Introduction
Gastric cancer (GC) refers to the deleterious
inflammation of the stomach wall, and this type of tumor accounted
for ~8.2% of tumor-associated deaths worldwide in 2018 (1). GC is a multifactorial disease that is
associated with the environment, including Helicobacter
pylori infection (2), smoking
and alcohol consumption (3). To
date, treatment options such as surgery, radiotherapy, chemotherapy
and medication are the most frequently therapeutic strategies for
patients with GC; however, these methods have not improved the
recurrence and the poor prognosis of GC (4). Gene therapy has also been utilized in
recent years to treat patients with GC (5). However, numerous target genes in GC
remain unknown. By exploring the molecular mechanism of GC, the
clinical outcomes of patients suffering from GC can be
improved.
The cadherin 3 (CDH3) gene is located on chromosome
16q22.1 and consists of 18 exons. It is found in a six-cadherin
bundle, and this calcium-dependent gene is comprised of five
extracellular repeats, a cytoplasmic tail and a transmembrane
region (6). The aberrant expression
of CDH3 has been frequently observed in various types of cancer,
including colorectal (7), pancreatic
(8) and esophageal cancer (9). More specifically, CDH3 has been
demonstrated to increase cell adhesion and invasion (10). However, only a few studies have been
conducted to investigate the tumorigenic role of CDH3 in GC cells
(11,12), revealing that CDH3 expression was
upregulated in GC tissues. Therefore, it is noteworthy to
investigate the impacts of CDH3 on GC.
MicroRNAs (miRNAs/miRs) can be referred to as
non-coding protein sequences with the capability of regulating,
decoding and controlling gene expression (13). Previous studies have revealed that
miRNAs regulate cell proliferation, invasion and apoptosis, as well
as serving a pivotal role in tumor progression (14,15). For
instance, miR-665 has been associated with lung cancer progression
by increasing cell proliferation, migration and invasion (16). Some studies have suggested that
miR-665 may be used as a potential molecular target for various
types of cancer, including pancreatic (17), breast (18) and ovarian carcinoma (19). Additionally, several studies on GC
have suggested that by targeting different genes, miR-665 may act
both as an oncogene and as a tumor inhibitor (20,21).
However, the mechanism of interaction between miR-665 and CDH3 in
GC requires further investigation.
The present study aimed to explore the effect of the
miR-665/CDH3 interactome on the pathogenesis of GC. The objective
included the investigation of potential bio-targets for GC
treatment. It was hypothesized that miR-665 could suppress GC cells
by targeting CDH3. The current study may be relevant in terms of
providing a theoretical basis for GC clinical therapy.
Materials and methods
Bioinformatics analysis
The mRNA expression profiles GSE118916 (22) and GSE79973 (23) were obtained from the Gene Expression
Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/gds/?term=), a public
database that stores gene expression microarrays. The upregulated
differentially expressed genes (DEGs) were screened from GSE118916
and GSE79973 using log2-fold-change (logFC)>1 and adjusted (adj)
P<0.05. STRING (https://string-db.org/) was used to analyze the Gene
Ontology (GO) enrichment of the upregulated DEGs. The expression
levels of the key genes in stomach adenocarcinoma (STAD) were
analyzed using The Cancer Genome Atlas (TCGA) database (https://portal.gdc.cancer.gov/).
Clinical samples and cell lines
All tissue samples, including tumor and non-tumor
specimens, were obtained from 30 patients with GC (16 male and 14
female; median age, 60 years; age range 29–72 years) at Henan
Provincial People's Hospital (Zhengzhou, China) between January
2019 and January 2020. All patients signed informed consent forms
to participate in the study. The ethical protocols of the present
study were approved by the Ethics Committee of Henan Provincial
People's Hospital. The diagnosis of GC included a clinical
examination, blood sampling, endoscopy (when clinically indicated)
and computed tomography scanning of the chest, abdomen and pelvis.
The tumor tissues were all confirmed by endoscopic biopsy or
surgical specimens. Patients with a history of cancer or severe
clinical symptoms and genetic diseases, and patients who received
preoperative radiochemotherapy were excluded from the study.
Non-tumor specimens were ≥1.5 cm from the tumor margins. All
collected samples were stored at the recommended temperature
(−80°C). The clinical features of patients who participated in the
study are shown in Table I. The
tumors were staged following the tumor-node-metastasis (TNM)
staging system of the International Union Against Cancer (24). CDH3 was divided into high and low
expression groups according to the median expression level of CDH3
(5.848). The association between CDH3 expression and clinical
characteristics in patients with GC are shown in Table SI.
 | Table I.Clinical characteristics of 30
patients with gastric cancer. |
Table I.
Clinical characteristics of 30
patients with gastric cancer.
|
Characteristics | N | Percentage, % |
|---|
| Age, years |
|
|
|
>60 | 18 | 60.0 |
|
≤60 | 12 | 40.0 |
| Sex |
|
|
|
Male | 16 | 53.3 |
|
Female | 14 | 46.7 |
| Tumor size, cm |
|
|
|
>5 | 19 | 63.3 |
| ≤5 | 11 | 36.7 |
| Histological
grade |
|
|
|
Low | 13 | 43.3 |
|
High | 17 | 56.7 |
| TNM stage |
|
|
| I | 5 | 16.7 |
| II | 7 | 23.3 |
|
III | 14 | 46.7 |
| IV | 4 | 13.3 |
| Lymph nodes
status |
|
|
|
Positive | 19 | 63.3 |
|
Negative | 11 | 36.7 |
|
Differentiation |
|
|
|
Poor | 17 | 56.7 |
|
Well/moderate | 13 | 43.3 |
All cell lines used were purchased from the American
Type Culture Collection, including GC cell lines (HGC-27, GTL-16,
MKN74 and AGS) and the gastric epithelial cell line (GES-1). MKN74
cells were maintained in F-12K medium, GES-1 cells were maintained
in RPMI-1640 medium and AGS, HGC-27 and GTL-16 cells were kept in
DMEM (Gibco; Thermo Fisher Scientific, Inc.). The media were
supplemented with 100 U/ml penicillin and 10% FBS (Gibco; Thermo
Fisher Scientific, Inc.). Cells were incubated at 37°C in an
atmosphere containing 5% CO2.
RNA extraction and reverse
transcription-quantitative (RT-q)PCR
RNA extraction from tissues and cells was performed
using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). cDNA was obtained using the PrimeScript First
Strand cDNA Synthesis kit (Takara Bio, Inc.) according to the
manufacturer's protocol. Subsequently, qPCR was performed using
SYBR® Premix Ex Taq™ (Takara Bio, Inc.) according to the
manufacturer's protocol. The thermocycling conditions were as
follows: Denaturation at 95°C for 20 sec, followed by 40 cycles of
denaturation at 95°C for 1 min and annealing/extension at 60°C 20
sec. The expression levels of miR-665, CDH3, collagen type XVIII α1
chain (COL18A1) and fibroblast activation protein (FAP) were
measured using the 2−ΔΔCq method, and U6 or
GAPDH were used as an endogenous reference for miRNA or mRNA,
respectively. The relative expression levels of miR-665, CDH3,
COL18A1 or FAP were reported in relation to an internal control
gene called ΔCt. The ΔCT and ΔΔCT values were calculated using the
following mathematical formulas:
ΔCt=CtmiRNA-126/CDH3-CtU6/GAPDH and
ΔΔCt=ΔCtcase-ΔCtcontrol (25). All the primer sequences used in the
study are listed in Table II.
 | Table II.Primer sequences used for reverse
transcription-quantitative PCR. |
Table II.
