Introduction
Advancing techniques for detection and targeted
therapy of colon cancer have increased patient 5-year survival
rates to 65% and estimated newly diagnosed cases per year was
134,490 in the United states in 2016 (1). Despite medical advancements, colon
cancer remains the fifth most common type of cancer and is the
fourth-leading cause of cancer-associated mortality worldwide
(2). The direct cost per patient
with colon cancer during the primary treatment period was estimated
to be >24,000 US dollars (3).
Current treatment strategies for colon cancer include surgery,
radiation therapy, chemotherapy and immunotherapy (4). However, a proportion of patients with
colon cancer require alternative treatments due to the adverse
effects of conventional therapy. Thus, it is important to identify
and develop novel therapeutic strategies and pathways to improve
treatment efficacy and reduce side effects.
Autophagy is a highly conserved metabolic process
that can occur in all cell types and is induced by increasing
oxidative stress, drug stimulation and nutrient depletion (5). Recently, autophagy has become a novel
target for cancer treatment and adjuvant therapy. The molecular
mechanisms underlying autophagy involve regulating the expression
of related proteins, including LC3B and p62 (6). LC3B expression is associated with
autophagosome formation, and p62 expression is involved in
autophagy substrate selection and serves as an adaptor for
intracellular targets (7). LC3B and
p62 expression are used to evaluate autophagy activity during tumor
progression (8). However, autophagy
plays a dual role in cancer. During the initial progression phase,
it serves as a tumor suppressor (9,10).
Conversely, in hypoxic environments, autophagy serves as a
developer (11). Thus, it is
important to identify the role of drug-induced autophagy in
cancer.
Direct inhibition of the key enzymes in glycolysis
to decrease ATP production is also a novel cancer treatment
strategy. For example, transketolase (TKT) is an enzyme of the
pentose phosphate pathway (PPP), which connects PPP to glycolysis
(12). In cancer cells, ATP is
produced by glycolysis rather than oxidative phosphorylation
(13). Furthermore, overexpression
of TKT is associated with tumorigenesis, such as the development of
hepatocellular carcinoma (14) and
peritoneal metastases of ovarian cancer (15). Recent studies have reported that
anti-TKT therapy inhibits cell migration and invasion in esophageal
cancer (16) and inhibits the
expression of the TKT-like-1 gene, which significantly decreases
TKT expression in human colon cancer cells and substantially
inhibits cell proliferation (17).
Furthermore, cryptotanshinone, a traditional oriental medicinal
agent, has been demonstrated to decrease non-small cell lung cancer
proliferation by suppressing TKT expression (18).
Itraconazole has been used as an antifungal drug in
the past few decades (19). The
primary mechanism by which itraconazole inhibits microorganism
growth is interference with ergosterol synthesis, which damages the
cell membrane integrity of fungal cells (20). Recently, induction of apoptosis or
autophagic apoptosis by itraconazole has been widely used as an
anticancer therapy (21,22). Preclinical data have demonstrated
that itraconazole can suppress tumor growth, and many clinical
trials have been performed to evaluate the effects of itraconazole
on different cancer treatments (23,24).
Itraconazole can induce autophagic cell death by activating the
hedgehog pathway to inhibit breast cancer cell proliferation
(25). However, the potential effect
of itraconazole on colon cancer and the role of autophagy in colon
cancer cell proliferation remain unclear.
The present study aimed to investigate the effects
of itraconazole on the antitumor development of colon cancer,
determine its underlying molecular mechanisms, and used a
nationwide database analysis to determine the associations between
basic experiments and clinical manifestations. The results
presented here provide evidence that itraconazole may act as a
promising drug for colon cancer therapy.
Materials and methods
Data collection
This observational cohort study used the cancer
registry dataset of the ‘2010 Longitudinal Generation Tracking
Database (LGTD 2010),’ established by the Health and Welfare Data
Science Center (HWDC), Ministry of Health and Welfare (MOHW) in
Taiwan. The Taiwan National Health Insurance (NHI) program has been
implemented since 1995 and offers comprehensive medical care
coverage to >99% of the country's 23 million inhabitants
(26).
The present study collected data from the LGTD 2010,
which were randomly selected from a cohort of 2 million individuals
covered by the NHI Program, between January 2011 and December 2011.
