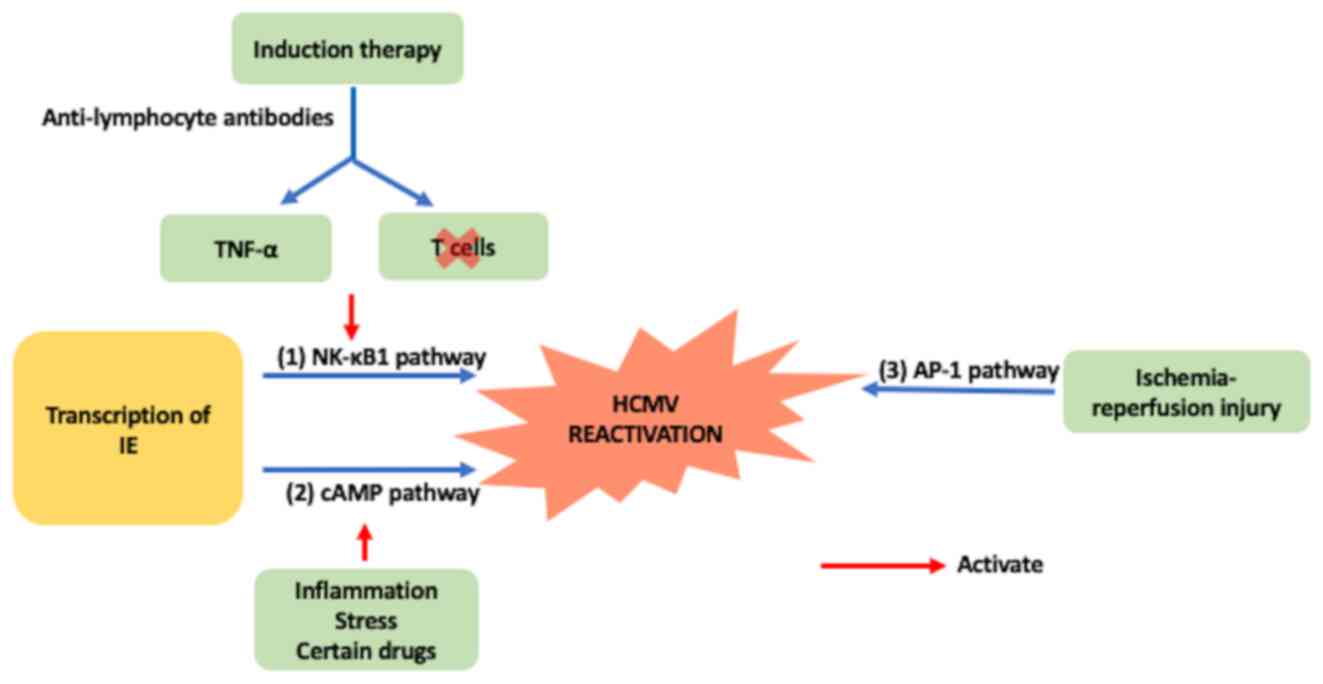
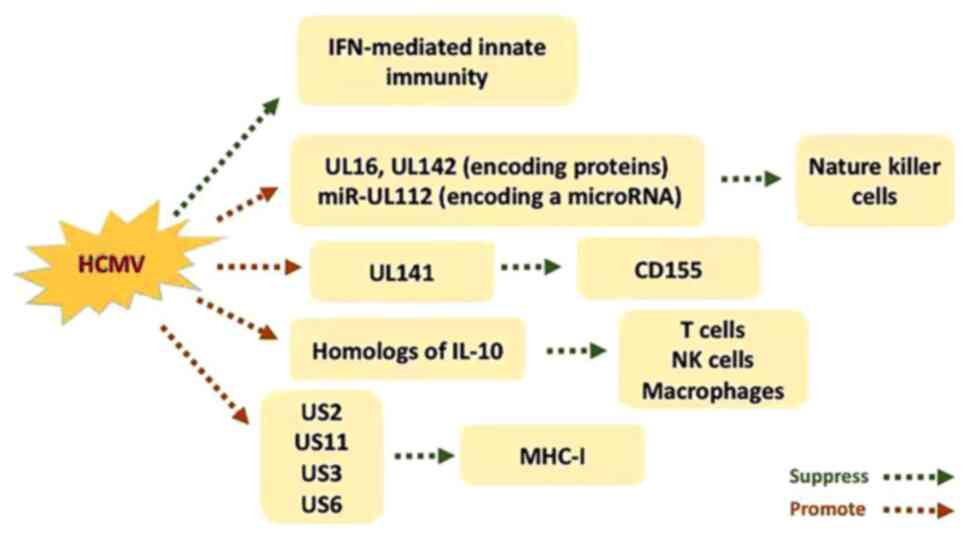
|
1
|
Ljungman P, de la Camara R, Robin C,
Crocchiolo R, Einsele H, Hill JA, Hubacek P, Navarro D, Cordonnier
C and Ward KN; 2017 European Conference on Infections in Leukaemia
group, : Guidelines for the management of cytomegalovirus infection
in patients with haematological malignancies and after stem cell
transplantation from the 2017 European Conference on Infections in
Leukaemia (ECIL 7). Lancet Infect Dis. 19:e260–e272. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gyurkocza B, Rezvani A and Storb RF:
Allogeneic hematopoietic cell transplantation: The state of the
art. Expert Rev Hematol. 3:285–299. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gratwohl A, Baldomero H, Aljurf M,
Pasquini MC, Bouzas LF, Yoshimi A, Szer J, Lipton J, Schwendener A,
Gratwohl M, et al: Hematopoietic stem cell transplantation: A
global perspective. JAMA. 303:1617–1624. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Borchers S, Luther S, Lips U, Hahn N,
Kontsendorn J, Stadler M, Buchholz S, Diedrich H, Eder M, Koehl U,
et al: Tetramer monitoring to assess risk factors for recurrent
cytomegalovirus reactivation and reconstitution of antiviral
immunity post allogeneic hematopoietic stem cell transplantation.
Transpl Infect Dis. 13:222–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Moss P and Rickinson A: Cellular
immunotherapy for viral infection after HSC transplantation. Nat
Rev Immunol. 5:9–20. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Haidar G and Singh N: Viral infections in
solid organ transplant recipients: Novel updates and a review of
the classics. Curr Opin Infect Dis. 30:579–588. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Enders G, Daiminger A, Lindemann L, Knotek
F, Bäder U, Exler S and Enders M: Cytomegalovirus (CMV)
seroprevalence in pregnant women, bone marrow donors and
adolescents in Germany, 1996–2010. Med Microbiol Immunol.
201:303–309. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ouédraogo AS, Yaméogo JT, Poda GEA,
Kientega Y and Ouédraogo Traore R: Prevalence of anti-CMV
antibodies in blood donors in Ouagadougou (Burkina Faso). Med Sante
Trop. 22:107–109. 2012.PubMed/NCBI
|
|
9
|
Revello MG, Vauloup-Fellous C,
Grangeot-Keros L, van Helden J, Dickstein Y, Lipkin I, Mühlbacher A
and Lazzarotto T: Clinical evaluation of new automated
cytomegalovirus IgM and IgG assays for the Elecsys(®)
analyser platform. Eur J Clin Microbiol Infect Dis. 31:3331–3339.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Strand KM, Odland ML, Iversen AC, Nordbø
SA, Vik T and Austgulen R: Cytomegalovirus antibody status at 17–18
weeks of gestation and pre-eclampsia: A case-control study of
pregnant women in Norway. BJOG. 119:1316–1323. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Amsler L, Verweij M and DeFilippis VR: The
tiers and dimensions of evasion of the type I interferon response
by human cytomegalovirus. J Mol Biol. 425:4857–4871. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2013. View Article : Google Scholar
|
|
13
|
Boeckh M and Ljungman P: How we treat
cytomegalovirus in hematopoietic cell transplant recipients. Blood.
