|
1
|
Zambo I and Veselý K: WHO classification
of tumours of soft tissue and bone 2013: The main changes compared
to the 3rd edition. Cesk Patol. 50:64–70. 2014.PubMed/NCBI
|
|
2
|
Gelderblom H, Blay JY, Seddon BM, Leahy M,
Ray-Coquard R, Sleijfer S, Kerst JM, Rutkowski P, Bauer S, Ouali M,
et al: Brostallicin versus doxorubicin as first-line chemotherapy
in patients with advanced or metastatic soft tissue sarcoma: an
European Organisation for Research and Treatment of Cancer Soft
Tissue and Bone Sarcoma Group randomised phase II and
pharmacogenetic study. Eur J Cancer. 50:388–396. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mirabello L, Troisi RJ and Savage SA:
International osteosarcoma incidence patterns in children and
adolescents, middle ages and elderly persons. Int J Cancer.
125:229–234. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Esiashvili N, Goodman M and Marcus RB Jr:
Changes in incidence and survival of Ewing sarcoma patients over
the past 3 decades: Surveillance Epidemiology and End Results data.
J Pediatr Hematol Oncol. 30:425–430. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goorin AM, Schwartzentruber DJ, Devidas M,
Gebhardt MC, Ayala AG, Harris MB, Helman LJ, Grier HE and Link MP;
Pediatric Oncology Group, : Presurgical chemotherapy compared with
immediate surgery and adjuvant chemotherapy for nonmetastatic
osteosarcoma: Pediatric Oncology Group Study POG-8651. J Clin
Oncol. 21:1574–1580. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nesbit ME Jr, Gehan EA, Burgert EO Jr,
Vietti TJ, Cangir A, Tefft M, Evans R, Thomas P, Askin FB and
Kissane JM: Multimodal therapy for the management of primary,
nonmetastatic Ewing's sarcoma of bone: A long-term follow-up of the
First Intergroup study. J Clin Oncol. 8:1664–1674. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Italiano A, Mir O, Cioffi A, Palmerini E,
Piperno-Neumann S, Perrin C, Chaigneau L, Penel N, Duffaud F, Kurtz
JE, et al: Advanced chondrosarcomas: Role of chemotherapy and
survival. Ann Oncol. 24:2916–2922. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
George B, Bresson D, Herman P and Froelich
S: Chordomas: A review. Neurosurg Clin N Am. 26:437–452. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Swann JB and Smyth MJ: Immune surveillance
of tumors. J Clin Invest. 117:1137–1146. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pardoll DM: The blockade of immune
checkpoints in cancer immunotherapy. Nat Rev Cancer. 12:252–264.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
McCoy KD and Le Gros G: The role of CTLA-4
in the regulation of T cell immune responses. Immunol Cell Biol.
77:1–10. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nishimura H and Honjo T: PD-1: An
inhibitory immunoreceptor involved in peripheral tolerance. Trends
Immunol. 22:265–268. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
He Y, Rivard CJ, Rozeboom L, Yu H, Ellison
K, Kowalewski A, Zhou C and Hirsch FR: Lymphocyte-activation
gene-3, an important immune checkpoint in cancer. Cancer Sci.
107:1193–1197. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cheng L and Ruan Z: Tim-3 and Tim-4 as the
potential targets for antitumor therapy. Hum Vaccin Immunother.
11:2458–2462. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Riha P and Rudd CE: CD28 co-signaling in
the adaptive immune response. Self Nonself. 1:231–240. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Redmond WL, Ruby CE and Weinberg AD: The
role of OX40-mediated co-stimulation in T-cell activation and
survival. Crit Rev Immunol. 29:187–201. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Palazón A, Teijeira A, Martínez-Forero I,
Hervás-Stubbs S, Roncal C, Peñuelas I, Dubrot J, Morales-Kastresana
A, Pérez-Gracia JL, Ochoa MC, et al: Agonist anti-CD137 mAb act on
tumor endothelial cells to enhance recruitment of activated T
lymphocytes. Cancer Res. 71:801–811. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dong C, Juedes AE, Temann UA, Shresta S,
Allison JP, Ruddle NH and Flavell RA: ICOS co-stimulatory receptor
is essential for T-cell activation and function. Nature.
