Introduction
Primary liver cancer is currently the fourth most
common malignant tumor type and the third leading cause of
mortality from cancer in China (1).
There are three pathological types of primary liver cancer,
including hepatocellular carcinoma (HCC), intrahepatic
cholangiocarcinoma (ICC) and a mixed HCC-ICC type. Of these, HCC
accounts for 85–90% of the cases (2). These cancers differ significantly in
pathogenesis, biological behavior, histological morphology,
treatment methods and prognosis (3).
The diverse pathological types coupled with their characteristic
rich blood supply dictate the specific surgical and anesthesia
techniques used for treatment (4).
Almost all liver cancer surgeries are inevitably
subject to prolonged anesthesia. Among all volatile anesthetics,
isoflurane may be used to induce and maintain general anesthesia to
eliminate the behavioral response of patients undergoing tumor
resection (5). However, several
studies have demonstrated that continuous isoflurane exposure
results in the alteration of tumor cell behavior, including
increased cell proliferation, invasion and metastasis. Huang et
al (6) reported that isoflurane
enhanced the proliferation and chemo-resistance of prostate cancer
cells through regulating the HIF-1α pathway. In non-small cell lung
cancer cells, exposure to isoflurane promoted cancer cell
proliferation by activating the Akt-mTOR signaling pathway
(7). In addition, it was reported
that, under exposure to isoflurane, melanoma grew faster in male
mice than in female mice (8). In
summary, these phenomena suggest that isoflurane exposure may be a
promoting factor in the progression of the majority of cancer types
(9).
SUMOylation, a form of protein post-translational
modification, frequently contributes toward the malignant
progression of tumors through involvement in various intracellular
processes, including transcriptional regulation, nuclear transport,
maintaining genomic integrity and signal transduction (10–13). In
general, SUMO1 mediates delayed reactions, while SUMO2 and SUMO3
are involved in acute cellular stress responses (14). To date, it has not been determined
whether isoflurane, a specific acute stress inducer, exerts its
cancer-promoting effect on HCC by activating the SUMO2/3 pathway.
In the present study, differences in SUMO2/3 protein expression
were observed in HCC cells following isoflurane treatment and the
effect of isoflurane on HCC cells was determined. The results
indicated that isoflurane may significantly induce the activation
of SUMO2/3 protein in HCC cells, characterized by an increase in
the conjugated form of SUMO2/3 during the acute phase of isoflurane
stimulation. Overexpression of SENP3 inhibited the binding of
SUMO2/3 to the target protein, which significantly inhibited the
isoflurane-induced activation of HCC cells. The results of the
present study provide potential targets for anesthesiologists to
decrease the side effects of anesthetic drugs during prolonged
liver cancer surgery.
Materials and methods
HCC cell culture and isoflurane
exposure
The human-derived hepatocellular carcinoma Hep3B
cell line was purchased from the American Type Culture Collection
(ATCC). The cells were placed in an incubator at 37°C with 5%
CO2 and cultured in Dulbecco's modified Eagle's medium
(DMEM), supplemented with 10% fetal bovine serum (FBS), 100 U/ml
penicillin and 100 µg/ml streptomycin (all Gibco; Thermo Fisher
Scientific, Inc.). Prior to isoflurane exposure, the Hep3B cells
were cultured in 60-mm plates at a density of 1×106
cells per plate for 12 h. The plates were then placed in a
1.5-liter airtight chamber with a continuous flow of 2% isoflurane
(Abbott Laboratories Ltd.), 21% oxygen, 5% carbon dioxide and 72%
nitrogen for 12 h, followed by a 36-h period of drug withdrawal.
The control group did not receive isoflurane treatment.
Gene transfection
A 2nd generation system was used for lentiviral
transfection. The overexpressing vector for pWPXLD-GFP-SENP3 or
pWPXLD-GFP (Biogot Technology, Co. Ltd.), the packaging plasmid,
psPAX2 (Addgene, Inc.), and the envelope plasmid, pMD2.G (Addgene,
Inc.), were transfected into 293T cells at the ratio of 4:3:1 for
production of lentiviral particles. Lipofectamine 2000™ (Thermo
Fisher Scientific, Inc.) was used as the transfection reagent, and
the ratio of transfection reagent to plasmid was 1:2.5. The
supernatant was filtered through a 0.45-µm filter and was
concentrated to 108 PFU/ml by passing through an
ultrafiltration tube (EMD Millipore). The concentrated virus was
then aliquoted, snap frozen in liquid nitrogen and stored at −80°C
as soon as possible to avoid loss of titer. In order to improve the
efficiency of infection, two infections were performed in total.
First, the concentrated virus was used to infect Hep3B cells with
20–30% confluence (5×105 cells) in a 60-mm dish with 8
mg/ml polybrene. The cells were incubated at 37°C with 5%
CO2 for ~18 h, or until the following morning.
Subsequently, the medium was carefully aspirated. The virus that
had been aliquoted and preserved was used to infect Hep3B cells
with 50–60% confluence with 8 mg/ml polybrene a second time.
Infected cells were selected using 1 mg/ml puromycin. After
successfully obtaining stable cell lines, 0.5 mg/ml puromycin was
used to maintain the stable cell system. After 15 days, gene
transfection efficiency was verified by western blot analysis.
Western blot analysis
Total protein was extracted from Hep3B cells after
0.5, 2, 6 and 12 h of isoflurane exposure, and under conditions of
12 h of isoflurane exposure, followed by drug withdrawal for a
period of 12 or 36 h through incubation in RIPA buffer (Beijing
Solarbio Science & Technology Co., Ltd.), supplemented with 1
mM phenylmethanesulfonyl fluoride and 20 mM N-ethylmaleimide.