113:5711–5719. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bankier AT, Beck S, Bohni R, Brown CM,
Cerny R, Chee MS, Hutchison CA III, Kouzarides T, Martignetti JA,
Preddie E, et al: The DNA sequence of the human cytomegalovirus
genome. DNA Seq. 2:1–12. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Murphy E and Shenk T: Human
cytomegalovirus genome. Curr Top Microbiol Immunol. 325:1–19.
2008.PubMed/NCBI
|
|
16
|
Scherer M, Schilling EM and Stamminger T:
The human CMV IE1 protein: An offender of PML NUCLEAR BOdies. Adv
Anat Embryol Cell Biol. 223:77–94. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fox HL, Dembowski JA and DeLuca NA: A
herpesviral immediate early protein promotes transcription
elongation of viral transcripts. mBio. 8:e00745–17. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gardner TJ and Tortorella D: Virion
glycoprotein-mediated immune evasion by human cytomegalovirus: A
sticky virus makes a slick getaway. Microbiol Mol Biol Rev.
80:663–677. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sezgin E, An P and Winkler CA: Host
genetics of cytomegalovirus pathogenesis. Front Genet. 10:6162019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheung AK, Gottlieb DJ, Plachter B,
Pepperl-Klindworth S, Avdic S, Cunningham AL, Abendroth A and
Slobedman B: The role of the human cytomegalovirus UL111A gene in
down-regulating CD4+ T-cell recognition of latently
infected cells: Implications for virus elimination during latency.
Blood. 114:4128–4137. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Slavuljica I, Busche A, Babić M, Mitrović
M, Gašparović I, Cekinović D, Markova Car E, Pernjak Pugel E,
Ciković A, Lisnić VJ, et al: Recombinant mouse cytomegalovirus
expressing a ligand for the NKG2D receptor is attenuated and has
improved vaccine properties. J Clin Invest. 120:4532–4545. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Griffiths PD: Burden of disease associated
with human cytomegalovirus and prospects for elimination by
universal immunisation. Lancet Infect Dis. 12:790–798. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wilkinson GW, Tomasec P, Stanton RJ,
Armstrong M, Prod'homme V, Aicheler R, McSharry BP, Rickards CR,
Cochrane D, Llewellyn-Lacey S, et al: Modulation of natural killer
cells by human cytomegalovirus. J Clin Virol. 41:206–212. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fang D and Zhu J: Molecular switches for
regulating the differentiation of inflammatory and IL-10-producing
anti-inflammatory T-helper cells. Cell Mol Life Sci. 77:289–303.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou L, Chong MM and Littman DR:
Plasticity of CD4+ T cell lineage differentiation.
Immunity. 30:646–655. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Opal SM and DePalo VA: Anti-inflammatory
cytokines. Chest. 117:1162–1172. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
McSharry BP, Avdic S and Slobedman B:
Human cytomegalovirus encoded homologs of cytokines, chemokines and
their receptors: Roles in immunomodulation. Viruses. 4:2448–2470.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Barel MT, Ressing M, Pizzato N, van
Leeuwen D, Le Bouteiller P, Lenfant F and Wiertz EJ: Human
cytomegalovirus-encoded US2 differentially affects surface
expression of MHC class I locus products and targets
membrane-bound, but not soluble HLA-G1 for degradation. J Immunol.
171:6757–6765. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Khan N, Bruton R, Taylor GS, Cobbold M,
Jones TR, Rickinson AB and Moss PA: Identification of
cytomegalovirus-specific cytotoxic T lymphocytes in vitro is
greatly enhanced by the use of recombinant virus lacking the US2 to
US11 region or modified vaccinia virus Ankara expressing individual
viral genes. J Virol. 79:2869–2879. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ciáurriz M, Zabalza A, Beloki L, Mansilla
C, Pérez-Valderrama E, Lachén M, Bandrés E, Olavarría E and Ramírez
N: The immune response to cytomegalovirus in allogeneic
hematopoietic stem cell transplant recipients. Cell Mol Life Sci.
72:4049–4062. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Minton K: Viral immunity: How CMV bypasses
immune memory. Nat Rev Immunol. 10:2882010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Reddehase MJ and Lemmermann NAW: Cellular
reservoirs of latent cytomegaloviruses. Med Microbiol Immunol.
208:391–403. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sinzger C, Digel M and Jahn G:
Cytomegalovirus cell tropism. Curr Top Microbiol Immunol.
325:63–83. 2008.PubMed/NCBI
|
|
34
|
Higdon LE, Trofe-Clark J, Liu S, Margulies
KB, Sahoo MK, Blumberg E, Pinsky BA and Maltzman JS:
Cytomegalovirus-responsive CD8+ T cells expand after
solid organ transplantation in the absence of CMV disease. Am J
Transplant. 17:2045–2054. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Albiero E, Amati E, Baumeister E, Einsele
H, Grigoleit GU and Rodeghiero F: Heterogeneity of Specific
CD4+ and CD8+ T cells stimulated by CMV pp65
and IE1 antigens. J Immunother. 39:329–342. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Styczynski J: Who is the patient at risk
of CMV recurrence: A review of the current scientific evidence with
a focus on hematopoietic cell transplantation. Infect Dis Ther.
7:1–16. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cho SY, Lee DG and Kim HJ: Cytomegalovirus
infections after hematopoietic stem cell transplantation: Current
status and future immunotherapy. Int J Mol Sci. 20:26662019.
View Article : Google Scholar
|
|
38
|
Kim SJ, Varghese TK, Zhang Z, Zhao LC,
Thomas G, Hummel M and Abecassis M: Renal ischemia/reperfusion
injury activates the enhancer domain of the human cytomegalovirus
major immediate early promoter. Am J Transplant. 5:1606–1613. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kurata K, Yakushijin K, Mizuno I, Gomyo H,
Okamura A, Ichikawa H, Sakai R, Mizutani Y, Kakiuchi S, Miyata Y,
et al: Early lymphocyte recovery predicts clinical outcome after
HSCT with mycophenolate mofetil prophylaxis in the Japanese
population. Int J Hematol. 108:58–65. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Theobald SJ, Khailaie S, Meyer-Hermann M,
Volk V, Olbrich H, Danisch S, Gerasch L, Schneider A, Sinzger C,
Schaudien D, et al: Signatures of T and B cell development,
functional responses and PD-1 upregulation after HCMV latent
infections and reactivations in Nod.Rag.Gamma mice humanized with
cord blood CD34+ cells. Front Immunol. 9:27342018.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Camargo JF, Wieder ED, Kimble E, Benjamin
CL, Kolonias DS, Kwon D, Chen XS and Komanduri KV: Deep functional
immunophenotyping predicts risk of cytomegalovirus reactivation
after hematopoietic cell transplantation. Blood. 133:867–877. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
van Gent R, Metselaar HJ and Kwekkeboom J:
Immunomodulation by hyperimmunoglobulins after solid organ
transplantation: Beyond prevention of viral infection. Transplant
Rev (Orlando). 31:78–86. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Prösch S, Wendt CE, Reinke P, Priemer C,
Oppert M, Krüger DH, Volk HD and Döcke WD: A novel link between
stress and human cytomegalovirus (HCMV) infection: Sympathetic
hyperactivity stimulates HCMV activation. Virology. 272:357–365.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Atabani SF, Smith C, Atkinson C, Aldridge
RW, Rodriguez-Perálvarez M, Rolando N, Harber M, Jones G, O'Riordan
A, Burroughs AK, et al: Cytomegalovirus replication kinetics in
solid organ transplant recipients managed by preemptive therapy. Am
J Transplant. 12:2457–2464. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yong MK, Lewin SR and Manuel O: Immune
monitoring for CMV in transplantation. Curr Infect Dis Rep.