The LGTD 2010 contains all information on diagnostic codes,
detailed prescriptions, dates of clinical visits, dates of
admission and discharge, and expenditures for the enrollees of 2
million beneficiaries (26). No
statistical differences was observed in the age and sex
distributions between the cohorts in the LGTD and Taiwan NHI
enrollees. All patients were regularly monitored after diagnosis
until death or at the last follow-up date. Follow-up was completed
on December 31, 2015 (27). From the
Taiwanese National Health Insurance Research Database (https://www.mohw.gov.tw/mp-2.html), data from
patients newly diagnosed with colon cancer were identified using
the International Classification of Diseases for Oncology codes
(C180-C189), treated with or without itraconazole, according to the
Anatomical Therapeutic Chemical code: J02AC02, between January 2011
and December 2015. A total of 5,221 patients newly diagnosed with
colon cancer were included in the present study, and confounding
factors, such as age, sex, clinical stage, surgery, radiation
therapy and chemotherapy were adjusted accordingly. Patients were
pathologically diagnosed with primary colon cancer based on Taiwan
Cancer Registry, a population-based cancer registry enrolled. The
exclusion criteria for this study were patients' age at diagnosis
was unknown; incomplete data of tumor stage, pathology and
treatment history; lacking clear records on survival status;
previous history of cancer; incomplete data on the American Joint
Committee on Cancer (AJCC) stage and cause of death. All staging
was performed according to the AJCC Staging System (7th edition)
(28). To protect the privacy of
individuals included in the database, the NHI only released data
with encoded identification numbers so that personnel without
authorization would not be able to associate any direct information
to the enrollees. The Bureau of NHI approved the application after
reviewing all the required medical documents. The present study was
approved by the Institutional Review Board of Kaohsiung Veterans
General Hospital (IRB no. KSVGH18-CT10-07) and performed in
accordance with the tenets of the Declaration of Helsinki (1975)
and its later amendments (2013). The requirement for written
informed consent was waived.
Cell culture
The human colon adenocarcinoma cell line, COLO 205,
was purchased from the Food Industry Research and Development
Institute, and the colorectal cell line, HCT 116, was kindly gifted
by Dr Tzu-Ming Jao from Kaohsiung Veterans General Hospital
(Kaohsiung, Taiwan). Both cell lines were maintained in DMEM
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% fetal
bovine serum (Sigma-Aldrich; Merck KGaA), 2 mM L-glutamine (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin and streptomycin
(Invitrogen; Thermo Fisher Scientific, Inc.), at 37°C with 5%
CO2.
To determine the role of autophagy on itraconazole
induced apoptosis in COLO 205 cells, 5 mM 3-MA (3-methyladenine,
Sigma-Aldrich; Merck KGaA) was co-cultured with 50 µM itraconazole
(Sigma-Aldrich; Merck KGaA) at 37°C for 24 h.
MTT assay
The MTT assay (Sigma-Aldrich; Merck KGaA) was
performed to assess the cytotoxic effect of itraconazole on
COLO-205 and HCT 116 cells. A total of 1×104 cells were
seeded into 96-well plates and cultured in DMEM (Gibco; Thermo
Fisher Scientific, Inc.) supplemented with 10% fetal bovine serum
(Sigma-Aldrich; Merck KGaA), 2 mM L-glutamine (Gibco; Thermo Fisher
Scientific, Inc.) and 1% penicillin and streptomycin (Invitrogen;
Thermo Fisher Scientific, Inc.), at 37°C with 5% CO2 for
overnight. Following incubation, 1, 2, 5, 10, 25, 30, 50, 60, 100,
125, 250 and 500 µM itraconazole were added into each well and
cells were incubated for an additional 24 h at 37°C. The culture
medium was subsequently replaced with complete medium (DMEM, Gibco;
Thermo Fisher Scientific, Inc.) containing 5 mg/ml MTT and
incubated for 2 h at 37°C. The purple formazan crystals were
dissolved using DMSO and data were subsequently measured at a
wavelength of 570 nm, using an ELISA reader (BioTek Instruments,
Inc.).
Small interfering (si)RNA
transfection
The commercial siRNA gene silencer (LC3B) and
scrambled siRNA oligo were purchased from Cell Signaling
Technology, Inc. (cat. nos. 6212S and 6568S). Briefly,
1×106 COLO 205 and HCT 116 cells were seeded into 6-well
plates, respectively, and cultured in complete medium overnight at
37°C. siRNA (100 nM) and scrambled siRNA (100 nM) were transfected
into cells using Lipofectamine® RNAiMAX (Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Following incubation at 37°C for 24 h, the supernatant was removed,
washed with PBS and incubated with different concentrations of
itraconazole in complete medium for an additional 24 h at 37°C.