Bicinchoninic acid assay (cat. no. PC0020; Beijing Solarbio Science
& Technology Co., Ltd.) was used for protein quantification.
Subsequently, 100 µg protein was separated on 4–12% SDS-PAGE gels,
transferred onto polyvinylidene fluoride membranes (EMD Millipore,
Inc.), blocked with 5% skimmed milk and 0.1% Tris-buffered saline
with 1 ml/l Tween-20 for 1 h at 37°C, and incubated with specific
antibodies overnight at 4°C following the parameters provided in
Table I. The membranes were then
incubated for 1 h at room temperature with horseradish
peroxidase-conjugated mouse anti-rabbit IgG (1:2,000; cat. no.
sc-2357; Santa Cruz Biotechnology, Inc.) or goat anti-chicken IgY
H&L (1:2,000; cat. no. ab6877; Abcam) secondary antibodies. A
Super Signal protein detection kit (cat. no. 34095; Pierce; Thermo
Fisher Scientific, Inc.) was used to detect protein signals
according to the manufacturer's protocol. Following development,
the value of each band was quantified by ImageJ version 1.48 image
analysis software (National Institutes of Health).
 | Table I.Information regarding all antibodies
used in the present study. |
Table I.
Information regarding all antibodies
used in the present study.
| Antibody | SUMO2/3 | SENP3 | GFP | β-actin | IgG | IgY |
|---|
| Supplier | Abcam | Abcam | Abcam | Abcam | Santa Cruz
Biotechnology, Inc. | Abcam |
| Catalog no. | ab3742 | ab124790 | ab13970 | ab8227 | sc-2357 | ab6877 |
| Dilution
factor | 1:1,000 | 1:2,000 | 1:5,000 | 1:1,000 | 1:2,000 | 1:2,000 |
Cell proliferation assay
The proliferation activity of tumor cells was
measured using the Cell Counting kit-8 (CCK-8; Shanghai Yeasen
Biotechnology Co., Ltd.). Hep3B cells were cultured in 96-well
plates for 24 h in normal medium, followed by a 12 h isoflurane
exposure. The culture media was then replaced with fresh media, and
cells were cultured for an additional 36 h. To determine the
proliferation rate of the tumor cells, CCK-8 (5 mg/ml) was added to
each well and the cells were incubated for 1 h, followed by
measuring the absorbance of each well at 450 nm using a microplate
reader (Bio-Rad Laboratories, Inc.). Finally, the absorbance values
were used to derive the relative cell number from a standard
curve.
Wound healing assay
When cell confluence in a 6-well plate reached 90%,
the Hep3B cells were exposed to 2% isoflurane for 12 h in DMEM
supplemented with 10% FBS. The plates were removed and the medium
was replaced with serum-free DMEM. The surface of the cells was
lightly scratched using a 200 µl pipette tip to create a wound
healing model. The scratch width was measured and recorded as S0
under a light microscope (magnification, ×200; Olympus cellSens
Entry 1.16; Olympus Corporation). After 36 h of routine culture in
serum-free DMEM, the distance between the two sides of the scratch
was measured again and recorded as S1. The actual healing distance
was calculated by S0-S1.
Invasion assay
The invasion capacity of the tumor cells was
detected using a Transwell culture chamber. In brief,
1×104 Hep3B cells were seeded into the upper chambers
precoated with Matrigel overnight at 37°C (BD Biosciences), and the
lower chambers remained empty. Next, the Transwell chamber was
transferred into an airtight chamber and exposed to 2% isoflurane
for 12 h. The Transwell culture chamber was then removed and the
medium in the upper chambers was replaced with serum-free DMEM, and
DMEM medium containing 10% FBS was added to the lower chambers.
After 36 h of culture at 37°C, the membrane of the upper chamber
was stained with crystal violet for 10 min at room temperature. The
number of invading cells was counted under an inverted light
microscope (magnification, ×200; Olympus cellSens Entry 1.16;
Olympus Corporation).
Statistical analysis
The data are presented as the mean ± standard
deviation and were analyzed using GraphPad Prism 6 software.
Comparisons between two groups were performed using a Student's
t-test. Differences between multiple groups were analyzed by a
one-way analysis of variance followed by Tukey's test. P<0.05
was considered to indicate a statistically significant difference.
All the experiments were repeated three times.
