Introduction
According to the 2019 Cancer Statistics Report from
the United States, prostate cancer ranks highest with regard to new
cases, and second highest in terms of cancer-associated deaths
amongst men (1). Prostate cancer is
a very common type of cancer, which threatens the health and life
of men. Additionally, the metastatic potential of prostate cancer,
and the risk of development of hormone-resistant recurrence is high
(2,3). The prevalence of prostate cancer
increases with age, and differs significantly with region, being
higher in Europe and America than in Asia. The mortality and
morbidity rates of patients with prostate cancer in Asian
countries, including China, have been increasing in recent years,
but remain lower than that of Western countries (4–6). The
risk factors for prostate cancer have not been fully clarified, but
may include age, ethnicity, family history, genetics, obesity and
levels of sex hormones (7).
Currently, the first-line chemotherapeutic drugs used to treat
prostate cancer are associated with serious side effects and, the
cancer may develop resistance when used long-term, particularly
when they are administered during the early stages of the disease.
The use of these drugs is thus restricted to specific cases
(8). Therefore, there is an urgent
need to identify novel agents to prevent and treat prostate cancer.
Candidate drugs should ideally be able to reduce proliferation,
migration and invasion of these highly metastatic tumors.
Natural compounds often exhibit inhibitory effects
on a variety of cellular signaling pathways and are thus
investigated to identify their bioactive molecules for use as
potential anticancer drugs (9).
Chinese herbal medicines are an important resource for discovery of
novel compounds which may assist in treating a range of diseases
and have formed an important part of the Chinese health care system
for several millennia.
A medicinal herb known as ‘Yan guan tou cao’ belongs
to the Carpesium cernuum L. family and is widely distributed
in the southern regions of China (10). It consists of an abundance of
chemical constituents and has been shown to possess several
biological effects, including antitumor (11–13),
anti-inflammatory (14,15), analgesic and detoxifying effects
(16). It primarily consists of
sesquiterpenoid lactones (17,18),
sterols and aromatic compounds (19), and glycosides (20). It has been reported that the
sesquiterpenoid lactones derived from C. cernuum L. are
effective cytotoxic (16) and
antitumor agents (21) IA (Fig. 1A), a known sesquiterpenoid (22), isolated from C. cernuum L.,
can inhibit the growth of A549, BGC-823, MCF-7 and other cancer
cell lines (23). However, to the
best of our knowledge, the mechanisms by which IA exerts its
effects on cancer cells have not been determined. Therefore, the
aim of the present study was to determine the antitumor effects of
IA in prostate cancer cells, and explore the potential underlying
molecular mechanism, by assessing the expression of key signaling
proteins associated with the major signal transduction
pathways.
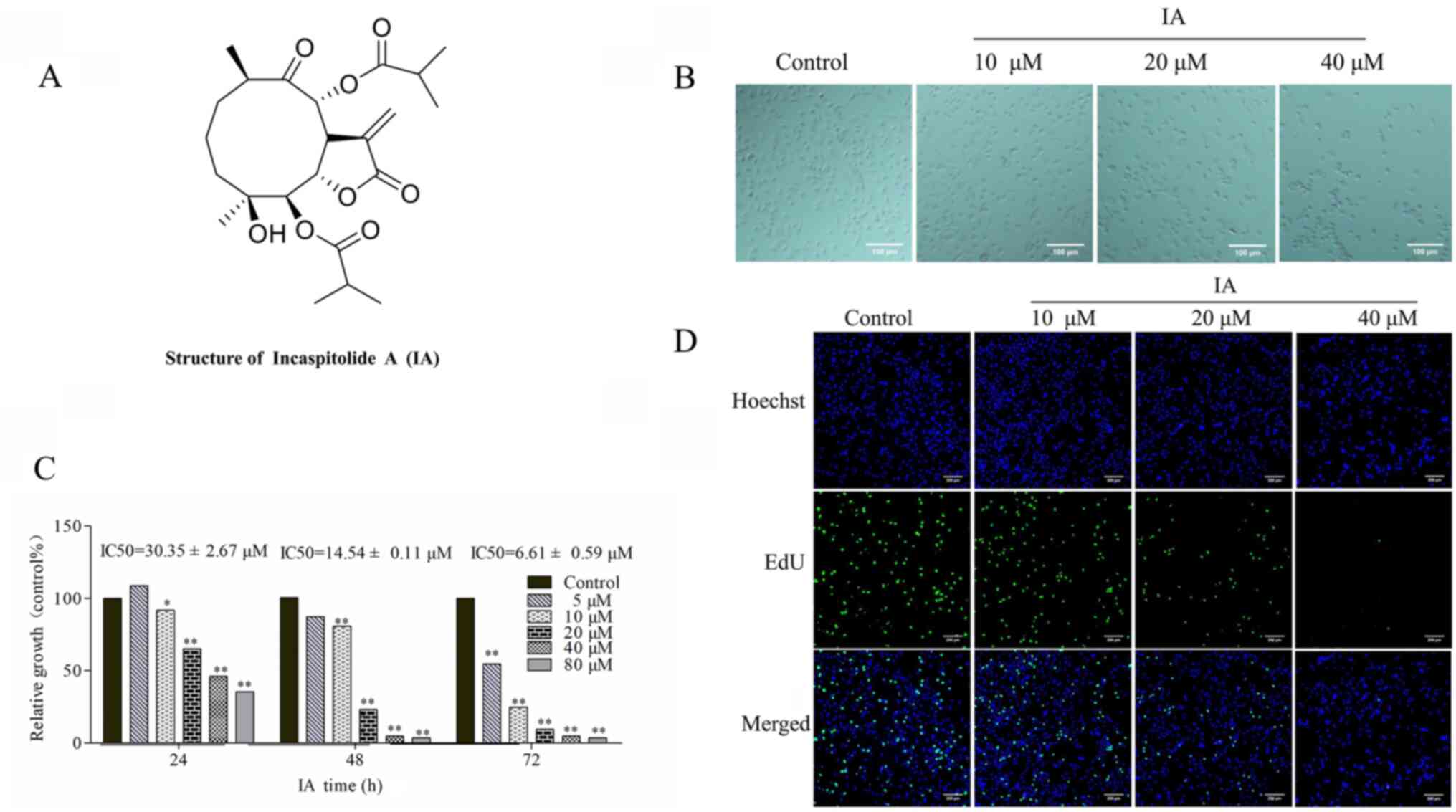 | Figure 1.Effect of IA on the morphology and
proliferation of PC-3 cells. (A) Structure of IA isolated from
Carpesium cernuum L.. (B) Morphology of PC-3 cells following
treatment with IA for 24 h. Phase-contrast images were captured
using an inverted light microscope, from three independent
experiments. Scale bar, 100 µm; magnification, ×200. (C) Cytotoxic
effects of IA on PC-3 cells. PC-3 cells were treated with 5–80 µM
IA for 24, 48 or 72 h. A Cell Counting Kit-8 assay was used to
