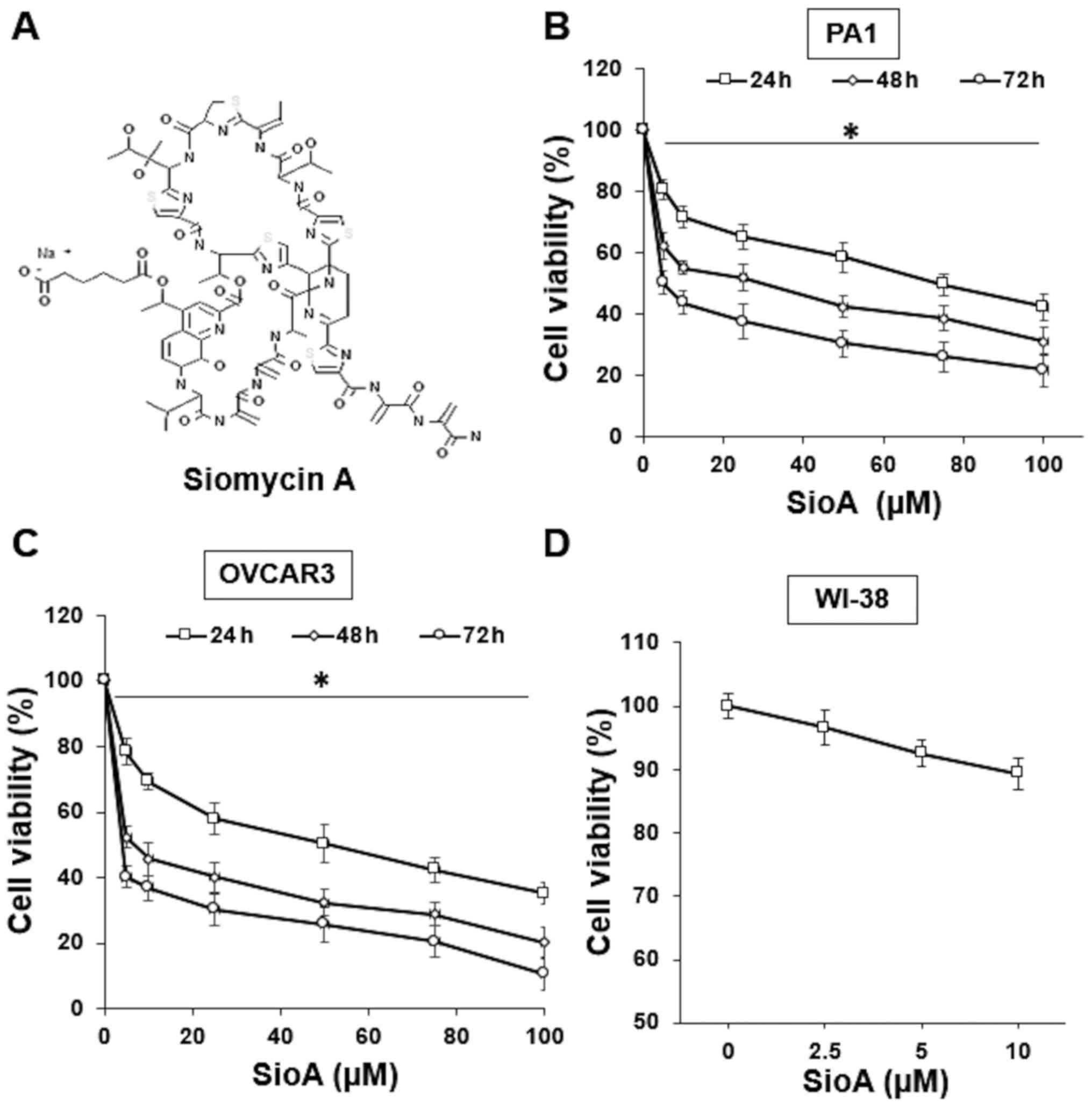
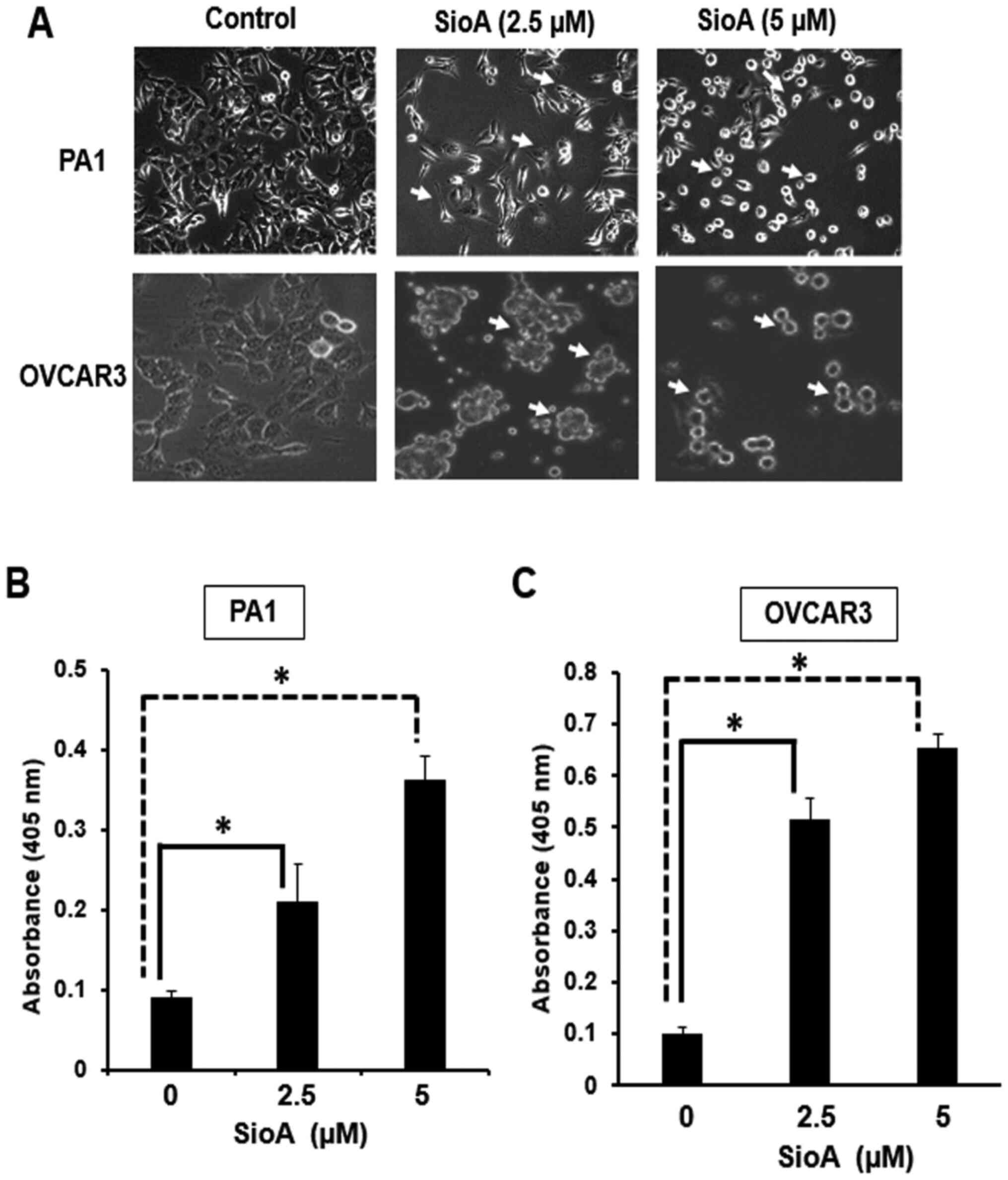
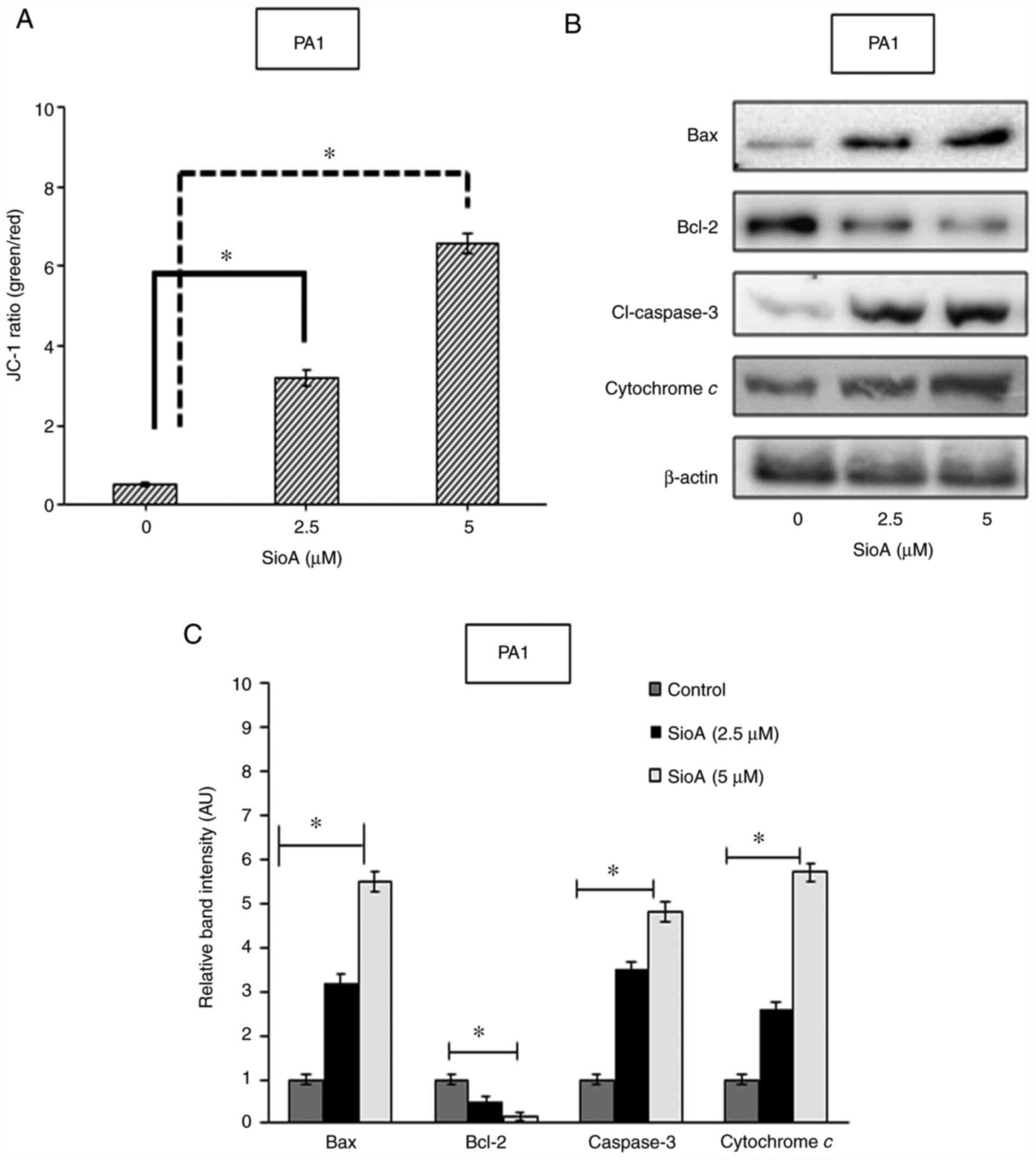
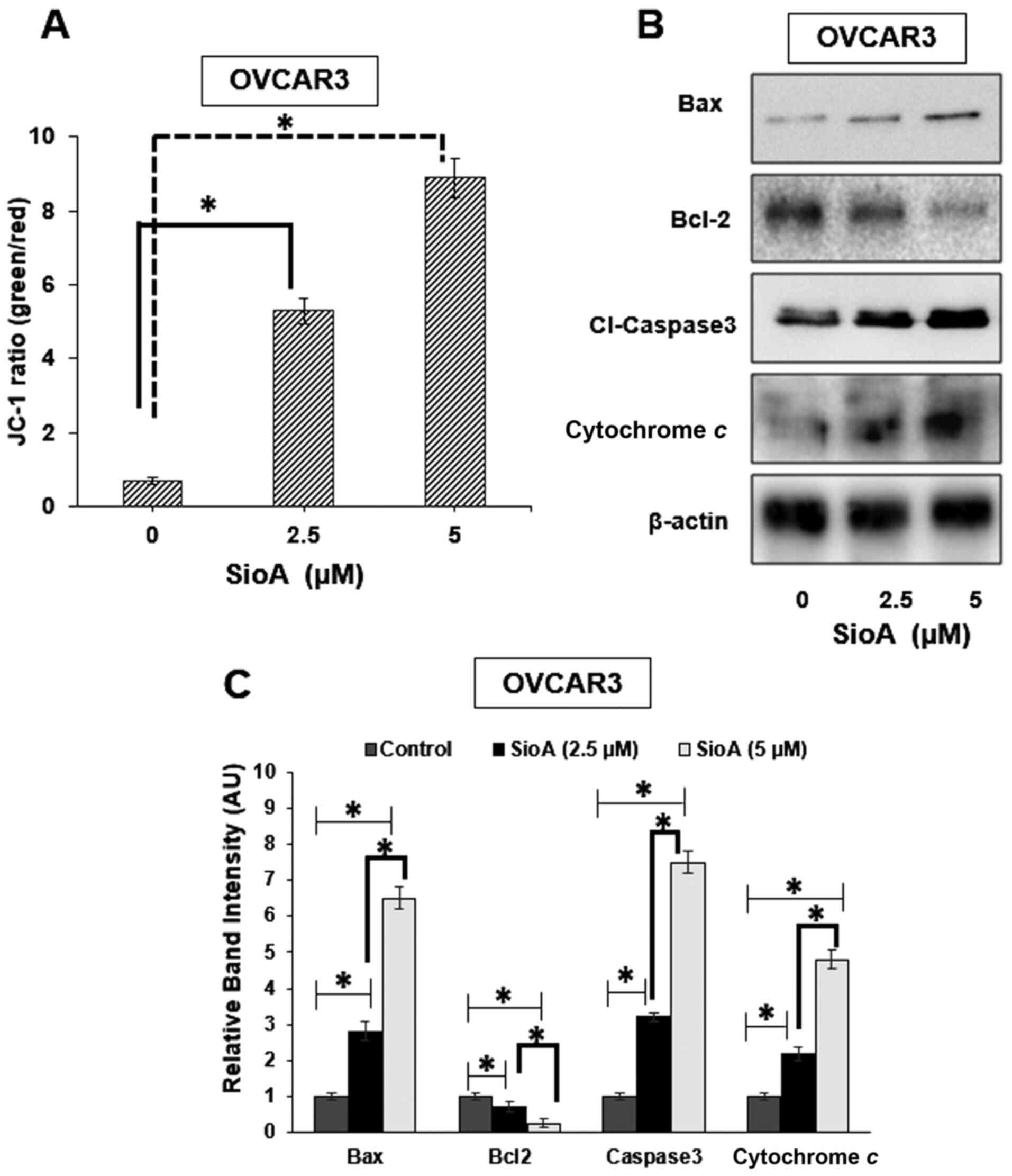
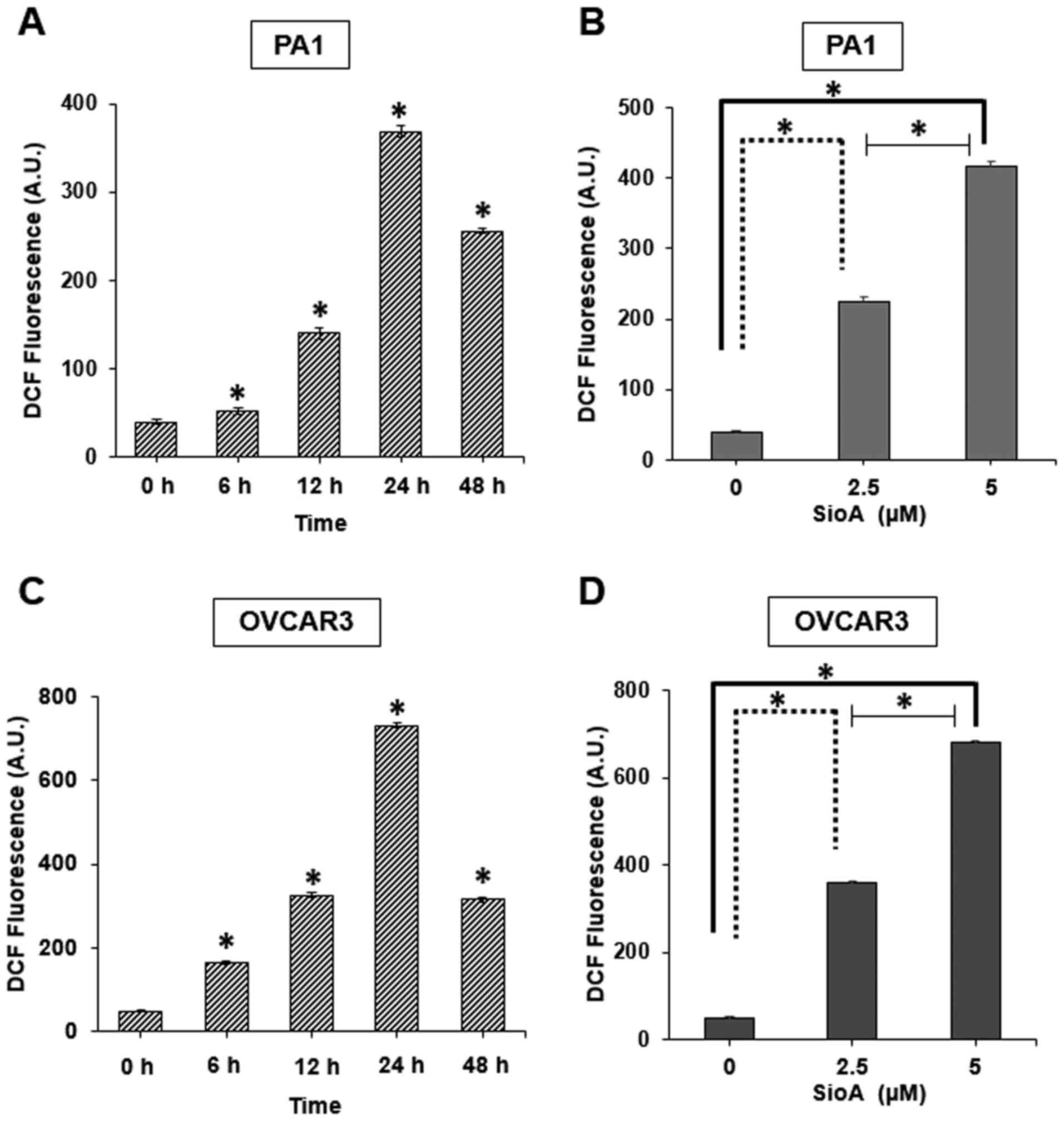
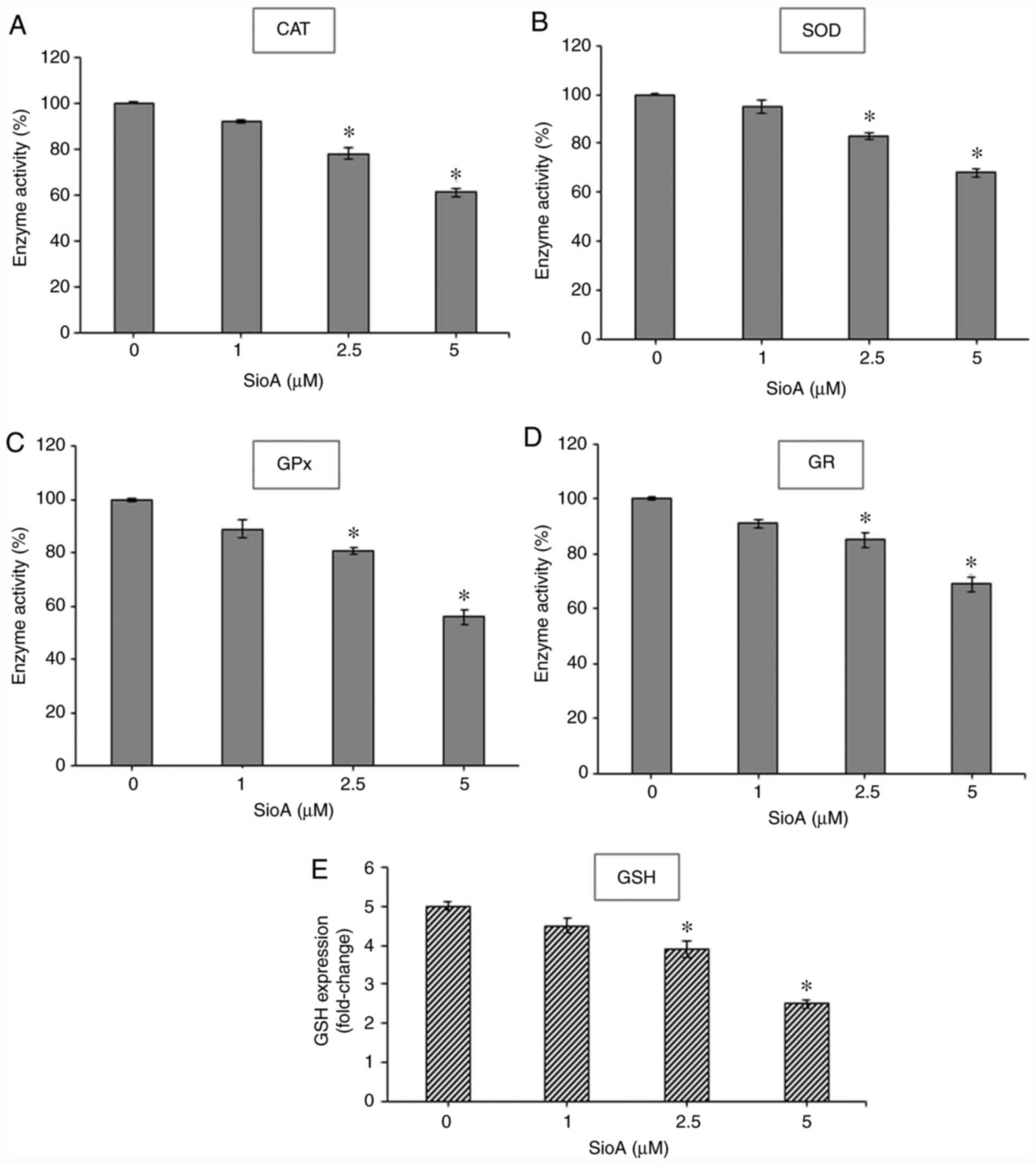
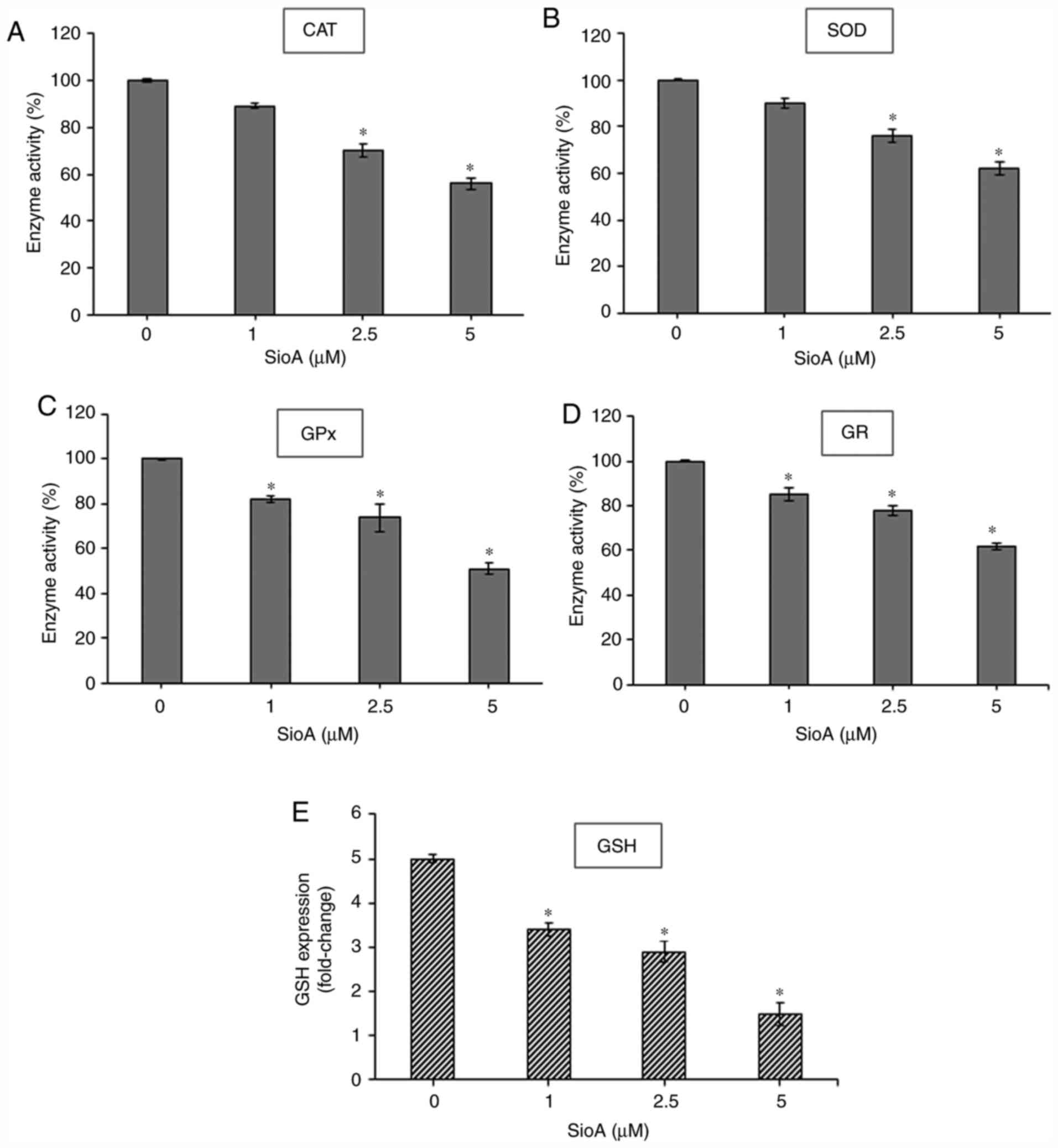
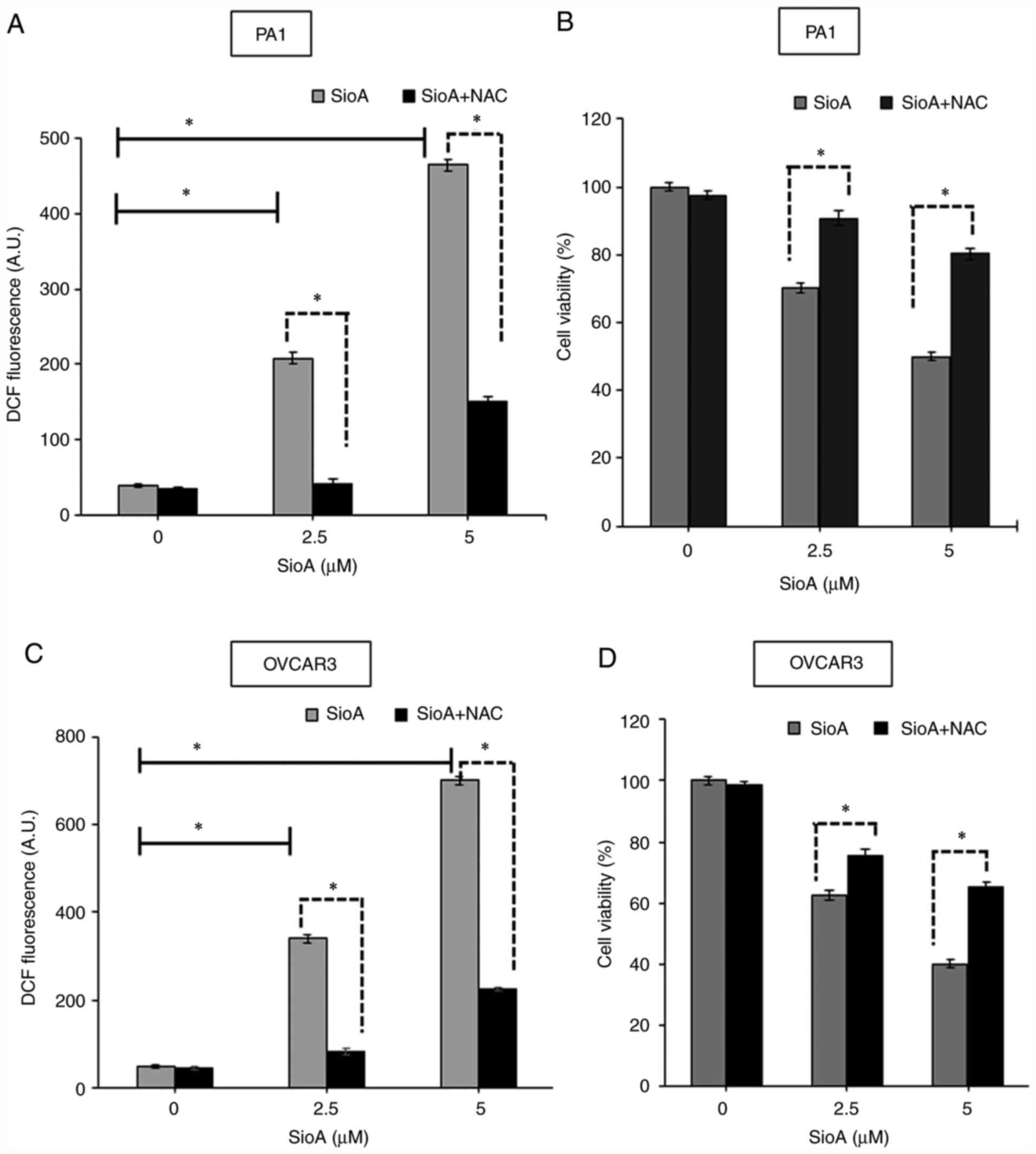
|
1
|
Zhang Y, Luo G, Li M, Guo P, Xiao Y, Ji H
and Hao Y: Global patterns and trends in ovarian cancer incidence:
Age, period and birth cohort analysis. BMC Cancer. 19:9842019.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Reid BM, Permuth JB and Sellers TA:
Epidemiology of ovarian cancer: A review. Cancer Biol Med. 14:9–32.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Webb PM and Jordan SJ: Epidemiology of
epithelial ovarian cancer. Best Pract Res Clin Obstet Gynaecol.
41:3–14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pavlik EJ, Smith C, Dennis TS, Harvey E,
Huang B, Chen Q, Piecoro DW, Burgess BT, McDowell A, Gorski J, et
al: Disease-specific survival of type I and type II epithelial
ovarian cancers-stage challenges categorical assignments of
indolence & aggressiveness. Diagnostics (Basel). 10:E562020.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Momenimovahed Z, Tiznobaik A, Taheri S and
Salehiniya H: Ovarian cancer in the world: Epidemiology and risk
factors. Int J Womens Health. 11:287–299. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ottevanger PB: Ovarian cancer stem cells
more questions than answers. Semin Cancer Biol. 44:67–71. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cornelison R, Llaneza DC and Landen CN:
Emerging therapeutics to overcome chemoresistance in epithelial
ovarian cancer: A mini-review. Int J Mol Sci. 18:21712017.
View Article : Google Scholar
|
|
8
|
Bast RC Jr, Hennessy B and Mills GB: The
biology of ovarian cancer: New opportunities for translation. Nat
Rev Cancer. 9:415–428. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nakano I, Joshi K, Visnyei K, Hu B,
Watanabe M, Lam D, Wexler E, Saigusa K, Nakamura Y, Laks DR, et al:
Siomycin A targets brain tumor stem cells partially through a
MELK-mediated pathway. Neuro Oncol. 13:622–634. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Koh B, Jeon H, Kim D, Kang D and Kim KR:
Effect of fibroblast co-culture on the proliferation, viability and
drug response of colon cancer cells. Oncol Lett. 17:2409–2417.
2019.PubMed/NCBI
|
|
11
|
Shen X, Mustafa M, Chen Y, Cao Y and Gao
J: Natural thiopeptides as a privileged scaffold for drug discovery
and therapeutic development. Med Chem Res. 28:1063–1098. 2019.
View Article : Google Scholar
|
|
12
|
Tüfekçi Ö, Yandım MK, Ören H, İrken G and
Baran Y: Targeting FoxM1 transcription factor in T-cell acute
lymphoblastic leukemia cell line. Leuk Res. 39:342–347. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hou Y, Zhu Q, Li Z, Peng Y, Yu X, Yuan B,
Liu Y, Liu Y, Yin L, Peng Y, et al: The FOXM1-ABCC5 axis
contributes to paclitaxel resistance in nasopharyngeal carcinoma
cells. Cell Death Dis. 8:e26592017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gartel AL: A new target for proteasome
inhibitors: FoxM1. Expert Opin Investig Drugs. 19:235–242. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nowak A, Zakłos-Szyda M, Żyżelewicz D,
Koszucka A and Motyl I: Acrylamide decreases cell viability, and
provides oxidative stress, DNA damage, and apoptosis in human colon
adenocarcinoma cell line caco-2. Molecules. 25:3682020. View Article : Google Scholar
|
|
16
|
Maity G, De A, Das A, Banerjee S, Sarkar S
and Banerjee SK: Aspirin blocks growth of breast tumor cells and
tumor-initiating cells and induces reprogramming factors of
mesenchymal to epithelial transition. Lab Invest. 95:702–717. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Harshkova D, Zielińska E and Aksmann A:
Optimization of a microplate reader method for the analysis of
changes in mitochondrial membrane potential in Chlamydomonas
reinhardtii cells using the fluorochrome JC-1. J Appl Phycol.