Results
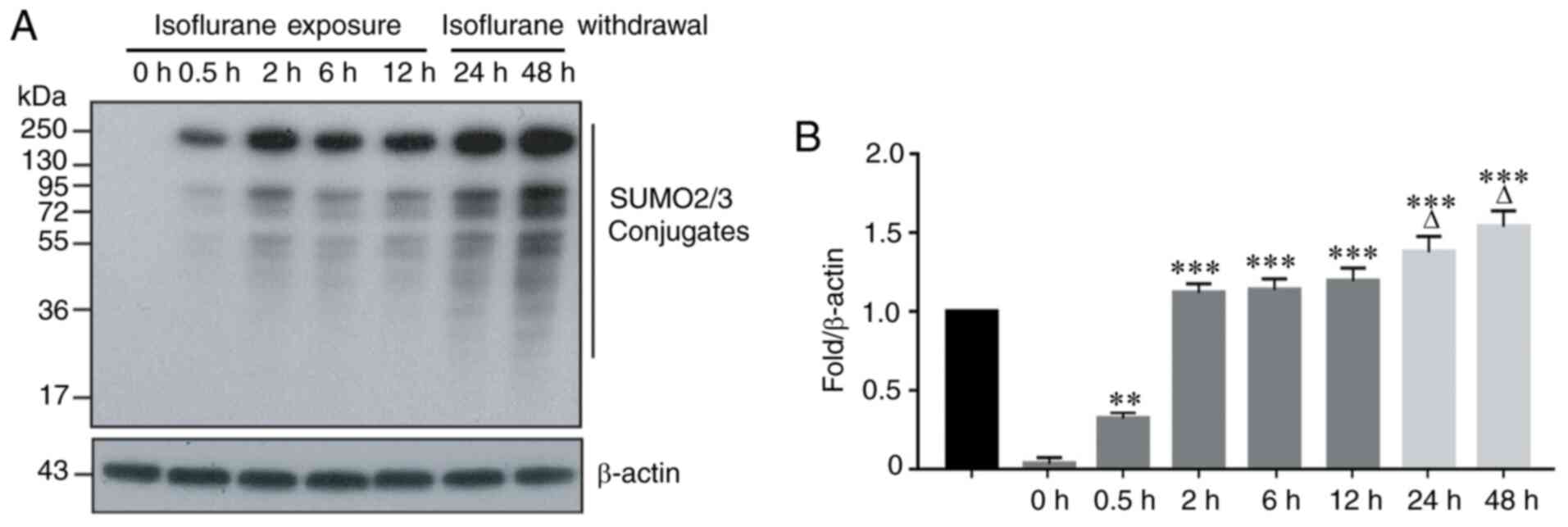
Isoflurane exposure significantly
increases the number of SUMO2/3 conjugates
SUMO2 and SUMO3 are two members of the SUMO family
that participate in the acute stress response. To observe the
effect of isoflurane exposure on SUMO2/3 protein expression, the
human-derived HCC Hep3B line was exposed to 2% isoflurane for 0.5,
2, 6 and 12 h. The results indicated that a short period of
isoflurane exposure resulted in significant changes in the
expression of SUMO2/3 protein in hepatoma cells. This was
manifested by an increased number of SUMO2/3 conjugates at 0.5 h of
isoflurane exposure, and this upward trend continued with prolonged
exposure to isoflurane (Fig. 1A and
B). To determine whether the number of SUMO2/3 conjugates
changes following the withdrawal of isoflurane, the HCC cells were
exposed to isoflurane for 12 h, and the cells were cultured in
fresh medium without isoflurane for an additional 12 h (total 24 h)
and 36 h (total 48 h). The results indicated that the number of
SUMO2/3 conjugates continued to increase until 48 h, and this
upward trend did not change following the withdrawal of isoflurane
(Fig. 1A and B).
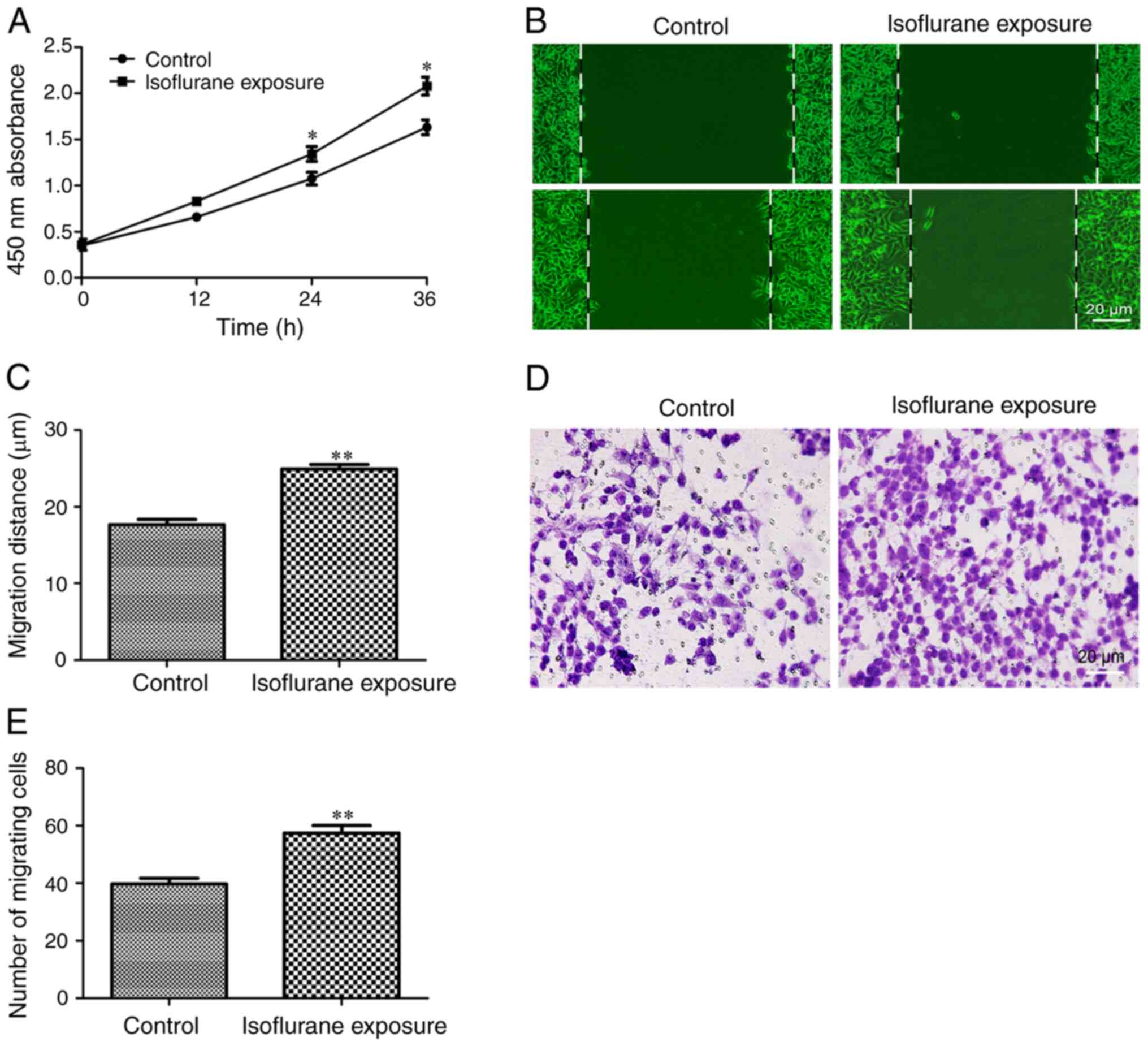
Isoflurane exposure promotes malignant
progression of liver cancer cells
Next, the effects of isoflurane exposure on the
behavior of liver cancer cells were investigated. The results of
the CCK-8 experiment demonstrated that HCC cells exhibited a faster
proliferation rate following exposure to isoflurane for 12 h
(Fig. 2A). The effects of isoflurane
on liver cancer cell migration and invasion were then examined. The
results indicated that the tumor cells exposed to isoflurane
exhibited a stronger migration (Fig. 2B
and C) and invasion ability (Fig. 2D
and E). These results indicated that isoflurane exposure
promotes the proliferation, migration and invasion of HCC cells and
induces the malignant progression of liver cancer.