measure the numbers of viable cells, and data are presented as a
percentage of treated cells to untreated cells. (D) PC-3 cells were
treated with 0, 10, 20 or 40 µM IA for 24 h, and then proliferation
was evaluated by an EdU staining assay. Scale bar, 200 µm;
magnification, ×100. Data are presented as the mean ± standard
deviation of three repeats. *P<0.05, **P<0.01 vs. control
group. IA, incaspitolide A. |
Material and methods
Materials
The entire plant of C. cernuum L. was
collected from Zhenning, Guizhou Province, China, and identified by
Professor Qing-wen Sun (Guiyang College of Traditional Chinese
Medicine). Primary extraction, isolation and purification of IA was
performed as described previously (24). IA was obtained (32.6 mg) from
fraction-5 (82.0 g) (Fr. 5). Based on the physical and spectral
data, it was identified as IA (25,26). The
purity of IA was analyzed by high performance liquid chromatography
(HPLC; Fig. S1 and Table SI). IA was dissolved in
chromatographic methanol to obtain a concentration of 1.00 mg/ml.
Chromatographic methanol alone was used as the control. HPLC
(LC-20AD; Shimadzu Corporation) was performed on a Waters SunFire
C18 column (150×4.6 mm, 5 µm) with mobile phase consisted of
methanol:water (65:35, V/V) at the flow rate of 1.0 ml/min at 210
nm and 23°C, and the injection volume was 10 µl.
Reagents
The primary reagents and kits used were the same as
those described previously (27).
Antibodies against phosphorylated (p-) PI3K (rabbit; cat. no.
3242), PI3K (rabbit; cat. no. 13666), p-Akt (rabbit; cat. no.
4060), Akt (rabbit; cat. no. 4691), cleaved-poly(ADP-ribose)
polymerase (PARP; rabbit; cat. no. 5625), PARP (rabbit; cat. no.
9532), X-linked inhibitor of apoptosis (xIAP; rabbit; cat. no.
14334), CDK2 (rabbit; cat. no. 10122), P53 (rabbit; cat. no.
10442), cyclin A2 (CCNA2; rabbit; cat. no. 18202), Caspase-3
(rabbit; cat. no. 9662) and GAPDH (rabbit; cat. no. 5174) were
purchased from Cell Signaling Technology, Inc. IA was dissolved in
DMSO and stored at a constant temperature of 4°C.
Cell culture
PC-3 cells were purchased from the American Type
Culture Collection (CRL-1435) and were immediately warmed to 37°C
and cultured in RPMI-1640 (Beijing Solarbio Science &
Technology Co., Ltd.) medium supplemented with 10% FBS (Gibco;
Thermo Fisher Scientific Inc.), 100 µg/ml penicillin and 100 µg/ml
streptomycin in a humidified incubator at 37°C with 5%
CO2.
Cell counting kit-8 (CCK-8)
assays
CCK-8 assays were used to analyze the proliferation
of PC-3 cells. PC-3 cells were cultured in 96-well plates with 150
µl RPMI-1640 for 24 h at 37°C. Control group cells were untreated
(0 µM IA), whereas the experimental group cells were treated with
5, 10, 20, 40 and 80 µM IA for 24, 48 and 72 h, at 37°C. The final
concentration of DMSO in the cells was <1% in all groups.
Subsequently, 10 µl CCK-8 solution was added, and cells were
cultured for a further 3 h, at which point 150 µl DMSO was added. A
microplate reader was used to measure the absorbance at 450 nm and
the IC50 values were calculated using GraphPad Prism
version 5.0 (GraphPad Software, Inc.). Experiments were repeated
three times.
EdU assay
EdU analysis was performed using the BeyoClick™
Edu-488 cell proliferation test kit (Beyotime Institute of
Biotechnology) according to the manufacturer's instructions to
observe individual proliferating cells. PC-3 cells
(5×105/well-5×106/well) were cultured in
6-well plates with 1 ml RPMI-1640 for 12 h at 37°C and then the
cells were treated with 0, 10, 20 and 40 µM IA for 24 h at 37°C.