Primer sequences used for reverse
transcription-quantitative PCR.
| Gene | Primer sequences
(5′-3′) |
|---|
| miR-665 | Forward:
GCCGAGACCAGGAGGCUGA |
|
| Reverse:
CTCAACTGGTGTCGTGGA |
| U6 | Forward:
ATTGGAACGATACAGAGAAGATT |
|
| Reverse: GGA
ACGCTTCACGAATTTG |
| CDH3 | Forward:
CAGGTGCTGAACATCACGGACA |
|
| Reverse:
CTTCAGGGACAAGACCACTGTG |
| COL18A1 | Forward:
GGAGAGATTGGCTTTCCTGGAC |
|
| Reverse:
CCTCATGCCAAATCCAAGGCTG |
| FAP | Forward:
GGAAGTGCCTGTTCCAGCAATG |
|
| Reverse:
TGTCTGCCAGTCTTCCCTGAAG |
| GAPDH | Forward:
AGCCACATCGCTCAGACAC |
|
| Reverse:
GCCCAATACGACCAAATCC |
Western blotting
GC cells were lysed using RIPA lysis buffer (Sangon
Biotech Co., Ltd.). Total protein concentration was quantified
using the BCA method. Proteins (30 µg/lane) were separated via 10%
SDS-PAGE and then transferred to PVDF membranes, which were blocked
with 5% BSA (Gibco; Thermo Fisher Scientific, Inc.) for 2 h at
25°C, and washed three times with TBS-0.1% Tween-20. After blocking
the membranes, primary antibodies against CDH3 (1:1,000; cat. no.
ab242060; Abcam) and GAPDH (1:2,000; cat. no. ab181602; Abcam) were
used to incubate the membranes overnight at 4°C. Subsequently,
HRP-conjugated goat anti-rabbit IgG (1:5,000; cat. no. ab97051;
Abcam) secondary antibody was incubated for 1 h at 25°C. The
protein signal was subsequently visualized using an ECL system
(Bio-Rad Laboratories, Inc.). Finally, the quantification of the
density of each band was performed using Gel-Pro Analyzer 4.0
(Media Cybernetics, Inc.).
Cell transfection
The small interfering RNA (siRNA) used as a
non-targeting negative control (si-NC) and the siRNA against CDH3
(si-CDH3) were obtained from Shanghai Tuoran Biological Technology
Co., Ltd. For CDH3 overexpression, full-length CDH3 was synthesized
and transformed into pcDNA3.1 (OE-CDH3), provided by Shanghai
Tuoran Biological Technology Co., Ltd., and the corresponding empty
vector was used as a NC (OE-NC). Using Lipofectamine®
3000 Transfection Reagent (Invitrogen; Thermo Fisher Scientific,
Inc.), the transfection of AGS and HGC-27 cells was performed using
50 nM siRNA or 2 µg/ml pcDNA3.1 in 24-well plates at 37°C for 48 h.
Subsequent experiments were performed 48 h after transfection. The
next procedure involved the selection of stable cell lines with
puromycin (4 µg/ml). Additionally, the miR-665 inhibitor, miR-665
mimic and the corresponding non-targeting NCs (mimic-NC and
inhibitor-NC) were obtained from Genewiz, Inc. The sequences of
si-CDH3, miR-665 inhibitor, miR-665 mimic and their corresponding
NCs are listed in Table III.
HGC-27 and AGS cells were seeded into 24-well plates
(2×105 cells/well). After the density of the cells was
50%, they were transfected with the miR-665 mimic, miR-665
inhibitor or corresponding negative controls at a concentration of
50 nM using Lipofectamine 3000 Transfection Reagent for 48 h at
37°C. Subsequent experiments were performed 48 h after
transfection.
 | Table III.Oligonucleotide sequences used for
cell transfection. |
Table III.
Oligonucleotide sequences used for
cell transfection.
|
Oligonucleotide | Primer sequences
(5′-3′) |
|---|
| si-CDH3 | Forward:
UUCAACAGCAACCAGCCUGUUUCCU |
|
| Reverse:
AGGAAACAGGCUGGUUGCUGUUGAA |
| si-NC | Forward:
CGGAAGGCCUAAUGCCGAAdTdT |
|
| Reverse:
UUCGGCAUUAGGCCUUCCGdTdG |
| miR-665
inhibitor |
AGGGGCCUCAGCCUCCUGGU |
| inhibitor-NC |
CAGUACUUUUGUGUAGUACAA |
| miR-665 mimic | Forward:
CAGCAGCACACUGUGGUUUGU |
|
| Reverse:
AGGGACCUCAGCCUCCUGGUUU |
| mimic-NC | Forward:
UUCUCCGAACGUGUCACGUTT |
|
| Reverse:
ACGUGACACGUUCGGAGAATT |
MTT assay
MTT colorimetric assay was used to detect cell
viability. HGC-27 and AGS cells were cultured into 96-well plates
at a density of 5×103 cells/well. MTT (5 mg/ml) was then
added to each well at four time points (0, 24, 48 and 72 h). After
4 h incubation at 37°C, acid isopropanol was added to each well.
The cell samples were then mixed until the dark blue crystals
dissolved completely. A microplate reader was used to read the
absorbance at 570 nm. The triplicate repeated readings for each
sample were averaged, and the medium background was subtracted from
the analytical readings to correct the absorbance.
BrdU ELISA assay
The BrdU Cell Proliferation Assay kit (cat. no.
6813; Cell Signaling Technology, Inc.) was used to detect cell
proliferation according to the manufacturer's protocol. First,
2×104 HGC-27 and AGS cells were cultured into each well
of a 96-well plate. The next day, the cells were serum-starved
overnight. Subsequently, the serum was added to the cells for 8 h.
After that, cells were incubated for 8–12 h without removing the
treatment media. The absorbance value was subsequently recorded at
450 nm using a microplate reader. BrdU was incorporated into the
DNA of dividing cells, and the intensity of the luminescence
recorded using a microplate reader was proportional to the amount
of incorporated BrdU in the cells to evaluate cell
proliferation.
Apoptosis assay
The FITC Annexin V Apoptosis Detection kit (BD
Biosciences) was used for apoptosis analysis according to the
manufacturer's protocol. After the cell lines were collected in 100
µl ice-cold PBS (6×104 cells), they were suspended with
the binding buffer (100 µl) and treated with Annexin V-FITC (5 µl)
and propidium iodide (5 µl) in the dark at 20–22°C for 15 min. The
next procedure was the addition of 400 µl of the same binding
buffer to the aforementioned binding buffer (100 µl). The cells
were detected and characterized by flow cytometry (FACSCalibur;
Becton, Dickinson and Company), and cells were analyzed using
FlowJo v10.6.2 (FlowJo LLC). Early apoptosis (Annexin
V-FITC+/PI−; Q4) and late apoptosis (Annexin
V-FITC+/PI+; Q2) were considered as the
apoptosis rate.
Caspase activity assay
This assay was performed using the caspase-3
activity assay kit (cat. no. 5723; Cell Signaling Technology, Inc.)
according to the manufacturer's protocol. The first step was
seeding AGS and HGC-27 cells into the 96-well plates
(2×104 cells/well) and culturing the cells at the
suggested operating conditions (37°C and 80% density). The
caspase-3 assay loading solution was then prepared by adding
N-Acetyl-Asp-Glu-Val-Asp-p-Nitroanilide (DEVD-pNA) substrates and
DL-Dithiothreitol (DTT) reagents. This preparation was performed
according to the manufacturer's instructions. After the cells were
collected, they were suspended with ice-cold cell lysis buffer
included in the aforementioned kit for 10 min and then incubated on
ice. Then, the caspase-3 assay loading solution (100 µl/well) was
added to the cells, and the mixture was incubated at 37°C for 2 h.
The absorbance at 405 nm was measured using a microplate
reader.
Cell adhesion assay
HGC-27 and AGS cells were transferred to a DMEM
containing 10% FBS and seeded into 96-well plates (2×104
cells/well). A total of ~30 µl/well of Collagen I solution (40
µg/ml) (Chrono-log Corporation) was transferred to the 96-well
plate and incubated at 4°C for 12 h. The next step was the removal
of the solution, followed by air-drying the plate at 20–22°C. The
cells were then washed with serum-free DMEM and cultured for 8 h at
37°C in serum-free DMEM. To dissociate them, 10 mM EDTA was added
to the cells for 10 min at 25°C. The cells were then washed three
times with PBS to remove EDTA and were suspended in DMEM with 0.1%
BSA (2×105 cells/ml; Gibco; Thermo Fisher Scientific,
Inc.). Subsequently, 100 µl cell suspension was added to the
96-well plate with air-dried Collagen I, and the cells were
incubated at 37°C for 60 min. After the cells adhered to the plate
surface, DMEM (100 µl) was added to eliminate other non-adherent
cells. Next, the cells were incubated at 37°C for 1 h with DMEM
containing 10% FBS. Subsequently, the MTT substrate (10 µl) was
added to each well, and the cells were incubated for 2 h at 30°C.