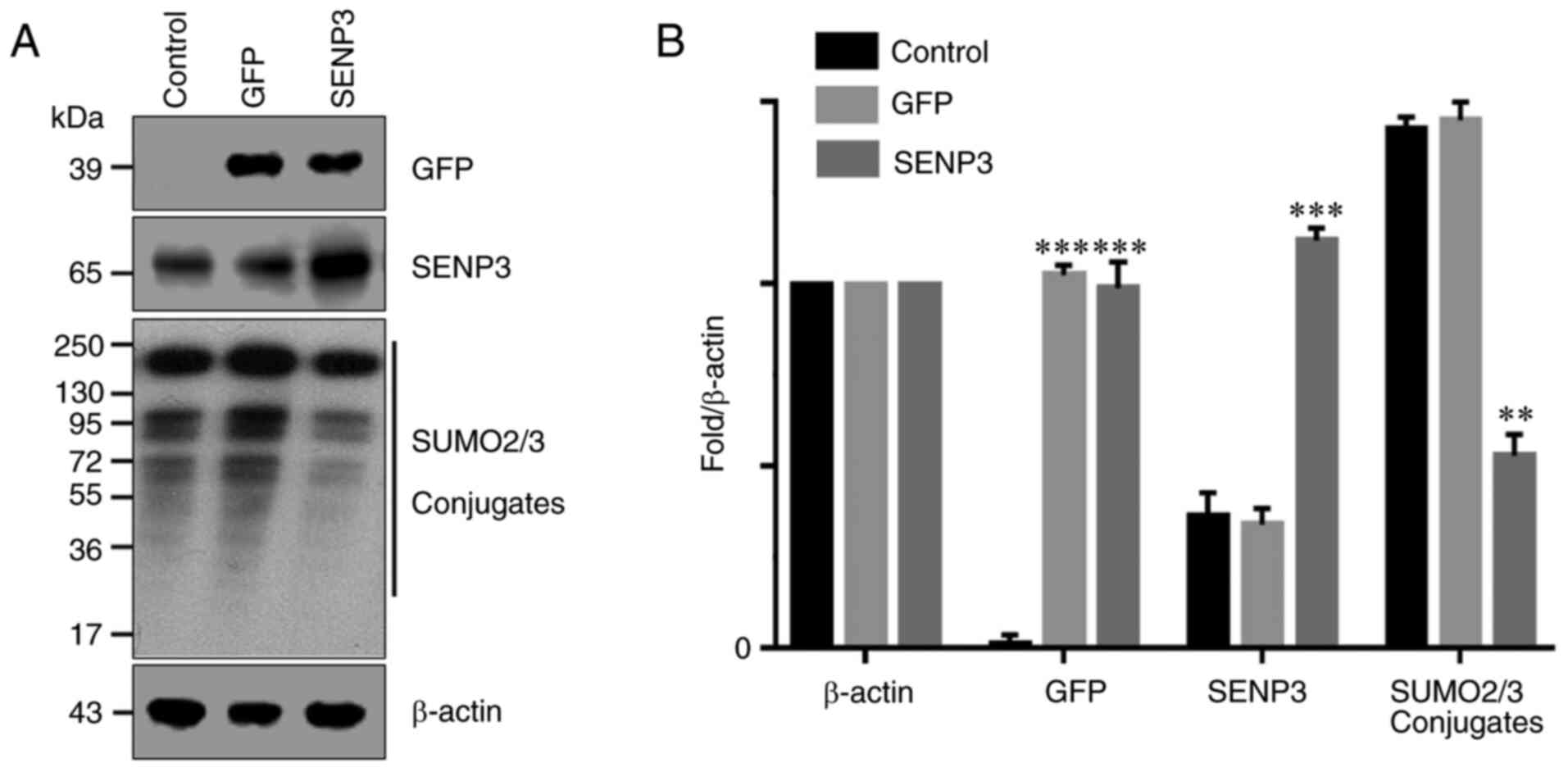
Overexpression of SENP3 effectively
inhibits the generation of SUMO2/3 conjugates
It has been reported that SENP3 may specifically
inhibit the binding of SUMO2/3 to its target proteins. Therefore,
SENP3 was overexpressed in HCC cells through gene transfection and
the resulting number of SUMO2/3 conjugates were detected by western
blot analysis. The results demonstrated that the control HCC cells
expressed lower levels of SENP3 protein (Fig. 3A and B). By contrast, gene
transfection significantly increased the expression level of SENP3
protein in HCC cells. As a result, the ability of SUMO2/3 to bind
to its target protein was inhibited and the number of SUMO2/3
conjugates was significantly decreased (Fig. 3A and B).