Fluorescence microscopy (magnification, ×100) was used to visualize
the EdU positive cells. EdU analysis was performed using ImageJ
software v.1.50 (National Institutes of Health).
Phase-contrast microscopy
The morphological changes associated with apoptosis
of PC-3 cells were observed in cells following the aforementioned
treatments using phase-contrast light microscopy.
Apoptosis assay
Apoptosis was evaluated by fluorescence microscopy
and by flow cytometry. The methods of culture, administration and
treatment were the same as that described for the cell cycle
distribution analysis. For the microscopy analysis, treated cells
were washed twice with cold PBS, after which the cells were stained
with Hoechst 33342 at 4°C for 20 min, and a fluorescence microscope
was used to observe the changes in nuclear morphology. For the flow
cytometry analysis, the cells were centrifuged at 1,000 × g/min at
4°C for 5 min, resuspended in 100 µl buffer, stained with an
Annexin V-FITC/PI double staining apoptosis kit (Dojindo Molecular
Technologies, Inc.) at room temperature for 10 min in the dark and
finally resuspended in 400 µl buffer. Cell apoptosis was analyzed
using a flow cytometer and the data obtained were analyzed using
CytExpert version X (Beckman Coulter, Inc.).
Cell cycle assay
PC-3 cells were cultured in 6-well plates and
treated with different concentrations of IA (0, 10, 20 and 40 µM)
for 24 h. The cells were digested and collected, washed with cold
PBS, fixed using 70% cold ethanol at 4°C for 24 h. The cells were
then washed twice with cold PBS and treated with RNase. After
staining with PI, flow cytometry analysis was used to determine the
cell cycle phase distribution, and the data obtained were analyzed
using ModFit software (BD Biosciences).
Western blotting
Treated cells were lysed using lysis buffer (0.01%
(v/v) Triton X-100, 20 mM Tris and 150 mM NaCl). Cells were scraped
from the plates, and the lysates were centrifuged at
1.2×104 × g at 4°C for 5 min, to obtain the total
protein. A BCA protein assay was used to determine the protein
concentration, and 10% SDS-PAGE was used to resolve protein samples
(50 µg). The proteins were transferred to PVDF membranes, after
which blocking, and incubation with the primary and secondary
antibodies was performed as described by Mao et al (28). The following primary and secondary
antibodies were used: p-PI3K (1:1,000), PI3K (1:1,000), p-Akt
(1:1,000), Akt (1:1,000), cleaved-PARP (1:1,000), PARP (1:1,000),
xIAP (1:1,000), CDK2 (1:1,000), P53 (1:1,000), CCNA2 (1:1,000),
Caspase-3 (1:1,000), GAPDH (1:1,000) and goat anti-rabbit antibody
(1:1,000). Densitometry analysis was performed using ImageJ version
1.50 (National Institutes of Health).
Statistical analysis
Data are presented as the mean ± standard deviation.
Multiple comparisons were performed using a one-way ANOVA with the
Dunnett's post hoc test. A Student's t-test was used to analyze
differences between two groups. P<0.05 was considered to
indicate a statistically significant difference. Data were analyzed
using GraphPad Prism version 5.0 (GraphPad Software, Inc.).
Results
Effect of IA on the morphology and
proliferation of PC-3 cells
As evident from its chemical structure, IA is a
sesquiterpenes lactone (Fig. 1A). In
the present study, IA was isolated from C. cernuum L. with a
purity >98% (Fig. S1 and
Table SI). After being treated with
IA for 24 h, the morphological changes of PC-3 cells were observed
using an inverted light microscope, and images were captured from
the three independent experiments. The majority of the cells
exhibited an irregular shape, and were smaller in size when treated
with 10, 20 or 40 µM IA for 24 h compared with the control
untreated PC-3 cells (Fig. 1B).
Based on the images (Fig. 1B), it
was hypothesized that survival was decreased, partly as a result of
detachment and loss of adhesion to the surface, thus losing the
effects of adhesion. As treatment concentration increased, the
cells eventually lost adhesion to the surface and floated freely
(Fig. 1B).
PC-3 cells were treated with IA (5-80 µM) for 24, 48
or 72 h, and proliferation was examined using a CCK-8 assay. The
IC50 values of IA were 30.35±2.67, 14.54±0.11 and
6.61±0.59 µM for 24, 48 and 72 h in PC-3 cells (Fig. 1C). Together, these results suggest
that the effects of IA on PC-3 cells were time and dose-dependent.
In subsequent experiments, cells were treated with IA for 24 h. To
confirm the inhibitory effect of IA on cell proliferation, EdU
staining and Hoechst staining were used. The cells were treated
with 0, 10, 20 or 40 µM. The results indicated that IA treatment
for 24 h markedly inhibited the number of actively proliferating
PC-3 cells compared with the untreated control; the numbers of
green fluorescent cells labeled with EdU were markedly decreased
following IA administration (Fig.
1D).