DMSO (100 µl) was then added to lyse the cells. Finally, the
absorbance was measured at 570 nm using a microplate reader. The
absorbance was positively associated with the number of adherent
cells, so the optical density value was used to evaluate the
adhesion ability of cells.
Luciferase assay
The miRNA upstream of CDH3 was predicted using
bioinformatics softwares, including GSE93415 (26) from Gene Expression Omnibus DataSets
(https://www.ncbi.nlm.nih.gov/gds/?term=), TargetScan
(http://www.targetscan.org/), starBase
(http://starbase.sysu.edu.cn/panCancer.php) and Venny
2.1.0 (http://bioinfogp.cnb.csic.es/tools/venny/index.html).
The pmiRGLO plasmid vector (Promega Corporation) used in the
present study contained both the firefly luciferase reporter gene
and the internal control gene, Renilla luciferase. The
pmiRGLO CDH3 3′-untranslated regions (3′-UTRs) wild-type (WT)
vectors and pmiRGLO CDH3 3′-UTR mutated (MUT) vectors with the
miR-665 binding sites replaced by a random nucleotide sequence were
constructed. A 24-well plate was then used to seed AGS and HGC-27
cells (2×105 cells/well). The next step was the
co-transfection of the cells with pmiRGLO CDH3-MUT or pmiRGLO
CDH3-WT and with either miR-665 mimic or mimic-NC using
Lipofectamine 3000 Transfection Reagent. After collecting the cells
transfected for 48 h, Renilla luciferase and firefly
luciferase activities were detected using the Dual Luciferase
Reporter Assay System (Thermo Fisher Scientific, Inc.). Firefly
luciferase activity was normalized to that of Renilla
luciferase activity.
Statistical analysis
GraphPad Prism 7.0 (GraphPad Software, Inc.) was
used for data analysis. To compare two variables in tissues, paired
Student's t-test was utilized. To compare two groups in cell
experiments, unpaired Student's t-test was used. To compare
multiple groups, one-way ANOVA with Dunnett's or Tukey's post-hoc
test was employed. Pearson correlation analysis was applied to
assess the correlation between CDH3 and miR-665 expression.
Fisher's exact test was used to determine the association between
CDH3 expression and clinicopathological variables. Each experiment
was performed thrice, and results were presented as the mean ±
standard error. P<0.05 was considered to indicate a
statistically significant difference.
Results
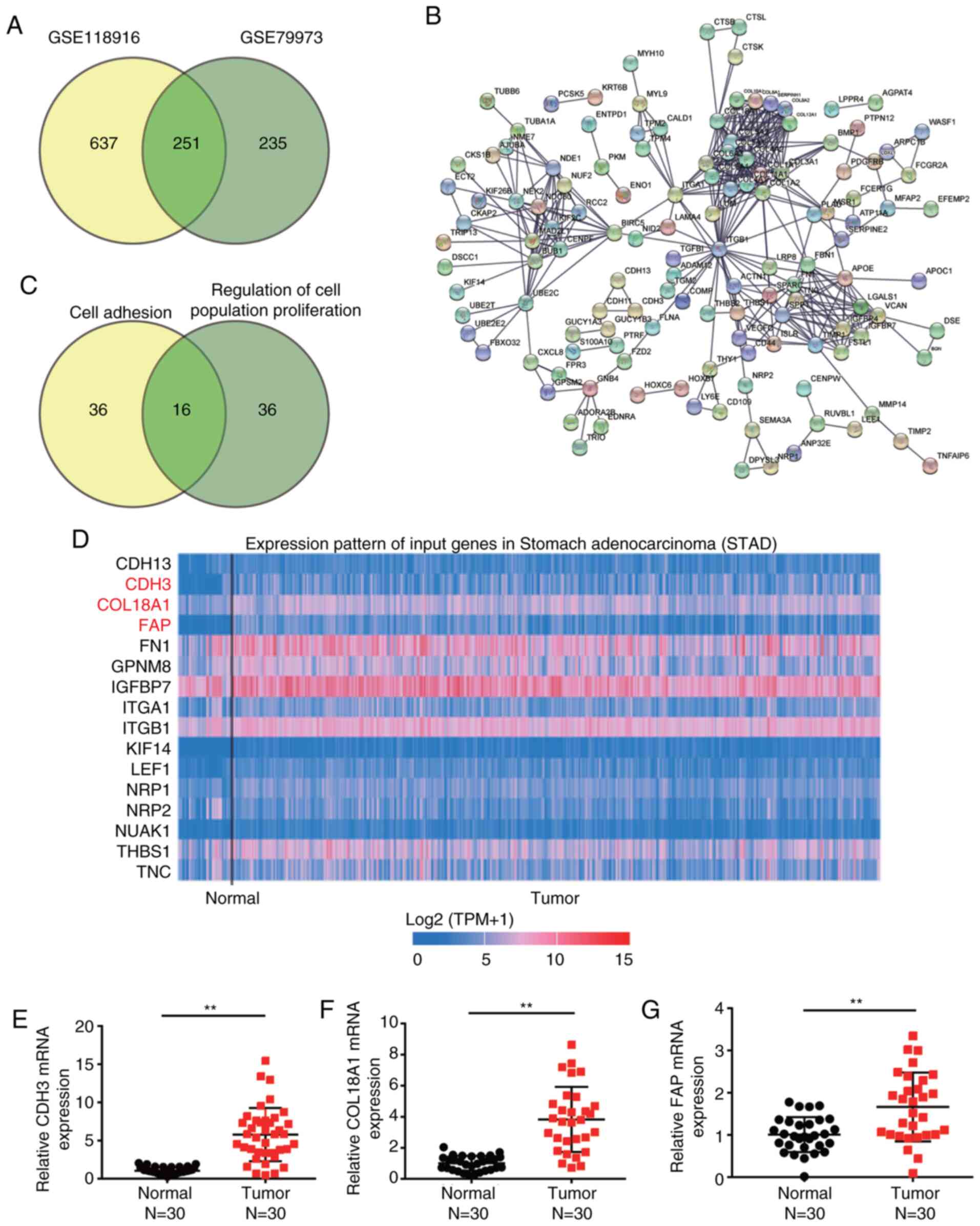
Identification of key genes involved
in GC
Two mRNA expression profiles (GSE118916 and
GSE79973) were downloaded from the GEO database. The profiles were
then applied to filter the DEGs involved in GC. A total of 251
upregulated DEGs were overlapped between GSE79973 and GSE118916,
with logFC values >1 and adj. P<0.05 (Fig. 1A). The 251 overlapped DEGs were
uploaded to STRING for GO enrichment analysis (Fig. 1B). After performing GO enrichment
analysis, ‘cell adhesion’ and ‘regulation of cell population
proliferation’ were significantly enriched following GO analysis,
and 16 genes were involved in these two biological processes
(Fig. 1C). By using TCGA analysis,
CDH3, COL18A1 and FAP were found to be significantly upregulated in
GC samples compared with in non-tumor samples (Fig. 1D). To identify and investigate these
key genes, the expression levels of CDH3, COL18A1 and FAP were
further detected in clinical tissues, revealing that they were all
upregulated in tumor tissues compared with in normal tissues
(Fig. 1E-G). Since CDH3 exhibited
the highest upregulation, it was selected to be further explored in
subsequent experiments.