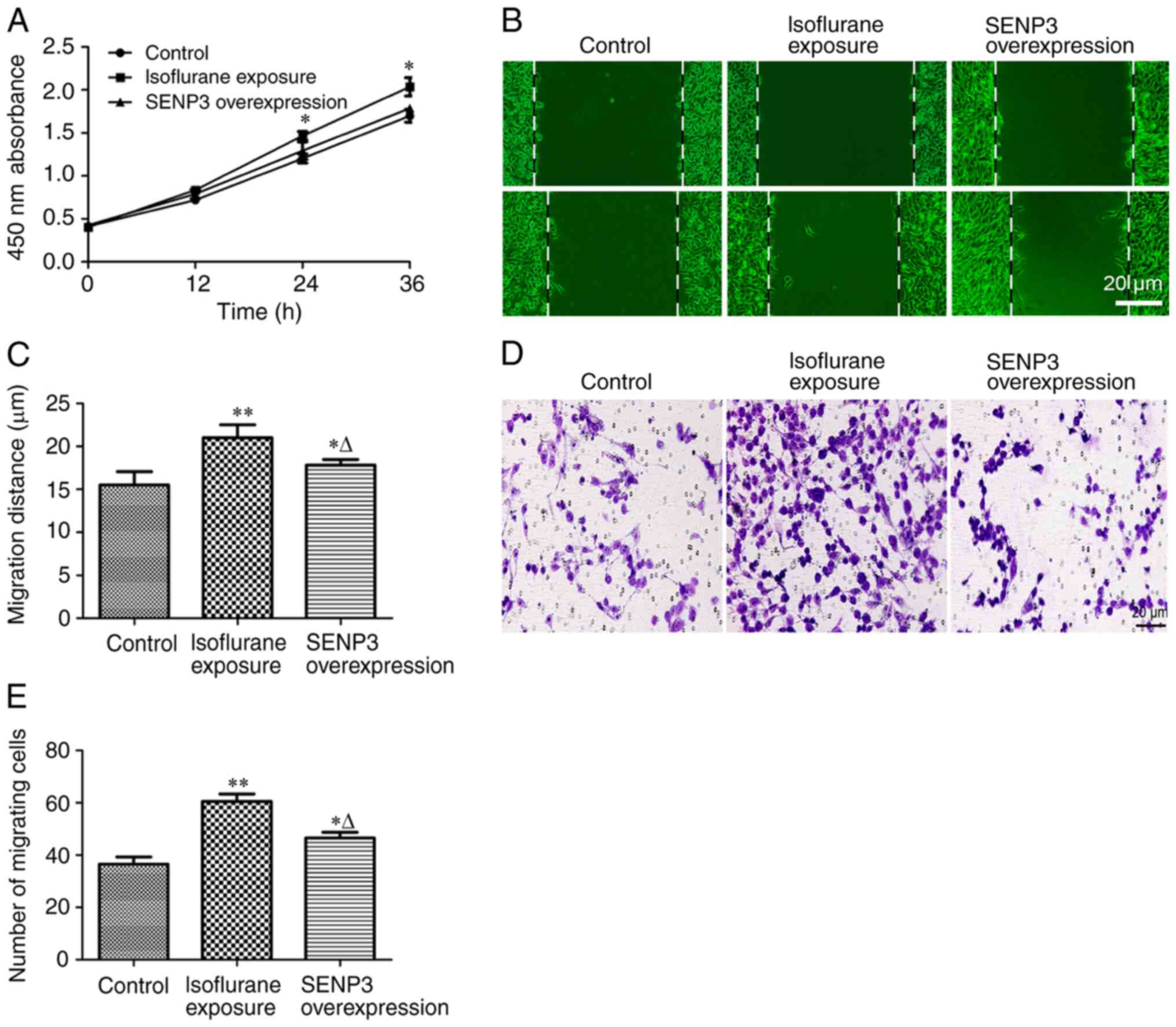
Inhibition of SUMO2/3 disrupts the
malignant progression of liver cancer cells exposed to
isoflurane
The effect of inhibiting the number of SUMO2/3
conjugates on the progression of liver cancer cells exposed to
isoflurane was investigated by overexpressing SENP3. The results of
cell proliferation experiments revealed that overexpression of
SENP3 antagonized isoflurane-induced proliferation of HCC cells
(Fig. 4A). Furthermore, inhibition
of SUMO2/3 conjugate formation by overexpression of SENP3
significantly inhibited the migration (Fig. 4B and C) and invasion (Fig. 4D and E) of HCC cells resulting from
isoflurane exposure. Taken together, these results indicated that
overexpression of SENP3 may effectively inhibit the generation of
SUMO2/3 conjugates and antagonize the malignant progression of
liver cancer cells induced by isoflurane exposure.
