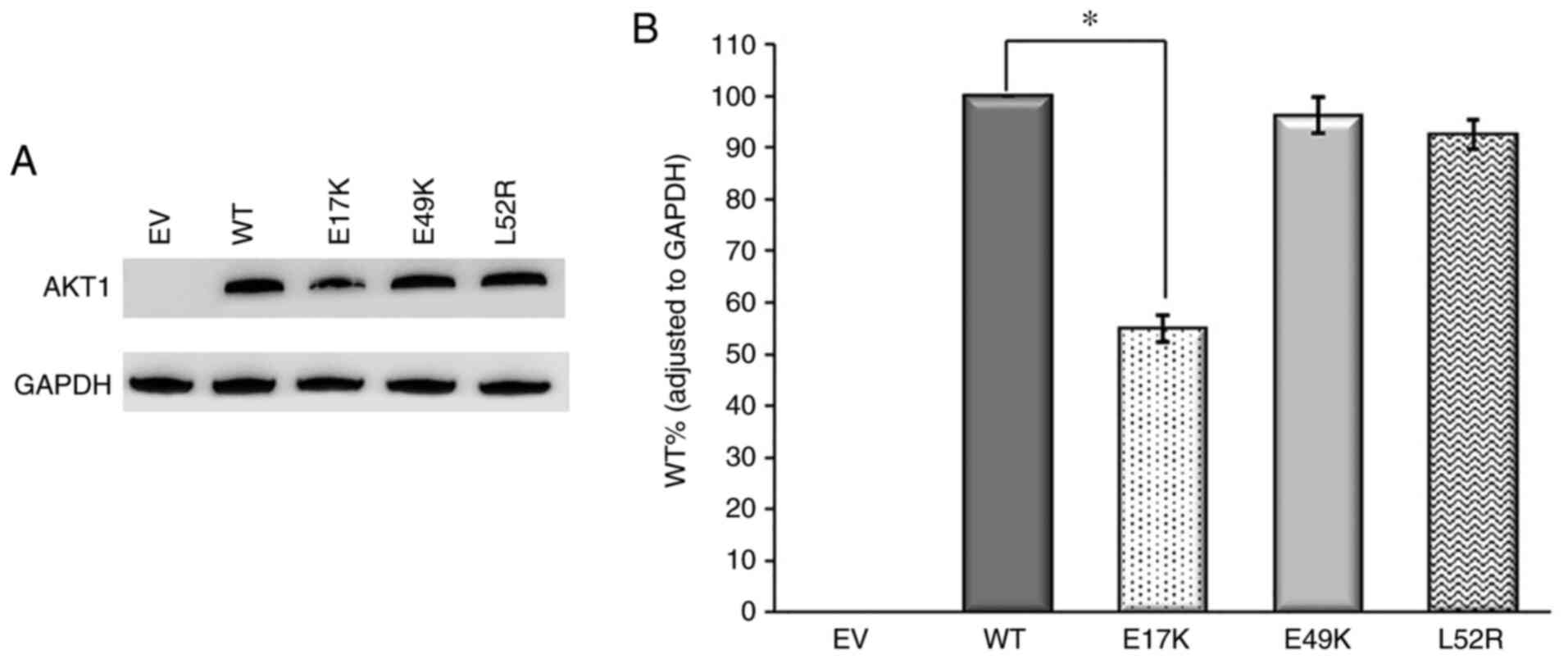
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar
|
|
2
|
Longley DB and Johnston PG: Molecular
mechanisms of drug resistance. J Pathol. 205:275–292. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Arqués O, Chicote I, Puig I, Tenbaum SP,
Argilés G, Dienstmann R, Fernández N, Caratù G, Matito J,
Silberschmidt D, et al: Tankyrase inhibition blocks Wnt/β-catenin
pathway and reverts resistance to PI3K and AKT inhibitors in the
treatment of colorectal cancer. Clin Cancer Res. 22:644–656. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Banck MS and Grothey A: Biomarkers of
resistance to epidermal growth factor receptor monoclonal
antibodies in patients with metastatic colorectal cancer. Clin
Cancer Res. 15:7492–7501. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Graham J, Muhsin M and Kirkpatrick P:
Cetuximab. Nat Rev Drug Discov. 3:549–550. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chong CR and Jänne PA: The quest to
overcome resistance to EGFR-targeted therapies in cancer. Nat Med.