409:97–101. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pende D, Cantoni C, Rivera P, Vitale M,
Castriconi R, Marcenaro S, Nanni M, Biassoni R, Bottino C, Moretta
A, et al: Role of NKG2D in tumor cell lysis mediated by human NK
cells: Cooperation with natural cytotoxicity receptors and
capability of recognizing tumors of nonepithelial origin. Eur J
Immunol. 31:1076–1086. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Overman MJ, McDermott R, Leach JL, Lonardi
S, Lenz HJ, Morse MA, Desai J, Hill A, Axelson M, Moss RA, et al:
Nivolumab in patients with metastatic DNA mismatch repair-deficient
or microsatellite instability-high colorectal cancer (CheckMate
142): An open-label, multicentre, phase 2 study. Lancet Oncol.
18:1182–1191. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Capdevila J, Wirth LJ, Ernst T, Ponce Aix
S, Lin CC, Ramlau R, Butler MO, Delord JP, Gelderblom H, Ascierto
PA, et al: PD-1 blockade in anaplastic thyroid carcinoma. J Clin
Oncol. 38:2620–2627. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Armand P, Lesokhin A, Borrello I,
Timmerman J, Gutierrez M, Zhu L, Popa McKiver M and Ansell SM: A
phase 1b study of dual PD-1 and CTLA-4 or KIR blockade in patients
with relapsed/refractory lymphoid malignancies. Leukemia. Jun
29–2020.(Epub ahead of print). doi: 10.1038/s41375-020-0939-1.
View Article : Google Scholar
|
|
23
|
Gabrilovich DI and Nagaraj S:
Myeloid-derived suppressor cells as regulators of the immune
system. Nat Rev Immunol. 9:162–174. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kondĕlková K, Vokurková D, Krejsek J,
Borská L, Fiala Z and Ctirad A: Regulatory T cells (TREG) and their
roles in immune system with respect to immunopathological
disorders. Acta Med (Hradec Kralove). 53:73–77. 2010. View Article : Google Scholar
|
|
25
|
Tada K, Kitano S, Shoji H, Nishimura T,
Shimada Y, Nagashima K, Aoki K, Hiraoka N, Honma Y, Iwasa S, et al:
Pretreatment immune status correlates with progression-free
survival in chemotherapy-treated metastatic colorectal cancer
patients. Cancer Immunol Res. 4:592–599. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kitano S, Postow MA, Ziegler CG, Kuk D,
Panageas KS, Cortez C, Rasalan T, Adamow M, Yuan J, Wong P, et al:
Computational algorithm-driven evaluation of monocytic
myeloid-derived suppressor cell frequency for prediction of
clinical outcomes. Cancer Immunol Res. 2:812–821. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gonda K, Shibata M, Ohtake T, Matsumoto Y,
Tachibana K, Abe N, Ohto H, Sakurai K and Takenoshita S:
Myeloid-derived suppressor cells are increased and correlated with
type 2 immune responses, malnutrition, inflammation, and poor