CDH3 expression is increased in GC
cells
The association between CDH3 expression and clinical
features in patients with GC was then analyzed. CDH3 expression was
associated with tumor size, histological grade, TNM stage and
advanced lymph nodes status (Table
SI). However, CDH3 expression was not associated with other
clinicopathological features, such as age, sex, differentiation,
smoking, drinking alcohol and H. pylori infection.
Furthermore, the expression levels of CDH3 were confirmed in the
gastric epithelial cells (GES-1) and the gastric adenocarcinoma
cells (AGS, MKN74, GTL-16 and HGC-27). CDH3 expression was
significantly increased in all GC cells compared with in GES-1
cells (Fig. 2A). Western blotting
was employed to analyze CDH3 protein expression. Consistent with
gene expression, CDH3 protein expression in GC cells was
significantly increased compared with in GES-1 cells (Fig. 2B). Since HGC-27 and AGS cell lines
exhibited the highest CDH3 expression, they were selected to
perform subsequent experiments. The transfection of si-CDH3 and
OE-CDH3 in AGS and HGC-27 cells was performed to assess the
function of CDH3 expression in GC cells. The transfection
efficiency was confirmed using RT-qPCR and western blotting. The
results revealed that overexpression of CDH3 led to a significant
upregulation of CDH3 mRNA and protein expression, while si-CDH3 led
to a significant downregulation of CDH3 mRNA and protein expression
(Figs. S1A, 2C and D).
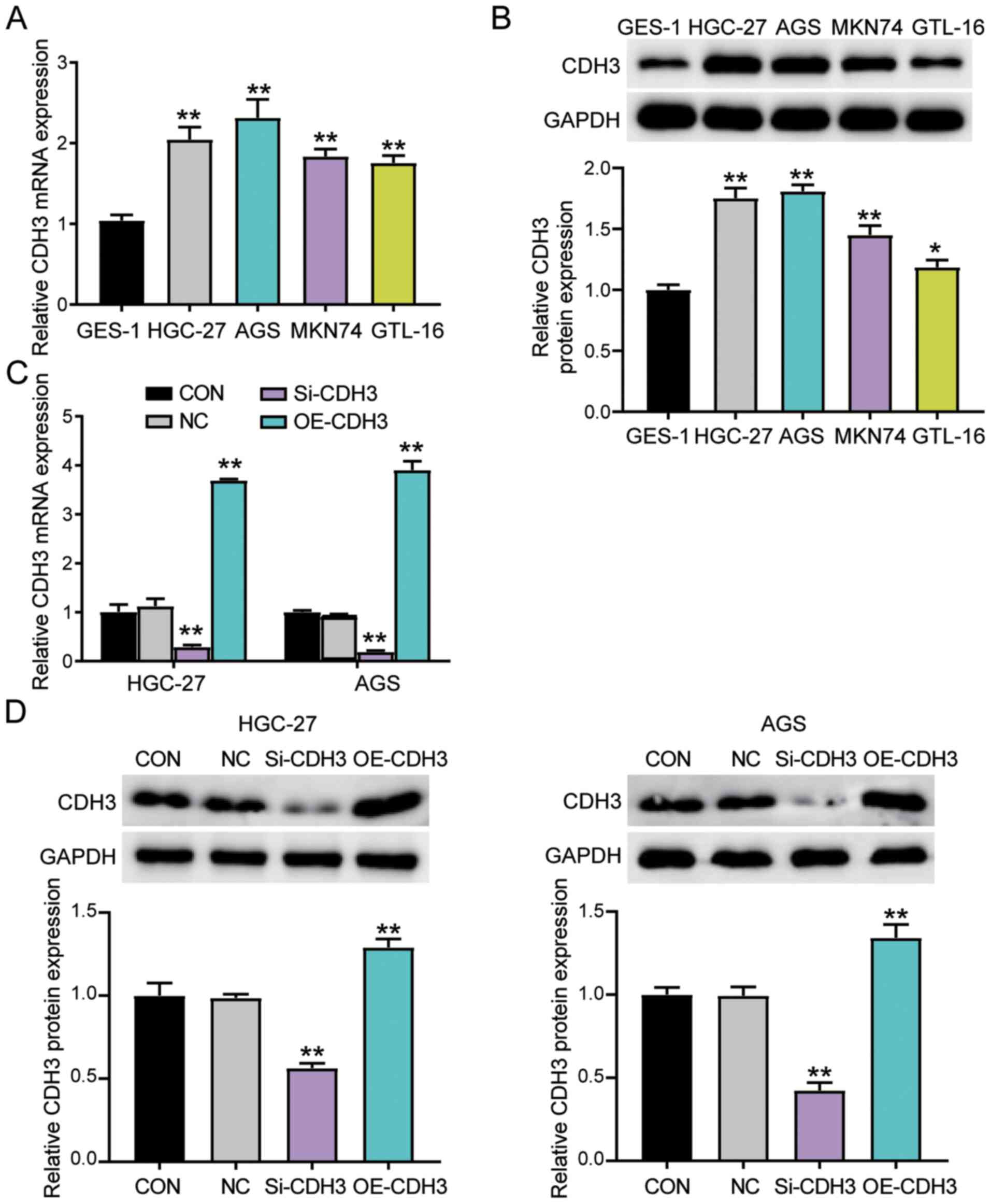 | Figure 2.Upregulation of CDH3 expression in GC
cells. (A) RT-qPCR detection of CDH3 mRNA expression in gastric
epithelial cells (GES-1) and gastric adenocarcinoma cell lines
(AGS, MKN74, GTL-16 and HGC-27). **P<0.001 vs. GES-1 cells. (B)
Measurement of CDH3 protein expression in GES-1, AGS, MKN74, GTL-16
and HGC-27 cell lines via western blotting. *P<0.05 and
**P<0.001 vs. GES-1 cells. (C) CDH3 mRNA expression was
identified using RT-qPCR in HGC-27 and AGS cells transfected with
Si-CDH3 or OE-CDH3. **P<0.001 vs. NC (D) CDH3 protein expression
was confirmed via western blotting in HGC-27 and AGS cells
transfected with Si-CDH3 or OE-CDH3. Data are presented as the mean
± SD (n=3), and at least three independent tests were performed for
every experiment. **P<0.001 vs. NC. CDH3, cadherin 3; Si-CDH3,
small interfering RNA-CDH3; OE-CDH3, overexpression-CDH3; CON,
blank control; NC, negative control of Si-CDH3 co-transfected with
negative control of OE-CDH3; GC, gastric cancer; RT-qPCR, reverse
transcription-quantitative PCR. |
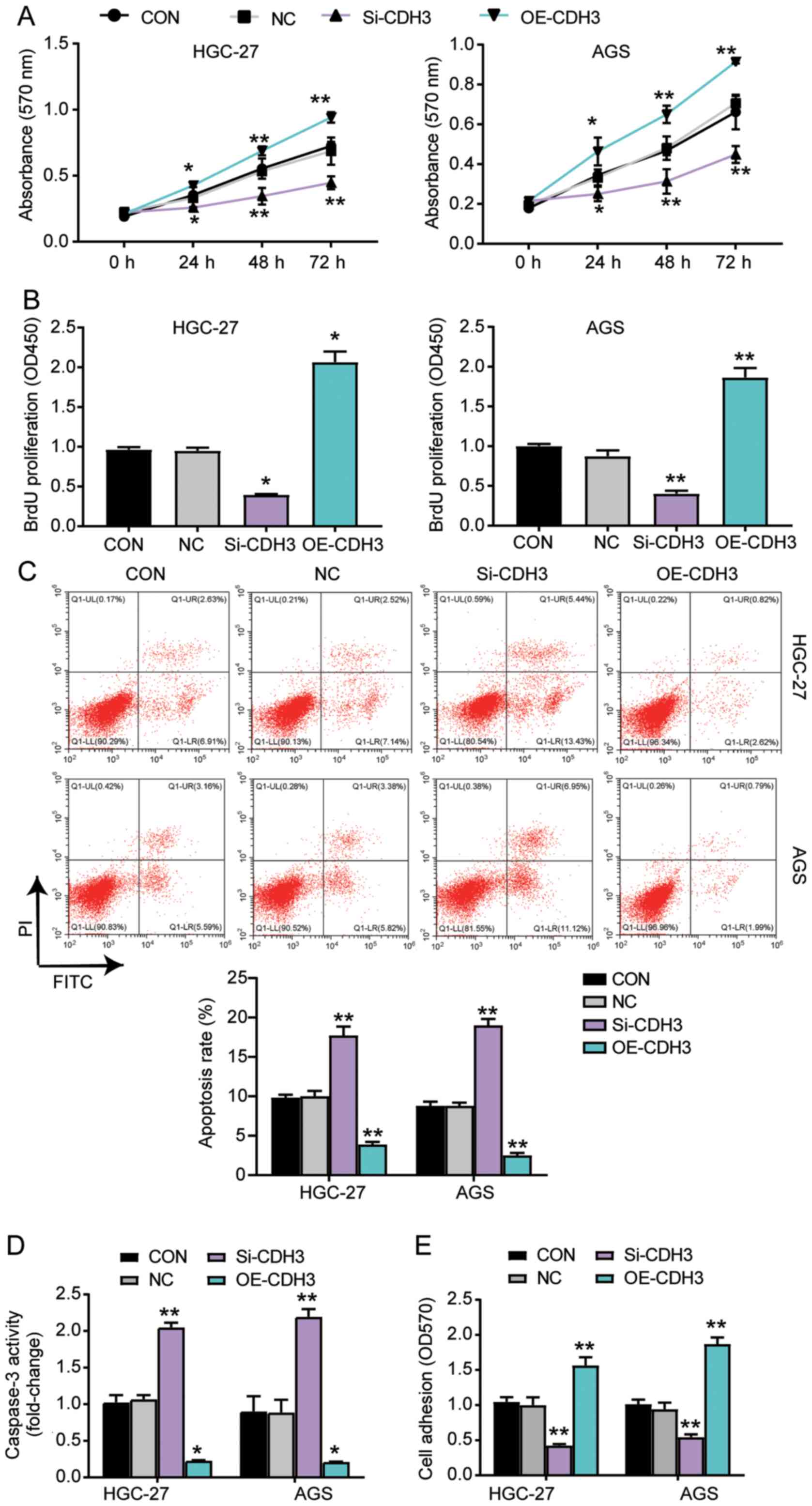
CDH3 acts as an oncogene in GC
cells
Several experiments were performed to explore the
function of CDH3 in GC cells. The MTT assay results indicated that
the cells transfected with OE-CDH3 presented higher cell viability
than control cells, while the cells transfected with si-CDH3
exhibited lower cell viability than control cells (Fig. 3A). The results of the BrdU ELISA
