Introduction
In women, breast cancer is the most diagnosed cancer
(24.5% of total cases), followed by colorectal (9.4%) and lung
(8.4%) cancers (1). Breast cancer is
also the leading cause of cancer-associated mortality (15.5% of
total cancer-associated mortalities) among females, followed by
lung (13.7%) and colorectal (9.5%) cancers (1). Currently, chemotherapy and surgery are
the main treatment approaches for breast cancer. However, the
recurrence and chemoresistance of breast cancer are obstacles to
its treatment, and the molecular mechanism underlying these
phenomena remain unclear (2). Using
a single biomarker is insufficient in adequately assessing tumor
growth, thus, identifying the molecular mechanisms underlying tumor
growth and cell proliferation, through multiple signaling pathways
consisting of a set of core regulatory elements, may be more
effective.
The Hippo signaling pathway, a novel and
evolutionarily conserved tumor suppression pathway first discovered
in Drosophila, regulates tissue growth and organ size by
attenuating cell proliferation and simultaneously promoting cell
death (3). In humans, the core Hippo
pathway consists of the scaffold protein merlin, encoded by the
neurofibromatosis type 2 (NF2) gene, KIBRA protein, WW and the C2
domain containing 1 (WWC1) gene and a cascade of kinases, including
the upstream sterile 20-like kinase 1 and 2 (MST1/2), the cofactor
Salvador homologue 1 (SAV1), MOB kinase activator 1A and B
(MOB1A/B), and the downstream large tumor suppressors 1 and 2
(LATS1/2). In response to high cell density, NF2 and WWC1 activate
the Hippo signaling pathway by binding to LATS1/2 to facilitate
their activation by MST1/2 (4,5).
Subsequently, MST1/2 binds to SAV1 to form an enzymatic complex
that phosphorylates and activates LATS1/2 kinases and their MOB1A/B
regulatory subunits. LATS1/2 phosphorylates the
transcriptionalcoactivator Yes-associated protein (YAP) and WW
domain-containing transcription regulator protein 1, also known as
transcriptional coactivator with PDZ-binding motif (TAZ), which in
turn translocate in the cytoplasm and are inactivated (6). In cancer, the Hippo signaling pathway
is abnormally inactivated, thus, LATS1/2 is not activated.
Following nuclear translocation, the YAP/TAZ complex binds to the
transcriptional enhancer factor domain (TEAD)/TEF to activate the
transcription of its target genes, including connective tissue
growth factor (CTGF), cysteine-rich angiogenic inducer 61 (CYR61),
glioma-associated oncogene family zinc finger 2 (Gli2) and
amphiregulin (AREG), which in turn promote tumor cell proliferation
and growth (5,7) (Fig. 1).
Previous studies have demonstrated that the YAP/TAZ complex is
often abnormally expressed in breast cancer, and its overactivation
eventually leads to several tumor-promoting processes, such as
epithelial-to-mesenchymal transition(EMT), cancer stem cell
generation and chemotherapy resistance (7,8).
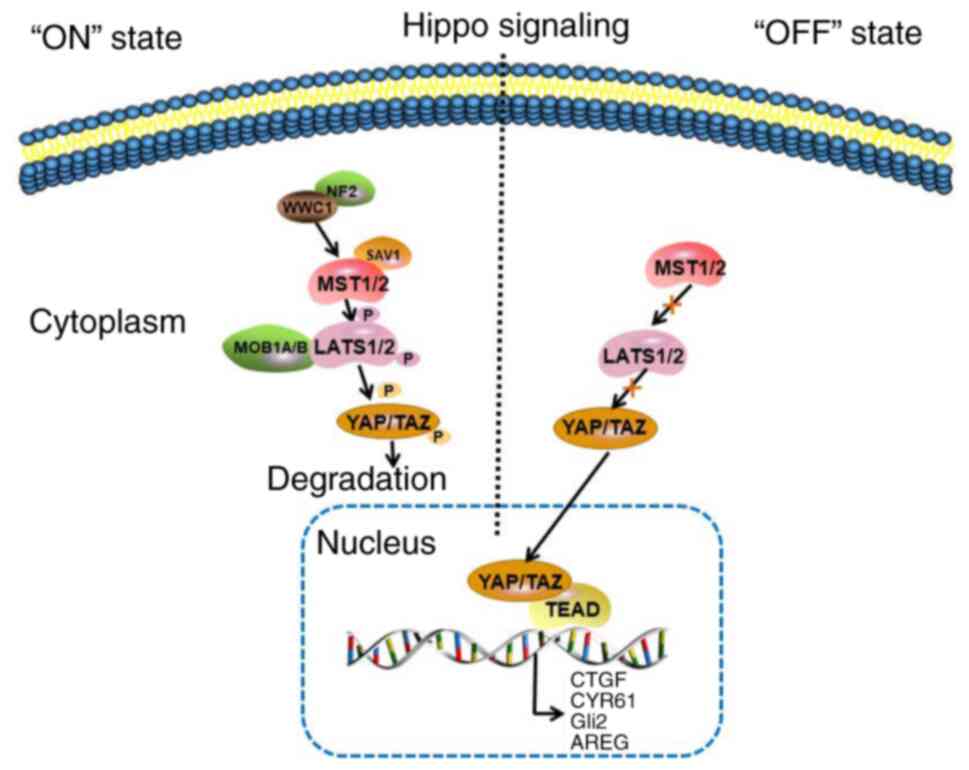 | Figure 1.Hippo signaling pathway. In response
to high cell density, NF2 and WWC1 activate the Hippo signaling
pathway by binding to LATS1/2 to facilitate their activation by
MST1/2. In turn, MST1/2 bind to the cofactor, SAV1, to form an
enzymatic complex that phosphorylates and activates the LATS1/2
kinases, and the MOB1A/B regulatory subunits of LAST1/2.
