Introduction
Glioma, which is the most common type of primary
brain and central nervous system (CNS) cancer, originates from
supporting or glial cells in the CNS (1). Glioma is classified into two subtypes:
Diffuse glioma and non-diffuse glioma, which exhibits slower growth
(2). Treatment for glioma mainly
comprises surgical resection, chemotherapy and radiation. In
addition, immunotherapies and targeted therapies have been
developed in recent years in an effort to increase the low survival
rate of patients (3). However,
further studies on the mechanisms of glioma progression are
required to identify and develop novel treatment strategies.
Long non-coding RNAs (lncRNAs) are transcripts of
>200 nucleotides in length that lack protein-coding ability
(4). lncRNAs have been reported to
serve roles in a wide variety of physiological and
pathophysiological functions by regulating gene and chromatin
dynamics, which subsequently promotes tumorigenesis and metastasis
(5). Therefore, lncRNAs have been
considered as novel biomarkers and therapeutic targets for various
types of cancer (6). Small nucleolar
RNA host gene 7 (SNHG7) was first identified in lymphoblastoid cell
lines by Chaudhry (7) in 2013. SNHG7
comprises 2,157 nucleotides and is located on chromosome 9q34.3
(8). The expression levels of SNHG7
were reported to be upregulated in numerous types of cancer such as
bladder, breast, colorectal, esophageal, gastric and prostate
cancer, and were associated with tumor progression, metastasis and
the suppression of apoptosis (9).
For example, Ren et al (10)
discovered that SNHG7 promoted the growth and progression of
glioblastoma. However, to the best of our knowledge, the underlying
mechanism of the effect of SNHG7 on the proliferation of glioma
cells remains poorly understood.
MicroRNAs (miRNAs/miRs) are small non-coding RNAs of
22 nucleotides in length, which also lack protein-coding ability
(11). miRNAs downregulate the
expression levels of target protein-coding genes by binding to the
3′-untranslated regions (UTRs) of mRNAs, post-transcriptionally
(12). An increasing number of
studies have demonstrated that miRNAs serve significant roles in
the progression of various types of cancer, including glioma. For
instance, Wang et al (13) reported
that miR-138-5p inhibited the progression of human colorectal
cancer by targeting telomerase reverse transcriptase expression. Ou
et al (14) reported that miR-138-5p
inhibited the proliferation of cervical cancer cells. The
lncRNASNP2 database was used in the present study to identify
putative binding sites complementary to SNHG7 in miR-138-5p.
Accumulating evidence has demonstrated that lncRNAs
can act as competing endogenous RNAs (ceRNAs) by sponging or
decoying target miRNAs involved in tumor proliferation (6,15). The
aim of the present study was to determine the clinical significance
and biological functions of SNHG7 in glioma.
Materials and methods
Patient studies
A total of 20 glioma tissues resected from patients
with glioma (11 male and 9 female; mean age ± SD, 55.3±12.5 years;
age range, 23–85 years) and 14 normal tissues from brains of
traffic accident victims (8 male and 6 female; mean age ± SD,
51.2±15.4 years; age range, 18–82 years) were acquired. The tissues
were obtained from the Department of Neurosurgery of Chenzhou First
People's Hospital (Chenzhou, China) between January 2016 and
December 2018. Samples were frozen in liquid nitrogen immediately
after collection. Based on the World Health Organization (WHO)
classification system (16), two
clinical pathologists pathologically diagnosed and divided the
glioma specimens into two groups: Low-grade and high-grade. The
low-grade group contained 9 stage I–II tumors, while the high-grade
group comprised 11 stage III–IV tumors. Patients had not received
chemotherapy or radiotherapy prior to surgical resection.
Experiments were performed according to the principles of the
Declaration of Helsinki. Experimental protocols were approved by
Chenzhou First People's Hospital and written informed consent was
obtained from all patients or their relatives prior to
participation in the study.
Cell lines and culture
Normal human astrocytes (NHA), glioma LN229, A172
and U251 cell lines, and a glioblastoma cell line of unknown
origin, U87 (cat. no. HTB14), were purchased from the American Type
Culture Collection. Cells were cultured in DMEM (HyClone; Cytiva)
supplemented with 10% FBS (HyClone; Cytiva), and maintained at 37°C
in a humidified atmosphere containing 5% CO2.
Cell transfection
Small interfering RNA (siRNA/si) sequences targeting
SNHG7 (si-SNHG7-1, 5′-GCCGCUUGUGUU CUUGAUUTT-3′; and si-SNHG7-2,
5′-CCTCTGGTGCCUCGUUCUGGAAACGAUTT-3′) and scrambled si-negative
control (NC; 5′-CCCAGUAAUAGGACACCAATT-3′) were purchased from
Guangzhou RiboBio Co., Ltd. The miR-138-5p inhibitor
(5′-CACACUCGGGUCGAAUGAAGGA-3′), miR-138-5p mimic
(5′-AGCUGGUGUUGUGAAUCAGGCCG-3′), scrambled mimic NC
(5′-UUCUCCGAACGUGUCACGU-3′) and scrambled inhibitor NC
(5′-CAGUACUUUUGUGUAGUA CAA-3′) were obtained from Shanghai
GenePharma Co., Ltd. Cell transfection was performed using
Lipofectamine® 3000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from cells and tissues using
TRIzol® and TRIzol™ LS reagent (both Invitrogen; Thermo
Fisher Scientific, Inc.), respectively, according to the
manufacturers' protocols. Total RNA was reverse transcribed into
cDNA to analyze lncRNA and mRNA expression using the PrimeScript™
RT reagent kit (Takara Bio, Inc.) according to the manufacturer's
protocol. qPCR was subsequently performed using a TB Green™ Premix
Ex Taq™ II kit (Takara Bio, Inc.), with GAPDH as the internal
control. The PCR was performed using the following conditions:
Initial denaturation at 95 °C for 30 sec, followed by 40 cycles of
95°C for 5 sec and 60°C for 30 sec, with final annealing and
extension at 60°C for 60 sec and 95°C for 15 sec. Total RNA was
reverse transcribed into cDNA to determine miRNA expression using a
miRcute Plus miRNA First-Strand cDNA kit (Tiangen Biotech Co.,
Ltd.) according to the manufacturer's protocol. qPCR was
subsequently performed using a miRcute Plus miRNA qPCR kit (SYBR
Green) (Tiangen Biotech Co., Ltd.), with U6 as the internal
control. qPCR was performed on an ABI 7500 Real-Time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The following
primer pairs (Sangon Biotech Co., Ltd.) were used for qPCR: SNHG7
forward, 5′-GTTGGGGTGTTGGCATTCTTGTT-3′ and reverse,
5′-TGGTCAGCCTGGTCACTCTGG-3′; EZH2 forward,
5′-AATCAGAGTACATGCGACTGAGA-3′ and reverse,
5′-GCTGTATCCTTCGCTGTTTCC-3′; GAPDH forward,
5′-GTCTCCTCTGACTTCAACAGCG-3′ and reverse,
5′-ACCACCCTGTTGCTGTAGCCAA-3′; miR-138-5p forward,
5′-AGCTGGTGTTGTGAATCAGGCCG-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′; and U6 forward, 5′-CTC
GCTTCGGCAGCACA-3′ and reverse, 5′-AACGCTTCACGA ATTTGCGT-3′. The
miR-138-5p reverse primer was obtained using the miRcute Plus miRNA
qPCR kit (SYBR Green). Expression levels were quantified using the
2−ΔΔCq method (17).