19:1389–1400. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
De Mattia E, Toffoli G, Polesel J,
D'Andrea M, Corona G, Zagonel V, Buonadonna A, Dreussi E and
Cecchin E: Pharmacogenetics of ABC and SLC transporters in
metastatic colorectal cancer patients receiving first-line FOLFIRI
treatment. Pharmacogenet Genomics. 23:549–557. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hsu HC, Thiam TK, Lu YJ, Yeh CY, Tsai WS,
You JF, Hung HY, Tsai CN, Hsu A, Chen HC, et al: Mutations of
KRAS/NRAS/BRAF predict cetuximab resistance in metastatic
colorectal cancer patients. Oncotarget. 7:22257–22270. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Carpten JD, Faber AL, Horn C, Donoho GP,
Briggs SL, Robbins CM, Hostetter G, Boguslawski S, Moses TY, Savage
S, et al: A transforming mutation in the pleckstrin homology domain
of AKT1 in cancer. Nature. 448:439–444. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Parikh C, Janakiraman V, Wu WI, Foo CK,
Kljavin NM, Chaudhuri S, Stawiski E, Lee B, Lin J, Li H, et al:
Disruption of PH-kinase domain interactions leads to oncogenic
activation of AKT in human cancers. Proc Natl Acad Sci.
109:19368–19373. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yi KH, Axtmayer J, Gustin JP, Rajpurohit A
and Lauring J: Functional analysis of non-hotspot AKT1 mutants
found in human breast cancers identifies novel driver mutations:
Implications for personalized medicine. Oncotarget. 4:29–34. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fruman DA, Chiu H, Hopkins BD, Bagrodia S,
Cantley LC and Abraham RT: The PI3K pathway in human disease. Cell.
170:605–635. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Malapelle U, Pisapia P, Sgariglia R,
Vigliar E, Biglietto M, Carlomagno C, Giuffrè G, Bellevicine C and
Troncone G: Less frequently mutated genes in colorectal cancer:
Evidences from next-generation sequencing of 653 routine cases. J
Clin Pathol. 69:767–771. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hechtman JF, Sadowska J, Huse JT, Borsu L,
Yaeger R, Shia J, Vakiani E, Ladanyi M and Arcila ME: AKT1 E17K in
colorectal carcinoma is associated with BRAF V600E but not MSI-H
status: A clinicopathologic comparison to PIK3CA helical and kinase
domain mutants. Mol Cancer Res. 13:1003–1008. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu Y, Savage RE, Eathiraj S, Meade J, Wick
MJ, Hall T, Abbadessa G and Schwartz B: Targeting AKT1-E17K and the
PI3K/AKT pathway with an allosteric AKT inhibitor, ARQ 092. PLoS
One. 10:e01404792015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gong J, Cho M, Sy M, Salgia R and Fakih M:
Molecular profiling of metastatic colorectal tumors using
next-generation sequencing: A single-institution experience.
Oncotarget. 8:42198–42213. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hagen T and Vidal-Puig A: Characterisation
of the phosphorylation of beta-catenin at the GSK-3 priming site
Ser45. Biochem Biophys Res Commun. 294:324–328. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cai ZX, Tang XD, Gao HL, Tang C,
Nandakumar V, Jones L, Ye H, Lou F, Zhang D, Sun H, et al: APC,
FBXW7, KRAS, PIK3CA, and TP53 gene mutations in human colorectal
cancer tumors frequently detected by next-generation DNA
sequencing. J Mol Genet Med. 8:1452014.
|
|
19
|
Jauhri M, Bhatnagar A, Gupta S, Shokeen Y,
Minhas S and Aggarwal S: Targeted molecular profiling of rare
genetic alterations in colorectal cancer using next-generation
sequencing. Med Oncol. 33:1062016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim S and Jeong S: Mutation hotspots in
the β-catenin gene: Lessons from the human cancer genome databases.
Mol Cells. 42:8–16. 2019.PubMed/NCBI
|
|
21
|
Anwar M, Kochhar R, Singh R, Bhatia A,
Vaiphei K, Mahmood A and Mahmood S: Frequent activation of the
β-catenin gene in sporadic colorectal carcinomas: A mutational
& expression analysis. Mol Carcinog. 55:1627–1638. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Frattini M, Saletti P, Romagnani E, Martin
V, Molinari F, Ghisletta M, Camponovo A, Etienne LL, Cavalli F and
Mazzucchelli L: PTEN loss of expression predicts cetuximab efficacy
in metastatic colorectal cancer patients. Br J Cancer.