Subsequently, LATS1/2 phosphorylate the transcriptional
coactivators, YAP and TAZ, which in turn translocate in the
cytoplasm and get inactivated. In cancer, the Hippo signaling is
abnormally inactivated and LATS1/2 are not activated by MST1/2. The
YAP/TAZ complex translocates to the nucleus and binds with TEAD to
activate the transcription of its target genes, including CTGF,
CYR61, Gli2 and AREG. NF2, neurofibromatosis type 2; WWC1, WW And
C2 domain containing 1 gene; LATS1/2, large tumor suppressors 1 and
2; MST1/2, upstream sterile 20-like kinase 1 and 2; SAV1, Salvador
homologue 1; MOB1A/B, MOB kinase activator 1A and B; YAP,
Yes-associated protein; TAZ, transcriptional coactivator with
PDZ-binding motif; TEAD, transcriptional enhancer factor domain;
CTGF, connective tissue growth factor; CYR61, cysteine-rich
angiogenic inducer 61; Gli2, glioma-associated oncogene family zinc
finger 2; AREG, amphiregulin. |
The present review summarizes the current research
on the key targets of the Hippo signaling pathway in the treatment
of breast cancer. Furthermore, the latest advances on the role of
the TEAD family in breast cancer are highlighted, and the role of
TEAD4 as a potential target for therapeutic intervention in breast
cancer is discussed.
Aberrant expression of various components of
the Hippo signaling pathway in breast cancer
Increasing evidence suggests that the expression of
the upstream kinases involved in the Hippo signaling pathway is
consistent in different subtypes of breast cancer. For example,
MST1 and MST2 expression levels are downregulated in human breast
cancer tissues compared with normal breast tissues (9). In addition, upregulated MST1 expression
is associated with high overall survival and disease-free survival
rates, suggesting that MST1 functions as a tumor suppressor in
breast cancer (10). Phosphorylation
of MST1/2 (p-MST1/2) promotes the proliferation and migration of
MDA-MB-231 cells (11), while the
nuclear localization of p-MST1/2 exhibits tumor suppressive effects
in human epidermal growth factor receptor 2-positive and
triple-negative breast cancer (TNBC) (12). The loss of LATS2 is associated with
embryonic lethality in mouse embryos, and it was hypothesized that
the embryonic lethality was caused by abnormal cell proliferation,
mitotic defects and accumulated genomic instability; however,
LATS1-null mice survive. LATS1 and LATS2 are considered key
regulators of cell fate as they modulate the functions of tumor
suppressive effectors, which exert a highly conserved function
during evolution (13).
In addition to the close association between the
kinase chain of the Hippo signaling pathway and breast cancer
progression, the transcription coactivators, YAP and TAZ, serve
important roles in the occurrence and development of breast cancer
(8,14,15). The
activity of the YAP/TAZ complex is associated with high
histological grade and breast cancer stem cell traits, and can
induce multiple processes involved in tumor progression and
metastasis of breast cancer (14–16). In
luminal type A breast cancer, low YAP1 expression is associated
with a low recurrence-free survival rate (14); however, this expression trend is
reversed in TNBC (15). In addition,
the protein expression levels and activity of TAZ are elevated in
prospective breast cancer stem cells and in poorly differentiated
human tumors (16). Consistently,
overexpression of TAZ in differentiated breast cancer cells can
induce cell transformation and confer tumorigenicity and migratory
ability (17). Nuclear TAZ
expression is strongly associated with the TNBC subtype, which is
further strengthened in the basal-like subtype. Furthermore, 90% of
breast carcinomas with morphological EMT features are TAZ-positive
(18). The downstream effectors of
TEAD transcription factors include the oncogenes, CYR61, CTGF, MYC,
AREG and Gli2 (19–23).
Upstream kinases as therapeutic targets for
breast cancer
Gene and drug therapies can inhibit the development
of breast cancer by affecting the expression of the MST1/2
downstream kinases. It has been reported that Ras association
domain-containing protein 6 (RASSF6), discs large homolog 5 (DLG5),
microtubule affinity regulating kinase 4 (MARK4) and
meningioma-associated protein (MAC30)act on the Hippo signaling
pathway MST1/2 upstream kinases (24–26).