Representative images were obtained from three independent
experiments.
Cell Counting Kit-8 (CCK-8) assay
Following transfection with LC3B or scrambled siRNA,
COLO 205 and HCT 116 cells were treated with different
concentrations of itraconazole in triplicate wells at 37°C for 24
h. Cell viability was assessed via the CCK-8 assay. Briefly, 100 µl
of cell suspension was seeded into a 96-well plate, and 10 µl of
CCK-8 reagent (Sigma-Aldrich; Merck KGaA) was added to each well
and incubated for 1 h. Optical density was measured at a wavelength
of 450 nm, using an enzyme microplate reader (Synergy HTX; BioTek
Instruments, Inc.).
Trypan blue exclusion assay
Following transfection with LC3B or scrambled siRNA,
COLO 205 and HCT 116 cells were treated with different
concentrations of itraconazole in triplicate wells for 24 h. Cells
were collected and resuspended in the culture medium. Subsequently,
cells were mixed with trypan blue at a ratio of 1:1 (Gibco; Thermo
Fisher Scientific, Inc.) and counted using a hemocytometer (Yung
Yuen Scientific Instrument Co., Ltd.).
Annexin V/PI staining
Itraconazole-induced cell apoptosis was detected via
Annexin V/PI staining. A total of 1×106 cells were
seeded into 6-well plates and incubated in complete medium
overnight at 37°C. Following attachment to the plate, cells were
treated with different concentrations of itraconazole for 24 h.
Annexin V/PI staining was performed according to the manufacturer's
instructions (AAT Bioquest, Inc.). The percentage of apoptotic
cells was detected using FACSCalibor (BD Biosciences).
Representative images are shown for three independent
experiments.
Cell cycle analysis
A total of 1×106 COLO 205 and HCT 116
cells were seeded into 6-well plates and treated with different
concentrations of itraconazole for 24 h. Following treatment, cells
were fixed with 100% ethanol overnight at −20°C. DNA in the nuclei
was stained with PI (50 µg/ml) and RNase A (0.5 µg/ml) for 30 min
at 4°C. Cell cycle distribution was analyzed using an Attune NxT
flow cytometer (Thermo Fisher Scientific, Inc.). Representative
images were obtained from three independent experiments.
Reverse transcription-quantitative
(RT-q)PCR
Following treatment with itraconazole for 24 h, mRNA
was extracted from COLO 205 and HCT 116 cells using the GENEzol™
TriRNA Pure kit (Geneaid, http://www.geneaid.com/Tri-RNA/GZXD), according to the
manufacturer's instructions. A total of 1 µg RNA was reverse
transcribed into cDNA using the cDNA reverse transcription kit
(Morrebio, http://www.light-biotech.com/pcr), according to the
manufacturer's instructions. The temperature protocol for RT was as
follows: 50°C for 15 min followed by 85°C for 5 sec, and the cDNA
products were used for the gene expression assay. The PCR primer
and the TaqMan probe were purchased from Thermo Fisher Scientific,
Inc. The primer and probe sequences used in the present study were
as follows: LC3B (Gene ID: Hs00797944_s1), p62 (Gene ID:
Hs02621445_s1) and GAPDH (Gene ID: Hs02786624_g1). Gene expression
was detected via qPCR (StepOnePlus™; Thermo Fisher Scientific,
Inc.) and the protocol was as follows: 10 µl 2× PCR master mix, 2
µl cDNA, 7 µl dd H2O and 1 µl probe were mixed in each
reaction. The following thermocycling conditions were used for
qPCR: 50°C for 2 min, 95°C for 2 min followed by 40 cycles of 95°C
for 1 sec and 60°C for 20 sec. LC3B and p62 expression levels were
calculated using the 2−ΔΔCq method and normalized to
β-actin (29).
Western blotting
A total of 1×106 COLO 205 and HCT 116
cells, were seeded into 6-well plates and treated with different
concentrations of itraconazole for 24 h. Following treatment, total
protein was extracted using RIPA buffer (Biomed, http://www.bio-protech.com.tw/products_detailed.php?id=347)
and quantified using the BCA kit (Invitrogen; Thermo Fisher
Scientific, Inc.). Equal amounts (25 µg/lane) of protein extracts
were subjected in 12% polyacrylamide gel and separated by
electrophoresis then transferred onto PVDF membranes and blocked
with 5% skim milk for 1 h at room temperature. Membranes were
washed three times with PBST (PBS with 0.05% Tween-20) and
incubated with primary antibodies against TKT (1:1,000; cat. no.