97:1139–1145. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Di Nicolantonio F, Martini M, Molinari F,
Sartore-Bianchi A, Arena S, Saletti P, De Dosso S, Mazzucchelli L,
Frattini M, Siena S and Bardelli A: Wild-type BRAF is required for
response to panitumumab or cetuximab in metastatic colorectal
cancer. J Clin Oncol. 26:5705–5712. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sartore-Bianchi A, Martini M, Molinari F,
Veronese S, Nichelatti M, Artale S, Di Nicolantonio F, Saletti P,
De Dosso S, Mazzucchelli L, et al: PIK3CA mutations in colorectal
cancer are associated with clinical resistance to EGFR-targeted
monoclonal antibodies. Cancer Res. 69:1851–1857. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Montagut C, Dalmases A, Bellosillo B,
Crespo M, Pairet S, Iglesias M, Salido M, Gallen M, Marsters S,
Tsai SP, et al: Identification of a mutation in the extracellular
domain of the epidermal growth factor receptor conferring cetuximab
resistance in colorectal cancer. Nat Med. 18:221–223. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xu JM, Wang Y, Wang YL, Wang Y, Liu T, Ni
M, Li MS, Lin L, Ge FJ, Gong C, et al: PIK3CA mutations contribute
to acquired cetuximab resistance in patients with metastatic
colorectal cancer. Clin Cancer Res. 23:4602–4616. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dekker E and Rex DK: Advances in CRC
prevention: Screening and surveillance. Gastroenterology.
154:1970–1984. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
van Brummelen EMJ, de Boer A, Beijnen JH
and Schellens JHM: BRAF mutations as predictive biomarker for
response to anti-EGFR monoclonal antibodies. Oncologist.
22:864–872. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Holohan C, Van Schaeybroeck S, Longley DB
and Johnston PG: Cancer drug resistance: An evolving paradigm. Nat
Rev Cancer. 13:714–726. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nemecek R, Berkovcova J, Radova L, Kazda
T, Mlcochova J, Vychytilova-Faltejskova P, Slaby O and Svoboda M:
Mutational analysis of primary and metastatic colorectal cancer
samples underlying the resistance to cetuximab-based therapy. Onco
Targets Ther. 9:4695–4703. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kumar SS, Price TJ, Mohyieldin O, Borg M,
Townsend A and Hardingham JE: KRAS G13D mutation and sensitivity to
cetuximab or panitumumab in a colorectal cancer cell line model.
Gastrointest Cancer Res. 7:23–26. 2014.PubMed/NCBI
|
|
32
|
Nakadate Y, Kodera Y, Kitamura Y,
Shirasawa S, Tachibana T, Tamura T and Koizumi F: KRAS mutation
confers resistance to antibody-dependent cellular cytotoxicity of
cetuximab against human colorectal cancer cells. Int J Cancer.
134:2146–2155. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Stachler MD, Rinehart E, Lindeman N, Odze
R and Srivastava A: Novel molecular insights from routine
genotyping of colorectal carcinomas. Hum Pathol. 46:507–513. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen D, Huang X, Cai J, Guo S, Qian W,
Wery JP and Li QX: A set of defined oncogenic mutation alleles
seems to better predict the response to cetuximab in CRC
patient-derived xenograft than KRAS 12/13 mutations. Oncotarget.
6:40815–40821. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xu G, Li K, Zhang N, Zhu B, Feng G and Fan
Q: Colon cancers carrying BRAF V600E and β-catenin T41A activating
mutations are resistant to numerous common anticancer drugs. Oncol
Lett. 15:4471–4476. 2018.PubMed/NCBI
|
|
36
|
Sparks AB, Morin PJ, Vogelstein B and
Kinzler KW: Mutational analysis of the APC/beta-catenin/Tcf pathway
in colorectal cancer. Cancer Res. 58:1130–1134. 1998.PubMed/NCBI
|
|
37
|
Abdelmaksoud-Damak R, Miladi-Abdennadher
I, Triki M, Khabir A, Charfi S, Ayadi L, Frikha M,
Sellami-Boudawara T and Mokdad-Gargouri R: Expression and mutation
pattern of β-catenin and adenomatous polyposis coli in colorectal
cancer patients. Arch Med Res. 46:54–62. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Askham JM, Platt F, Chambers PA, Snowden
H, Taylor CF and Knowles MA: AKT1 mutations in bladder cancer:
Identification of a novel oncogenic mutation that can co-operate
with E17K. Oncogene. 29:150–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu C, Li Y, Semenov M, Han C, Baeg GH,
Tan Y, Zhang Z, Lin X and He X: Control of beta-catenin
phosphorylation/degradation by a dual-kinase mechanism. Cell.