Previous studies have demonstrated that RASSF6 (24) and DLG5 (25) can promote the phosphorylation of
MST1/2 and LATS1, and inhibit YAP expression. Conversely, MARK4 can
bind and phosphorylate MST and SAV, and attenuate the formation of
the MST-SAV and LATS complexes in MDA-MB-231 cells (11). In addition, MAC30 inhibits MST1/2 and
LATS1/2 expression levels in MDA-MB-157 and MDA-MB-231 cells
(26). It has also been demonstrated
that neoadjuvant therapy activates the nuclear localization of
p-MST1/2, resulting in tumor suppressive effects (12).
LATS1/2 are considered important kinases involved in
the Hippo signaling pathway, thus, drug and gene therapies
targeting LATS1/2 have been extensively studied (27–29).
Phospholipase D (PLD), phosphatidic acid (PA) and vascular
endothelial growth factor (VEGF) can regulate the activity of
LATS1/2. PLD activates PA, which in turn directly interacts with
LATS to disrupt the formation of the LATS-MOB1 complex, thereby
promoting the carcinogenic activity of YAP (27). Another study demonstrated that
VEGF-NRP2 signaling can activate GTPase Rac1, which inhibits the
Hippo kinase, LATS, and activates TAZ and stem-like traits in
MDA-MB-231 cells (28). Conversely,
resveratrol phosphorylates and inactivates YAP by activating LATS1
in TNBC cells (29).
Kinases are regulated and activated by various
genes, and are also degraded in cells. Protein kinases are degraded
by E3 ubiquitin ligase and are deubiquitinated by deubiquitinating
enzymes to maintain protein stability (30–34). A
study revealed that breast cancer type 1 susceptibility protein
could turn ‘off’ the Hippo signaling pathway via ubiquitination of
NF2 (30). Furthermore, NF2 can
inhibit the interaction between the E3 ubiquitin ligase,
CRL4DCAF1 and LATS1; however, when derepressed,
CRL4DCAF1 can ubiquitylate and block LATS1/2 in the cell
nucleus (31). Similarly, it has
been reported that Itch, a HECT class E3 ubiquitin ligase, can form
a complex with LATS1 in MDA-MB-231 cells through the PPxY motifs of
LATS1 and the WW domains of Itch (32). In addition, a study demonstrated that
E3 ubiquitin-protein ligase seven in absentia homolog 2
destabilizes LATS2 in response to hypoxia (33). It has also been reported that
deubiquitinase ubiquitin specific peptidase 9 X-linked strongly
interacts with LATS kinase and enhances its stability (34).
Transcriptional coactivators YAP/TAZ as
therapeutic targets for breast cancer
Several studies have focused on the phosphorylation
of YAP/TAZ, and the interaction between YAP/TAZ and TEAD to
activate transcription of their target genes. It has been
demonstrated that leukemia inhibitory factor receptor can promote
YAP phosphorylation and its cytoplasmic retention, eventually
resulting in YAP inactivation (35).
Another study reported that the loss of FAT atypical cadherin 1
promotes the resistance of estrogen receptor (ER)-positive breast
cancer to cyclin-dependent kinase (CDK) 4/6 inhibitors through the
accumulation of YAP/TAZ transcription factors on the CDK6 promoter
(36). Similarly, the loss of
Ca2+-ATPase isoform 2 (37) and extracellular regulated protein
kinase 1 (38) can activate YAP/TAZ
expression. Τhe concept that regular exercise can prevent breast
cancer is based on scientific evidence suggesting that the serum
levels of catecholamines, including epinephrine and norepinephrine,
increase following exercise, thus resulting in YAP1 phosphorylation
and its cytoplasmic retention, eventually promoting its tumor
suppressive effects (39).
The ubiquitination and deubiquitination of YAP/TAZ
can also affect breast cancer development (40,41). A
study demonstrated that YAP1 is polyubiquitinated and degraded by
the E3 ubiquitin-protein ligase, RNF187, thus acquiring tumor
suppressive effects in TNBC (40).
In addition to ubiquitination and deubiquitination, SUMOylation is
also considered a common modified degradation method. The ubiquitin
thioesterase protein, OTUB2, was demonstrated to be poly-SUMOlated
on K233, thus allowing its binding with the YAP/TAZ complex,
resulting in the deubiquitination and activation of the complex
(41) (Table I).
 | Table I.Gene and drug therapies targeting the
Hippo signaling pathway. |
Table I.