20:42018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Blyth E, Withers B, Clancy L and Gottlieb
D: CMV-specific immune reconstitution following allogeneic stem
cell transplantation. Virulence. 7:967–980. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Król L, Stuchlý J, Hubáček P, Keslová P,
Sedláček P, Starý J, Hrušák O and Kalina T: Signature profiles of
CMV-specific T-cells in patients with CMV reactivation after
hematopoietic SCT. Bone Marrow Transplant. 46:1089–1098. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lilleri D, Gerna G, Zelini P, Chiesa A,
Rognoni V, Mastronuzzi A, Giorgiani G, Zecca M and Locatelli F:
Monitoring of human cytomegalovirus and virus-specific T-cell
response in young patients receiving allogeneic hematopoietic stem
cell transplantation. PLoS One. 7:e416482012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Reusser P, Riddell SR, Meyers JD and
Greenberg PD: Cytotoxic T-lymphocyte response to cytomegalovirus
after human allogeneic bone marrow transplantation: Pattern of
recovery and correlation with cytomegalovirus infection and
disease. Blood. 78:1373–1380. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ogonek J, Varanasi P, Luther S, Schweier
P, Kühnau W, Göhring G, Dammann E, Stadler M, Ganser A, Borchers S,
et al: Possible impact of cytomegalovirus-specific CD8+
T cells on immune reconstitution and conversion to complete donor
chimerism after allogeneic stem cell transplantation. Biol Blood
Marrow Transplant. 23:1046–1053. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hill GR, Kuns RD, Raffelt NC, Don AL,
Olver SD, Markey KA, Wilson YA, Tocker J, Alexander WS, Clouston
AD, et al: SOCS3 regulates graft-versus-host disease. Blood.
116:287–296. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Alexander WS: Suppressors of cytokine
signalling (SOCS) in the immune system. Nat Rev Immunol. 2:410–416.
2002. View
Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lee TH, Lee JY, Park S, Shin SH, Yahng SA,
Yoon JH, Lee SE, Cho BS, Kim YJ, Lee S, et al: Expression of SOCS1
and SOCS3 genes in human graft-versus-host disease after allogeneic
hematopoietic stem cell transplantation. Blood Res. 48:16–23. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Shin SH, Lee JY, Lee TH, Park SH, Yahng
SA, Yoon JH, Lee SE, Cho BS, Lee DG, Kim YJ, et al: SOCS1 and SOCS3
are expressed in mononuclear cells in human cytomegalovirus viremia
after allogeneic hematopoietic stem cell transplantation. Blood
Res. 50:40–45. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Meyers JD, Flournoy N and Thomas ED: Risk
factors for cytomegalovirus infection after human marrow
transplantation. J Infect Dis. 153:478–488. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kuo HT, Ye X, Sampaio MS, Reddy P and
Bunnapradist S: Cytomegalovirus serostatus pairing and deceased
donor kidney transplant outcomes in adult recipients with antiviral
prophylaxis. Transplantation. 90:1091–1098. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Nichols WG, Corey L, Gooley T, Davis C and
Boeckh M: High risk of death due to bacterial and fungal infection
among cytomegalovirus (CMV)-seronegative recipients of stem cell
transplants from seropositive donors: Evidence for indirect effects
of primary CMV infection. J Infect Dis. 185:273–282. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Schnitzler MA, Woodward RS, Brennan DC,
Spitznagel EL, Dunagan WC and Bailey TC: Impact of cytomegalovirus
serology on graft survival in living related kidney
transplantation: Implications for donor selection. Surgery.
121:563–568. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gerstenkorn C, Balupuri S, Mohamed MA,
Manas DM, Ali S, Kirby J and Talbot D: The impact of
cytomegalovirus serology for 7-year graft survival in cadaveric
kidney transplantation-the Newcastle experience. Transpl Int. 13
(Suppl 1):S372–S374. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Harvala H, Stewart C, Muller K, Burns S,
Marson L, MacGilchrist A and Johannessen I: High risk of
cytomegalovirus infection following solid organ transplantation
despite prophylactic therapy. J Med Virol. 85:893–898. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Amouzegar A, Dey BR and Spitzer TR:
Peripheral blood or bone marrow stem cells? Practical
considerations in hematopoietic stem cell transplantation. Transfus
Med Rev. 33:43–50. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Trenschel R, Ross S, Hüsing J, Ottinger H,
Elmaagacli A, Roggendorf M, Schaefer UW and Runde V: Reduced risk
of persisting cytomegalovirus pp65 antigenemia and cytomegalovirus
interstitial pneumonia following allogeneic PBSCT. Bone Marrow
Transplant. 25:665–672. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Uppuluri R, Subburaj D, Jayaraman D,
Swaminathan VV, Mullanfiroze K, Vaidhyanathan L and Raj R:
Cytomegalovirus reactivation posthematopoietic stem cell
transplantation (HSCT) and type of graft: A step toward
rationalizing CMV testing and positively impacting the economics of
HSCT in developing countries. Pediatr Blood Cancer. 64:2017.doi:
10.1002/pbc.26639. View Article : Google Scholar
|
|
64
|
Boeckh M, Gooley TA, Myerson D, Cunningham
T, Schoch G and Bowden RA: Cytomegalovirus pp65 antigenemia-guided
early treatment with ganciclovir versus ganciclovir at engraftment
after allogeneic marrow transplantation: A randomized double-blind
study. Blood. 88:4063–4071. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Gatault P, Al-Hajj S, Noble J, Chevallier
E, Piollet M, Forconi C, Gaudy-Graffin C, Thibault G,
Miquelestorena-Standley E, Halimi JM, et al: CMV-infected kidney
grafts drive the expansion of blood-borne CMV-specific T cells
restricted by shared class I HLA molecules via presentation on
donor cells. Am J Transplant. 18:1904–1913. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sylwester AW, Mitchell BL, Edgar JB,
Taormina C, Pelte C, Ruchti F, Sleath PR, Grabstein KH, Hosken NA,
Kern F, et al: Broadly targeted human cytomegalovirus-specific
CD4+ and CD8+ T cells dominate the memory
compartments of exposed subjects. J Exp Med. 202:673–685. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Essa S, Pacsa A, Said T, Nampoory MR,
Raghupathy R, Johny KV, Al-Nakib W and Al-Mosawy M: Is combined
pretransplantation seropositivity of kidney transplant recipients
for cytomegalovirus antigens (pp150 and pp28) a predictor for
protection against infection? Med Princ Pract. 17:66–70. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wills MR, Okecha G, Weekes MP, Gandhi MK,
Sissons PJ and Carmichael AJ: Identification of naive or
antigen-experienced human CD8+ T cells by expression of
costimulation and chemokine receptors: Analysis of the human
cytomegalovirus-specific CD8+ T cell response. J
Immunol. 168:5455–5464. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Maecker HT and Maino VC: Analyzing T-cell
responses to cytomegalovirus by cytokine flow cytometry. Hum
Immunol. 65:493–499. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Baumann NS, Welten SPM, Torti N, Pallmer
K, Borsa M, Barnstorf I, Oduro JD, Cicin-Sain L and Oxenius A:
Early primed KLRG1-CMV-specific T cells determine the size of the
inflationary T cell pool. PLoS Pathog. 15:e10077852019. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Scheinberg P, Melenhorst JJ, Brenchley JM,
Hill BJ, Hensel NF, Chattopadhyay PK, Roederer M, Picker LJ, Price
DA, Barrett AJ and Douek DC: The transfer of adaptive immunity to
CMV during hematopoietic stem cell transplantation is dependent on
the specificity and phenotype of CMV-specific T cells in the donor.