108:837–847. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jhawer M, Goel S, Wilson AJ, Montagna C,
Ling YH, Byun DS, Nasser S, Arango D, Shin J, Klampfer L, et al:
PIK3CA mutation/PTEN expression status predicts response of colon
cancer cells to the epidermal growth factor receptor inhibitor
cetuximab. Cancer Res. 68:1953–1961. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Levy EM, Sycz G, Arriaga JM, Barrio MM,
Von Euw EM, Morales SB, González M, Mordoh J and Bianchini M:
Cetuximab-mediated cellular cytotoxicity is inhibited by HLA-E
membrane expression in colon cancer cells. Innate Immun. 15:91–100.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Balin-Gauthier D, Delord JP, Rochaix P,
Mallard V, Thomas F, Hennebelle I, Bugat R, Canal P and Allal C: In
vivo and in vitro antitumor activity of oxaliplatin in combination
with cetuximab in human colorectal tumor cell lines expressing
different level of EGFR. Cancer Chemother Pharmacol. 57:709–718.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Shigeta K, Hayashida T, Hoshino Y,
Okabayashi K, Endo T, Ishii Y, Hasegawa H and Kitagawa Y:
Expression of epidermal growth factor receptor detected by
cetuximab indicates its efficacy to inhibit in vitro and in vivo
proliferation of colorectal cancer cells. PLoS One. 8:e663022013.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Luca T, Barresi V, Privitera G, Musso N,
Caruso M, Condorelli DF and Castorina S: In vitro combined
treatment with cetuximab and trastuzumab inhibits growth of colon
cancer cells. Cell Prolif. 47:435–447. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bardelli A and Jänne PA: The road to
resistance: EGFR mutation and cetuximab. Nat Med. 18:199–200. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nakamura M, Aoyama T, Ishibashi K, Tsuji
A, Takinishi Y, Shindo Y, Sakamoto J, Oba K and Mishima H:
Randomized phase II study of cetuximab versus irinotecan and
cetuximab in patients with chemo-refractory KRAS codon G13D
metastatic colorectal cancer (G13D-study). Cancer Chemother
Pharmacol. 79:29–36. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yao H, Ashihara E and Maekawa T: Targeting
the Wnt/β-catenin signaling pathway in human cancers. Expert Opin
Ther Targets. 15:873–887. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sebio A, Kahn M and Lenz HJ: The potential
of targeting Wnt/β-catenin in colon cancer. Expert Opin Ther
Targets. 18:611–615. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ciombor KK, Wu C and Goldberg RM: Recent
therapeutic advances in the treatment of colorectal cancer. Annu
Rev Med. 66:83–95. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Misset JL, Bleiberg H, Sutherland W,
Bekradda M and Cvitkovic E: Oxaliplatin clinical activity: A
review. Crit Rev Oncol Hematol. 35:75–93. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Toscano F, Parmentier B, Fajoui ZE,
Estornes Y, Chayvialle JA, Saurin JC and Abello J: p53 dependent
and independent sensitivity to oxaliplatin of colon cancer cells.
Biochem Pharmacol. 74:392–406. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Panczyk M: Pharmacogenetics research on
chemotherapy resistance in colorectal cancer over the last 20
years. World J Gastroenterol. 20:9775–9827. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Longley DB, Harkin DP and Johnston PG:
5-Fluorouracil: Mechanisms of action and clinical strategies. Nat
Rev Cancer. 3:330–338. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Feldman M, Friedman LS and Brandt LJ:
Sleisenger and Fordtran's gastrointestinal and liver disease,
pathophysiology, diagnosis, management. 11th edition. Elsevier
Saunders; Philadelphia: pp. 2292–2294. 2016
|
|
55
|
Cunningham D, Humblet Y, Siena S, Khayat
D, Bleiberg H, Santoro A, Bets D, Mueser M, Harstrick A, Verslype
C, et al: Cetuximab monotherapy and cetuximab plus irinotecan in
irinotecan-refractory metastatic colorectal cancer. N Engl J Med.
351:337–345. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Prewett M, Deevi DS, Bassi R, Fan F, Ellis
LM, Hicklin DJ and Tonra JR: Tumors established with cell lines
selected for oxaliplatin resistance respond to oxaliplatin if
combined with cetuximab. Clin Cancer Res. 13:7432–7440. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ge XJ, Jiang JY, Wang M, Li MY, Zheng LM,
Feng ZX and Liu L: Cetuximab enhances the efficiency of irinotecan
through simultaneously inhibiting the MAPK signaling and ABCG2 in
colorectal cancer cells. Pathol Res Pract. 216:1527982020.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Van Cutsem E, Kohne CH, Láng I, Folprecht
G, Nowacki MP, Cascinu S, Shchepotin I, Maurel J, Cunningham D,
Tejpar S, et al: Cetuximab plus irinotecan, fluorouracil, and
leucovorin as first-line treatment for metastatic colorectal
cancer: Updated analysis of overall survival according to tumor
KRAS and BRAF mutation status. J Clin Oncol. 29:2011–2019. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Van der Jeught K, Xu HC, Li YJ, Lu XB and
Ji G: Drug resistance and new therapies in colorectal cancer. World
J Gastroenterol. 24:3834–3848. 2018. View Article : Google Scholar : PubMed/NCBI
|