Gene and drug therapies targeting the
Hippo signaling pathway.
| Gene and drug
therapies | Hippo target | Mechanisms | (Refs.) |
|---|
| RASSF6, DLG5 | MST1/2 and
LATS1 | Promotes the
phosphorylation of MST1/2 and LATS1 | (24,25) |
| MARK4 | MST and SAV | Weakens the
formation of the MST-SAV and LATS complex | (11) |
| MAC30 | MST1/2 and
LATS1/2 | Attenuates MST1/2
and LATS1/2 levels | (26) |
| Neoadjuvant
therapy | MST1/2 | Activates the
nuclear localization of pMST1/2 | (12) |
| VEGF | LATS1/2 | Activates GTPase
Rac1, thereby inhibiting LATS | (28) |
| Resveratrol | LATS1 | Activates
LATS1 | (29) |
| BRCA1 | NF2 | Contributes to the
inactivation of the Hippo signaling pathway through NF2
ubiquitination | (30) |
|
CRL4DCAF1, Itch | LATS1/2 | Ubiquitylates and
inhibits LATS1/2 | (31) |
| SIAH2 | LATS1/2 | Inactivates LATS1/2
via proteases | (33) |
| USP9X | LATS | Interacts with LATS
kinase and enhances the stability of LATS | (34) |
| LIFR | YAP | Promotes YAP
phosphorylation and cytoplasmic retention | (35) |
| FAT1 | YAP/TAZ | The loss of FAT1
promotes YAP/TAZ expression | (36) |
| SPCA2, ERK1 | YAP/TAZ | Activates YAP/TAZ
expression | (37,38) |
| EPI and NE | YAP1 | Through YAP1
phosphorylation and cytoplasmic retention | (39) |
| RNF187 | YAP1 | Polyubiquitinates
and degrades YAP1 | (40) |
| OTUB2 | YAP/TAZ | Deubiquitinates and
activates YAP/TAZ | (41) |
TEAD4 as a promising target for breast
cancer treatment
Although several studies have focused on
investigating the therapeutic potential of the regulation of YAP
phosphorylation and its nuclear localization (35,39), the
regulation and function of the TEAD family have not been
extensively studied. TEAD is an important transcription factor that
binds to YAP/TAZ in the nucleus. The mammalian encoding TEAD family
consists of four homologous genes, namely TEAD1-4. Following
binding of YAP to TEAD, it depends on the DNA binding domain of
TEAD to initiate transcription of the downstream target genes
(5,7,8). It has
been demonstrated that the oncogenes, interleukin 6 (42), angiomotin (43) and activator protein-1 (44) can promote the formation of the
YAP/TAZ-TEAD complex, and facilitate the proliferation and invasion
of breast cancer cells. Furthermore, the YAP/TAZ-TEAD complex
increases programmed cell death 1 ligand 1 promoter activity to
enhance the cell invasive ability and immune evasion in breast
cancer (45). Notably, it has been
reported that Ski activates the non-classical Hippo signaling
pathway by binding to the TAZ-TEAD complex and recruiting the
transcriptional repressor nuclear receptor corepressor 1 to the
TAZ-TEAD complex to inhibit TAZ transcription (46).
Previous studies have demonstrated that the TEAD
family, particularly TEAD4, is upregulated in various tumor
tissues, including gastric, colon, breast and prostate cancers, and
is associated with poor prognosis (47–49).
TEAD1 and TEAD4 play crucial roles in cancer by promoting tumor
growth (50–53). A study revealed that TEAD4 expression
is upregulated in breast cancer, thus serving as a prognostic
marker for breast cancer (50). In
MCF-7 cells, the YAP1-TEAD4 complex binds to the enhancer region of
ERα to activate the expression of the enhancer RNA transcription
markers by recruiting the mediator complex subunit 1, a component