Nucleic and cytoplasmic
separation
Nucleic and cytoplasmic fragments were extracted
from cells using a PARIS™ kit (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Briefly, collected cells were lysed in cell fractionation buffer,
then the nucleic and cytoplasmic fractions were isolated by
centrifugation. The supernatant containing the cytoplasmic fraction
was transferred into a clean tube in the absence of RNase.
Disruption buffer was used to lyse the pellet containing the
nucleic fraction, which was subsequently mixed with the cytoplasmic
fraction and lysis/binding solution. Subsequently, 100% ethanol was
added the mixture, and the sample was filtered and washed with
washing solution. The elution solution was used to obtain
cytoplasmic and nucleic mRNAs. U6 and GAPDH were used as the
positive controls for the nucleic and cytoplasmic extracts,
respectively.
Bioinformatics analysis
Prediction of putative binding sites complementary
to SNHG7 in miR-138-5p was performed using the lncRNASNP2 database
(http://bioinfo.life.hust.edu.cn/lncRNASNP). Putative
targeting sequences between miR-138-5p and EZH2 were predicted
using TargetScan 7.2 (http://www.targetscan.org).
Cell Counting Kit-8 (CCK-8) assay
Glioma cells were cultured in 96-well plates and
transfected as aforementioned. Following incubation for 24, 48 or
72 h, 20 µl CCK-8 (Dojindo Molecular Technologies, Inc.) was added
to each well for 4 h. The absorbance was measured at a wavelength
of 490 nm using a Elx800 microplate reader (BioTek Instruments,
Inc.; Agilent Technologies, Inc.). According to the absorbance
values, cell proliferation was calculated as a percentage of the
control.
Flow cytometry
Following transfection for 48 h, U87 and A172 cells
were collected for cell cycle analysis. Briefly, cells were fixed
in 75% ethanol overnight at −20°C and then stained with PI (BD
Biosciences) for 30 min in the dark at 37°C. The cell cycle
distribution was analyzed using a flow cytometer (Attune NxT;
Thermo Fisher Scientific, Inc.). All data were analyzed using
FlowJo v10 software (FlowJo LLC). All experiments were
independently performed in triplicate.
Western blotting
Total protein was extracted from glioma cells using
a Protein Extraction kit (Nanjing KeyGen Biotech Co., Ltd.) and
quantified using a BCA protein assay kit (Sigma-Aldrich; Merck
KGaA). Proteins (40 µg/lane) were separated by 10% SDS-PAGE and
transferred onto PVDF membranes (Bio-Rad Laboratories, Inc.). The
membranes were blocked with 5% skimmed milk for 1.5 h at room
temperature. Membranes were subsequently incubated with the
following primary antibodies at 4°C overnight: Rabbit polyclonal
anti-EZH2 (1:2,000; cat. no. 21800-1-AP; ProteinTech Group, Inc.)
and rabbit polyclonal anti-GAPDH (1:1,000; cat. no. 25778; Santa
Cruz Biotechnology, Inc.). Following the primary antibody
incubation, the membranes were incubated with an HRP-conjugated
goat anti-rabbit secondary antibody (1:5,000; cat. no. ab6721;
Abcam) at room temperature for 2 h. Protein bands were visualized
using the ECL reagent (EMD Millipore).
Dual luciferase reporter assay
Following the prediction of the putative binding
sites of miR-138-5p in SNHG7/EZH2, pMIR-REPORT vectors (Guangzhou
RiboBio Co., Ltd.) containing wild-type (WT) or mutant (MT) 3′-UTR
sequences of SNHG7 or EZH2 were constructed. Cells were
co-transfected with miR-138-5p mimic or mimic NC and SNHG7/EZH2-WT
or SNHG7/EZH2-MT for 48 h using Lipofectamine® 3000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). Relative
luciferase activity was measured using a Luc-Pair™ Duo-Luciferase
assay kit (Shanghai Yeasen Biotechnology Co., Ltd.).
RNA immunoprecipitation (RIP)
assay
The RIP assay was performed using a Magna
RNA-Binding Protein Immunoprecipitation kit (EMD Millipore)
according to the manufacturer's protocol. Whole cell lysates were
incubated with anti-IgG negative control antibody (derived from
normal mice; EMD Millipore) or anti-Argonaute 2 (Ago2) antibody
(from humans; 1:50; EMD Millipore) and RIP buffer containing
magnetic beads. Each sample was subsequently incubated with
proteinase K buffer. The immunoprecipitated RNAs were isolated and
SNHG7 and miR-138-5p expression was detected using RT-qPCR, as
aforementioned.
Colony formation assay
Transfected cells were cultured in 6-well plates
overnight in medium supplemented with 10% FBS at 37°C.
Subsequently, cells were incubated for 14 days at 37°C. Following
the incubation, cells were fixed with methanol for 10 min at room
temperature and stained with 0.1% crystal violet for 20 min at room
temperature. Colonies were counted under a light microscope.
Statistical analysis
Statistical analysis was performed using SPSS 21.0
software (IBM Corp.) and data are presented as the mean ± SD of
three independent experiments. Statistical differences between two
groups were determined using an unpaired two-tailed Student's
t-test, while statistical differences between multiple groups were
determined using a one-way ANOVA followed by Tukey's post hoc test.