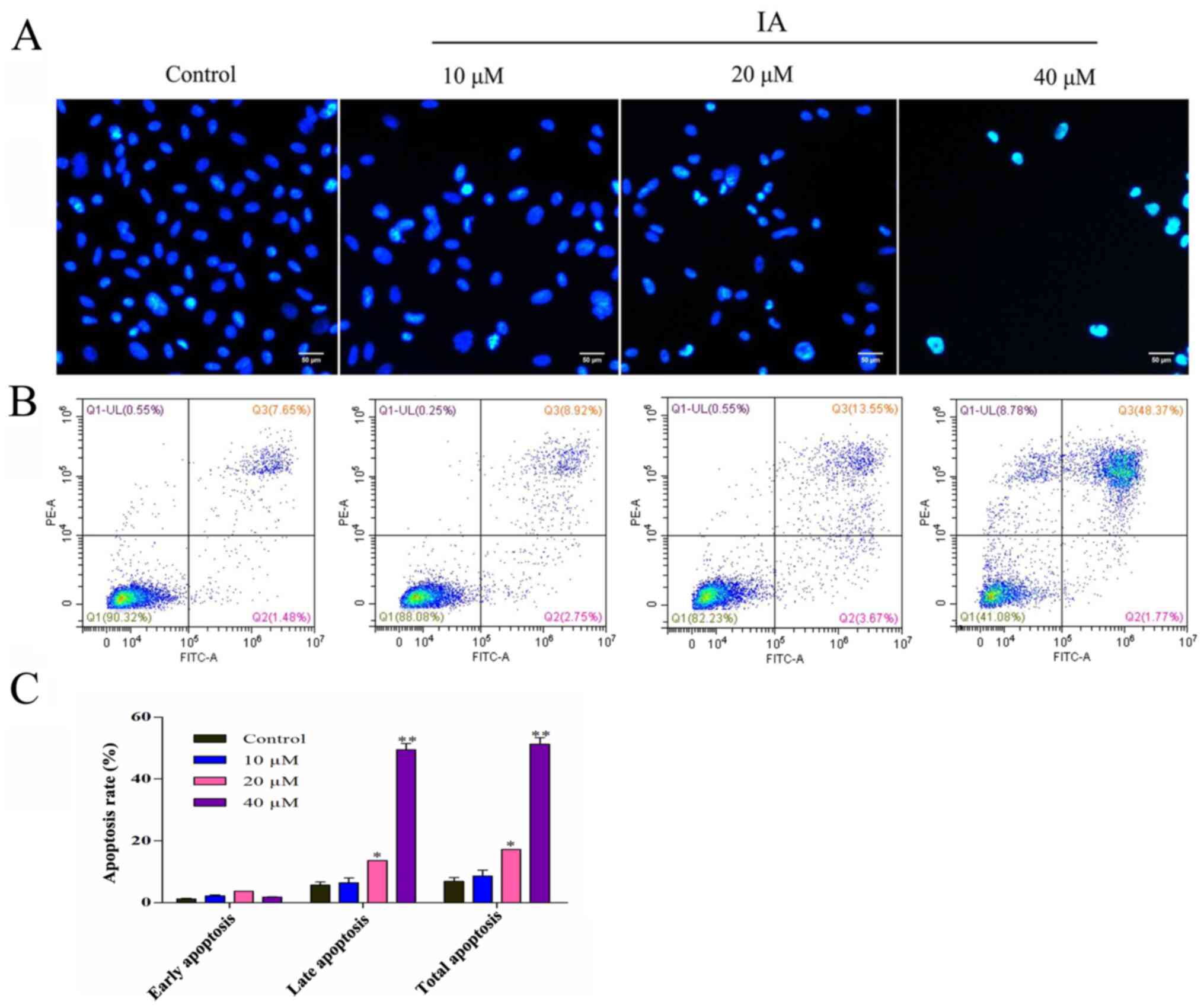
Effects of IA on apoptosis of PC-3
cells
Hoechst-stained cells were analyzed using
fluorescence microscopy to assess the nuclear changes and the
formation of apoptotic bodies in the cells treated with IA for 24
h. Compared with the control group, the different concentrations of
10, 20 or 40 µM IA treatment group exhibited a decreasing nuclei
trend (Fig. 2A).
Apoptosis was further evaluated by annexin V/PI
double staining and flow cytometry. PC-3 cells were either
untreated (control) or treated with 10, 20 or 40 µM IA for 24 h.
The apoptotic rate of PC-3 cells increased significantly following
IA treatment in a dose-dependent manner (Fig. 2B and C). The apoptotic rates of PC-3
cells treated with 0, 10, 20 or 40 µM IA were 9.13, 11.67, 17.22
and 50.14%, respectively. These results indicated that IA promoted
cell apoptosis in a dose-dependent manner.