of the enhancer activation mechanism. ERα is subsequently activated
by binding to the estrogen ligand 17b-estradiol (E2), thereby
regulating CTGF expression (51).
Notably, complementation experiments using wild-type TEAD4 and
YAP-binding mutant TEAD4, Y429H, demonstrated that both factors can
reverse EMT, increase the expression levels of the mesenchymal
marker vimentin, and enhance the cell invasive and migratory
abilities in a YAP-independent manner (52,53).
Thus, nuclear TEAD4 expression is considered a biomarker for colon
cancer progression and poor prognosis (52,53). It
has also been reported that the activation of glucocorticoids
promotes the interaction between glucocorticoid receptor and TEAD4,
eventually resulting in the formation of a complex. This complex is
subsequently recruited to the TEAD4 promoter to enhance its own
expression and promote breast cancer stem cell-like properties
(54). Thus, TEAD4 knockdown can
inhibit glucocorticoid-induced chemotherapy resistance in breast
cancer, suggesting new properties of TEAD4 in breast cancer
treatment (54). Analysis of the
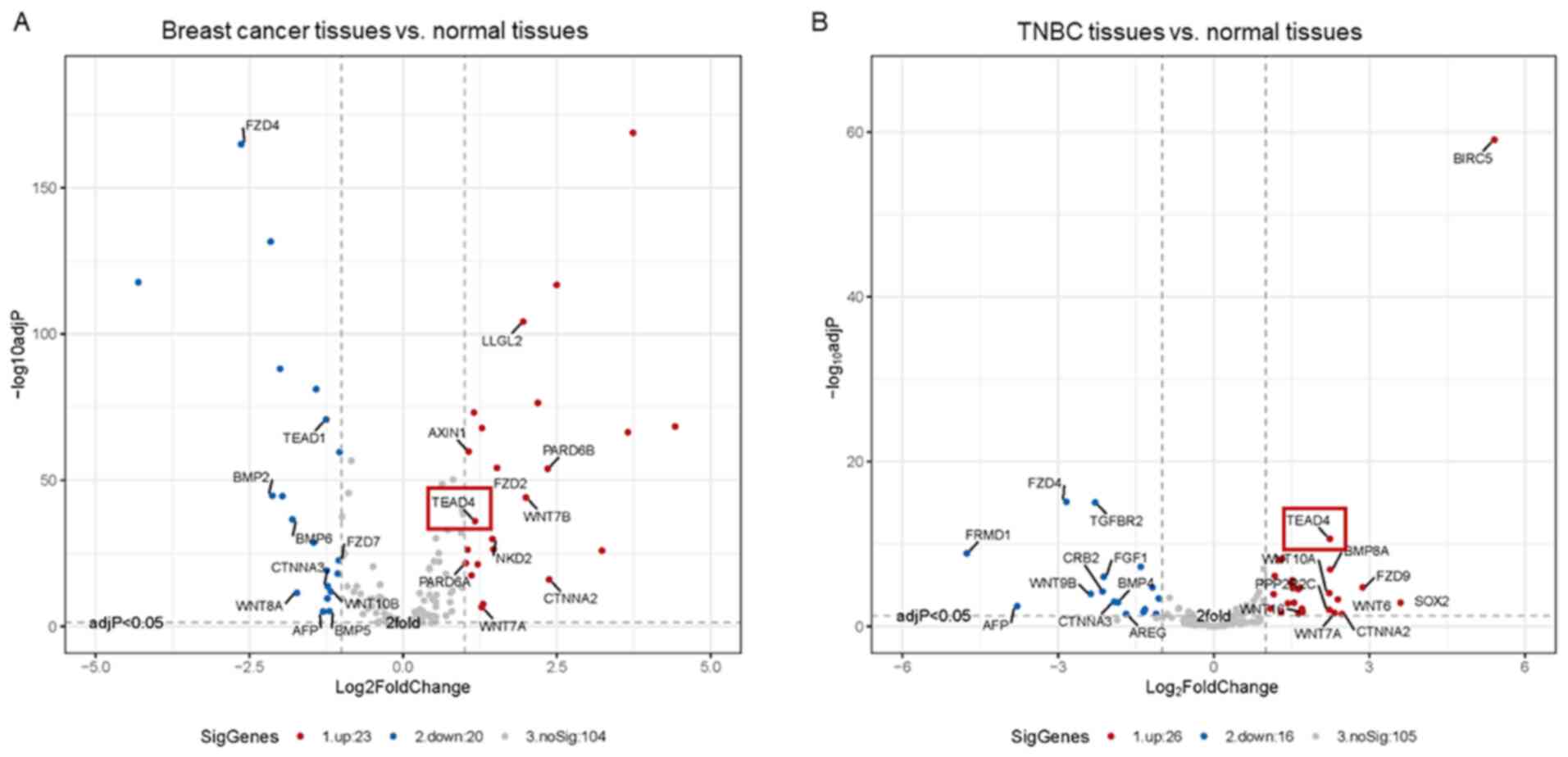
Hippo signaling pathway-related differentially expressed genes
between breast cancer tissues and normal tissues revealed that only
TEAD4 is overexpressed among all members of the TEAD family
(Fig. 2A). Notably, TEAD4 expression
is upregulated in TNBC tissues compared with the normal breast
tissues (Fig. 2Β). Taken together,
these results suggest that the use of agents targeting TEAD4 and
inhibiting the formation of the YAP/TAZ-TEADs complex, exert
promising results in in vitro experiments and animal models
of different types of cancer. Thus, TEAD4 may be used as a
potential target for the therapeutic intervention of tumors.
Conclusions
Breast cancer is the most common type of cancer and
the leading cause of cancer-associated mortality among women
worldwide. The treatment of breast cancer has gained significant
global attention. The Hippo signaling pathway includes a cascade of
signals that control tissue growth and organ size by attenuating
cell proliferation and promoting cell death. When the Hippo
signaling pathway is activated, the transcriptional activity of its
target genes, which exert a pivotal effect on carcinogenesis, is
repressed. Conversely, when the Hippo signaling pathway is
aberrantly inactivated, the transcriptional activity of its target
genes is enhanced, resulting in uncontrolled cell
proliferation.
With regards to the upstream kinase cascade in the
Hippo signaling pathway, the expression of oncogenes, tumor
suppressor genes, as well as the ubiquitinating and
deubiquitinating modifications can disrupt the function of these
cascades, eventually affecting the human body. Increasing evidence
suggest that the transcriptional coactivators, YAP and TAZ, serve
important role in the Hippo signaling pathway. Studies on the
regulation of the phosphorylation and nuclear localization of
YAP/TAZ confirm that these coactivators play a key role in the
development of breast cancer.
The TEAD family is the most important family of
transcription factors for the transcription of YAP/TAZ
coactivators. It is generally accepted that following YAP/TAZ
activation, the transcriptional activity of TEADs increases.