Discussion
In China, liver cancer accounts for the second
highest mortality rate from malignant tumors in cities and
represents the highest mortality rate in certain rural areas
(15). Guangxi Province has
experienced a particularly high incidence of liver cancer in
mainland China (16). At present,
surgery remains the primary treatment method for liver cancer
(17). The pathological type, tumor
volume, intrahepatic location, invasive disease and distant
metastasis affect the duration of liver cancer surgery and expose
patients to anesthesia for extended periods of time (17). It is generally believed that
short-term exposure to narcotic drugs does not cause significant
adverse effects in patients (18).
However, inappropriate drug selection, long-term anesthetic drug
exposure and the quality of patient liver function, may affect
long-term prognosis (19).
Therefore, elderly and frail patients and those with abnormal liver
function are given extra attention by anesthesiologists.
Among narcotic drugs, halogen anesthetics, including
sevoflurane, isoflurane and enflurane, are the main gas anesthetics
that are widely used clinically (20). They are inhaled in gaseous form,
incorporated into the blood circulation and transported to the
central nervous system where they exert an anesthetic effect.
Halogen anesthetics have several advantages, including a strong
effect, rapid induction of anesthesia, quick patient recovery and
ease of use (21). Therefore, they
have remained a cornerstone in general anesthesia.
However, a problem that should not be overlooked is
that inhaled anesthetics exhibit liver toxicity. Halothane is the
most commonly used drug that causes liver toxicity (22). Compared with halothane, the incidence
of liver toxicity with sevoflurane, enflurane, isoflurane and other
halogenated inhalation anesthetics is significantly decreased, but
not completely eradicated (23).
These halogenated inhalation anesthetics are oxidized and
metabolized by P4502E1 isoenzymes to produce substances similar to
the intermediate products of halothane metabolism (24). Therefore, these drugs exhibit similar
activity coupled with mild hepatotoxicity. A recent study revealed
that insulin-like growth factor 1 (IGF-1) levels are downregulated
following exposure to isoflurane anesthesia (25). IGF-1 may be involved in the liver
injury mechanism induced by exposure to isoflurane (25). However, another group demonstrated
that isoflurane inhibits hepatic carcinoma growth and
aggressiveness, and promotes apoptosis through the
PI3K/Akt-mediated NF-κB signaling pathway (9). Although these results appear to be
contradictory, the results were obtained using primary cultured
cells from patients receiving general anesthesia, isoflurane,
intravenous medication or even tissue damage caused by surgery. We
hypothesize that different methods of anesthesia result in
different experimental conclusions. Lai et al (5) reported that during open hepatectomy for
HCC, the use of propofol anesthesia was associated with longer
survival times compared with desflurane. Patients receiving
propofol anesthesia exhibited a significant decrease in distant
metastasis and local recurrence (5).
Another issue that should be considered is that
specific types of inhaled anesthetics, or long-term exposure to
certain anesthetics, may cause unpredictable consequences in tumors
(26). The present study focused on
the biological response of HCC cells following isoflurane exposure.
The identification of novel molecular mechanisms may lead to
interventions against these molecular targets to decrease the
adverse effects of isoflurane on HCC cells. SUMOylation, mediated
by members of the SUMO family, is widely involved in various
cellular responses to external stimuli and internal
microenvironmental changes (27).
SUMO1 is involved in delayed cellular responses, whereas SUMO2/3 is
primarily involved in acute stress responses (14,28). In
general, simple liver cancer surgery lasts 2–5 h, while complex
liver cancer resection plus lymph node dissection may require 10 h
or more. Therefore, in the present study, changes in SUMO2/3
conjugates were investigated at different time points during early
isoflurane exposure. The results of the present study indicated
that isoflurane exposure for 0.5 h may induce a significant
increase in SUMO2/3 conjugates, and this upward trend continued
with increased isoflurane exposure time. Considering that patients
undergoing refractory liver cancer surgery may receive anesthesia
for more than ten h, the maximum exposure time of isoflurane was
limited to 12 h, and the subsequent changes in SUMO2/3 conjugates
following isoflurane withdrawal were observed. The results
demonstrated that the increased number of SUMO2/3 conjugates did
not abate with the withdrawal of isoflurane. By contrast, the
number of SUMO2/3 conjugates continued to increase up to 48 h after
detection. The effect of isoflurane exposure on the HCC cell
phenotype was then investigated. The results demonstrated that
isoflurane exposure increased the proliferation, migration and
invasion activity of HCC cells.
It is generally recognized that SENP3 may
effectively dissociate SUMO2/3 from its substrates (29). Therefore, in follow-up experiments,
SENP3 was overexpressed in HCC cells by gene transfection, which
effectively inhibited the generation of SUMO2/3 conjugates. The
SUMO2/3 substrates that have been identified thus far include Akt,
HIF-1α, Oct4, CDK6 and dozens of other proteins involved in tumor
cell proliferation, migration and invasion (30–35). A
recent study in rats demonstrated that increased levels of Heat
shock protein 27 resulting from SUMO2/3-mediated SUMOylation serve
an important role in the progression of primary hepatocellular
carcinoma (36). This finding may
provide insight for the development of novel therapeutic drugs to
treat primary hepatocellular carcinoma (36).
In the present study, the specific proteins that are
activated by isoflurane exposure to generate conjugates with
SUMO2/3 were not investigated, but such investigations should be
made in the future by targeting SUMO2/3 with proteomics technology.
Nevertheless, it remains likely that isoflurane participates in the
malignant progression of HCC cells by activating SUMO2/3 and
increasing the formation of conjugates. Inhibiting the generation
of SUMO2/3 conjugates by overexpressing SENP3 also antagonizes the
progression of cancer cells resulting from isoflurane exposure.