SC-390179; Santa Cruz Biotechnology, Inc.), Cleaved caspase-3
(1:1,000; cat. no. 9664; Cell Signaling Technology, Inc.),
Caspase-3 (1:1,000; cat. no. E-AB-30756; Elabscience, http://www.elabscience.com/PDF/Cate81/E-AB-30756-Elabscience.pdf),
Bax (1:1,000; cat. no. 2772; Cell Signaling Technology, Inc.), LC3B
(1:1,000; cat. no. 83506; Cell Signaling Technology, Inc.), p62
(1:1,000; cat. no. 5114; Cell Signaling Technology, Inc.), Beclin-1
(1:1,000; cat. no. 3738; Cell Signaling Technology, Inc.) and
β-actin (1:5,000; cat. no. E-AB-20034; Elabscience) overnight at
4°C. The membranes were washed three times with PBST and
subsequently incubated with anti-mouse IgG, HRP-linked antibody
(1:5,000; cat. no. 7076P2; Cell Signaling Technology, Inc.) or goat
anti-rabbit IgG antibody (1:5,000; cat. no. A0545; Sigma-Aldrich;
Merck KGaA) for 1 h at room temperature. Protein bands were
visualized using the ECL western blot detection kit (Thermo Fisher
Scientific, Inc.) and analyzed using Alliance Q9 software (V18.11;
UVITEC; http://www.uvitec.co.uk/alliance-q9-advanced).
Representative images and band quantification were obtained from
three independent experiments.
Confocal microscopy
To detect autophagosome formation, 1×105
COLO 205 and HCT 116 cells were seeded onto cover glasses in a
6-well plate. Following treatment with itraconazole for 24 h, cells
were fixed with 4% paraformaldehyde (Sigma-Aldrich; Merck KGaA) for
15 min at room temperature and permeabilized with 0.5% Triton X-100
(Sigma-Aldrich; Merck KGaA) in PBS for 15 min at room temperature.
Cells were subsequently blocked with 1% BSA for 1 h at room
temperature. Cells were incubated with LC3B primary antibody (Cell
Signaling Technology, Inc.) overnight at 4°C, followed by
incubation with FITC-conjugated secondary antibody (Jackson
ImmunoResearch Laboratories, Inc.) for 1 h at room temperature.
DAPI was used for nuclear staining. Cells were observed under a
Zeiss LSM 5 Pascal confocal microscope (Carl Zeiss). Number of LC3
puncta per cell was further quantified using ImageJ software
(Version 1.50i; National Institutes of Health) (30). Representative images were obtained
from three independent experiments.
Statistical analysis
Statistical analysis was performed using SAS
software (version 9.3; SAS Institute, Inc.) and SPSS software
(version 20; IBM Corp.). Descriptive statistics were used to
analyze baseline demographic data and the distribution of each
variable among the study population. Continuous variables are
presented as the mean ± SD. Survival analysis was performed using
the Kaplan-Meier method and log-rank test. Univariate Cox
regression analysis was performed to assess the association between
survival and the impact of itraconazole. Hazard ratios (HRs) and
their 95% confidence intervals (CIs) from the Cox regression
analyses were used to estimate the relative risk. Then stratified
survival analyses were also performed for different groups based on
multivariate Cox regression analysis. The results of the control
and treatment with different concentrations of itraconazole were
analyzed via one-way ANOVA followed by Tukey's post hoc test, and
performed using GraphPad Prism software (version 6.0; GraphPad
Software, Inc.). P<0.05 was considered to indicate a
statistically significant difference.
Results
Baseline demographic
characteristics
In the present study, a total of 5,221 patients with
colon cancer were identified using ICD-O3 codes between 2011 and
2015, and data were analyzed. The demographic characteristics of
all patients, including age, sex, primary tumor location, AJCC
clinical stage, treatment, itraconazole use and mortality are
presented in Table I. The average
age of all patients was 65.6±13.7 years (age range, 21–85 years).