Notably, it has been reported that the function of TEADs is
independent of YAP/TAZ. TCGA database analysis revealed that among
the members of the TEAD family, TEAD4 expression is significantly
upregulated in breast cancer, particularly in TNBC tissues compared
with normal breast tissues. In conclusion, TEAD4 may act as a
potential target for the treatment of breast cancer by targeting
the Hippo signaling pathway.
Acknowledgements
Not applicable.
Funding
The present review was supported by the Natural
Science Foundation of China (grant nos. 81873052 and 81503301).
Availability of data and materials
The datasets generated and analyzed during the
current study are available in the TCGA repository https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga.
Authors' contributions
YW drafted and revised the initial manuscript. CH
conceived and modified the manuscript. YW, ML and JL analyzed the
data from TCGA database. YW and CH confirmed the authenticity of
all the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 2021.(Epub ahead of
print). View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gadag S, Sinha S, Nayak Y, Garg S and
Nayak UY: Combination therapy and nanoparticulate systems: Smart
approaches for the effective treatment of breast cancer.
Pharmaceutics. 12:5242020. View Article : Google Scholar
|
|
3
|
Zanconato F, Cordenonsi M and Piccolo S:
YAP/TAZ at the roots of cancer. Cancer Cell. 29:783–803. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yin F, Yu J, Zheng Y, Chen Q, Zhang N and
Pan D: Spatial organization of Hippo signaling at the plasma
membrane mediated by the tumor suppressor Merlin/NF2. Cell.
154:1342–1355. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Maugeri-Sacca M and De Maria R: The Hippo
pathway in normal development and cancer. PharmacolTher. 186:60–72.
2018.
|
|
6
|
Moroishi T, Hansen CG and Guan KL: The
emerging roles of YAP and TAZ in cancer. Nat Rev Cancer. 15:73–79.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Maugeri-Sacca M, Barba M, Pizzuti L, Vici
P, Di Lauro L, Dattilo R, Vitale I, Bartucci M, Mottolese M and De
Maria R: The Hippo transducers TAZ and YAP in breast cancer:
Oncogenic activities and clinical implications. Expert Rev Mol Med.
17:e142015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lamar JM, Stern P, Liu H, Schindler JW,
Jiang ZG and Hynes RO: The Hippo pathway target, YAP, promotes
metastasis through its TEAD-interaction domain. Proc Natl Acad Sci
USA. 109:E2441–E2450. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang Y, Li J, Gao Y, Luo Y, Luo H, Wang L,
Yi Y, Yuan Z and Jim Xiao ZX: Hippo kinases regulate cell junctions
to inhibit tumor metastasis in response to oxidative stress. Redox
Biol. 26:1012332019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lin XY, Cai FF, Wang MH, Pan X, Wang F,
Cai L, Cui RR, Chen S and Biskup E: Mammalian sterile 20-like
kinase 1 expression and its prognostic significance in patients
with breast cancer. Oncol Lett. 14:5457–5463. 2017.PubMed/NCBI
|
|
11
|
Heidary Arash E, Shiban A, Song S and
Attisano L: MARK4 inhibits Hippo signaling to promote proliferation
and migration of breast cancer cells. EMBO Rep. 18:420–436. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ercolani C, Di Benedetto A, Terrenato I,
Pizzuti L, Di Lauro L, Sergi D, Sperati F, Buglioni S, Ramieri MT,
Mentuccia L, et al: Expression of phosphorylated Hippo pathway
kinases (MST1/2 and LATS1/2) in HER2-positive and triple-negative
breast cancer patients treated with neoadjuvant therapy. Cancer
Biol Ther. 18:339–346. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Furth N and Aylon Y: The LATS1 and LATS2
tumor suppressors: Beyond the Hippo pathway. Cell Death Differ.
24:1488–1501. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lehn S, Tobin NP, Sims AH, Stal O,
Jirstrom K, Axelson H and Landberg G: Decreased expression of
Yes-associated protein is associated with outcome in the luminal a
breast cancer subgroup and with an impaired tamoxifen response. BMC
Cancer. 14:1192014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Maugeri-Sacca M and De Maria R: Hippo
pathway and breast cancer stem cells. Crit Rev Oncol Hematol.
99:115–122. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cordenonsi M, Zanconato F, Azzolin L,
Forcato M, Rosato A, Frasson C, Inui M, Montagner M, Parenti AR,
Poletti A, et al: The Hippo transducer TAZ confers cancer stem
cell-related traits on breast cancer cells. Cell. 147:759–772.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bartucci M, Dattilo R, Moriconi C,
Pagliuca A, Mottolese M, Federici G, Benedetto AD, Todaro M, Stassi
G, Sperati F, et al: TAZ is required for metastatic activity and
chemoresistance of breast cancer stem cells. Oncogene. 34:681–690.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Diaz-Martin J, Lopez-Garcia MA,
Romero-Perez L, Atienza-Amores MR, Pecero ML, Castilla MA, Biscuola
M, Santon A and Palacios J: Nuclear TAZ expression associates with
the triple-negative phenotype in breast cancer. EndocrRelat Cancer.