Spearman's rank correlation was used for correlation analysis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
lncRNA SNHG7 expression levels are
upregulated in glioma and promote the proliferation of glioma
cells
To determine the role of SNHG7 in glioma, the
expression levels of SNHG7 in tissues from normal brains, low-grade
glioma (stage I–II) and high-grade glioma (stage III–IV) were
analyzed. The results revealed that SNHG7 expression levels were
significantly upregulated in glioma tissues compared with in normal
tissues; in addition, the expression levels of SNHG7 were
significantly increased in high grade glioma (stage III–IV) tissues
compared with in low-grade glioma (stage I–II) tissues (Fig. 1A). Thus, SNHG7 expression may be
positively associated with tumor grade. The expression levels of
SNHG7 were also determined in cell lines, including NHAs, U251,
LN229, U87 and A172 cells. Compared with in NHAs, SNHG7 expression
was significantly upregulated in glioma cell lines, including in
U251 (P<0.05) and LN229 (P<0.05), and especially in U87
(P<0.01) and A172 (P<0.01) (Fig.
1B). Therefore, the latter two cell lines (U87 and A172) were
chosen for subsequent experiments. RT-qPCR analysis revealed that
SNHG7 expression was successfully knocked down in U87 and A172 cell
lines following transfection with both si-SNHG7-1 and si-SNHG7-2
(Fig. 1C). The results of the CCK-8
assay demonstrated that SNHG7-knockdown significantly decreased
cell proliferation in U87 and A172 cell lines compared with in
cells transfected with si-NC (Fig.
1D). In addition, the results of the colony formation assay
further indicated that SNHG7-knockdown significantly decreased the
number of colonies formed from glioma cells compared with those
formed from glioma cells transfected with si-NC (Fig. 1E). Flow cytometry analysis of the
cell cycle distribution revealed that SNHG7-knockdown arrested U87
and A172 cells in the G1 phase (Fig. 1F).
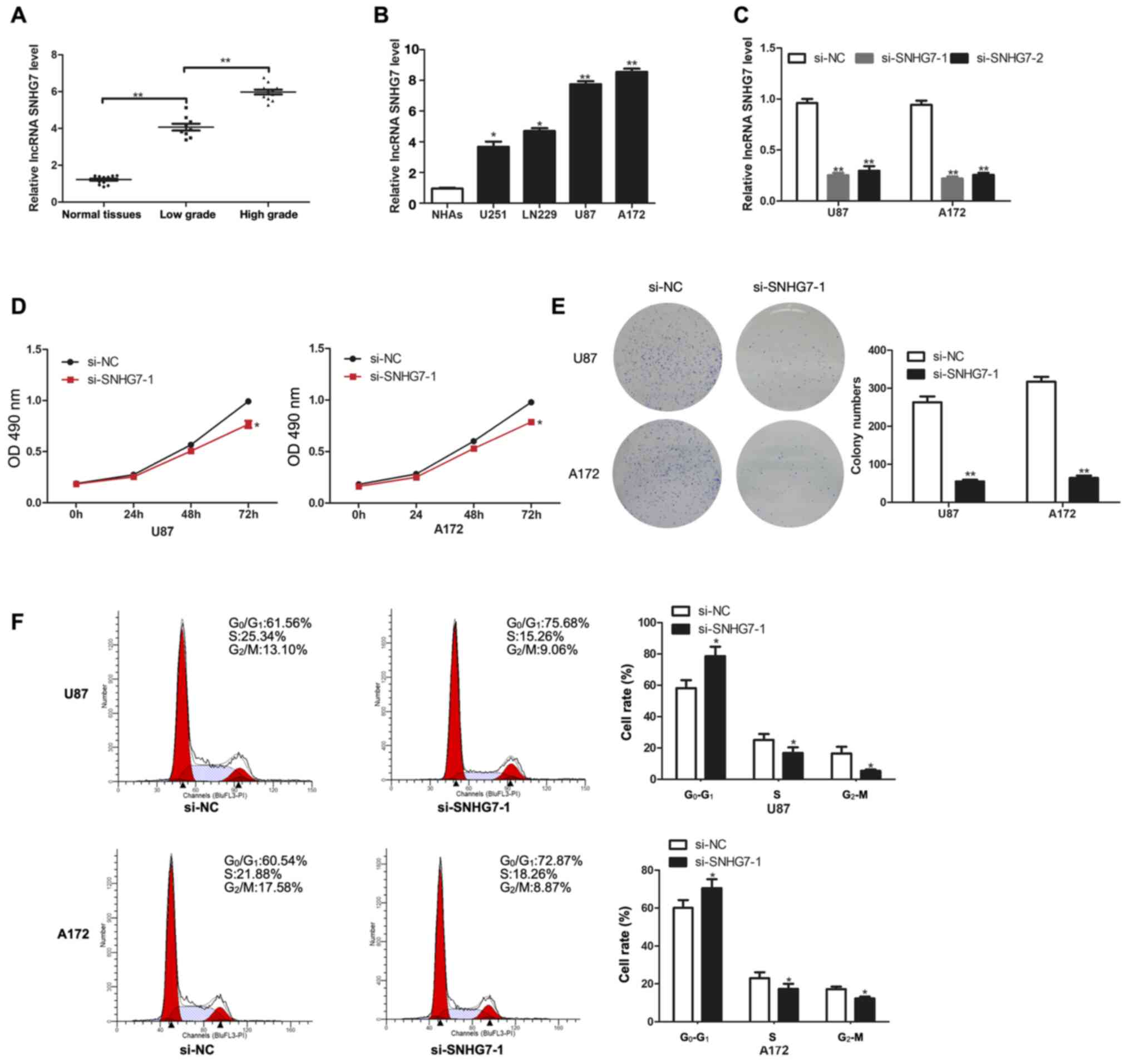 | Figure 1.lncRNA SNHG7 expression is
upregulated in glioma tissues and cell lines, and promotes
proliferation. (A) RT-qPCR analysis of SNHG7 expression in 14
normal tissues, 9 low-grade (stage I–II) glioma tissues and 11
high-grade (stage III–IV) glioma tissues. (B) RT-qPCR analysis of
SNHG7 expression in NHAs, U251, LN229, U87 and A172 cell lines. (C)
RT-qPCR analysis of SNHG7 expression for transfection efficiency in
U87 and A172 cells. (D) Cell proliferation was determined by Cell
Counting Kit-8 assay after transfection with si-SNHG7. (E) Colony
formation assay was performed to determine colony-forming ability.