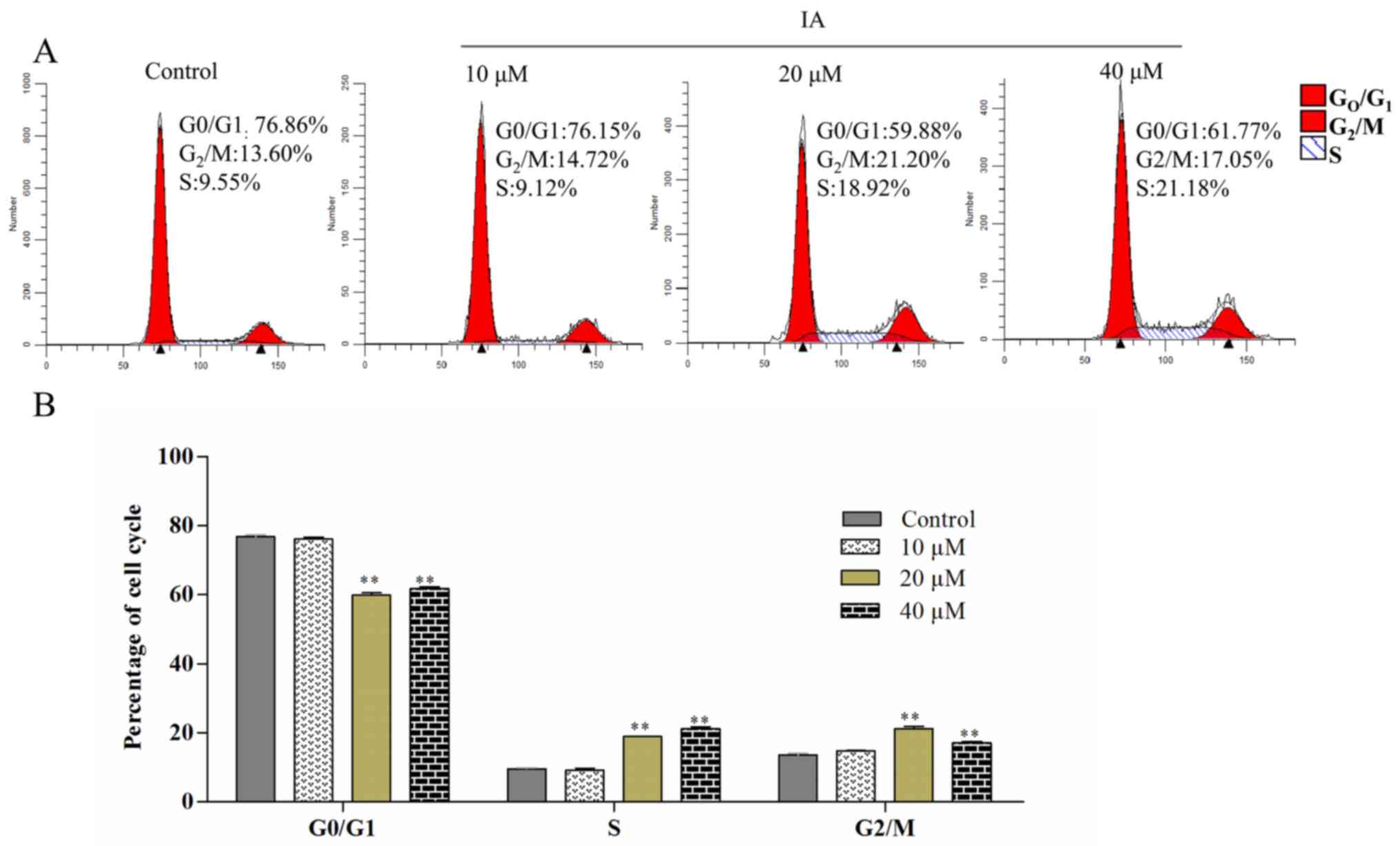
Effects of IA on cell cycle
progression in PC-3 cells
The effect of IA on PC-3 cell cycle phase
distribution was detected by flow cytometry. Compared with the
control group, the cells treated with different concentrations of
IA exhibited a decrease in the percentage of
G1/G0 phase cells and an increase in the
proportion of cells in the S phase (Fig.
3A and B). Following treatment with 0, 10, 20 or 40 µM IA for
24 h, the percentage of PC-3 cells in the S phase was 9.55±0.58,
9.12±1.06, 18.92±0.16, 21.18±1.05%, respectively. In addition, the
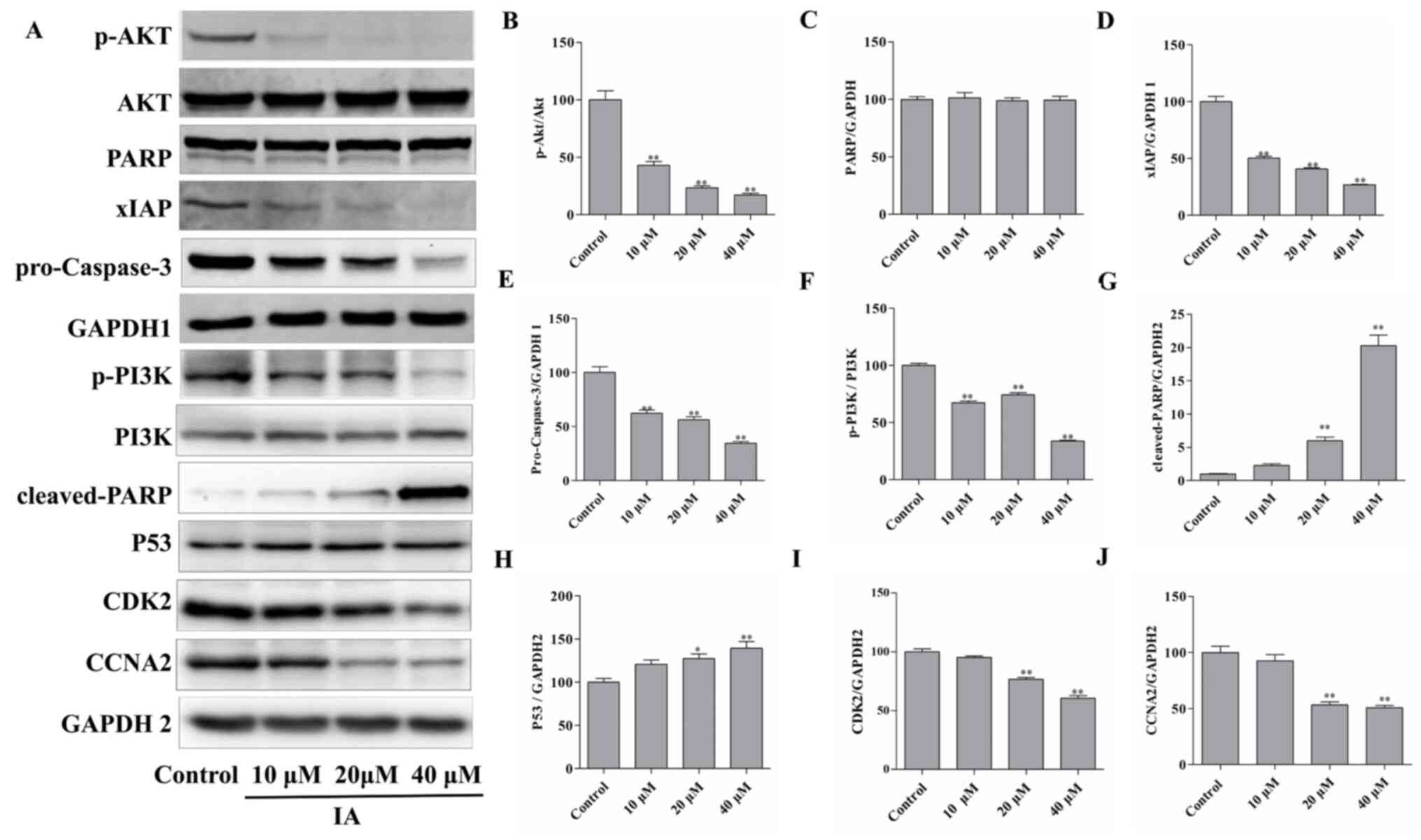
protein expression levels of cell cycle-related proteins were
examined by western blotting. As the concentration of IA was
increased, the protein expression levels of CDK2 and CCNA2 were
significantly decreased, whereas the protein expression levels of
P53 were significantly increased (Fig.
4A and H-J). These results suggested that the cell cycle was
arrested at the S phase, and the effects of IA were
dose-dependent.
Effects of IA on the PI3K/Akt/xIAP
signaling pathway in PC-3 cells
PC-3 cells were treated with 0, 10, 20 or 40 µM IA
for 24 h. Western blot analysis revealed that IA treatment resulted
in significant changes in the expression levels of key proteins
associated with the PI3K/Akt/xIAP signaling pathway. IA treatment
decreased the protein expression levels of p-PI3K, p-Akt,
pro-caspase-3 and xIAP significantly compared with the control
cells, whereas the levels of cleaved-PARP significantly increased,
all in a dose-dependent manner (Fig.
4A-G). Thus, it was hypothesized that IA may induce apoptosis
of PC-3 cells by inhibiting the activity of the PI3K/Akt/xIAP
signaling pathway.
Discussion
Prostate cancer is one of the primary types of
cancer threatening the health and life of men. There is an urgent
need to identify novel therapeutic agents to treat prostate cancer,
and a well-established source of potential compounds are natural
products. Recently, there has been increased interest in the
anticancer activity of various sesquiterpenoid lactones.
The results of the present study demonstrated that
the survival rates of PC-3 cells decreased following IA treatment,
in a dose and time-dependent manner. The proliferation of PC-3
cells was also reduced by IA treatment in a time-dependent manner.
Thus, for subsequent experiments, the PC-3 cells were treated with
0, 10, 20 or 40 µM IA for 24 h. After 24-h treatment, the total
number of PC-3 cells was decreased, as well as active
proliferation, both in a dose-dependent manner.
The pathogenesis of cancer has several aspects,
which are closely associated with cell cycle arrest and inhibition