assay revealed that the proliferation of cells transfected with
OE-CDH3 increased by 2-fold, while that of the cells transfected
with si-CDH3 declined by 50% compared with control cells (Fig. 3B). The apoptosis level was
ascertained using the Caspase-3 Activity Assay kit and Annexin
V-FITC Apoptosis Detection kit (Fig. 3C
and D). The results demonstrated that the apoptosis level and
caspase 3 activity in cells transfected with si-CDH3 significantly
increased compared with that in control cells, while there was a
decrease in the apoptosis and caspase 3 activity of cells
transfected with OE-CDH3 (Fig. 3C and
D). Additionally, the adhesion ability of the OE-CDH3 group
exhibited a >1.5-fold increase, while that of cells transfected
with si-CDH3 decreased by 50% compared with control cells (Fig. 3E). Overall, the present results
suggested that the overexpression of CDH3 enhanced the adhesion and
proliferation of GC cells, but inhibited the apoptosis of GC
cells.
miR-665 targets the CDH3 3′-UTR in
GC
The GSE93415 profile was obtained from the GEO
database and was used to screen the downregulated differentially
expressed miRNAs with logFC <-1 and adj. P<0.05. TargetScan
and starBase were used to predict the miRNAs binding to the CDH3
3′-UTR. Venny 2.1.0 analysis indicated an overlap of hsa-miR-665
(Fig. 4A). The TargetScan results
revealed that miR-665 matched the position 593–599 of the CDH3
3′-UTR (Fig. 4B). Next, the
luciferase activity detected in AGS and HGC-27 cells confirmed that
the miR-665 mimic in the CDH3-WT group downregulated the luciferase
activity by 60% compared with cells transfected with the mimic-NC;
in contrast to the CDH3-WT cells, miR-665 mimic had no significant
effect on the cells in the CDH3-MUT group (Fig. 4C). Additionally, miR-665 expression
was decreased by 60% in tumor tissues compared with in adjacent
non-tumor tissues (Fig. 4D).
Furthermore, an inverse correlation was detected between CDH3 and
miR-665 expression in GC tissues (Fig.
4E). miR-665 expression was also analyzed in GC cells,
revealing that miR-665 expression in AGS and HGC-27 GC cells was
decreased by 50% compared with that in GES-1 cells (Fig. 4F). After transfection, miR-665
expression in the cells transfected with the miR-665 mimic
increased by 4-fold, while it decreased by 80% in the cells
transfected with the miR-665 inhibitor compared with in the cells
in the control group (Figs. S1B and
4G). Western blot analysis indicated
that CDH3 protein expression in the miR-665 inhibitor-transfected
cells was significantly increased, while it was significantly
decreased in the cells transfected with the miR-665 mimic (Fig. 4H). Overall, these results indicated
that by targeting CDH3 mRNA, miR-665 regulated GC cells.
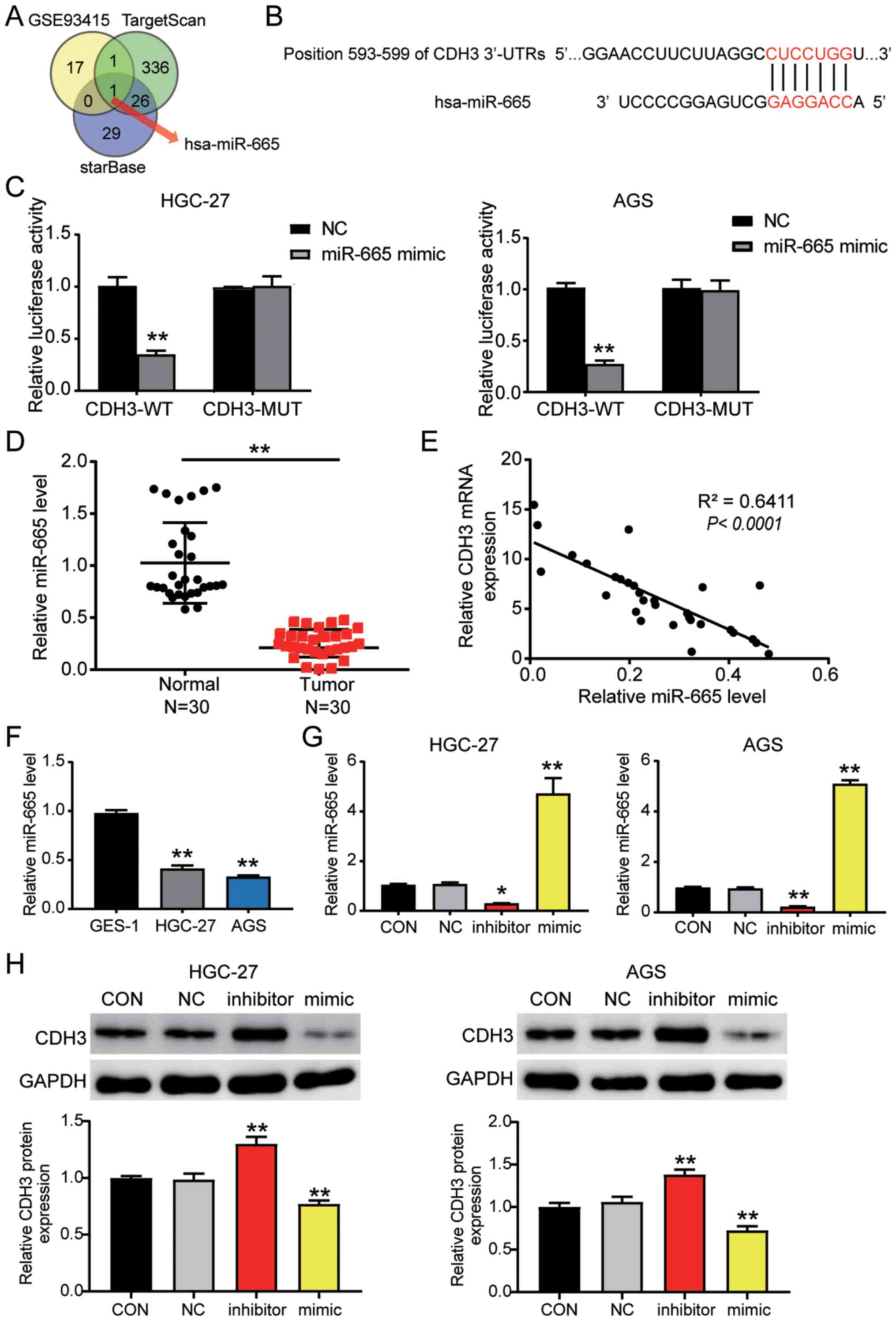 | Figure 4.CDH3 is a candidate target gene of
miR-665. (A) miR-665 was identified by Venny 2.1.0 analysis.