In conclusion, it was confirmed that short-term
(<12 h) isoflurane exposure results in the activation of SUMO2/3
and increases the formation of conjugates, thereby promoting the
malignant progression of HCC cells. Therefore, on the basis of the
results of the present study, we hypothesize that targeted
inhibition of SUMO2/3 conjugate formation may antagonize the
progression of HCC caused by isoflurane exposure. However, the
Hep3B cell line was derived from an 8-year-old black male with HCC
(37) and is different from HCC
cells derived from adults (38,39).
Therefore, further studies are required using additional liver
cancer cell lines, including those derived from adults, various
tumor types and animal experiments. Furthermore, as the role of
SUMO2/3 expression in the progression of HCC cells remains unclear,
further investigations are required in future studies.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from
National Natural Science Foundation of China (grant nos. 81901526
and 81900407), Tianjin Natural Science Foundation of China (grant
nos. 18JCQNJC12800, 19JCZDJC35200, 19JCQNJC12100 and
19JCQNJC11900), Tianjin Special Project of New Generation
Artificial Intelligence Technology (grant no. 18ZXZNSY00260) and
Binhai Health and Family Planning Commission Science and Technology
Projects (grant nos. 2019BWKQ030 and 2019BWKQ029).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
WW designed the experiments. PW, XL, CZ, ZJ and NX
performed the experiments and collected data. WW and SS analyzed
and interpreted the data. PW, XL and CZ drafted the manuscript. WW
agreed to be accountable for all aspects of the work in ensuring
that questions related to the accuracy or integrity of any part of
the work are appropriately investigated and resolved. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chao J, Zhao S and Sun H:
Dedifferentiation of hepatocellular carcinoma: Molecular mechanisms
and therapeutic implications. Am J Transl Res. 12:2099–2109.
2020.PubMed/NCBI
|
|
2
|
Garin E, Palard X and Rolland Y:
Personalised dosimetry in radioembolisation for HCC: Impact on
clinical outcome and on trial design. Cancers (Basel). 12:15572020.
View Article : Google Scholar
|
|
3
|
Tellapuri S, Sutphin PD, Beg MS, Singal AG
and Kalva SP: Staging systems of hepatocellular carcinoma: A
review. Indian J Gastroenterol. 37:481–491. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Glantzounis GK, Paliouras A, Stylianidi
MC, Milionis H, Tzimas P, Roukos D, Pentheroudakis G and Felekouras
E: The role of liver resection in the management of intermediate
and advanced stage hepatocellular carcinoma. A systematic review.
Eur J Surg Oncol. 44:195–208. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lai HC, Lee MS, Lin C, Lin KT, Huang YH,
Wong CS, Chan SM and Wu ZF: Propofol-based total intravenous
anaesthesia is associated with better survival than desflurane
anaesthesia in hepatectomy for hepatocellular carcinoma: A
retrospective cohort study. Br J Anaesth. 123:151–160. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huang H, Benzonana LL, Zhao H, Watts HR,
Perry NJ, Bevan C, Brown R and Ma D: Prostate cancer cell
malignancy via modulation of HIF-1α pathway with isoflurane and
propofol alone and in combination. Br J Cancer. 111:1338–1349.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang W and Shao X: Isoflurane promotes
non-small cell lung cancer malignancy by activating the
Akt-mammalian target of rapamycin (mTOR) signaling pathway. Med Sci
Monit. 22:4644–4650. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Meier A, Gross ETE, Schilling JM, Seelige
R, Jung Y, Santosa E, Searles S, Lin T, Tu XM, Patel HH and Bui JD:
Isoflurane impacts murine melanoma growth in a sex-specific,
immune-dependent manner: A brief report. Anesth Analg.
126:1910–1913. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hu J, Hu J, Jiao H and Li Q: Anesthetic
effects of isoflurane and the molecular mechanism underlying
isoflurane-inhibited aggressiveness of hepatic carcinoma. Mol Med
Rep. 18:184–192. 2018.PubMed/NCBI
|
|
10
|
Seeler JS and Dejean A: SUMO and the
robustness of cancer. Nat Rev Cancer. 17:184–197. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Flotho A and Melchior F: Sumoylation: A
regulatory protein modification in health and disease. Annu Rev
Biochem. 82:357–385. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Geiss-Friedlander R and Melchior F:
Concepts in sumoylation: A decade on. Nat Rev Mol Cell Biol.
8:947–956. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Henley JM, Craig TJ and Wilkinson KA:
Neuronal SUMOylation: Mechanisms, physiology, and roles in neuronal
dysfunction. Physiol Rev. 94:1249–1285. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chang SC and Ding JL: Ubiquitination and
SUMOylation in the chronic inflammatory tumor microenvironment.
Biochim Biophys Acta Rev Cancer. 1870:165–175. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Qiu G, Jin Z, Chen X and Huang J:
Interpretation of guidelines for the diagnosis and treatment of
primary liver cancer (2019 edition) in China. Glob Health Med.
2:306–311. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pang Y, Kartsonaki C, Turnbull I, Guo Y,
Chen Y, Clarke R, Bian Z, Bragg F, Millwood IY, Yang L, et al:
Adiposity in relation to risks of fatty liver, cirrhosis and liver
cancer: A prospective study of 0.5 million Chinese adults. Sci Rep.
9:7852019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
McCarty TR, Echouffo-Tcheugui JB, Lange A,
Haque L and Njei B: Impact of bariatric surgery on outcomes of
patients with nonalcoholic fatty liver disease: A nationwide
inpatient sample analysis, 2004–2012. Surg Obes Relat Dis.