Blood. 114:5071–5080. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Liu J, Chang YJ, Yan CH, Xu LP, Jiang ZF,
Zhang XH, Liu KY and Huang XJ: Poor CMV-specific CD8+ T central
memory subset recovery at early stage post-HSCT associates with
refractory and recurrent CMV reactivation. J Infect. 73:261–270.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
LaMattina JC, Mezrich JD, Hofmann RM,
Foley DP, D'Alessandro AM, Sollinger HW and Pirsch JD: Alemtuzumab
as compared to alternative contemporary induction regimens. Transpl
Int. 25:518–526. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wang QY, Dong YJ, Liang ZY, Yin Y, Liu W,
Xu WL, Sun YH, Han N, Li Y and Ren HY: Clinical analysis for
patients with AML treated after Allo-HSCT. Zhongguo Shi Yan Xue Ye
Xue Za Zhi. 28:1105–1114. 2020.(In Chinese). PubMed/NCBI
|
|
75
|
Kanter J, Pallardó L, Gavela E, Escudero
V, Beltrán S, Morales A, Avila A and Crespo JF: Cytomegalovirus
infection renal transplant recipients: Risk factors and outcome.
Transplant Proc. 41:2156–2158. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Hosseini-Moghaddam SM, Rotstein C and
Husain S: Effects of the intensity of immunosuppressive therapy on
outcome of treatment for CMV disease in organ transplant
recipients. Am J Transplant. 11:4072011. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Kobashigawa JA, Miller LW, Russell SD,
Ewald GA, Zucker MJ, Goldberg LR, Eisen HJ, Salm K, Tolzman D, Gao
J, et al: Tacrolimus with mycophenolate mofetil (MMF) or sirolimus
vs. cyclosporine with MMF in cardiac transplant patients: 1-year
report. Am J Transplant. 6:1377–1386. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Viganò M, Dengler T, Mattei MF, Poncelet
A, Vanhaecke J, Vermes E, Kleinloog R, Li Y, Gezahegen Y and
Delgado JF; RAD A2411 Study Investigators, : Lower incidence of
cytomegalovirus infection with everolimus versus mycophenolate
mofetil in de novo cardiac transplant recipients: A randomized,
multicenter study. Transpl Infect Dis. 12:23–30. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Axelrod D, Leventhal JR, Gallon LG, Parker
MA and Kaufman DB: Reduction of CMV disease with steroid-free
immunosuppresssion in simultaneous pancreas-kidney transplant
recipients. Am J Transplant. 5:1423–1429. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Ekberg H, Bernasconi C, Nöldeke J, Yussim
A, Mjörnstedt L, Erken U, Ketteler M and Navrátil P: Cyclosporine,
tacrolimus and sirolimus retain their distinct toxicity profiles
despite low doses in the Symphony study. Nephrol Dial Transplant.
25:2004–2010. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Muffly L, Sheehan K, Armstrong R, Jensen
K, Tate K, Rezvani AR, Miklos D, Arai S, Shizuru J, Johnston L, et
al: Infusion of donor-derived CD8+ memory T cells for
relapse following allogeneic hematopoietic cell transplantation.
Blood Adv. 2:681–690. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Teijaro JR: Cytokine storms in infectious
diseases. Semin Immunopathol. 39:501–503. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Miller W, Flynn P, McCullough J, Balfour
HH Jr, Goldman A, Haake R, McGlave P, Ramsay N and Kersey J:
Cytomegalovirus infection after bone marrow transplantation: An
association with acute graft-v-host disease. Blood. 67:1162–1167.
1986. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Stern A and Papanicolaou GA: CMV
prevention and treatment in transplantation: What's New in 2019.
Curr Infect Dis Rep. 21:452019. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Murata M, Ikegame K, Morishita Y, Ogawa H,
Kaida K, Nakamae H, Ikeda T, Nishida T, Inoue M, Eto T, et al:
Low-dose thymoglobulin as second-line treatment for
steroid-resistant acute GvHD: An analysis of the JSHCT. Bone Marrow
Transplant. 52:252–257. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Lin HC, Han SM, Hwang WL, Chou CW, Chang
KH, Shi ZY and Jerry Teng CL: Cytomegalovirus infection and
treatment in allogeneic hematopoietic stem cell transplantation: A
retrospective study from a single institution in an endemic area.
Turk J Haematol. 34:159–166. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Xue H, Hu Y, Feng S, Liu Z and Gao F: Risk
factor analysis for cytomegalovirus infection after peripheral
blood allogeneic hematopoietic stem cell transplantation. J China
Med Univ. 48:417–420. 2019.
|
|
88
|
Fuji S, Einsele H and Kapp M:
Cytomegalovirus disease in hematopoietic stem cell transplant
patients: Current and future therapeutic options. Curr Opin Infect
Dis. 30:372–376. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Park SY, Lee SO, Choi SH, Kim YS, Woo JH,
Baek S, Sung H, Kim MN, Kim DY, Lee JH, et al: Efficacy and safety
of low-dose ganciclovir preemptive therapy in allogeneic
haematopoietic stem cell transplant recipients compared with
conventional-dose ganciclovir: A prospective observational study. J
Antimicrob Chemother. 67:1486–1492. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Zhu MX, Wan WL, Li HS, Wang J, Wang YF, Hu
K and Ke XY: Early immune reconstitution after hematopoietic stem
cell transplantation. Beijing Da Xue Xue Bao Yi Xue Ban.
48:515–522. 2016.(In Chinese). PubMed/NCBI
|
|
91
|
Yong MK, Cameron PU, Slavin M, Morrissey
CO, Bergin K, Spencer A, Ritchie D, Cheng AC, Samri A, Carcelain G,
et al: Identifying cytomegalovirus complications using the
quantiferon-CMV assay after allogeneic hematopoietic stem cell
transplantation. J Infect Dis. 215:1684–1694. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wagner-Drouet E, Teschner D, Wolschke C,
Janson D, Schäfer-Eckart K, Gärtner J, Mielke S, Schreder M, Kobbe
G, Kondakci M, et al: Standardized monitoring of
cytomegalovirus-specific immunity can improve risk stratification
of recurrent cytomegalovirus reactivation after hematopoietic stem
cell transplantation. Haematologica. 106:363–374. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
El Haddad L, Ariza-Heredia E, Shah DP,
Jiang Y, Blanchard T, Ghantoji SS, El Chaer F, El-Haddad D, Prayag
A, Nesher L, et al: The ability of a cytomegalovirus ELISPOT assay
to predict outcome of low-level CMV reactivation in hematopoietic
cell transplant recipients. J Infect Dis. 219:898–907. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Krawczyk A, Ackermann J, Goitowski B,
Trenschel R, Ditschkowski M, Timm J, Ottinger H, Beelen DW, Grüner
N and Fiedler M: Assessing the risk of CMV reactivation and
reconstitution of antiviral immune response post bone marrow
transplantation by the QuantiFERON-CMV-assay and real time PCR. J
Clin Virol. 99-100:61–66. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Cao K, Marin D, Sekine T, Rondon G, Zhao
W, Smith NT, Daher M, Wang Q, Li L, Saliba RM, et al: Donor NKG2C
copy number: An independent predictor for CMV reactivation after
double cord blood transplantation. Front Immunol. 9:24442018.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Albert E, Solano C, Giménez E, Focosi D,
Pérez A, Macera L, Piñana JL, Boluda JCH, Maggi F and Navarro D:
The kinetics of torque teno virus plasma DNA load shortly after
engraftment predicts the risk of high-level CMV DNAemia in
allogeneic hematopoietic stem cell transplant recipients. Bone
Marrow Transplant. 53:180–187. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Takahashi R, Nakatsukasa H, Shiozawa S and
Yoshimura A: SOCS1 is a key molecule that prevents regulatory T
cell plasticity under inflammatory conditions. J Immunol.
199:149–158. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Lodding IP, Schultz HH, Jensen JU, Kirkby
N, Perch M, Andersen C, Lundgren JD and Iversen M: Cytomegalovirus
viral load in bronchoalveolar lavage to diagnose lung transplant
associated CMV pneumonia. Transplantation. 102:326–332. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Bhatia J, Shah BV, Mehta AP, Deshmukh M,
Sirsat RA and Rodrigues C: Comparing serology, antigenemia assay
and polymerase chain reaction for the diagnosis of cytomegalovirus
infection in renal transplant patients. J Assoc Physicians India.