TargetScan and starBase were employed to ascertain the miRNAs
binding to CDH3. GSE93415 was used to identify the miRNAs with low
expression in GC. (B) TargetScan revealed the predicted binding
sequences of CDH3 3′-UTR. (C) Dual luciferase assay kit was
utilized in CDH3-WT and CDH3-MUT cells transfected with NC or
miR-665 mimic. **P<0.001 vs. NC. (D) RT-qPCR was used to
identify miR-665 expression in GC and adjacent normal tissues
(n=30). **P<0.001. (E) Pearson's correlation analysis revealed
the negative correlation between CDH3 and miR-665 expression in GC
samples. RT-qPCR was used to detect the expression levels of
miR-665 in (F) GES-1, HGC-27 and AGS cells, and (G) AGS and HGC-27
cells transfected with miR-665 inhibitor or miR-665 mimic. (H)
Measurement of CDH3 protein expression in HGC-27 and AGS cells
transfected with miR-665 inhibitor or miR-665 mimic. Data are
presented as the mean ± SD (n=3), and at least three independent
tests were performed for every experiment. *P<0.05 and
**P<0.001 vs. NC. CDH3, cadherin 3; miR/miRNA, microRNA; WT,
wild-type; MUT, mutant; CON, blank control; NC, negative control of
miR-665 inhibitor co-transfected with negative control of miR-665
mimic; UTR, untranslated region; RT-qPCR, reverse
transcription-quantitative PCR; GC, gastric cancer. |
miR-665 targeting CDH3 inhibits GC
progression
MTT assay were performed to study the function of
miR-665 in GC cells. The results indicated that the cells
transfected with the miR-665 inhibitor exhibited enhanced cell
viability; additionally, the miR-665 inhibitor could reverse the
suppressive effect of si-CDH3 on cell viability (Fig. 5A). Additionally, the results of the
BrdU ELISA assay revealed a 2-fold increase in the proliferation of
the cells transfected with the miR-665 inhibitor, and the miR-665
inhibitor reversed the suppressive effect of si-CDH3 on cell
proliferation (Fig. 5B). The
apoptosis level was identified using the two aforementioned kits,
and the results indicated that apoptosis was suppressed by the
miR-665 inhibitor, while the apoptosis in the group co-transfected
with si-CDH3 and miR-665 inhibitor was similar to that in the
control group (Fig. 5C and D). The
cells transfected with the miR-665 inhibitor also exhibited a
1.5-fold increase in cell adhesion, and the negative effect of
si-CDH3 on cell adhesion was relieved by the miR-665 inhibitor
(Fig. 5E). Overall, the current
results suggested that by targeting CDH3, miR-665 could inhibit
cell proliferation and adhesion, but promote the apoptosis of GC
cells.
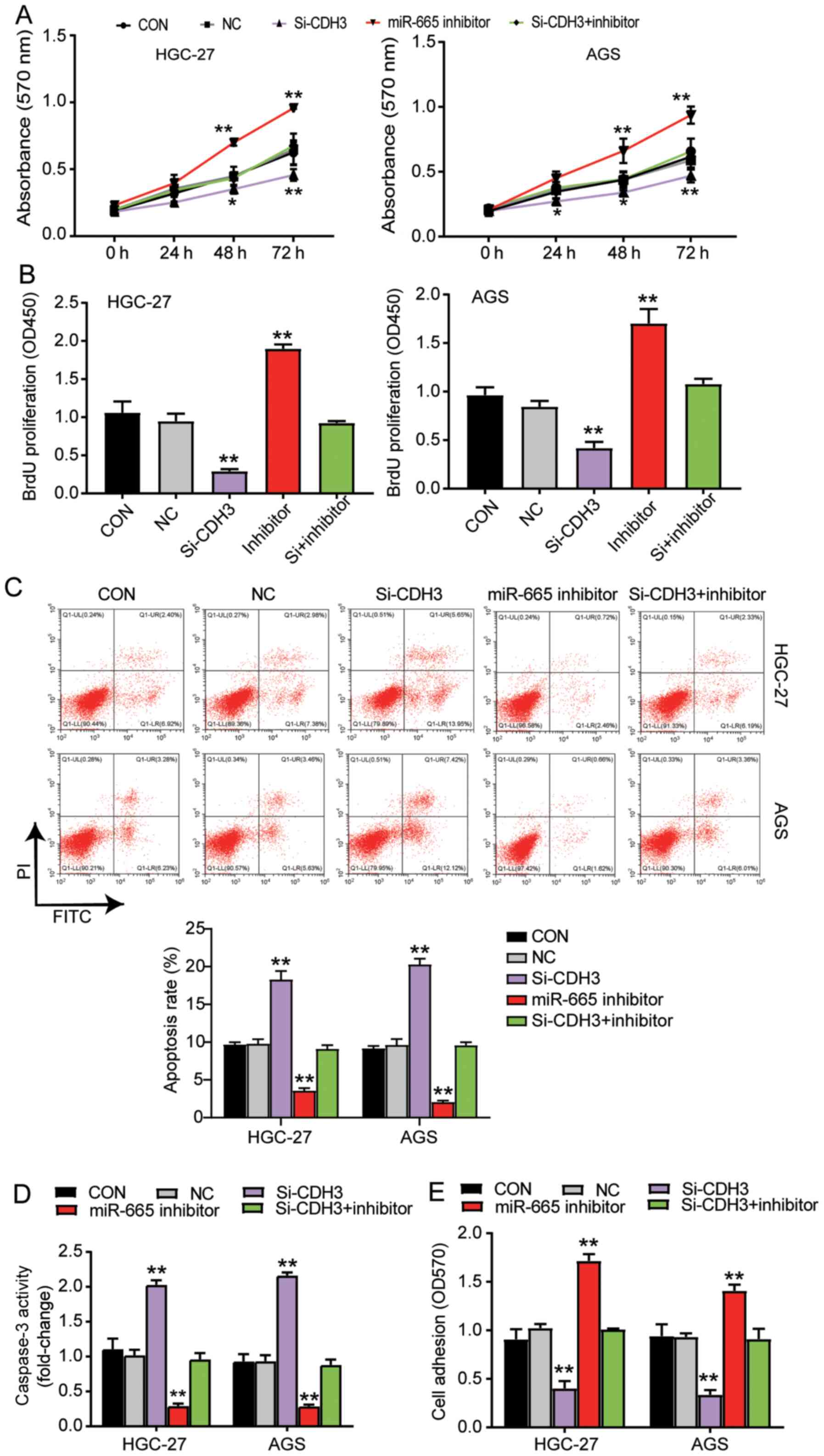 | Figure 5.miR-665 targeting CDH3 inhibits
proliferation and adhesion, and promotes apoptosis of gastric
cancer cells. (A) Cell viability was ascertained in AGS and HGC-27
cells transfected with Si-CDH3, miR-665 inhibitor or
Si-CDH3+miR-665 inhibitor by MTT assay. (B) Cell proliferation was
analyzed in AGS and HGC-27 cells transfected with Si-CDH3, miR-665
inhibitor or Si-CDH3+miR-665 inhibitor by BrdU ELISA assay.