The proportions of stage I+II and stage III+IV were 42 and 58%,
respectively. Most patients underwent surgery, and the proportions
of patients who received radiation therapy and chemotherapy were 2
and 38%, respectively. Notably, only 1% of all patients received
itraconazole treatment. The mortality rate of all patients with
colon cancer was 34%.
 | Table I.Baseline characteristics of patients
with colon cancer (n=5,221). |
Table I.
Baseline characteristics of patients
with colon cancer (n=5,221).
| Characteristic | Patients, n
(%) |
|---|
| Age, years (mean ±
SD) | 65.6±13.7 |
| Sex |
|
|
Male | 2,921 (56) |
|
Female | 2,300 (44) |
| AJCC clinical
stage |
|
|
I+II | 2,206 (42) |
|
III+IV | 3,015 (58) |
| Surgery |
|
|
Yes | 4,095 (78) |
| No | 1,126 (22) |
| Radiation
therapy |
|
|
Yes | 88 (2) |
| No | 5,133 (98) |
| Chemotherapy |
|
|
Yes | 1,977 (38) |
| No | 3,244 (62) |
| Drug |
|
|
Yes | 62 (1) |
| No | 5,159 (99) |
| Mortality |
|
|
Yes | 1,780 (34) |
| No | 3,441 (66) |
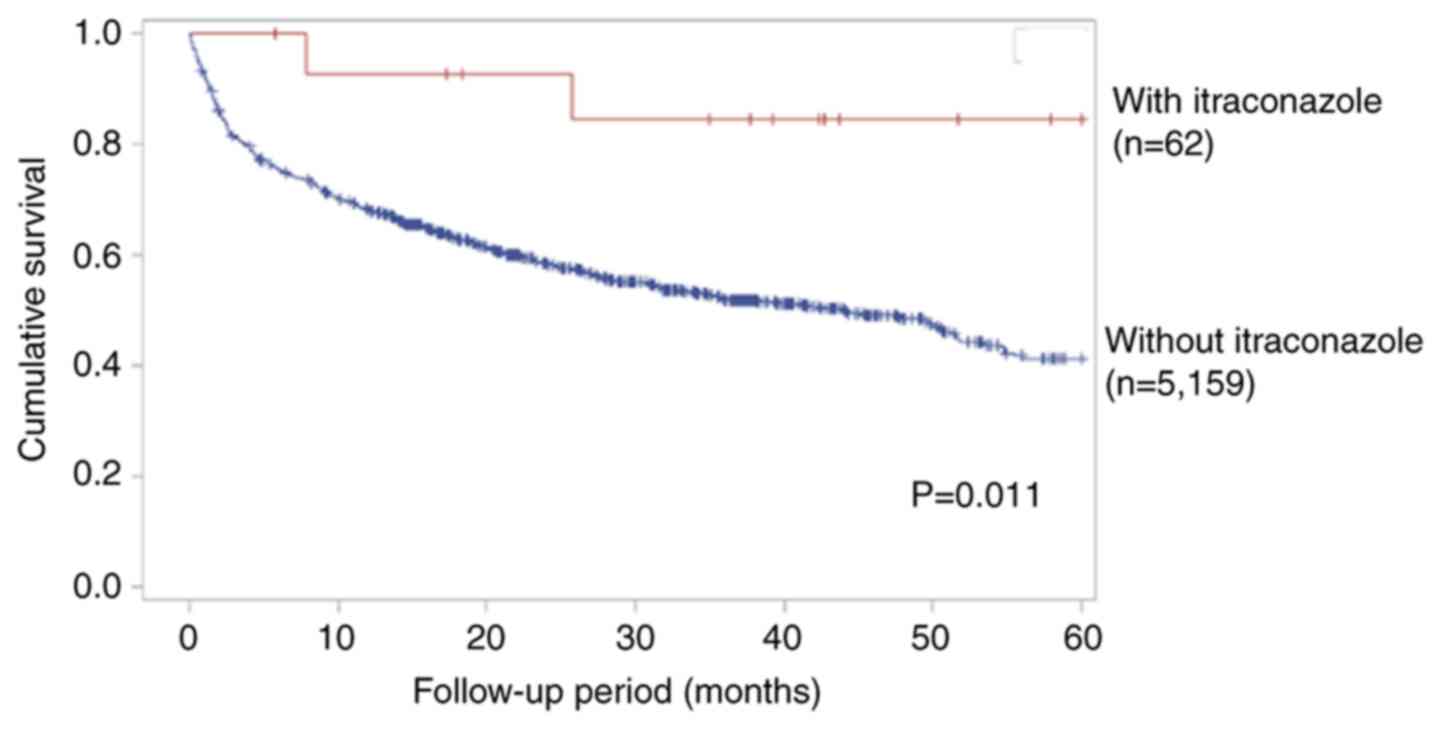
Itraconazole increases the 5-year
survival rate in patients with late-stage colon cancer who receive
chemotherapy
The univariate Cox proportional hazard model was
used to assess the HR for patients with colon cancer, in the
presence and absence of itraconazole treatment. Statistically
significant HRs for the 5-year survival rate were observed for age
(HR, 1.03; 95% CI, 1.01–1.04; P<0.001), AJCC clinical stage (HR,
3.08; 95% CI, 2.75–3.44; P<0.001), surgery (HR, 4.15; 95% CI,
3.77–4.57; P<0.001) and radiation therapy (HR, 0.43; 95% CI,
0.33–0.58; P<0.001) (Table II).