(F) Flow cytometry was used to evaluate the effects of si-SNHG7 on
cell cycle distribution. *P<0.05 and **P<0.01 vs. NHAs or
si-NC. OD, optical density; NHAs, normal human astrocytes; RT-qPCR,
reverse transcription-quantitative PCR; si, small interfering RNA;
NC, negative control; lncRNA; long non-coding RNA; SNHG7, small
nucleolar RNA host gene 7. |
SNHG7 directly targets miR-138-5p
To determine the underlying mechanism of SNHG7 in
glioma, RT-qPCR was used to analyze SNHG7 expression in the nucleus
and cytoplasm. The results demonstrated that SNHG7 was mainly
localized in the cytoplasm of U87 and A172 cell lines (Fig. 2A). Based on these results, it was
hypothesized that SNHG7 may function as a ceRNA. Bioinformatics
analysis using the lncRNASNP2 database predicted that miR-138-5p
contained potential binding sites for SNHG7 (Fig. 2B). miR-138-5p expression levels were
upregulated in glioma cells following the transfection of U87 and
A172 cells with si-SNHG7-1 compared with si-NC (Fig. 2C). miR-138-5p expression was
significantly increased following transfection with the miR-138-5p
mimic compared with the miR-NC (Fig.
2D). The results of the dual luciferase reporter assay
demonstrated that the relative luciferase activity was
significantly decreased following the co-transfection of cells with
the miR-138-5p mimic and SNHG7-WT, but not SNHG7-MT vectors
(Fig. 2E). These findings indicated
the potential association between SNHG7 and miR-138-5p. It was
subsequently investigated whether miR-138-5p and SNHG7 existed in
the same RNA-induced silencing complex using a RIP assay. The
results indicated significant enrichment of SNHG7 in the anti-Ago2
group compared with the anti-IgG group; similar results were
obtained for miR-138-5p (Fig. 2F).
Additionally, miR-138-5p expression was analyzed in 14 normal brain
tissues, nine low-grade glioma (stage I–II) tissues and 11
high-grade glioma (stage III–IV) tissues. miR-138-5p expression
levels were significantly downregulated in glioma tissues compared
with in normal tissues, and significantly lower expression levels
of miR-138-5p were observed in high-grade tissues compared with in
low-grade tissues (Fig. 2G).
Furthermore, Pearson's correlation analysis identified a negative
correlation between miR-138-5p and SNHG7 expression in glioma
tissues (Fig. 2H). These findings
suggested that SNHG7 may directly target miR-138-5p.
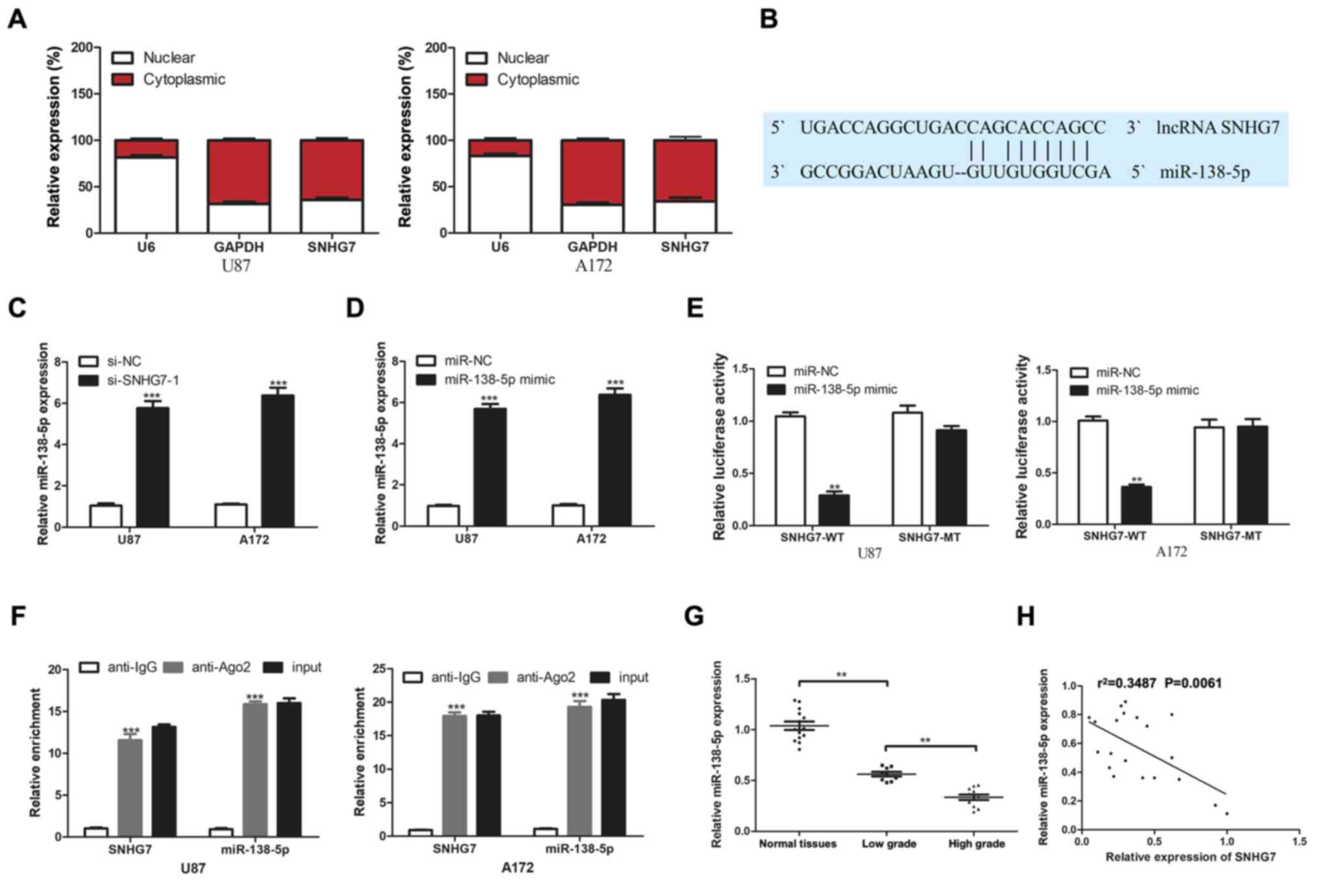 | Figure 2.lncRNA SNHG7 targets miR-138-5p. (A)
Nuclear and cytoplasmic SNHG7 percentage was analyzed by RT-qPCR.