Apoptosis was confirmed in AGS and HGC-27 cells transfected with
Si-CDH3, miR-665 inhibitor or Si-CDH3+miR-665 inhibitor by (C) FITC
apoptosis detection kit and (D) caspase-3 activity assay kit. (E)
Cell adhesion was detected in HGC-27 and AGS cells transfected with
miR-665 inhibitor, Si-CDH3 or Si-CDH3+miR-665 inhibitor using a
cell adhesion assay kit. Data are presented as the mean ± SD, and
at least three independent tests were performed for every
experiment. *P<0.05 and **P<0.001 vs. NC. CDH3, cadherin 3;
miR, microRNA; Si-CDH3, small interfering RNA-CDH3; CON, blank
control; NC, negative control of miR-665 inhibitor co-transfected
with negative control of Si-CDH3; OD, optical density. |
Discussion
The present study confirmed the upregulation of CDH3
expression and the downregulation of miR-665 expression in GC
tissues and cell lines. The overexpression of CDH3 increased cell
proliferation and adhesion levels, and decreased the apoptosis
level. On the other hand, the miR-665 mimic restrained the
proliferation and adhesion of GC cells, and promoted their
apoptosis. Moreover, CDH3 seemed to be a direct target of miR-665
in GC cells, meaning that miR-665 may inhibit CDH3 expression to
suppress the progression of GC.
In the last three decades, several studies have
reported the ability of CDH3 to accelerate the growth of multiple
types of cancer, including lung (27), esophageal (9), colorectal (7) and breast cancer (28). A previous study has also suggested
the involvement of CDH3 in promoting the motility of pancreatic
cancer cells by activating Rho-family GTPases (8). Another study revealed that CDH3
upregulation associated with CDH3 promoter hypomethylation
significantly influenced the proliferation of breast cancer cells
(29). Moreover, in some studies,
CDH3 expression was upregulated in GC cells (11,12). The
detection of the CDH3 demethylation level in 36 primary gastric
carcinoma samples revealed that 25/36 (69%) samples had an abnormal
demethylation level, which may result in the promotion of gastric
carcinoma (11). Another study
reported the upregulation of CDH3 expression in 20/28 (71%) GC
tissues based on a cDNA microarray, and immunohistochemical
analyses further confirmed an increase in CDH3 protein expression
in GC tissues (12). Consistent with
the aforementioned studies, the present study demonstrated that
upregulated CDH3 expression in GC tissues and cells augmented cell
proliferation and adhesion, and decreased apoptosis.
Furthermore, miR-665 has been associated with
numerous human malignancies, including pancreatic (17), breast (18), cervical (30) and ovarian cancer (31). Previous studies indicated that this
miRNA could promote the occurrence of cancer, as well as suppress
cancer. For instance, by targeting Homeobox Protein Hox-A10,
miR-665 inhibited the progression of ovarian cancer (19). Additionally, miR-665 sponged by RHPN1
Antisense RNA 1 suppressed tumor development by preventing the
activation of AKT3 (32).
Nonetheless, miR-665 could facilitate the tumorigenesis of ovarian
cancer cells by targeting SRC Kinase Signaling Inhibitor 1
(31). In addition, miR-665
overexpression enhanced the proliferation, migration and invasion
of non-small cell lung cancer cells (16). Regarding GC, miR-665 has been
documented in several studies to suppress the progression of GC
(20,33). After binding to Protein Phosphatase 2
Regulatory Subunit Bα, this miRNA inhibited the proliferation,
invasion and epithelial-mesenchymal transition (EMT) of GC cells
(20). In addition, miR-665 attached
to AKT3 suppressed GC cells by decreasing proliferation and
increasing apoptosis. Additionally, miR-665 was observed to be a
target of LINC00565, which accelerated the development of GC by
upregulating AKT3 expression (33).
However, by inhibiting its target gene suppressor of cytokine
signaling 3 (SOCS3), miR-665 promoted the EMT of gastric
adenocarcinoma cells (21). In the
present study, miR-665 binding to CDH3 inhibited cell proliferation
and adhesion, but enhanced apoptosis in GC cells.
A single miRNA may have a number of targets
(34). More specifically, miR-665 is
involved in numerous pathways involved in GC, since it can target
AKT3 (33), Yes-associated protein 1
(YAP1) (35), cysteine-rich
transmembrane bone morphogenetic protein regulator 1 (CRIM1)
(36) and SOCS3 (21). However, in the current study, the
effect of miR-665 on AKT3, YAP1, CRIM1 and SOCS3 was not explored.
While the current study has investigated the proliferation,
adhesion and apoptosis of GC cells, it is important to note that
cancer occurrence is a complicated pathway interaction. The present
study only confirmed the association between miR-665 and CDH3 in GC
cells by analyzing the enrichment of target genes, so the lack of
miRNA enrichment analysis and of survival analysis on the target
genes and associated miRNAs are the other major limitations of the
study. In the future, the potential signaling pathways and survival
analysis of the target genes and associated miRNAs in GC should be
further explored. Additionally, the current study only observed
tumor characteristics at the cellular level. However, further
studies are required to verify the current findings in animal
models.
In conclusion, to the best of our knowledge, the
present study was the first to demonstrate an interaction between
miR-665 and CDH3, and to reveal that miR-665 may suppress the
progression of GC by targeting CDH3. The present findings may offer
further insight into the prognosis and treatment of GC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by Basic and
Frontier projects of Science and Technology Department of Henan
Province (grant no. 142300410050), Joint project of Henan
Provincial Health and Family Planning Commission (grant no.
20170123), Henan Health and Family Planning Research and Innovation
Talent Project (grant no. 51282) and Joint project of the National
Natural Science Foundation of China (grant no. U1604174).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request. The datasets generated and/or analyzed during the current
study are available in the Gene Expression Omnibus database
(https://www.ncbi.nlm.nih.gov/gds/?term=).
Authors' contributions
XF and SD performed the experiments and data
analysis. YB and LZ conceived and designed the study. LZ and SD
assessed the raw data to ensure its legitimacy. XF reviewed and
edited the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Henan Provincial People's Hospital (Zhengzhou, China;
approval no. 2019-04). All patients signed written informed
consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang F, Meng W, Wang B and Qiao L:
Helicobacter pylori-induced gastric inflammation and gastric
cancer. Cancer Lett. 345:196–202. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dong J and Thrift AP: Alcohol, smoking and
risk of oesophago-gastric cancer. Best Pract Res Clin
Gastroenterol. 31:509–517. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Marin JJ, Al-Abdulla R, Lozano E, Briz O,
Bujanda L, Banales JM and Macias RI: Mechanisms of resistance to
chemotherapy in gastric cancer. Anticancer Agents Med Chem.
16:318–334. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Biagioni A, Skalamera I, Peri S, Schiavone
N, Cianchi F, Giommoni E, Magnelli L and Papucci L: Update on
gastric cancer treatments and gene therapies. Cancer Metastasis
Rev. 38:537–548. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sprecher E, Bergman R, Richard G, Lurie R,
Shalev S, Petronius D, Shalata A, Anbinder Y, Leibu R, Perlman I,
et al: Hypotrichosis with juvenile macular dystrophy is caused by a
mutation in CDH3, encoding P-cadherin. Nat Genet. 29:134–136. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kumara HMCS, Bellini GA, Caballero OL,
Herath SAC, Su T, Ahmed A, Njoh L, Cekic V and Whelan RL:
P-Cadherin (CDH3) is overexpressed in colorectal tumors and has
potential as a serum marker for colorectal cancer monitoring.