22:443–454. 2015. View Article : Google Scholar
|
|
19
|
Huang YT, Lan Q, Lorusso G, Duffey N and
Ruegg C: The matricellular protein CYR61 promotes breast cancer
lung metastasis by facilitating tumor cell extravasation and
suppressing anoikis. Oncotarget. 8:9200–9215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Di Benedetto A, Mottolese M, Sperati F,
Ercolani C, Di Lauro L, Pizzuti L, Vici P, Terrenato I, Sperduti I,
Shaaban AM, et al: The Hippo transducers TAZ/YAP and their target
CTGF in male breast cancer. Oncotarget. 7:43188–43198. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Elster D, Jaenicke LA, Eilers M and von
Eyss B: TEAD activity is restrained by MYC and stratifies human
breast cancer subtypes. Cell Cycle. 15:2551–2556. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xiang G, Liu F, Liu J, Meng Q, Li N and
Niu Y: Prognostic role of Amphiregulin and the correlation with
androgen receptor in invasive breast cancer. Pathol Res Pract.
215:1524142019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Niu J, Ma J, Guan X, Zhao X, Li P and
Zhang M: Correlation between doppler ultrasound blood flow
parameters and angiogenesis and proliferation activity in breast
cancer. Med Sci Monit. 25:7035–7041. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
He Z, Zhao TT, Jin F, Li JG, Xu YY, Dong
HT, Liu Q, Xing P, Zhu GL, Xu H and Miao ZF: Downregulation of
RASSF6 promotes breast cancer growth and chemoresistance through
regulation of Hippo signaling. BiochemBiophys Res Commun.
503:2340–2347. 2018. View Article : Google Scholar
|
|
25
|
Liu J, Li J, Li P, Wang Y, Liang Z, Jiang
Y, Li J, Feng C, Wang R, Chen H, et al: Loss of DLG5 promotes
breast cancer malignancy by inhibiting the Hippo signaling pathway.
Sci Rep. 7:421252017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song GQ and Zhao Y: MAC30 knockdown
involved in the activation of the Hippo signaling pathway in breast
cancer cells. Biol Chem. 399:1305–1311. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Han H, Qi R, Zhou JJ, Ta AP, Yang B,
Nakaoka HJ, Seo G, Guan KL, Luo R and Wang W: Regulation of the
Hippo pathway by phosphatidic Acid-Mediated Lipid-Protein
Interaction. Mol Cell. 72:328–340.e8. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Elaimy AL, Guru S, Chang C, Ou J, Amante
JJ, Zhu LJ, Goel HL and Mercurio AM: VEGF-neuropilin-2 signaling
promotes stem-like traits in breast cancer cells by TAZ-mediated
repression of the Rac GAP β2-chimaerin. Sci Signal.
11:eaao68972018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kim YN, Choe SR, Cho KH, Cho DY, Kang J,
Park CG and Lee HY: Resveratrol suppresses breast cancer cell
invasion by inactivating a RhoA/YAP signaling axis. Exp Mol Med.
49:e2962017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Verma S, Yeddula N, Soda Y, Zhu Q, Pao G,
Moresco J, Diedrich JK, Hong A, Plouffe S, Moroishi T, et al:
BRCA1/BARD1-dependent ubiquitination of NF2 regulates Hippo-YAP1
signaling. Proc Natl Acad Sci USA. 116:7363–7370. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li W, Cooper J, Zhou L, Yang C,
Erdjument-Bromage H, Zagzag D, Snuderl M, Ladanyi M, Hanemann CO,
Zhou P, et al: Merlin/NF2 loss-driven tumorigenesis linked to
CRL4(DCAF1)-mediated inhibition of the hippo pathway kinases Lats1
and 2 in the nucleus. Cancer Cell. 26:48–60. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ho KC, Zhou Z, She YM, Chun A, Cyr TD and
Yang X: Itch E3 ubiquitin ligase regulates large tumor suppressor 1
stability [corrected]. Proc Natl Acad Sci USA. 108:4870–4875. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ma B, Chen Y, Chen L, Cheng H, Mu C, Li J,
Gao R, Zhou C, Cao L, Liu J, et al: Hypoxia regulates Hippo
signalling through the SIAH2 ubiquitin E3 ligase. Nat Cell Biol.
17:95–103. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Toloczko A, Guo F, Yuen HF, Wen Q, Wood
SA, Ong YS, Chan PY, Shaik AA, Gunaratne J, Dunne MJ, et al:
Deubiquitinating enzyme USP9X suppresses tumor growth via LATS
kinase and core components of the Hippo pathway. Cancer Res.
77:4921–4933. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen D, Sun Y, Wei Y, Zhang P, Rezaeian
AH, Teruya-Feldstein J, Gupta S, Liang H, Lin HK, Hung MC and Ma L:
LIFR is a breast cancer metastasis suppressor upstream of the
Hippo-YAP pathway and a prognostic marker. Nat Med. 18:1511–1517.