(B) Bioinformatics prediction of miR-138-5p targeting sites in
SNHG7 3′-untranlsated region using lncRNASNP2. (C) Relative
miR-138-5p expression was detected via RT-qPCR after transfection
with si-NC or si-SNHG7-1. (D) Relative miR-138-5p expression was
detected via RT-qPCR after transfection with miR-NC or miR-138-5p
mimic. (E) Luciferase activity was analyzed by luciferase reporter
assay for the targeting of SNHG7 to miR-138-5p. (F) Enrichment
analysis of SNHG7 and miR-138-5p in immunoprecipitates of Ago2 and
control IgG by RNA immunoprecipitation. (G) miR-138-5p expression
was determined by RT-qPCR in 9 low-grade (stage I–II) glioma
tissues, 11 high-grade (stage III–IV) glioma tissues and 14 normal
tissues. (H) Negative correlation between SNHG7 and miR-138-5p in
glioma tissues. **P<0.01 and ***P<0.001 vs. si-NC, miR-NC or
anti-IgG. RT-qPCR, reverse transcription-quantitative PCR; si,
small interfering RNA; NC, negative control; lncRNA; long
non-coding RNA; SNHG7, small nucleolar RNA host gene 7; miR,
microRNA; WT, wild-type; MT, mutant; Ago2, Argonaute 2. |
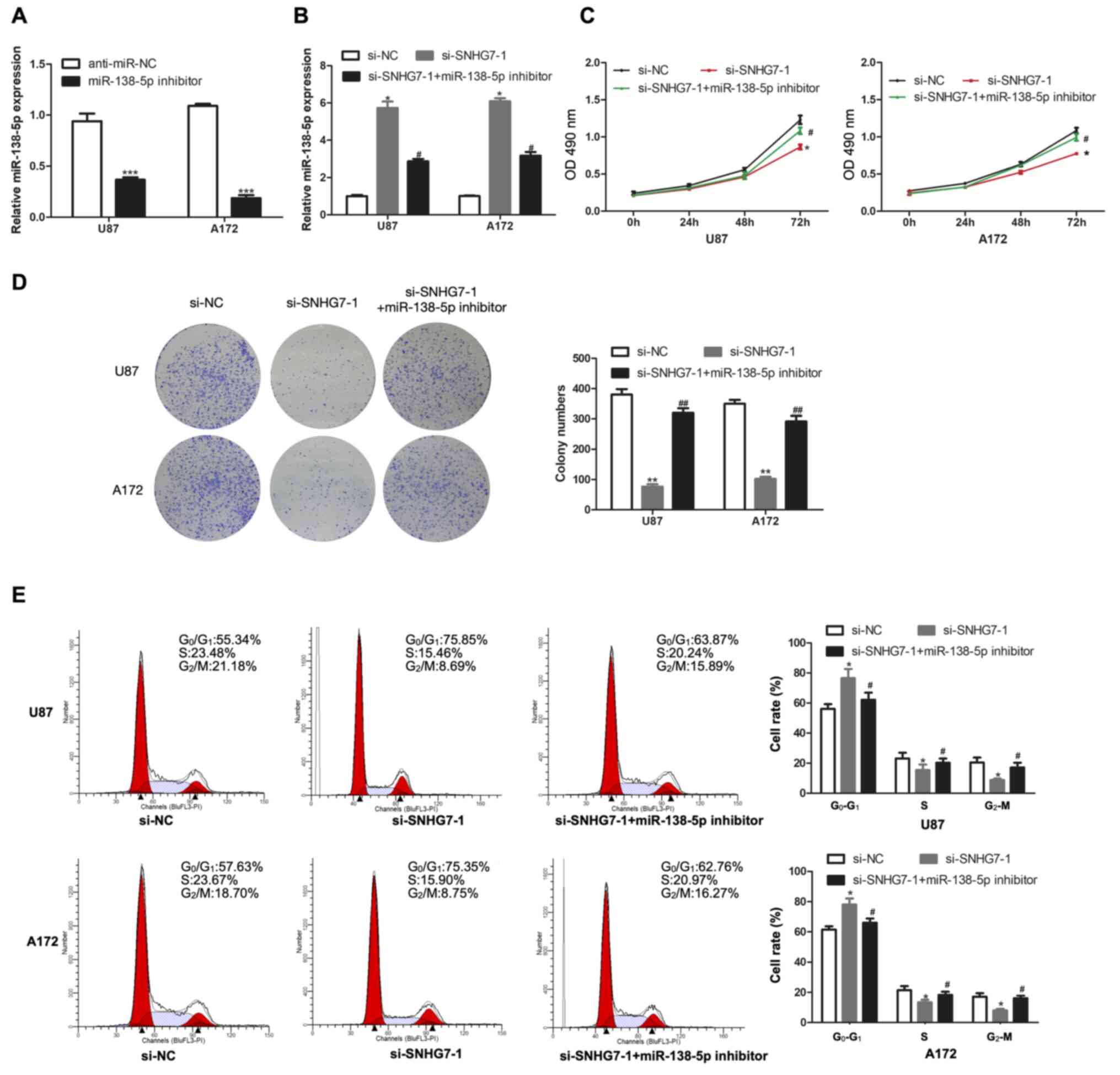
Transfection with the miR-138-5p
inhibitor reverses the effect of SNHG7-knockdown in glioma
cells
To further investigate whether SNHG7 exerted
biological effects on glioma cells by targeting miR-138-5p,
miR-138-5p expression was knocked down in U87 and A172 cells by
transfection with a miR-138-5p inhibitor, and the transfection
efficiency was analyzed using RT-qPCR (Fig. 3A). Compared with the transfection
with si-SNHG7 alone, miR-138-5p expression was downregulated to a
further extent in U87 and A172 cells co-transfected with si-SNHG7
and miR-138-5p inhibitor (Fig. 3B).
The results of the CCK-8 assay demonstrated that transfection with
the miR-138-5p inhibitor reversed the SNHG7-knockdown-induced
inhibition of proliferation in U87 and A172 cells (Fig. 3C). Notably, the same trend was also
observed in the colony formation assay (Fig. 3D). Compared with the si-SNHG7 group,
the number of cells in the G1 phase of the cell cycle
was decreased in the si-SNHG7 and miR-138-5p inhibitor group
(Fig. 3E). Overall, these results
suggested that transfection with the miR-138-5p inhibitor may
reverse the effect of SNHG7-knockdown in glioma cells.