prognosis in patients with breast cancer. Oncol Lett. 14:1766–1774.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Arihara F, Mizukoshi E, Kitahara M, Takata
Y, Arai K, Yamashita T, Nakamoto Y and Kaneko S: Increase in
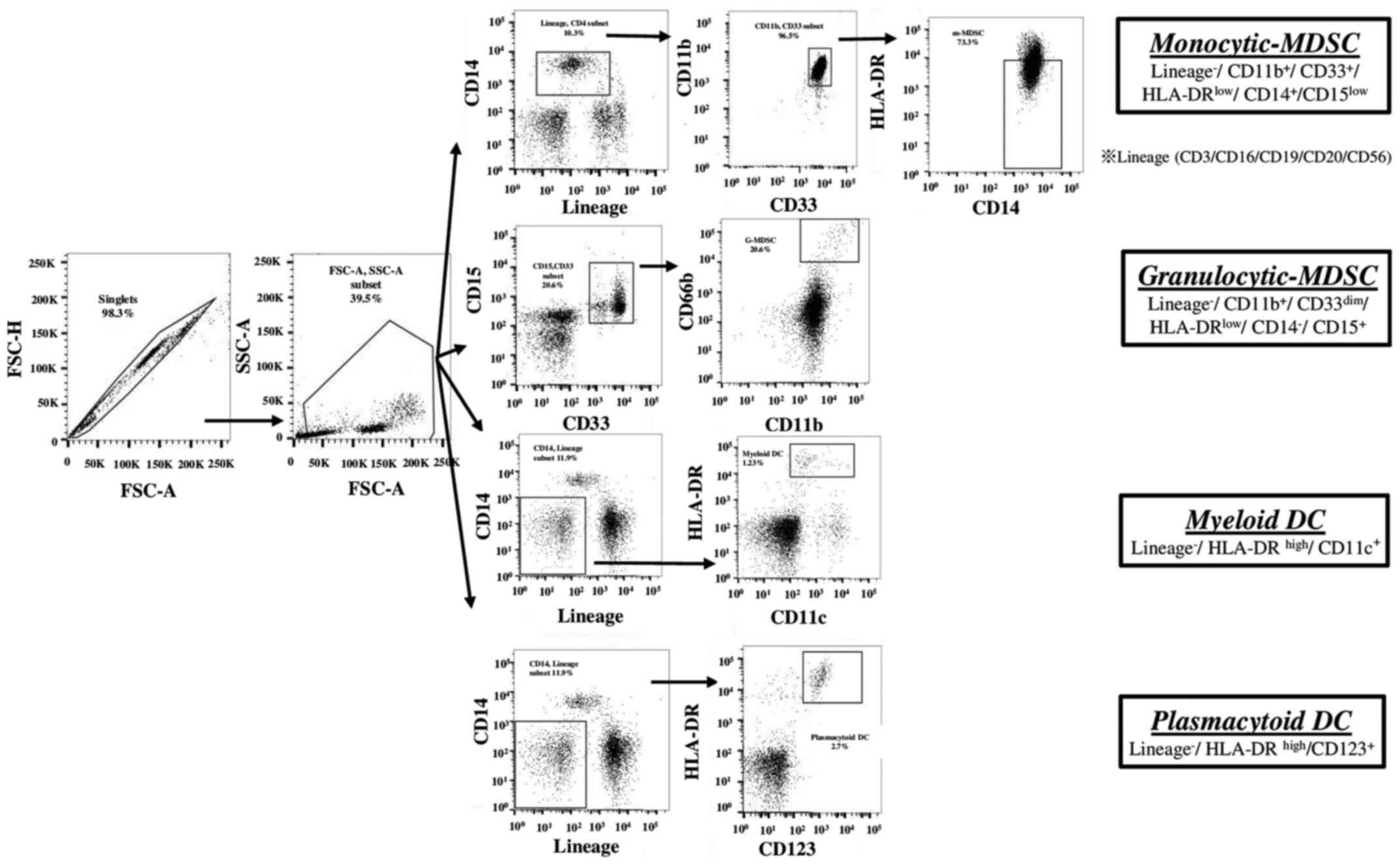
CD14+HLA-DR−/low myeloid-derived suppressor
cells in hepatocellular carcinoma patients and its impact on
prognosis. Cancer Immunol Immunother. 62:1421–1430. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kotsakis A, Harasymczuk M, Schilling B,
Georgoulias V, Argiris A and Whiteside TL: Myeloid-derived
suppressor cell measurements in fresh and cryopreserved blood
samples. J Immunol Methods. 381:14–22. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gabitass RF, Annels NE, Stocken DD, Pandha
HA and Middleton GW: Elevated myeloid-derived suppressor cells in
pancreatic, esophageal and gastric cancer are an independent
prognostic factor and are associated with significant elevation of
the Th2 cytokine interleukin-13. Cancer Immunol Immunother.
60:1419–1430. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vetsika EK, Koinis F, Gioulbasani M,
Aggouraki D, Koutoulaki A, Skalidaki E, Mavroudis D, Georgoulias V
and Kotsakis A: A circulating subpopulation of monocytic
myeloid-derived suppressor cells as an independent
prognostic/predictive factor in untreated non-small lung cancer
patients. J Immunol Res. 2014:6592942014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shoji H, Tada K, Kitano S, Nishimura T,
Shimada Y, Nagashima K, Aoki K, Hiraoka N, Honma Y, Iwasa S, et al:
The peripheral immune status of granulocytic myeloid-derived
suppressor cells correlates the survival in advanced gastric cancer
patients receiving cisplatin-based chemotherapy. Oncotarget.
8:95083–95094. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wesolowski R, Markowitz J and Carson WE
III: Myeloid derived suppressor cells - a new therapeutic target in
the treatment of cancer. J Immunother Cancer. 1:102013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhou J, Liu M, Sun H, Feng Y, Xu L, Chan
AW, Tong JH, Wong J, Chong CC, Lai PB, et al: Hepatoma-intrinsic
CCRK inhibition diminishes myeloid-derived suppressor cell
immunosuppression and enhances immune-checkpoint blockade efficacy.
Gut. 67:931–944. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sade-Feldman M, Kanterman J, Klieger Y,
Ish-Shalom E, Olga M, Saragovi A, Shtainberg H, Lotem M and
Baniyash M: Clinical significance of circulating
CD33+CD11b+HLA-DR− myeloid cells
in patients with stage IV melanoma treated with Ipilimumab. Clin
Cancer Res. 22:5661–5672. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhu C, Anderson AC, Schubart A, Xiong H,
Imitola J, Khoury SJ, Zheng XX, Strom TB and Kuchroo VK: The Tim-3
ligand galectin-9 negatively regulates T helper type 1 immunity.