Subgroup analysis was performed to assess the effect of
itraconazole on patients with late-stage colon cancer who received
chemotherapy. Following adjustment for age, sex, surgery and
radiation therapy, the benefit of itraconazole treatment in
reducing the risk of mortality was observed (HR, 0.27; 95% CI,
0.07–1.09; P=0.067; Table III).
Although there was not statistically significant, however the trend
in reducing the risk of mortality could still be noted. The 5-year
survival rate also increased in patients treated with itraconazole
group, according to Kaplan-Meier curve by log-rank test with
significant statistical difference. (P<0.05; Fig. 1). Taken together, these results
suggest that itraconazole can increase the 5-year survival rate in
patients with late-stage colon cancer.
 | Table II.Univariate Cox regression analysis in
patients with colon cancer. |
Table II.
Univariate Cox regression analysis in
patients with colon cancer.
| Variable | HR (95% CI) | P-value |
|---|
| Age, years | 1.03
(1.01–1.04) |
<0.001a |
| Sex |
| 0.541 |
|
Male | Reference |
|
|
Female | 0.97
(0.88–1.07) |
|
| AJCC clinical
stage |
|
<0.001a |
|
I+II | Reference |
|
|
III+IV | 3.08
(2.75–3.44) |
|
| Surgery |
|
<0.001a |
|
Yes | Reference |
|
| No | 4.15
(3.77–4.57) |
|
| Radiation
therapy |
|
<0.001a |
|
Yes | Reference |
|
| No | 0.43
(0.33–0.58) |
|
| Chemotherapy |
| 0.493 |
|
Yes | Reference |
|
| No | 0.97
(0.88–1.06) |
|
| Drug |
| 0.434 |
|
Yes | Reference |
|
| No | 0.83
(0.52–1.32) |
|
 | Table III.Stratification multivariate Cox
regression analysis in patients with colon cancer, with advanced
AJCC clinical stage (III+IV). |
Table III.
Stratification multivariate Cox
regression analysis in patients with colon cancer, with advanced
AJCC clinical stage (III+IV).