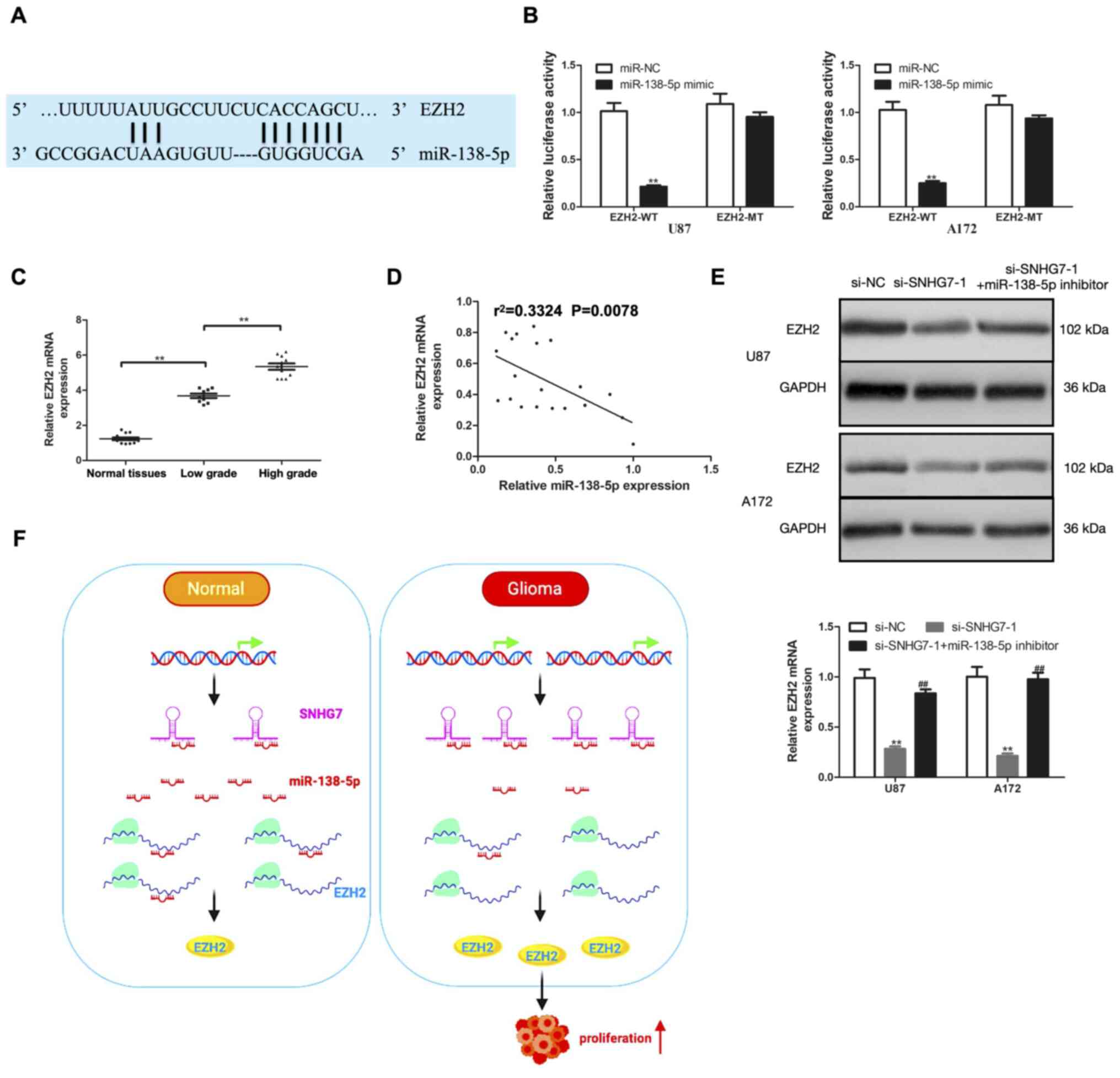
SNHG7 regulates EZH2 expression by
acting as a ceRNA and sponging miR-138-5p
miRNAs are known to act at the post-transcriptional
level by directly regulating target gene expression and modulating
subsequent protein expression (18).
To determine whether SNHG7 regulated EZH2 expression by functioning
as a ceRNA and sponging miR-138-5p, bioinformatics analysis was
first performed using TargetScan, which predicted the presence of
potential miR-138-5p targeting sites in EZH2 (Fig. 4A). According to the results of the
dual luciferase reporter assay, the relative luciferase activity
was significantly decreased by co-transfection of the miR-138-5p
mimic and EZH2-WT vector in U87 and A172 cells, while the relative
luciferase activity was unaffected in U87 and A172 cells
co-transfected with miR-138-5p mimic and EZH2-MT (Fig. 4B). EZH2 mRNA expression in glioma and
normal tissues was subsequently determined using RT-qPCR. The
results revealed that EZH2 mRNA expression was significantly
upregulated in glioma tissues compared with normal tissues;
moreover, compared with in low-grade glioma (stage I–II) tissues,
EZH2 mRNA expression was significantly upregulated in high-grade
glioma (stage III–IV) tissues (Fig.
4C). In addition, EZH2 mRNA expression was negatively
correlated with miR-138-5p expression (Fig. 4D). The role of miR-138-5p in
modulating the effects of SNHG7 on EZH2 were further investigated.
SNHG7-knockdown markedly downregulated the expression levels of
EZH2; however, the expression levels were reversed following the
transfection with the miR-138-5p inhibitor (Fig. 4E). These findings suggested that
SNHG7 may serve as a ceRNA and may sponge miR-138-5p to modulate
EZH2 expression and promote glioma cell proliferation (Fig. 4F).
Discussion
Glioma is classified into four grades (grade I–IV)
according to the level of tumor malignancy by the WHO (19). Standard treatment for glioma includes
surgical resection, radiotherapy and chemotherapy. Further
potential treatments targeting specific signaling pathways and
antigenic profiles of glioma are currently under investigation,
which may improve the survival of patients (20). The expression levels of lncRNAs are
dysregulated in glioma and are involved in its molecular pathology
(10,21).
lncRNAs have been found to influence the prognosis
and treatment of numerous types of cancer (22,23).
Previous studies have revealed that SNHG7 is involved in the
progression of several types of cancer. For example, Zeng et al
(24) reported that SNHG7 acted as
an oncogene to promote cervical cancer cell proliferation and
invasion. In addition, Cheng et al (25) identified an oncogenic role for SNHG7
in promoting the proliferation, migration and invasion of
pancreatic cancer cells. Yao et al (26) demonstrated that SNHG7 exerted a
tumorigenic role in hepatic cancer cell proliferation and
metastasis. The results of the present study revealed that the
expression levels of SNHG7 were upregulated in glioma tissues and
cells, and were positively associated with tumor grade. Moreover,
SNHG7-knockdown decreased the proliferation of glioma cells. These
results are consistent with the findings of the aforementioned
studies, thereby suggesting that SNHG7 may act as an oncogenic
lncRNA to promote cell proliferation in glioma.