of apoptosis. The results of the present study revealed that, in
cells treated with IA, the cell cycle was arrested in the S phase
and apoptosis was induced based on the appearance of apoptotic
bodies in the nuclei. The apoptotic rate increased in a
dose-dependent manner. Apoptosis is a crucial physiological process
and is required for normal development and maintenance of tissue
homeostasis. Additionally, downregulation of apoptosis has been
extensively shown to be crucially involved in the development of
cancer (29). The results of the
Hoechst staining analyses demonstrated that IA induced apoptosis in
PC-3 cells, in a dose-dependent manner. The sensitivity of prostate
cancer cells to IA, and the extent of apoptosis was confirmed by
flow cytometry. It was shown that exposure to 40 µM IA resulted in
48.37% of cells being in a late apoptotic or necrotic state, higher
than the 1.77% of early apoptotic cells, suggesting that IA
treatment accelerated cell death.
Several signal transduction pathways can regulate
the same physiological process, and thus a high degree of
coordination in cell growth and programmed cell death pathways is
required (30,31). Apoptosis signaling pathways have been
shown to be promising targets for the development of novel
anticancer drugs (32). It is
well-established that the PI3K/Akt pathway is a fundamental
intracellular signaling pathway involved in cell cycle regulation
(33), and there is a direct
relationship between cellular quiescence, proliferation and cancer
cell viability (34). Activation of
PI3K promotes the conversion of diphosphoinositide into
triphosphoinositide, resulting in activation of Akt and xIAP
(35). The inhibitory effects of IA
on the PI3K/Akt/xIAP pathway were examined in PC-3 cells, and the
expression levels of proteins associated with this pathway were
measured in cells treated with different concentrations of IA.
Firstly, IA treatment was demonstrated to inhibit the PI3K/Akt/xIAP
signaling pathway. In addition, P53 expression was significantly
upregulated, while CDK2 and CCNA2 were downregulated following IA
treatment, consistent with the cell cycle arrest and cell
proliferation inhibition phenotype obtained from the functional
assays. Furthermore, the downregulation of xIAP and the
upregulation of cleaved-PARP were consistent with the increased
apoptosis observed by flow cytometry. AKT is a key regulator of
cell growth and survival, which is essential for cancer growth
(36). xIAP, a known caspase
inhibitor, can inhibit apoptosis induced by different stimuli
(37). Apoptosis of cancer cells can
be induced by downregulation of p-Akt and xIAP (38,39).