52:297–300. 2004.PubMed/NCBI
|
|
100
|
Griscelli F, Barrois M, Chauvin S, Lastere
S, Bellet D and Bourhis JH: Quantification of human cytomegalovirus
DNA in bone marrow transplant recipients by real-time PCR. J Clin
Microbiol. 39:4362–4369. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Drew WL: Laboratory diagnosis of
cytomegalovirus infection and disease in immunocompromised
patients. Curr Opin Infect Dis. 20:408–411. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Pollack M, Heugel J, Xie H, Leisenring W,
Storek J, Young JA, Kukreja M, Gress R, Tomblyn M and Boeckh M: An
international comparison of current strategies to prevent
herpesvirus and fungal infections in hematopoietic cell transplant
recipients. Biol Blood Marrow Transplant. 17:664–673. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Malouli D, Hansen SG, Nakayasu ES,
Marshall EE, Hughes CM, Ventura AB, Gilbride RM, Lewis MS, Xu G,
Kreklywich C, et al: Cytomegalovirus pp65 limits dissemination but
is dispensable for persistence. J Clin Invest. 124:1928–1944. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Lee H, Park KH, Ryu JH, Choi AR, Yu JH,
Lim J, Han K, Kim SI, Yang CW, Chung BH and Oh EJ: Cytomegalovirus
(CMV) immune monitoring with ELISPOT and QuantiFERON-CMV assay in
seropositive kidney transplant recipients. PLoS One.
12:e01894882017. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Lisboa LF, Asberg A, Kumar D, Pang X,
Hartmann A, Preiksaitis JK, Pescovitz MD, Rollag H, Jardine AG and
Humar A: The clinical utility of whole blood versus plasma
cytomegalovirus viral load assays for monitoring therapeutic
response. Transplantation. 91:231–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Gerna G, Lilleri D, Caldera D, Furione M,
Zenone Bragotti L and Alessandrino EP: Validation of a DNAemia
cutoff for preemptive therapy of cytomegalovirus infection in adult
hematopoietic stem cell transplant recipients. Bone Marrow
Transplant. 41:873–879. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Boeckh M, Stevens-Ayers T, Travi G, Huang
M-L, Cheng GS, Xie H, Leisenring W, Erard V, Seo S, Kimball L, et
al: Cytomegalovirus (CMV) DNA quantitation in bronchoalveolar
lavage fluid from hematopoietic stem cell transplant recipients
with CMV pneumonia. J Infect Dis. 215:1514–1522. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Beam E, Germer JJ, Lahr B, Yao JDC, Limper
AH, Binnicker MJ and Razonable RR: Cytomegalovirus (CMV) DNA
quantification in bronchoalveolar lavage fluid of immunocompromised
patients with CMV pneumonia. Clin Transplant. 32:2018.doi:
10.1111/ctr.13149. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Hebart H, Rudolph T, Loeffler J,
Middeldorp J, Ljubicic T, Jahn G and Einsele H: Evaluation of the
NucliSens CMV pp67 assay for detection and monitoring of human
cytomegalovirus infection after allogeneic stem cell
transplantation. Bone Marrow Transplant. 30:181–187. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Rea F, Potena L, Yonan N, Wagner F and
Calabrese F: Cytomegalovirus hyper immunoglobulin for CMV
prophylaxis in thoracic transplantation. Transplantation. 100
(Suppl 3):S19–S26. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Kotton CN: CMV: Prevention, diagnosis and
therapy. Am J Transplant. 13 (Suppl 3):S24–S40. 2013. View Article : Google Scholar
|
|
112
|
Griffiths PD: An explanation for
posttransplant late-onset disease associated with CMV prophylaxis.
Rev Med Virol. 29:e20802019. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Vamvakas EC: Is white blood cell reduction
equivalent to antibody screening in preventing transmission of
cytomegalovirus by transfusion? A review of the literature and
meta-analysis. Transfus Med Rev. 19:181–199. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Bowden RA, Sayers M, Flournoy N, Newton B,
Banaji M, Thomas ED and Meyers JD: Cytomegalovirus immune globulin
and seronegative blood products to prevent primary cytomegalovirus
infection after marrow transplantation. N Engl J Med.
314:1006–1010. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Ljungman P, Larsson K, Kumlien G, Aschan
J, Barkholt L, Gustafsson-Jernberg A, Lewensohn-Fuchs I and Ringdén
O: Leukocyte depleted, unscreened blood products give a low risk
for CMV infection and disease in CMV seronegative allogeneic stem
cell transplant recipients with seronegative stem cell donors.
Scand J Infect Dis. 34:347–350. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Boeckh M, Bowden RA, Storer B, Chao NJ,
Spielberger R, Tierney DK, Gallez-Hawkins G, Cunningham T, Blume
KG, Levitt D and Zaia JA: Randomized, placebo-controlled,
double-blind study of a cytomegalovirus-specific monoclonal
antibody (MSL-109) for prevention of cytomegalovirus infection
after allogeneic hematopoietic stem cell transplantation. Biol
Blood Marrow Transplant. 7:343–351. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Raanani P, Gafter-Gvili A, Paul M,
Ben-Bassat I, Leibovici L and Shpilberg O: Immunoglobulin
prophylaxis in hematopoietic stem cell transplantation: Systematic
review and meta-analysis. J Clin Oncol. 27:770–781. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Alexander BT, Hladnik LM, Augustin KM,
Casabar E, McKinnon PS, Reichley RM, Ritchie DJ, Westervelt P and
Dubberke ER: Use of cytomegalovirus intravenous immune globulin for
the adjunctive treatment of cytomegalovirus in hematopoietic stem
cell transplant recipients. Pharmacotherapy. 30:554–561. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Malagola M, Greco R, Santarone S, Natale
A, Iori AP, Quatrocchi L, Barbieri W, Bruzzese A, Leotta S, Carotti
A, et al: CMV management with specific immunoglobulins: A
multicentric retrospective analysis on 92 allotransplanted
patients. Mediterr J Hematol Infect Dis. 11:e20190482019.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Grossi P, Mohacsi P, Szabolcs Z and Potena
L: Cytomegalovirus immunoglobulin after thoracic transplantation:
An overview. Transplantation. 100 (Suppl 3):S1–S4. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Hammerstrom AE, Lombardi LR, Pingali SR,
Rondon G, Chen J, Milton DR, Chemaly RF, Champlin RE, Gulbis A and
Ciurea SO: Prevention of cytomegalovirus reactivation in
haploidentical stem cell transplantation. Biol Blood Marrow
Transplant. 24:353–358. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Boeckh M, Nichols WG, Chemaly RF,
Papanicolaou GA, Wingard JR, Xie H, Syrjala KL, Flowers ME,
Stevens-Ayers T, Jerome KR and Leisenring W: Valganciclovir for the
prevention of complications of late cytomegalovirus infection after
allogeneic hematopoietic cell transplantation: A randomized trial.
Ann Intern Med. 162:1–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Green ML, Leisenring W, Xie H, Mast TC,
Cui Y, Sandmaier BM, Sorror ML, Goyal S, Özkök S, Yi J, et al:
Cytomegalovirus viral load and mortality after haemopoietic stem
cell transplantation in the era of pre-emptive therapy: A
retrospective cohort study. Lancet Haematol. 3:e119–e127. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Wloch MK, Smith LR, Boutsaboualoy S, Reyes
L, Han C, Kehler J, Smith HD, Selk L, Nakamura R, Brown JM, et al:
Safety and immunogenicity of a bivalent cytomegalovirus DNA vaccine
in healthy adult subjects. J Infect Dis. 197:1634–1642. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Schleiss MR: Cytomegalovirus vaccines
under clinical development. J Virus Erad. 2:198–207. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Kharfan-Dabaja MA, Boeckh M, Wilck MB,
Langston AA, Chu AH, Wloch MK, Guterwill DF, Smith LR, Rolland AP
and Kenney RT: A novel therapeutic cytomegalovirus DNA vaccine in
allogeneic haemopoietic stem-cell transplantation: A randomised,
double-blind, placebo-controlled, phase 2 trial. Lancet Infect Dis.