It is well established that lncRNAs act as ceRNAs,
which sponge and suppress miRNA expression, thereby alleviating
their inhibitory effects on mRNA target genes. For example, Wang et
al (27) indicated that lncRNA 01121
acted as a ceRNA to promote cell proliferation, migration and
invasion in breast cancer. Fan et al (28) discovered that lncRNA FYVE, RhoGEF and
PH domain containing 5-antisense RNA 1 upregulated fibroblast
growth factor receptor like 1 expression and increased the
proliferation of non-small cell lung cancer cells via sponging
hsa-miR-107. The present study identified complementary binding
sites for SNHG7 in miR-138-5p using the lncRNASNP2 database. In
addition, the relative luciferase activity of the SNHG7-WT vector
was decreased compared with the SNHG7-MT vector following the
co-transfection with the miR-138-5p mimic. The results of the RIP
assay revealed a marked enrichment of SNHG7 in the anti-Ago2 group
compared with the anti-IgG group, and similar findings were
observed for miR-138-5p. In addition, miR-138-5p expression was
significantly upregulated in glioma cells transfected with si-SNHG7
compared with si-NC. miR-138-5p expression was significantly
downregulated in glioma tissues and was negatively associated with
SNHG7 expression. Furthermore, the transfection with the miR-138-5p
inhibitor reversed the effects induced by SNHG7-knockdown in glioma
cells. The present findings revealed the importance of the
association between SNHG7 and miR-138-5p in glioma tumorigenesis.
Thus, the current study suggested that SNHG7 may play a
carcinogenic role in glioma by promoting cell proliferation, which
may be partially rescued by miR-150-5p.
EZH2, which encodes a histone methyltransferase, is
an evolutionary conserved gene. EZH2 has been found to have
analogous structural motifs and domains (29). As part of the polycomb repressive
complex 2, EZH2 catalyzes the tri-methylation of lysine 27 on the
Histone H3 protein, which silences downstream target genes
(29). Through this mechanism, EZH2
can regulate the cell cycle, cell proliferation, cell
differentiation and cancer progression (30). An increasing number of studies have
reported that EZH2 plays an oncogenic role in tumor progression
(31–33). For instance, Zhang et al (34) revealed that the knockdown of EZH2
inhibited cell cycle progression in glioma cells. Chen et al
(35) demonstrated that EZH2, by
binding to lncRNA nuclear paraspeckle assembly transcript 1, could
promote glioma cell proliferation and invasion. Wu et al (36) indicated that EZH2 was recruited and
downregulated by HOXA distal transcript-antisense RNA 2, which
facilitated glioma progression. In the present study, the binding
sequences between miR-138-5p and EZH2 were identified using
TargetScan. The relative luciferase activity of the EZH2-WT vector
was decreased compared with the EZH2-MT vector following the
co-transfection with the miR-138-5p mimic. The current results
indicated that miR-138-5p may target EZH2. In addition, EZH2 mRNA
expression was upregulated in glioma tissues and positively
associated with tumor grade. Moreover, EZH2 expression was
inversely correlated with miR-138-5p expression in glioma tissues.
The knockdown of miR-138-5p reversed SNHG7-knockdown-induced
downregulation of EZH2 expression and inhibition of cell
proliferation in glioma cells.
In conclusion, the findings of the present study
suggested that lncRNA SNHG7 may promote the proliferation of glioma
by acting as a ceRNA and sponging miR-138-5p to regulate EZH2
expression. The current results indicated that the
SNHG7/miR-138-5p/EZH2 signaling axis may represent potential
targets for the treatment of glioma. However, the present study had
several limitations. For example, the underlying mechanisms of
SNHG7 were not investigated in vivo. Therefore, animal experiments
should be performed to further explore the mechanisms of SNHG7 in
glioma and its potential role as a ceRNA.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 82000614), the
Natural Science Foundation of Hunan Province (grant no.
2020JJ5876), the Science and Technology Plan Project of Changsha
(grant no. kq2004146), the Hunan Provincial Science and Technology
Plan Project (grant no. 2019JJ80066) and the Scientific Research
Project of Health and Family Planning Commission of Hunan Province
of China (grant no. B20-17202).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YD, XM and HZ conceived and designed the
experiments. LC and XM performed most of the experiments. ZL
collected and analyzed the patient data. YD and HZ statistically
analyzed the data. LC wrote the manuscript. YD, XM and HZ confirm
the authenticity of all the raw data. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
Experimental protocols were approved by Chenzhou
First People's Hospital (approval no. 2016-S250) and written
informed consent was obtained from all patients or their relatives
prior to participation in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Davis ME: Epidemiology and Overview of
Gliomas. Semin Oncol Nurs. 34:420–429. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wesseling P and Capper D: WHO 2016
Classification of gliomas. Neuropathol Appl Neurobiol. 44:139–150.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bush NA, Chang SM and Berger MS: Current
and future strategies for treatment of glioma. Neurosurg Rev.
40:1–14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ulitsky I and Bartel DP: lincRNAs:
Genomics, evolution, and mechanisms. Cell. 154:26–46. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sanchez Calle A, Kawamura Y, Yamamoto Y,
Takeshita F and Ochiya T: Emerging roles of long non-coding RNA in
cancer. Cancer Sci. 109:2093–2100. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bhan A, Soleimani M and Mandal SS: Long
Noncoding RNA and Cancer: A New Paradigm. Cancer Res. 77:3965–3981.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chaudhry MA: Expression pattern of small
nucleolar RNA host genes and long non-coding RNA in X-rays-treated
lymphoblastoid cells. Int J Mol Sci. 14:9099–9110. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ota T, Suzuki Y, Nishikawa T, Otsuki T,
Sugiyama T, Irie R, Wakamatsu A, Hayashi K, Sato H, Nagai K, et al:
Complete sequencing and characterization of 21,243 full-length
human cDNAs. Nat Genet. 36:40–45. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou Y, Tian B, Tang J, Wu J, Wang H, Wu
Z, Li X, Yang D, Zhang B, Xiao Y, et al: SNHG7: A novel vital
oncogenic lncRNA in human cancers. Biomed Pharmacother.