31:3691–3697. 2019. View Article : Google Scholar
|
|
18
|
Halliwell B and Whiteman M: Measuring
reactive species and oxidative damage in vivo and in cell culture:
How should you do it and what do the results mean? Br J Pharmacol.
142:231–255. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Beers RF Jr and Sizer IW: A
spectrophotometric method for measuring the breakdown of hydrogen
peroxide by catalase. J Biol Chem. 195:133–140. 1952. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Marklund S and Marklund G: Involvement of
superoxide anion radical in the autoxidation of pyrogallol and a
convenient assay for superoxide dismutase. Eur J Biochem.
47:469–474. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhu H, Zhang L, Itoh K, Yamamoto M, Ross
D, Trush MA, Zweier JL and Li Y: Nrf2 controls bone marrow stromal
cell susceptibility to oxidative and electrophilic stress. Free
Radic Biol Med. 41:132–143. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Forman HJ, Zhang H and Rinna A:
Glutathione: Overview of its protective roles, measurement, and
biosynthesis. Mol Aspects Med. 30:1–12. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Das A, Chakrabarty S, Choudhury D and
Chakrabarti G: 1, 4-Benzoquinone (PBQ) induced toxicity in lung
epithelial cells is mediated by the disruption of the microtubule
network and activation of caspase-3. Chem Res Toxicol.
23:1054–1066. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ishiguro T, Ishiguro M, Ishiguro R and
Iwai S: Cotreatment with dichloroacetate and omeprazole exhibits a
synergistic antiproliferative effect on malignant tumors. Oncol
Lett. 3:726–728. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tipgomut C, Wongprommoon A, Takeo E,
Ittiudomrak T, Puthong S and Chanchao C: Melittin induced g1 cell
cycle arrest and apoptosis in chago-k1 human bronchogenic carcinoma
cells and inhibited the differentiation of THP-1 cells into
tumour-associated macrophages. Asian Pac J Cancer Prev.
19:3427–3434. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bustamante J, Caldas Lopes E, Garcia M, Di
Libero E, Alvarez E and Hajos SE: Disruption of mitochondrial
membrane potential during apoptosis induced by PSC 833 and CsA in
multidrug-resistant lymphoid leukemia. Toxicol Appl Pharmacol.
199:44–51. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Finucane DM, Bossy-Wetzel E, Waterhouse
NJ, Cotter TG and Green DR: Bax-induced caspase activation and
apoptosis via cytochromec release from mitochondria is inhibitable
by Bcl-xL. J Biol Chem. 274:2225–2233. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liou GY and Storz P: Reactive oxygen
species in cancer. Free Radic Res. 44:479–496. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Valko M, Rhodes CJ, Moncol J, Izakovic M
and Mazur M: Free radicals, metals and antioxidants in oxidative
stress-induced cancer. Chem Biol Interact. 160:1–40. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Filomeni G, Rotilio G and Ciriolo MR: Cell
signalling and the glutathione redox system. Biochem Pharmacol.
64:1057–1064. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Helm CW and States JC: Enhancing the
efficacy of cisplatin in ovarian cancer treatment-could arsenic
have a role. J Ovarian Res. 2:22009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang B, Wang W, Meng HY, Chen J and Yuan
LJ: Effects and mechanism of siomycin A on the growth and apoptosis
of MiaPaCa-2 cancer cells. Oncol Lett. 18:2869–2876.
2019.PubMed/NCBI
|
|
33
|
Guo X, Liu A, Hua H, Lu H, Zhang D, Lin Y,
Sun Q, Zhu X, Yan G and Zhao F: Siomycin a induces apoptosis in
human lung adenocarcinoma A549 cells by suppressing the expression
of foxm1. Nat Prod Commun. 10:1603–1606. 2015.PubMed/NCBI
|
|
34
|
Radhakrishnan SK, Bhat UG, Hughes DE, Wang
IC, Costa RH and Gartel AL: Identification of a chemical inhibitor
of the oncogenic transcription factor forkhead box M1. Cancer Res.
66:9731–9735. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lee VS, McRobb LS, Moutrie V, Santos ED
and Siu TL: Effects of FOXM1 inhibition and ionizing radiation on
melanoma cells. Oncol Lett. 16:6822–6830. 2018.PubMed/NCBI
|