The results of the present study demonstrated that
IA could inhibit the antiapoptotic mechanism of the cells through
the regulation of expression of various proteins, and the results
were consistent with previous studies (35,40,41) of
the PI3K/Akt/xIAP pathway. The present results highlight a
potential candidate compound for the management of prostate cancer
and provide a theoretical basis for the pathogenesis of prostate
cancer.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Dr Guowei Wang and
Dr Yunbin Jiang (College of Pharmaceutical Sciences, Southwest
University) for their helpful discussion and critical reading of
the manuscript.
Funding
This study was supported by the Science and
Technology Department of Guizhou Province [grant no. QKHJC
(2016)1001], the Education Department of Guizhou Province [grant
no. QJH KY (2020) 063] and the Science and Technology Bureau of
Anshun [grant no. ASKS (2018) 8].
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
MC conceived and designed the study. YH performed
the majority of the experiments and wrote the manuscript. CY and HG
performed experiments. JM and LZ analyzed the data. JM and CY
confirm the authenticity of all the raw data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Darnel AD, Behmoaram E, Vollmer RT, Corcos
J, Bijian K, Sircar K, Su J, Jiao J, Alaoui-Jamali MA and Bismar
TA: Fascin regulates prostate cancer cell invasion and is
associated with metastasis and biochemical failure in prostate
cancer. Clin Cancer Res. 15:1376–1383. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang J, Park JS, Wei Y, Rajurkar M, Cotton
JL, Fan Q, Lewis BC, Ji H and Mao J: TRIB2 acts downstream of
Wnt/TCF in liver cancer cells to regulate YAP and C/EBPα function.
Mol Cell. 51:211–225. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tye BK: MCM proteins in DNA replication.
Annu Rev Biochem. 68:649–686. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Matsuda T and Saika K: Comparison of time
trends in prostate cancer incidence (1973–2002) in Asia, from
cancer incidence in five continents, Vols IV–IX. Jpn J Clin Oncol.
39:468–469. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu X, Yu C, Bi Y and Zhang ZJ: Trends and
age-period-cohort effect on incidence and mortality of prostate
cancer from 1990 to 2017 in China. Public Health. 172:70–80. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhu X, Albertsen PC, Andriole GL, Roobol
MJ, Schröder FH and Vickers AJ: Risk-based prostate cancer
screening. Eur Urol. 61:652–661. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kallifatidis G, Hoy JJ and Lokeshwar BL:
Bioactive natural products for chemoprevention and treatment of
castration-resistant prostate cancer. Semin Cancer Biol.
40-41:160–169. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sarkar FH, Li Y, Wang Z and Kong D:
Cellular signaling perturbation by natural products. Cell Signal.
21:1541–1547. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Editorial Committee of Flora of China, .
Flora of China. 75. Beijing, China: Science Press; pp. 2961979
|
|
11
|
Kim MR, Hwang BY, Jeong ES, Lee YM, Yoo
HS, Chung YB, Hong JT and Moon DC: Cytotoxic germacranolide
sesquiterpene lactones from Carpesium triste var.
manshuricum. Arch Pharm Res. 30:556–560. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li XW, Weng L, Gao X, Zhao Y, Pang F, Liu
JH, Zhang HF and Hu JF: Antiproliferative and apoptotic
sesquiterpene lactones from Carpesium faberi. Bioorg Med
Chem Lett. 21:366–372. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dang H, Li H, Ma C, Wang Y, Tian J, Deng
L, Wang D, Jing X, Luo K, Xing W, et al: Identification of
Carpesium cernuum extract as a tumor migration inhibitor
based on its biological response profiling in breast cancer cells.
Phytomedicine. 64:1530722019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Koppula S, Kim WJ, Jiang J, Shim DW, Oh
NH, Kim TJ, Kang TB and Lee KH: Carpesium macrocephalum
attenuates lipopolysaccharide-induced inflammation in macrophages
by regulating the NF-κB/IκB-α, Akt, and STAT signaling pathways. Am
J Chin Med. 41:927–943. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim EJ, Jin HK, Kim YK, Lee HY, Lee SY,
Lee KR, Zee OP, Han JW and Lee HW: Suppression by a sesquiterpene
lactone from Carpesium divaricatum of inducible nitric oxide
synthase by inhibiting nuclear factor-kappaB activation. Biochem
Pharmacol. 61:903–910. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
State Administration of Traditional
Chinese Medicine. Chinese materia medica. Shanghai Scientific and
Technical Publishers; Shanghai, China: pp. 7601999
|
|
17
|
Chung IM and Moon HI: Antiplasmodial
activities of sesquiterpene lactone from Carpesium cernum. J
Enzyme Inhib Med Chem. 24:131–135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kim JJ, Chung IM, Jung JC, Kim MY and Moon
HI: In vivo antiplasmodial activity of 11(13)-dehydroivaxillin from
Carpesium ceruum. J Enzyme Inhib Med Chem. 24:247–250. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang JP, Wang GW, Tian XH, Yang YX, Liu
QX, Chen LP, Li HL and Zhang WD: The genus Carpesium: A review of
its ethnopharmacology, phytochemistry and pharmacology. J
Ethnopharmacol. 163:173–191. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ma JP, Tan CH and Zhu DY: Glycosidic
constituents from Carpesium cernuum L. J Asian Nat Prod Res.