2012. View
Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li Z, Razavi P, Li Q, Toy W, Liu B, Ping
C, Hsieh W, Sanchez-Vega F, Brown DN, Da Cruz Paula AF, et al: Loss
of the FAT1 tumor suppressor promotes resistance to CDK4/6
inhibitors via the hippo pathway. Cancer Cell. 34:893–905.e8. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dang DK, Makena MR, Llongueras JP, Prasad
H, Ko M, Bandral M and Rao R: A Ca2+-ATPase regulates
E-cadherin biogenesis and epithelial-mesenchymal transition in
breast cancer cells. Mol Cancer Res. 17:1735–1747. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yu S, Zhang M, Huang L, Ma Z, Gong X, Liu
W, Zhang J, Chen L, Yu Z, Zhao W, et al: ERK1 indicates good
prognosis and inhibits breast cancer progression by suppressing
YAP1 signaling. Aging (Albany NY). 11:12295–12314. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Dethlefsen C, Hansen LS, Lillelund C,
Andersen C, Gehl J, Christensen JF, Pedersen BK and Hojman P:
Exercise-induced catecholamines activate the Hippo tumor suppressor
pathway to reduce risks of breast cancer development. Cancer Res.
77:4894–4904. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Z, Kong Q, Su P, Duan M, Xue M, Li X,
Tang J, Gao Z, Wang B, Li Z, et al: Regulation of Hippo signaling
and triple negative breast cancer progression by an ubiquitin
ligase RNF187. Oncogenesis. 9:362020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang Z, Du J, Wang S, Shao L, Jin K, Li
F, Wei B, Ding W, Fu P, van Dam H, et al: OTUB2 promotes cancer
metastasis via Hippo-independent activation of YAP and TAZ. Mol
Cell. 73:7–21.e7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Hou L, Xie S, Li G, Xiong B, Gao Y, Zhao
X, Hu J, Deng S and Jiang J: IL-6 triggers the migration and
invasion of oestrogen receptor-negative breast cancer cells via
regulation of Hippo pathways. Basic Clin PharmacolToxicol.
123:549–557. 2018. View Article : Google Scholar
|
|
43
|
Lv M, Lv M, Chen L, Qin T, Zhang X, Liu P
and Yang J: Angiomotin promotes breast cancer cell proliferation
and invasion. Oncol Rep. 33:1938–1946. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zanconato F, Forcato M, Battilana G,
Azzolin L, Quaranta E, Bodega B, Rosato A, Bicciato S, Cordenonsi M
and Piccolo S: Genome-wide association between YAP/TAZ/TEAD and
AP-1 at enhancers drives oncogenic growth. Nat Cell Biol.
17:1218–1227. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Janse van Rensburg HJ, Azad T, Ling M, Hao
Y, Snetsinger B, Khanal P, Minassian LM, Graham CH, Rauh MJ and
Yang X: The Hippo pathway component TAZ promotes immune evasion in
human cancer through PD-L1. Cancer Res. 78:1457–1470. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Rashidian J, Le Scolan E, Ji X, Zhu Q,
Mulvihill MM, Nomura D and Luo K: Ski regulates Hippo and TAZ
signaling to suppress breast cancer progression. Sci Signal.
8:ra142015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Holden JK and Cunningham CN: Targeting the
Hippo pathway and cancer through the TEAD family of transcription
factors. Cancers (Basel). 10:812018. View Article : Google Scholar
|
|
48
|
Qi Y, Yu J, Han W, Fan X, Qian H, Wei H,
Tsai YH, Zhao J, Zhang W, Liu Q, et al: A splicing isoform of TEAD4
attenuates the Hippo-YAP signalling to inhibit tumour
proliferation. Nat Commun. 7:ncommss118402016. View Article : Google Scholar
|
|
49
|
Gibault F, Sturbaut M, Bailly F, Melnyk P
and Cotelle P: Targeting transcriptional enhanced associate domains
(TEADs). J Med Chem. 61:5057–5072. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhou Y, Huang T, Cheng AS, Yu J, Kang W
and To KF: The TEAD family and its oncogenic role in promoting
tumorigenesis. Int J Mol Sci. 17:1382016. View Article : Google Scholar
|
|
51
|
Zhu C, Li L, Zhang Z, Bi M, Wang H, Su W,
Hernandez K, Liu P, Chen J, Chen M, et al: A non-canonical role of
YAP/TEAD is required for activation of estrogen-regulated enhancers
in breast cancer. Mol Cell. 75:791–806.e8. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu Y, Wang G, Yang Y, Mei Z, Liang Z, Cui
A, Wu T, Liu CY and Cui L: Increased TEAD4 expression and nuclear
localization in colorectal cancer promote epithelial-mesenchymal
transition and metastasis in a YAP-independent manner. Oncogene.
35:2789–2800. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yu MH and Zhang W: TEAD1 enhances
proliferation via activating SP1 in colorectal cancer. Biomed
Pharmacother. 83:496–501. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
He L, Yuan L, Sun Y, Wang P, Zhang H, Feng
X, Wang Z, Zhang W, Yang C, Zeng YA, et al: Glucocorticoid receptor
signaling activates TEAD4 to promote breast cancer progression.
Cancer Res. 79:4399–4411. 2019. View Article : Google Scholar : PubMed/NCBI
|