14:74–80. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pinto RZ, Maher CG, Ferreira ML, Ferreira
PH, Hancock M, Oliveira VC, McLachlan AJ and Koes B: Drugs for
relief of pain in patients with sciatica: Systematic review and
meta-analysis. BMJ. 344:e4972012. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Komaki Y, Komaki F, Micic D, Ido A and
Sakuraba A: Risk of colorectal cancer in chronic liver diseases: A
systematic review and meta-analysis. Gastrointest Endosc.
86:93–104.e5. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Antipass A, Austin A, Awad S, Hughes D and
Idris I: Evaluation of liver function tests and risk score
assessment to screen patients for significant liver disease prior
to bariatric and metabolic surgery. Obes Surg. 30:2840–2843. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ikuta S, Tanimura K, Yasui C, Aihara T,
Yoshie H, Iida H, Beppu N, Kurimoto A, Yanagi H, Mitsunobu M and
Yamanaka N: Chronic liver disease increases the risk of
linezolid-related thrombocytopenia in methicillin-resistant
Staphylococcus aureus-infected patients after digestive surgery. J
Infect Chemother. 17:388–391. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Esser T, Keilhoff G and Ebmeyer U:
Anesthesia specific differences in a cardio-pulmonary resuscitation
rat model; halothane versus sevoflurane. Brain Res. 1652:144–150.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dugan CM, Fullerton AM, Roth RA and Ganey
PE: Natural killer cells mediate severe liver injury in a murine
model of halothane hepatitis. Toxicol Sci. 120:507–518. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yin H, Cheng L, Langenbach R and Ju C:
Prostaglandin I(2) and E(2) mediate the protective effects of
cyclooxygenase-2 in a mouse model of immune-mediated liver injury.
Hepatology. 45:159–169. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu Y, Xiao X, Li G, Bu J, Zhou W and Zhou
S: Isoflurane anesthesia induces liver injury by regulating the
expression of insulin-like growth factor 1. Exp Ther Med.
13:1608–1613. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Feng D, Wang Y, Xu Y, Luo Q, Lan B and Xu
L: Interleukin 10 deficiency exacerbates halothane induced liver
injury by increasing interleukin 8 expression and neutrophil
infiltration. Biochem Pharmacol. 77:277–284. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Delgado TC, Lopitz-Otsoa F and
Martínez-Chantar ML: Post-translational modifiers of liver kinase
B1/serine/threonine kinase 11 in hepatocellular carcinoma. J
Hepatocell Carcinoma. 6:85–91. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jin ZL, Pei H, Xu YH, Yu J and Deng T: The
SUMO-specific protease SENP5 controls DNA damage response and
promotes tumorigenesis in hepatocellular carcinoma. Eur Rev Med
Pharmacol Sci. 20:3566–3573. 2016.PubMed/NCBI
|
|
29
|
Kim DH, Kwon S, Byun S, Xiao Z, Park S, Wu
SY, Chiang CM, Kemper B and Kemper JK: Critical role of
RanBP2-mediated SUMOylation of small heterodimer partner in
maintaining bile acid homeostasis. Nat Commun. 7:121792016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xia W, Tian H, Cai X, Kong H, Fu W, Xing
W, Wang Y, Zou M, Hu Y and Xu D: Inhibition of SUMO-specific
protease 1 induces apoptosis of astroglioma cells by regulating
NF-κB/Akt pathways. Gene. 595:175–179. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lin CH, Liu SY and Lee EH: SUMO
modification of Akt regulates global SUMOylation and substrate
SUMOylation specificity through Akt phosphorylation of Ubc9 and
SUMO1. Oncogene. 35:595–607. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Risso G, Pelisch F, Pozzi B, Mammi P,
Blaustein M, Colman-Lerner A and Srebrow A: Modification of Akt by
SUMO conjugation regulates alternative splicing and cell cycle.
Cell Cycle. 12:3165–3174. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Carbia-Nagashima A, Gerez J, Perez-Castro
C, Paez-Pereda M, Silberstein S, Stalla GK, Holsboer F and Arzt E:
RSUME, a small RWD-containing protein, enhances SUMO conjugation
and stabilizes HIF-1alpha during hypoxia. Cell. 131:309–323. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wei F, Schöler HR and Atchison ML:
Sumoylation of Oct4 enhances its stability, DNA binding, and
transactivation. J Biol Chem. 282:21551–21560. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xu Y, Li J, Zuo Y, Deng J, Wang LS and
Chen GQ: SUMO-specific protease 1 regulates the in vitro and in
vivo growth of colon cancer cells with the upregulated expression
of CDK inhibitors. Cancer Lett. 309:78–84. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ge H, Du J, Xu J, Meng X, Tian J, Yang J
and Liang H: SUMOylation of HSP27 by small ubiquitin-like modifier
2/3 promotes proliferation and invasion of hepatocellular carcinoma
cells. Cancer Biol Ther. 18:552–559. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Knowles BB, Howe CC and Aden DP: Human
hepatocellular carcinoma cell lines secrete the major plasma
proteins and hepatitis B surface antigen. Science. 209:497–499.
1980. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Knasmuller S, Parzefall W, Sanyal R, Ecker
S, Schwab C, Uhl M, Mersch-Sundermann V, Williamson G, Hietsch G,
Langer T, et al: Use of metabolically competent human hepatoma
cells for the detection of mutagens and antimutagens. Mutat Res.
402:185–202. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Qiu GH, Xie X, Xu F, Shi X, Wang Y and
Deng L: Distinctive pharmacological differences between liver
cancer cell lines HepG2 and Hep3B. Cytotechnology. 67:1–12. 2015.
View Article : Google Scholar : PubMed/NCBI
|