10:565–569. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang GW, Qin JJ, Cheng XR, Shen YH, Shan
L, Jin HZ and Zhang WD: Inula sesquiterpenoids: Structural
diversity, cytotoxicity and anti-tumor activity. Expert Opin
Investig Drugs. 23:317–345. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang FY, Li XQ, Qi S, Yao S, Ke CQ, Tang
CP, Liu HC, Geng MY and Ye Y: Sesquiterpene lactones from Inula
cappa. Phytochem Lett. 5:639–642. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wu JW, Tang CP, Cai YY, Ke CQ, Lin LG, Yao
S and Ye Y: Cytotoxic germacrane-type sesquiterpene lactones from
the whole plant of Inula cappa. Chin Chem Lett. 28:927–930.
2017. View Article : Google Scholar
|
|
24
|
Yan C, Zhang WQ, Sun M, Liang W, Wang TY,
Zhang YD and Ding X: Carpescernolides A and B, rare oxygen
bridge-containing Sesquiterpenes lactones from Carpesium
cernuum. Tetrahedron Lett. 59:4063–4066. 2018. View Article : Google Scholar
|
|
25
|
Zhang T, Si JG, Zhang QB, Ding G and Zou
ZM: New highly oxygenated germacranolides from Carpesium
divaricatum and their cytotoxic activity. Sci Rep. 6:272372016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bohlmann F, Singh P and Jakupovic J:
Further ineupatorolide-like germacranolides from Inula
cuspidata. Phytochemistry. 21:157–160. 1982. View Article : Google Scholar
|
|
27
|
Wang R, Dong ZY, Lan XZ, Liao ZH and Chen
M: Sweroside alleviated LPS-induced inflammation via SIRT1
mediating NF-κB and FOXO1 signaling pathways in RAW264.7 cells.
Molecules. 24:8722019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mao J, Yi M, Tao Y, Huang Y and Chen M:
Costunolide isolated from Vladimiria souliei inhibits the
proliferation and induces the apoptosis of HepG2 cells. Mol Med
Rep. 19:1372–1379. 2019.PubMed/NCBI
|
|
29
|
Yin PH, Liu X, Qiu YY, Cai JF, Qin JM, Zhu
HR and Li Q: Anti-tumor activity and apoptosis-regulation
mechanisms of bufalin in various cancers: New hope for cancer
patients. Asian Pac J Cancer Prev. 13:5339–5343. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Guo Y, Balasubramanian B, Zhao ZH and Liu
WC: Marine algal polysaccharides alleviate aflatoxin B1-induced
bursa of Fabricius injury by regulating redox and apoptotic
signaling pathway in broilers. Poult Sci. 100:844–857. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu WC, Guo Y, Zhao ZH, Jha R and
Balasubramanian B: Algae-derived polysaccharides promote growth
performance by improving antioxidant capacity and intestinal
barrier function in broiler chickens. Front Vet Sci. 7:6013362020.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rao L and White E: Bcl-2 and the ICE
family of apoptotic regulators: Making a connection. Curr Opin
Genet Dev. 7:52–58. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jin D, Yang JP, Hu JH, Wang LN and Zou J:
MCP-1 stimulates spinal microglia via PI3K/Akt pathway in bone
cancer pain. Brain Res. 1599:158–167. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Petrulea MS, Plantinga TS, Smit JW,
Georgescu CE and Netea-Maier RT: PI3K/Akt/mTOR: A promising
therapeutic target for non-medullary thyroid carcinoma. Cancer
Treat Rev. 41:707–713. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pramanik KC, Kudugunti SK, Fofaria NM,
Moridani MY and Srivastava SK: Caffeic acid phenethyl ester
suppresses melanoma tumor growth by inhibiting PI3K/AKT/XIAP
pathway. Carcinogenesis. 34:2061–2070. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vyas VK, Ghate M and Goel A: Pharmacophore
modeling, virtual screening, docking and in silico ADMET analysis
of protein kinase B (PKB β) inhibitors. J Mol Graph Model.
42:17–25. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Deveraux QL, Takahashi R, Salvesen GS and
Reed JC: X-linked IAP is a direct inhibitor of cell-death
proteases. Nature. 388:300–304. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sasaki H, Sheng Y, Kotsuji F and Tsang BK:
Down-regulation of X-linked inhibitor of apoptosis protein induces
apoptosis in chemoresistant human ovarian cancer cells. Cancer Res.
60:5659–5666. 2000.PubMed/NCBI
|
|
39
|
Han B, Jiang P, Li Z, Yu Y, Huang T, Ye X
and Li X: Coptisine-induced apoptosis in human colon cancer cells
(HCT-116) is mediated by PI3K/Akt and mitochondrial-associated
apoptotic pathway. Phytomedicine. 48:152–160. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chang F, Lee JT, Navolanic PM, Steelman
LS, Shelton JG, Blalock WL, Franklin RA and McCubrey JA:
Involvement of PI3K/Akt pathway in cell cycle progression,
apoptosis, and neoplastic transformation: A target for cancer
chemotherapy. Leukemia. 17:590–603. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang W, Shang X, Zhang C, Gao X, Robinson
B and Liu J: The effects of carvedilol on cardiac function and the
AKT/XIAP signaling pathway in diabetic cardiomyopathy rats.
Cardiology. 136:204–211. 2017. View Article : Google Scholar : PubMed/NCBI
|