12:290–299. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Griffiths PD, Stanton A, McCarrell E,
Smith C, Osman M, Harber M, Davenport A, Jones G, Wheeler DC,
O'Beirne J, et al: Cytomegalovirus glycoprotein-B vaccine with MF59
adjuvant in transplant recipients: A phase 2 randomised
placebo-controlled trial. Lancet. 377:1256–1263. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Vincenti F, Budde K, Merville P, Shihab F,
Ram Peddi V, Shah M, Wyburn K, Cassuto-Viguier E, Weidemann A, Lee
M, et al: A randomized, phase 2 study of ASP0113, a DNA-based
vaccine, for the prevention of CMV in CMV-seronegative kidney
transplant recipients receiving a kidney from a CMV-seropositive
donor. Am J Transplant. 18:2945–2954. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Singh N, Winston DJ, Razonable RR, Lyon
GM, Silveira FP, Wagener MM, Stevens-Ayers T, Edmison B, Boeckh M
and Limaye AP: Effect of preemptive therapy vs. antiviral
prophylaxis on cytomegalovirus disease in seronegative liver
transplant recipients with seropositive donors: A randomized
clinical trial. JAMA. 323:1378–1387. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Einsele H, Ehninger G, Hebart H,
Wittkowski KM, Schuler U, Jahn G, Mackes P, Herter M, Klingebiel T,
Löffler J, et al: Polymerase chain reaction monitoring reduces the
incidence of cytomegalovirus disease and the duration and side
effects of antiviral therapy after bone marrow transplantation.
Blood. 86:2815–2820. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Tomblyn M, Chiller T, Einsele H, Gress R,
Sepkowitz K, Storek J, Wingard JR, Young JA, Boeckh MJ; Center for
International Blood and Marrow Researchm, ; et al: Guidelines for
preventing infectious complications among hematopoietic cell
transplantation recipients: A global perspective. Biol Blood Marrow
Transplant. 15:1143–1238. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Natori Y, Alghamdi A, Tazari M, Miller V,
Husain S, Komatsu T, Griffiths P, Ljungman P, Orchanian-Cheff A,
Kumar D, et al: Use of viral load as a surrogate marker in clinical
studies of cytomegalovirus in solid organ transplantation: A
systematic review and meta-analysis. Clin Infect Dis. 66:617–631.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Locatelli F, Bertaina A, Bertaina V and
Merli P: Cytomegalovirus in hematopoietic stem cell transplant
recipients-management of infection. Expert Rev Hematol.
9:1093–1105. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Limaye AP, Stapleton RD, Peng L, Gunn SR,
Kimball LE, Hyzy R, Exline MC, Files DC, Morris PE, Frankel SK, et
al: Effect of ganciclovir on IL-6 levels among
cytomegalovirus-seropositive adults with critical Illness: A
randomized clinical trial. JAMA. 318:731–740. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Paya C, Humar A, Dominguez E, Washburn K,
Blumberg E, Alexander B, Freeman R, Heaton N and Pescovitz MD;
Valganciclovir Solid Organ Transplant Study Group, : Efficacy and
safety of valganciclovir vs. oral ganciclovir for prevention of
cytomegalovirus disease in solid organ transplant recipients. Am J
Transplant. 4:611–620. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Winston DJ, Ho WG, Bartoni K, Du Mond C,
Ebeling DF, Buhles WC and Champlin RE: Ganciclovir prophylaxis of
cytomegalovirus infection and disease in allogeneic bone marrow
transplant recipients. Results of a placebo-controlled,
double-blind trial. Ann Intern Med. 118:179–184. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Griffiths P, Whitley R, Snydman DR, Singh
N and Boeckh M; International Herpes Management Forum, :
Contemporary management of cytomegalovirus infection in transplant
recipients: Guidelines from an IHMF workshop, 2007. Herpes.
15:4–12. 2008.PubMed/NCBI
|
|
138
|
Winston DJ, Yeager AM, Chandrasekar PH,
Snydman DR, Petersen FB and Territo MC; Valacyclovir
Cytomegalovirus Study Group, : Randomized comparison of oral
valacyclovir and intravenous ganciclovir for prevention of
cytomegalovirus disease after allogeneic bone marrow
transplantation. Clin Infect Dis. 36:749–758. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Rubin RH, Lynch P, Pasternack MS,
Schoenfeld D and Medearis DN Jr: Combined antibody and ganciclovir
treatment of murine cytomegalovirus-infected normal and
immunosuppressed BALB/c mice. Antimicrob Agents Chemother.
33:1975–1979. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Lukacova V, Goelzer P, Reddy M, Greig G,
Reigner B and Parrott N: A physiologically based pharmacokinetic
model for ganciclovir and its prodrug valganciclovir in adults and
children. AAPS J. 18:1453–1463. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Weiner RS, Bortin MM, Gale RP, Gluckman E,
Kay HE, Kolb HJ, Hartz AJ and Rimm AA: Interstitial pneumonitis
after bone marrow transplantation. Assessment of risk factors. Ann
Intern Med. 104:168–175. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Maffini E, Giaccone L, Festuccia M,
Brunello L, Busca A and Bruno B: Treatment of CMV infection after
allogeneic hematopoietic stem cell transplantation. Expert Rev
Hematol. 9:585–596. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Ritchie BM, Barreto JN, Barreto EF, Crow
SA, Dierkhising RA, Jannetto PJ, Tosh PK and Razonable RR:
Relationship of ganciclovir therapeutic drug monitoring with
clinical efficacy and patient safety. Antimicrob Agents Chemother.
63:e01855–18. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Kim ST, Lee MH, Kim SY, Kim SJ, Kim DH,
Jang JH, Kim K, Kim WS and Jung CW: A randomized trial of
preemptive therapy for prevention of cytomegalovirus disease after
allogeneic hematopoietic stem cell transplantation. Int J Hematol.
91:886–891. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
El Chaer F, Shah DP and Chemaly RF: How I
treat resistant cytomegalovirus infection in hematopoietic cell
transplantation recipients. Blood. 128:2624–2636. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Einsele H, Reusser P, Bornhäuser M, Kalhs
P, Ehninger G, Hebart H, Chalandon Y, Kröger N, Hertenstein B and
Rohde F: Oral valganciclovir leads to higher exposure to
ganciclovir than intravenous ganciclovir in patients following
allogeneic stem cell transplantation. Blood. 107:3002–3008. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Barkam C, Kamal H, Dammann E, Diedrich H,
Buchholz S, Eder M, Krauter J, Ganser A and Stadler M: Improving
safety of preemptive therapy with oral valganciclovir for
cytomegalovirus infection after allogeneic hematopoietic stem cell
transplantation. Bone Marrow Res. 2012:8746012012. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Moretti S, Zikos P, Van Lint MT, Tedone E,
Occhini D, Gualandi F, Lamparelli T, Mordini N, Berisso G, Bregante
S, et al: Forscarnet vs. ganciclovir for cytomegalovirus (CMV)
antigenemia after allogeneic hemopoietic stem cell transplantation
(HSCT): A randomised study. Bone Marrow Transplant. 22:175–180.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Meesing A and Razonable RR: New
developments in the management of cytomegalovirus infection after
transplantation. Drugs. 78:1085–1103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Marty FM, Ljungman P, Chemaly RF, Maertens
J, Dadwal SS, Duarte RF, Haider S, Ullmann AJ, Katayama Y, Brown J,
et al: Letermovir prophylaxis for cytomegalovirus in
hematopoietic-cell transplantation. N Engl J Med. 377:2433–2444.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Bowman LJ, Melaragno JI and Brennan DC:
Letermovir for the management of cytomegalovirus infection. Expert
Opin Investig Drugs. 26:235–241. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Phoompoung P, Ferreira VH, Tikkanen J,
Husain S, Viswabandya A, Kumar D and Humar A: Letermovir as salvage
therapy for Cytomegalovirus infection in transplant recipients.