124:1099212020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ren J, Yang Y, Xue J, Xi Z, Hu L, Pan SJ
and Sun Q: Long noncoding RNA SNHG7 promotes the progression and
growth of glioblastoma via inhibition of miR-5095. Biochem Biophys
Res Commun. 496:712–718. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ha M and Kim VN: Regulation of microRNA
biogenesis. Nat Rev Mol Cell Biol. 15:509–524. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Carthew RW and Sontheimer EJ: Origins and
Mechanisms of miRNAs and siRNAs. Cell. 136:642–655. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang X, Zhao Y, Cao W, Wang C, Sun B, Chen
J, Li S, Chen J, Cui M, Zhang B, et al: miR-138-5p acts as a tumor
suppressor by targeting hTERT in human colorectal cancer. Int J
Clin Exp Pathol. 10:11516–11525. 2017.PubMed/NCBI
|
|
14
|
Ou L, Wang D, Zhang H, Yu Q and Hua F:
Decreased Expression of miR-138-5p by lncRNA H19 in Cervical Cancer
Promotes Tumor Proliferation. Oncol Res. 26:401–410. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: A ceRNA hypothesis: The Rosetta Stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Louis DN, Perry A, Reifenberger G, von
Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD,
Kleihues P and Ellison DW: The 2016 World Health Organization
Classification of Tumors of the Central Nervous System: A summary.
Acta Neuropathol. 131:803–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Δ Δ C(T)) Method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Beermann J, Piccoli MT, Viereck J and Thum
T: Non-coding RNAs in Development and Disease: Background,
Mechanisms, and Therapeutic Approaches. Physiol Rev. 96:1297–1325.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Komori T: The 2016 WHO Classification of
Tumours of the Central Nervous System: The Major Points of
Revision. Neurol Med Chir (Tokyo). 57:301–311. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Weller M, Wick W, Aldape K, Brada M,
Berger M, Pfister SM, Nishikawa R, Rosenthal M, Wen PY, Stupp R, et
al: Glioma. Nat Rev Dis Primers. 1:150172015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pop S, Enciu AM, Necula LG and Tanase C:
Long non-coding RNAs in brain tumours: Focus on recent epigenetic
findings in glioma. J Cell Mol Med. 22:4597–4610. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xi J, Sun Q, Ma L and Kang J: Long
non-coding RNAs in glioma progression. Cancer Lett. 419:203–209.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Vecera M, Sana J, Lipina R, Smrcka M and
Slaby O: Long Non-Coding RNAs in Gliomas: From Molecular Pathology
to Diagnostic Biomarkers and Therapeutic Targets. Int J Mol Sci.
19:E27542018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zeng J, Ma YX, Liu ZH and Zeng YL: lncRNA
SNHG7 contributes to cell proliferation, invasion and prognosis of
cervical cancer. Eur Rev Med Pharmacol Sci. 23:9277–9285.
2019.PubMed/NCBI
|
|
25
|
Cheng D, Fan J, Ma Y, Zhou Y, Qin K, Shi M
and Yang J: lncRNA SNHG7 promotes pancreatic cancer proliferation
through ID4 by sponging miR-342-3p. Cell Biosci. 9:282019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yao X, Liu C, Liu C, Xi W, Sun S and Gao
Z: lncRNA SNHG7 sponges miR-425 to promote proliferation,
migration, and invasion of hepatic carcinoma cells via
Wnt/β-catenin/EMT signalling pathway. Cell Biochem Funct.
37:525–533. 2019. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Z, Wang P, Cao L, Li F, Duan S, Yuan
G, Xiao L, Guo L, Yin H, Xie D, et al: Long Intergenic Non-Coding
RNA 01121 Promotes Breast Cancer Cell Proliferation, Migration, and
Invasion via the miR-150-5p/HMGA2 Axis. Cancer Manag Res.
11:10859–10870. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fan Y, Li H, Yu Z, Dong W, Cui X, Ma J and
Li S: Long non-coding RNA FGD5-AS1 promotes non-small cell lung
cancer cell proliferation through sponging hsa-miR-107 to
up-regulate FGFRL1. Biosci Rep. 40:BSR201933092020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gan L, Yang Y, Li Q, Feng Y, Liu T and Guo
W: Epigenetic regulation of cancer progression by EZH2: From
biological insights to therapeutic potential. Biomark Res.
6:102018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gall Trošelj K, Novak Kujundzic R and
Ugarkovic D: Polycomb repressive complex's evolutionary conserved
function: The role of EZH2 status and cellular background. Clin
Epigenetics. 8:552016. View Article : Google Scholar
|
|
31
|
Liu Y, Sun J, Yu J, Ge W, Xiao X, Dai S
and Xiang Q: lncRNA CACS15 accelerates the malignant progression of
ovarian cancer through stimulating EZH2-induced inhibition of APC.
Am J Transl Res. 11:6561–6568. 2019.PubMed/NCBI
|
|
32
|
Xu M, Chen X, Lin K, Zeng K, Liu X, Xu X,
Pan B, Xu T, Sun L, He B, et al: lncRNA SNHG6 regulates EZH2
expression by sponging miR-26a/b and miR-214 in colorectal cancer.
J Hematol Oncol. 12:32019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao L, Sun H, Kong H, Chen Z, Chen B and
Zhou M: The lncrna-TUG1/EZH2 Axis Promotes Pancreatic Cancer Cell
Proliferation, Migration and EMT Phenotype Formation Through
Sponging Mir-382. Cell Physiol Biochem. 42:2145–2158. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang K, Sun X, Zhou X, Han L, Chen L, Shi
Z, Zhang A, Ye M, Wang Q, Liu C, et al: Long non-coding RNA HOTAIR
promotes glioblastoma cell cycle progression in an EZH2 dependent
manner. Oncotarget. 6:537–546. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen Q, Cai J, Wang Q, Wang Y, Liu M, Yang
J, Zhou J, Kang C, Li M and Jiang C: Long Noncoding RNA NEAT1,
Regulated by the EGFR Pathway, Contributes to Glioblastoma
Progression Through the WNT/β-Catenin Pathway by Scaffolding EZH2.
Clin Cancer Res. 24:684–695. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu L, Zhu X, Song Z, Chen D, Guo M, Liang
J, Ding D, Wang W and Yan D: Long Non-Coding RNA HOXA-AS2 Enhances
The Malignant Biological Behaviors In Glioma By Epigenetically
Regulating RND3 Expression. OncoTargets Ther. 12:9407–9419. 2019.
View Article : Google Scholar : PubMed/NCBI
|