Oncoscience. 4:139–147. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Taniuchi K, Nakagawa H, Hosokawa M,
Nakamura T, Eguchi H, Ohigashi H, Ishikawa O, Katagiri T and
Nakamura Y: Overexpressed P-cadherin/CDH3 promotes motility of
pancreatic cancer cells by interacting with p120ctn and activating
rho-family GTPases. Cancer Res. 65:3092–3099. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu D, Wu K, Yang Y, Zhu D, Zhang C and
Zhao S: Long noncoding RNA ADAMTS9-AS2 suppresses the progression
of esophageal cancer by mediating CDH3 promoter methylation. Mol
Carcinog. 59:32–44. 2020. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Albergaria A, Ribeiro AS, Pinho S,
Milanezi F, Carneiro V, Sousa B, Sousa S, Oliveira C, Machado JC,
Seruca R, et al: ICI 182,780 induces P-cadherin overexpression in
breast cancer cells through chromatin remodelling at the promoter
level: A role for C/EBPbeta in CDH3 gene activation. Hum Mol Genet.
19:2554–2566. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hibi K, Kitamura YH, Mizukami H, Goto T,
Sakuraba K, Sakata M, Saito M, Ishibashi K, Kigawa G, Nemoto H and
Sanada Y: Frequent CDH3 demethylation in advanced gastric
carcinoma. Anticancer Res. 29:3945–3947. 2009.PubMed/NCBI
|
|
12
|
Imai K, Hirata S, Irie A, Senju S, Ikuta
Y, Yokomine K, Harao M, Inoue M, Tsunoda T, Nakatsuru S, et al:
Identification of a novel tumor-associated antigen, cadherin
3/P-cadherin, as a possible target for immunotherapy of pancreatic,
gastric, and colorectal cancers. Clin Cancer Res. 14:6487–6495.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hornstein E, Mansfield JH, Yekta S, Hu JK,
Harfe BD, McManus MT, Baskerville S, Bartel DP and Tabin CJ: The
microRNA miR-196 acts upstream of Hoxb8 and Shh in limb
development. Nature. 438:671–674. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Boehm M and Slack FJ: MicroRNA control of
lifespan and metabolism. Cell Cycle. 5:837–840. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xia J, Li D, Zhu X, Xia W, Qi Z, Li G and
Xu Q: Upregulated miR-665 expression independently predicts poor
prognosis of lung cancer and facilitates tumor cell proliferation,
migration and invasion. Oncol Lett. 19:3578–3586. 2020.PubMed/NCBI
|
|
17
|
Zhou B, Guo W, Sun C, Zhang B and Zheng F:
Linc00462 promotes pancreatic cancer invasiveness through the
miR-665/TGFBR1-TGFBR2/SMAD2/3 pathway. Cell Death Dis. 9:7062018.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhao XG, Hu JY, Tang J, Yi W, Zhang MY,
Deng R, Mai SJ, Weng NQ, Wang RQ, Liu J, et al: miR-665 expression
predicts poor survival and promotes tumor metastasis by targeting
NR4A3 in breast cancer. Cell Death Dis. 10:4792019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu J, Jiang Y, Wan Y, Zhou S, Thapa S and
Cheng W: MicroRNA-665 suppresses the growth and migration of
ovarian cancer cells by targeting HOXA10. Mol Med Rep.
18:2661–2668. 2018.PubMed/NCBI
|
|
20
|
Zhang M, Wang S, Yi A and Qiao Y:
microRNA-665 is down-regulated in gastric cancer and inhibits
proliferation, invasion, and EMT by targeting PPP2R2A. Cell Biochem
Funct. 38:409–418. 2020. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tang H, Long Q, Zhuang K, Yan Y, Han K,
Guo H and Lu X: miR-665 promotes the progression of gastric
adenocarcinoma via elevating FAK activation through targeting SOCS3
and is negatively regulated by lncRNA MEG3. J Cell Physiol.
235:4709–4719. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li L, Zhu Z, Zhao Y, Zhang Q, Wu X, Miao
B, Cao J and Fei S: FN1, SPARC, and SERPINE1 are highly expressed
and significantly related to a poor prognosis of gastric
adenocarcinoma revealed by microarray and bioinformatics. Sci Rep.
9:78272019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jin Y, He J, Du J, Zhang RX, Yao HB and
Shao QS: Overexpression of HS6ST2 is associated with poor prognosis
in patients with gastric cancer. Oncol Lett. 14:6191–6197.
2017.PubMed/NCBI
|
|
24
|
Sobin LH and Compton CC: TNM seventh
edition: What's new, what's changed: Communication from the
International Union Against Cancer and the American Joint Committee
on Cancer. Cancer. 116:5336–5339. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sierzega M, Kaczor M, Kolodziejczyk P,
Kulig J, Sanak M and Richter P: Evaluation of serum microRNA
biomarkers for gastric cancer based on blood and tissue pools
profiling: The importance of miR-21 and miR-331. Br J Cancer.
117:266–273. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hsiao TF, Wang CL, Wu YC, Feng HP, Chiu
YC, Lin HY, Liu KJ, Chang GC, Chien KY, Yu JS and Yu CJ:
Integrative omics analysis reveals soluble cadherin-3 as a survival
predictor and an early monitoring marker of EGFR tyrosine kinase
inhibitor therapy in lung cancer. Clin Cancer Res. 26:3220–3229.
2020.PubMed/NCBI
|
|
28
|
Albergaria A, Resende C, Nobre AR, Ribeiro
AS, Sousa B, Machado JC, Seruca R, Paredes J and Schmitt F:
CCAAT/enhancer binding protein β (C/EBPβ) isoforms as
transcriptional regulators of the pro-invasive CDH3/P-cadherin gene
in human breast cancer cells. PLoS One. 8:e557492013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Paredes J, Albergaria A, Oliveira JT,
Jerónimo C, Milanezi F and Schmitt FC: P-cadherin overexpression is
an indicator of clinical outcome in invasive breast carcinomas and
is associated with CDH3 promoter hypomethylation. Clin Cancer Res.
11:5869–5877. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cao L, Jin H, Zheng Y, Mao Y, Fu Z, Li X
and Dong L: DANCR-mediated microRNA-665 regulates proliferation and
metastasis of cervical cancer through the ERK/SMAD pathway. Cancer
Sci. 110:913–925. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhou P, Xiong T, Yao L and Yuan J:
MicroRNA-665 promotes the proliferation of ovarian cancer cells by
targeting SRCIN1. Exp Ther Med. 19:1112–1120. 2020.PubMed/NCBI
|
|
32
|
Zhao J, Yang T, Ji J, Zhao F, Li C and Han
X: RHPN1-AS1 promotes cell proliferation and migration via
miR-665/Akt3 in ovarian cancer. Cancer Gene Ther. 28:33–41. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hu J, Ni G, Mao L, Xue X, Zhang J, Wu W,
Zhang S, Zhao H, Ding L and Wang L: LINC00565 promotes
proliferation and inhibits apoptosis of gastric cancer by targeting
miR-665/AKT3 axis. Onco Targets Ther. 12:7865–7875. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luo C, Tetteh PW, Merz PR, Dickes E,
Abukiwan A, Hotz-Wagenblatt A, Holland-Cunz S, Sinnberg T, Schittek
B, Schadendorf D, et al: miR-137 inhibits the invasion of melanoma
cells through downregulation of multiple oncogenic target genes. J
Invest Dermatol. 133:768–775. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin X, Huang C, Chen Z, Wang H and Zeng Y:
CircRNA_100876 is upregulated in gastric cancer (GC) and promotes
the GC cells' growth, migration and invasion via miR-665/YAP1
signaling. Front Genet. 11:5462752020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu KZ, Zhang CD, Zhang C, Pei JP and Dai
DQ: miR-665 suppresses the epithelial-mesenchymal transition and
progression of gastric cancer by targeting CRIM1. Cancer Manag Res.
12:3489–3501. 2020. View Article : Google Scholar : PubMed/NCBI
|