Transplantation. 104:404–409. 2019. View Article : Google Scholar
|
|
153
|
Popping S, Dalm VASH, Lübke N,
Cristanziano VD, Kaiser R, Boucher CAB and Van Kampen JJA:
Emergence and persistence of letermovir-resistant cytomegalovirus
in a patient with primary immunodeficiency. Open Forum Infectious
Diseases. 6:ofz3752019. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Griffiths P and Lumley S: Cytomegalovirus.
Curr Opin Infect Dis. 27:554–559. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Beadle JR, Hartline C, Aldern KA,
Rodriguez N, Harden E, Kern ER and Hostetler KY: Alkoxyalkyl esters
of cidofovir and cyclic cidofovir exhibit multiple-log enhancement
of antiviral activity against cytomegalovirus and herpesvirus
replication in vitro. Antimicrob Agents Chemother. 46:2381–2386.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Bidanset DJ, Beadle JR, Wan WB, Hostetler
KY and Kern ER: Oral activity of ether lipid ester prodrugs of
cidofovir against experimental human cytomegalovirus infection. J
Infect Dis. 190:499–503. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
157
|
Kern ER, Collins DJ, Wan WB, Beadle JR,
Hostetler KY and Quenelle DC: Oral treatment of murine
cytomegalovirus infections with ether lipid esters of cidofovir.
Antimicrob Agents Chemother. 48:3516–3522. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Chemaly RF, Hill JA, Voigt S and Peggs KS:
In vitro comparison of currently available and investigational
antiviral agents against pathogenic human double-stranded DNA
viruses: A systematic literature review. Antiviral Res. 163:50–58.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Gagelmann N, Ljungman P, Styczynski J and
Kröger N: Comparative efficacy and safety of different antiviral
agents for cytomegalovirus prophylaxis in allogeneic hematopoietic
cell transplantation: A systematic review and meta-analysis. Biol
Blood Marrow Transplant. 24:2101–2109. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Hostetler KY: Alkoxyalkyl prodrugs of
acyclic nucleoside phosphonates enhance oral antiviral activity and
reduce toxicity: Current state of the art. Antiviral Res.
82:A84–A98. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Painter W, Robertson A, Trost LC, Godkin
S, Lampert B and Painter G: First pharmacokinetic and safety study
in humans of the novel lipid antiviral conjugate CMX001, a
broad-spectrum oral drug active against double-stranded DNA
viruses. Antimicrob Agents Chemother. 56:2726–2734. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Aldern KA, Ciesla SL, Winegarden KL and
Hostetler KY: Increased antiviral activity of
1-O-hexadecyloxypropyl-[2-(14)C]cidofovir in MRC-5 human lung
fibroblasts is explained by unique cellular uptake and metabolism.
Mol Pharmacol. 63:678–681. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Ciesla SL, Trahan J, Wan WB, Beadle JR,
Aldern KA, Painter GR and Hostetler KY: Esterification of cidofovir
with alkoxyalkanols increases oral bioavailability and diminishes
drug accumulation in kidney. Antiviral Res. 59:163–171. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Tippin TK, Morrison ME, Brundage TM and
Momméja-Marin H: Brincidofovir is not a substrate for the human
organic anion transporter 1: A mechanistic explanation for the lack
of nephrotoxicity observed in clinical studies. Ther Drug Monit.
38:777–786. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Marty FM, Winston DJ, Chemaly RF, Mullane
KM, Shore TB, Papanicolaou GA, Chittick G, Brundage TM, Wilson C,
Morrison ME, et al: A randomized, double-blind, placebo-controlled
phase 3 trial of oral brincidofovir for cytomegalovirus prophylaxis
in allogeneic hematopoietic cell transplantation. Biol Blood Marrow
Transplant. 25:369–381. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Papanicolaou GA, Silveira FP, Langston AA,
Pereira MR, Avery RK, Uknis M, Wijatyk A, Wu J, Boeckh M, Marty FM
and Villano S: Maribavir for refractory or resistant
cytomegalovirus infections in hematopoietic-cell or solid-organ
transplant recipients: A randomized, dose-ranging, Double-blind,
phase 2 study. Clin Infect Dis. 68:1255–1264. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Frange P and Leruez-Ville M: Maribavir,
brincidofovir and letermovir: Efficacy and safety of new antiviral
drugs for treating cytomegalovirus infections. Med Mal Infect.
48:495–502. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Marty FM and Boeckh M: Maribavir and human
cytomegalovirus-what happened in the clinical trials and why might
the drug have failed? Curr Opin Virol. 1:555–562. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Piret J and Boivin G: Clinical development
of letermovir and maribavir: Overview of human cytomegalovirus drug
resistance. Antiviral Res. 163:91–105. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Bright PD, Gompels M, Donati M and
Johnston S: Successful oral treatment of Ganciclovir resistant
cytomegalovirus with Maribavir in the context of primary
immunodeficiency: First case report and review. J Clin Virol.
87:12–16. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Maertens J, Cordonnier C, Jaksch P, Poiré
X, Uknis M, Wu J, Wijatyk A, Saliba F, Witzke O and Villano S:
Maribavir for preemptive treatment of cytomegalovirus reactivation.
N Engl J Med. 381:1136–1147. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Avery RK, Marty FM, Strasfeld L, Lee I,
Arrieta A, Chou S, Tatarowicz W and Villano S: Oral maribavir for
treatment of refractory or resistant cytomegalovirus infections in
transplant recipients. Transpl Infect Dis. 12:489–496. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Germi R, Mariette C, Alain S, Lupo J,
Thiebaut A, Brion JP, Epaulard O, Saint Raymond C, Malvezzi P and
Morand P: Success and failure of artesunate treatment in five
transplant recipients with disease caused by drug-resistant
cytomegalovirus. Antiviral Res. 101:57–61. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Avery RK, Mossad SB, Poggio E, Lard M,
Budev M, Bolwell B, Waldman WJ, Braun W, Mawhorter SD, Fatica R, et
al: Utility of leflunomide in the treatment of complex
cytomegalovirus syndromes. Transplantation. 90:419–426. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Feuchtinger T, Opherk K, Bethge WA, Topp
MS, Schuster FR, Weissinger EM, Mohty M, Or R, Maschan M, Schumm M,
et al: Adoptive transfer of pp65-specific T cells for the treatment
of chemorefractory cytomegalovirus disease or reactivation after
haploidentical and matched unrelated stem cell transplantation.
Blood. 116:4360–4367. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Walter EA, Greenberg PD, Gilbert MJ, Finch
RJ, Watanabe KS, Thomas ED and Riddell SR: Reconstitution of
cellular immunity against cytomegalovirus in recipients of
allogeneic bone marrow by transfer of T-cell clones from the donor.
N Engl J Med. 333:1038–1044. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Blyth E, Clancy L, Simms R, Ma CK, Burgess
J, Deo S, Byth K, Dubosq MC, Shaw PJ, Micklethwaite KP and Gottlieb
DJ: Donor-derived CMV-specific T cells reduce the requirement for
CMV-directed pharmacotherapy after allogeneic stem cell
transplantation. Blood. 121:3745–3758. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Peggs KS, Verfuerth S, Pizzey A, Khan N,
Guiver M, Moss PA and Mackinnon S: Adoptive cellular therapy for
early cytomegalovirus infection after allogeneic stem-cell
transplantation with virus-specific T-cell lines. Lancet.