Nat Immunol. 6:1245–1252. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gorman JV and Colgan JD: Regulation of T
cell responses by the receptor molecule Tim-3. Immunol Res.
59:56–65. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yan J, Zhang Y, Zhang JP, Liang J, Li L
and Zheng L: Tim-3 expression defines regulatory T cells in human
tumors. PLoS One. 8:e580062013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Arai Y, Saito H and Ikeguchi M:
Upregulation of TIM-3 and PD-1 on CD4+ and
CD8+ T cells associated with dysfunction of
cell-mediated immunity after colorectal cancer operation. Yonago
Acta Med. 55:1–9. 2012.PubMed/NCBI
|
|
41
|
Anderson AC: Tim-3, a negative regulator
of anti-tumor immunity. Curr Opin Immunol. 24:213–216. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ge W, Li J, Fan W, Xu D and Sun S: Tim-3
as a diagnostic and prognostic biomarker of osteosarcoma. Tumour
Biol. Jul 3–2017.(Epub ahead of print).
doi.org/10.1177/1010428317715643. View Article : Google Scholar
|
|
43
|
Zhuang X, Zhang X, Xia X, Zhang C, Liang
X, Gao L, Zhang X and Ma C: Ectopic expression of TIM-3 in lung
cancers: A potential independent prognostic factor for patients
with NSCLC. Am J Clin Pathol. 137:978–985. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Dorfman DM, Hornick JL, Shahsafaei A and
Freeman GJ: The phosphatidylserine receptors, T cell immunoglobulin
mucin proteins 3 and 4, are markers of histiocytic sarcoma and
other histiocytic and dendritic cell neoplasms. Hum Pathol.
41:1486–1494. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yang ZZ, Grote DM, Ziesmer SC, Niki T,
Hirashima M, Novak AJ, Witzig TE and Ansell SM: IL-12 upregulates
TIM-3 expression and induces T cell exhaustion in patients with
follicular B cell non-Hodgkin lymphoma. J Clin Invest.
122:1271–1282. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Feng ZM and Guo SM: Tim-3 facilitates
osteosarcoma proliferation and metastasis through the NF-κB pathway
and epithelial-mesenchymal transition. Genet Mol Res. Sep
2–2016.(Epub ahead of print). doi: 10.4238/gmr.15037844. View Article : Google Scholar
|
|
47
|
Groh V, Wu J, Yee C and Spies T:
Tumour-derived soluble MIC ligands impair expression of NKG2D and
T-cell activation. Nature. 419:734–738. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Raulet DH, Gasser S, Gowen BG, Deng W and
Jung H: Regulation of ligands for the NKG2D activating receptor.
Annu Rev Immunol. 31:413–441. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zafirova B, Wensveen FM, Gulin M and Polić
B: Regulation of immune cell function and differentiation by the
NKG2D receptor. Cell Mol Life Sci. 68:3519–3529. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu Y, Zhou L, Zong J, Ye Y, Chen G, Chen
Y, Liao X, Guo Q, Qiu S, Lin S, et al: Decreased expression of the
NKG2D ligand ULBP4 may be an indicator of poor prognosis in
patients with nasopharyngeal carcinoma. Oncotarget. 8:42007–42019.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cho H, Chung JY, Kim S, Braunschweig T,
Kang TH, Kim J, Chung EJ, Hewitt SM and Kim JH: MICA/B and ULBP1
NKG2D ligands are independent predictors of good prognosis in
cervical cancer. BMC Cancer. 14:9572014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen J, Xu H and Zhu XX: Abnormal
expression levels of sMICA and NKG2D are correlated with poor
prognosis in pancreatic cancer. Ther Clin Risk Manag. 12:11–18.
2015.PubMed/NCBI
|
|
53
|
Gabrilovich DI: Myeloid-derived suppressor
cells. Cancer Immunol Res. 5:3–8. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tcyganov E, Mastio J, Chen E and
Gabrilovich DI: Plasticity of myeloid-derived suppressor cells in
cancer. Curr Opin Immunol. 51:76–82. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kumar V, Patel S, Tcyganov E and
Gabrilovich DI: The nature of myeloid-derived suppressor cells in
the tumor microenvironment. Trends Immunol. 37:208–220. 2016.
View Article : Google Scholar : PubMed/NCBI
|