362:1375–1377. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Neuenhahn M, Albrecht J, Odendahl M,
Schlott F, Dössinger G, Schiemann M, Lakshmipathi S, Martin K,
Bunjes D, Harsdorf S, et al: Transfer of minimally manipulated
CMV-specific T cells from stem cell or third-party donors to treat
CMV infection after allo-HSCT. Leukemia. 31:2161–2171. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Celilova S, Toret E, Adaklı Aksoy B, Ovalı
E and Bozkurt C: CMV Specific T cells for treatment of CMV
infection after hematopoietic stem cell transplantation in a
pediatric case: First application in Turkey. Turk J Haematol.
37:65–67. 2020.PubMed/NCBI
|
|
181
|
Tzannou I, Papadopoulou A, Naik S, Leung
K, Martinez CA, Ramos CA, Carrum G, Sasa G, Lulla P, Watanabe A, et
al: Off-the-shelf virus-specific T cells to treat BK virus, human
herpesvirus 6, cytomegalovirus, Epstein-Barr virus, and adenovirus
infections after allogeneic hematopoietic stem-cell
transplantation. J Clin Oncol. 35:3547–3557. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Kállay K, Kassa C, Réti M, Karászi É,
Sinkó J, Goda V, Stréhn A, Csordás K, Horváth O, Szederjesi A, et
al: Early experience with CliniMACS prodigy CCS (IFN-gamma) system
in selection of virus-specific T cells from third-party donors for
pediatric patients with severe viral infections after hematopoietic
stem cell transplantation. J Immunother. 41:158–163. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Ogonek J, Verma K, Schultze-Florey C,
Varanasi P, Luther S, Schweier P, Kühnau W, Göhring G, Dammann E,
Stadler M, et al: Characterization of high-avidity
cytomegalovirus-specific T cells with differential tetramer binding
coappearing after allogeneic stem cell transplantation. J Immunol.
199:792–805. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Bao L, Cowan MJ, Dunham K, Horn B, McGuirk
J, Gilman A and Lucas KG: Adoptive immunotherapy with CMV-specific
cytotoxic T lymphocytes for stem cell transplant patients with
refractory CMV infections. J Immunother. 35:293–298. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Koehne G, Hasan A, Doubrovina E, Prockop
S, Tyler E, Wasilewski G and O'Reilly RJ: Immunotherapy with donor
T cells sensitized with overlapping pentadecapeptides for treatment
of persistent cytomegalovirus infection or viremia. Biol Blood
Marrow Transplant. 21:1663–1678. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Pei XY, Zhao XY, Chang YJ, Liu J, Xu LP,
Wang Y, Zhang XH, Han W, Chen YH and Huang XJ:
Cytomegalovirus-specific T-cell transfer for refractory
cytomegalovirus infection after haploidentical stem cell
transplantation: The quantitative and qualitative immune recovery
for cytomegalovirus. J Infect Dis. 216:945–956. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Peggs KS, Thomson K, Samuel E, Dyer G,
Armoogum J, Chakraverty R, Pang K, Mackinnon S and Lowdell MW:
Directly selected cytomegalovirus-reactive donor T cells confer
rapid and safe systemic reconstitution of virus-specific immunity
following stem cell transplantation. Clin Infect Dis. 52:49–57.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Micklethwaite K, Hansen A, Foster A, Snape
E, Antonenas V, Sartor M, Shaw P, Bradstock K and Gottlieb D: Ex
vivo expansion and prophylactic infusion of CMV-pp65
peptide-specific cytotoxic T-lymphocytes following allogeneic
hematopoietic stem cell transplantation. Biol Blood Marrow
Transplant. 13:707–714. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Houghtelin A and Bollard CM:
Virus-specific T cells for the immunocompromised patient. Front
Immunol. 8:12722017. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Einsele H, Roosnek E, Rufer N, Sinzger C,
Riegler S, Löffler J, Grigoleit U, Moris A, Rammensee HG, Kanz L,
et al: Infusion of cytomegalovirus (CMV)-specific T cells for the
treatment of CMV infection not responding to antiviral
chemotherapy. Blood. 99:3916–3922. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Perruccio K, Tosti A, Burchielli E, Topini
F, Ruggeri L, Carotti A, Capanni M, Urbani E, Mancusi A, Aversa F,
et al: Transferring functional immune responses to pathogens after
haploidentical hematopoietic transplantation. Blood. 106:4397–4406.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Gary R, Aigner M, Moi S, Schaffer S,
Gottmann A, Maas S, Zimmermann R, Zingsem J, Strobel J, Mackensen
A, et al: Clinical-grade generation of peptide-stimulated
CMV/EBV-specific T cells from G-CSF mobilized stem cell grafts. J
Transl Med. 16:1242018. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Friedenstein AJ, Deriglasova UF, Kulagina
NN, Panasuk AF, Rudakowa SF, Luriá EA and Ruadkow IA: Precursors
for fibroblasts in different populations of hematopoietic cells as
detected by the in vitro colony assay method. Exp Hematol. 2:83–92.
1974.PubMed/NCBI
|
|
194
|
Méndez-Ferrer S, Michurina TV, Ferraro F,
Mazloom AR, Macarthur BD, Lira SA, Scadden DT, Ma'ayan A,
Enikolopov GN and Frenette PS: Mesenchymal and haematopoietic stem
cells form a unique bone marrow niche. Nature. 466:829–834. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Martínez-Peinado P, Pascual-García S,
Roche E and Sempere-Ortells JM: Differences of clonogenic
mesenchymal stem cells on immunomodulation of lymphocyte subsets. J
Immunol Res. 2018:72327172018. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Wolff D, Schleuning M, von Harsdorf S,
Bacher U, Gerbitz A, Stadler M, Ayuk F, Kiani A, Schwerdtfeger R,
Vogelsang GB, et al: Consensus conference on clinical practice in
chronic GVHD: Second-line treatment of chronic Graft-versus-Host
disease. Biol Blood Marrow Transplant. 17:1–17. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Tse WT, Pendleton JD, Beyer WM, Egalka MC
and Guinan EC: Suppression of allogeneic T-cell proliferation by
human marrow stromal cells: Implications in transplantation.
Transplantation. 75:389–397. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Malcherek G, Jin N, Huckelhoven AG, Mani
J, Wang L, Gern U, Diehlmann A, Wuchter P, Schmitt A, Chen B, et
al: Mesenchymal stromal cells inhibit proliferation of
virus-specific CD8+ T cells. Leukemia. 28:2388–2394.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Elmaagacli AH, Steckel NK, Koldehoff M,
Hegerfeldt Y, Trenschel R, Ditschkowski M, Christoph S, Gromke T,
Kordelas L, Ottinger HD, et al: Early human cytomegalovirus
replication after transplantation is associated with a decreased
relapse risk: Evidence for a putative virus-versus-leukemia effect
in acute myeloid leukemia patients. Blood. 118:1402–1412. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Inagaki J, Noguchi M, Kurauchi K, Tanioka
S, Fukano R and Okamura J: Effect of cytomegalovirus reactivation
on relapse after allogeneic hematopoietic stem cell transplantation
in pediatric acute leukemia. Biol Blood Marrow Transplant.
22:300–306. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Litjens NHR, van der Wagen L, Kuball J and
Kwekkeboom J: Potential beneficial effects of cytomegalovirus
infection after transplantation. Front Immunol. 9:3892018.
View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Yoon JH, Lee S, Kim HJ, Jeon YW, Lee SE,
Cho BS, Lee DG, Eom KS, Kim YJ, Min CK, et al: Impact of
cytomegalovirus reactivation on relapse and survival in patients
with acute leukemia who received allogeneic hematopoietic stem cell
transplantation in first remission. Oncotarget. 7:17230–17241.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Jang JE, Kim SJ, Cheong JW, Hyun SY, Kim
YD, Kim YR, Kim JS and Min YH: Early CMV replication and subsequent
chronic GVHD have a significant anti-leukemic effect after
allogeneic HSCT in acute myeloid leukemia. Ann Hematol. 94:275–282.
2015. View Article : Google Scholar : PubMed/NCBI
|