| Variable | Adjusted HR (95%
CI) | P-value |
|---|
| Chemotherapy |
|
|
| Drug |
|
|
|
Yes | 0.27
(0.07–1.09) | 0.067 |
| No | Reference |
|
| No
chemotherapy |
|
|
| Drug |
|
|
|
Yes | 1.06
(0.47–2.38) | 0.883 |
| No | Reference |
|
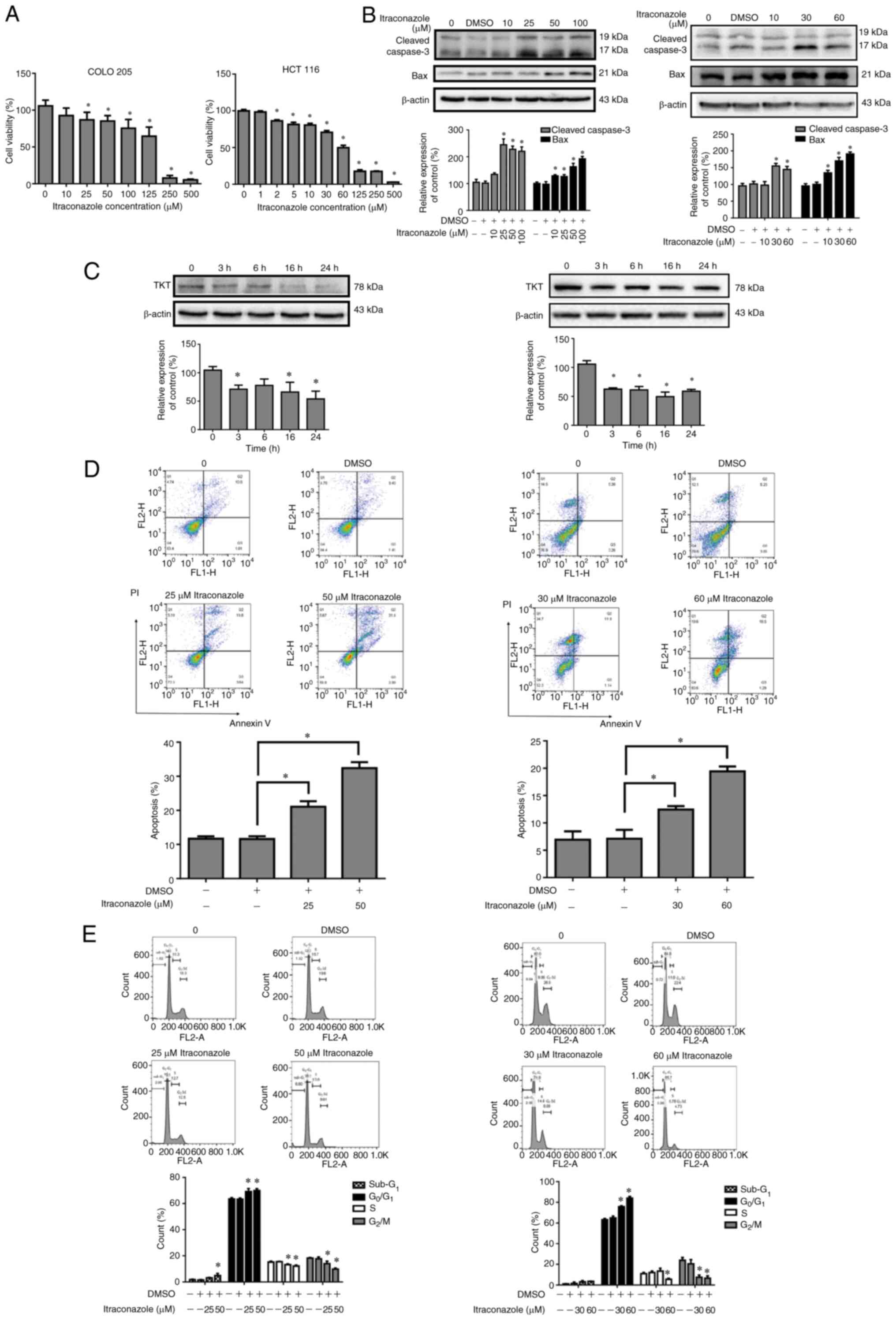
Itraconazole inhibits the
proliferation of colon cancer cells and induces apoptosis and cell
cycle arrest
To further investigate the effects and underlying
molecular mechanisms of itraconazole on colon cancer, the effect of
itraconazole on the proliferation of COLO 205 and HCT 116 cells was
assessed. Cells were treated with different concentrations of
itraconazole for 24 h, and cell viability was assessed via the MTT
assay. The results demonstrated that cell viability significantly
decreased following treatment with itraconazole, in a
dose-dependent manner (P<0.05; Fig.
2A). Apoptosis-related proteins and TKT expression were
detected in COLO 205 and HCT 116 cells treated with itraconazole.
The results demonstrated that the expression levels of cleaved
caspase-3 and Bax increased after itraconazole treatment, in a
dose-response manner for 24 h (P<0.05; Fig. 2B). TKT expression was decreased
following treatment with itraconazole in a time-dependent manner
(P<0.05; Fig. 2C). Annexin V-PI
staining demonstrated that COLO 205 and HCT 116 cells treated with
itraconazole significantly induced cell apoptosis (P<0.05;
Fig. 2D). Furthermore, treatment
with itraconazole significantly induced subG1 phase,
G1 arrest and decreased the number of cells in the
G2/M phase (P<0.05; Fig.
2E). Collectively, these results suggest that itraconazole
decreases colon cancer proliferation and TKT expression and induces
apoptosis.
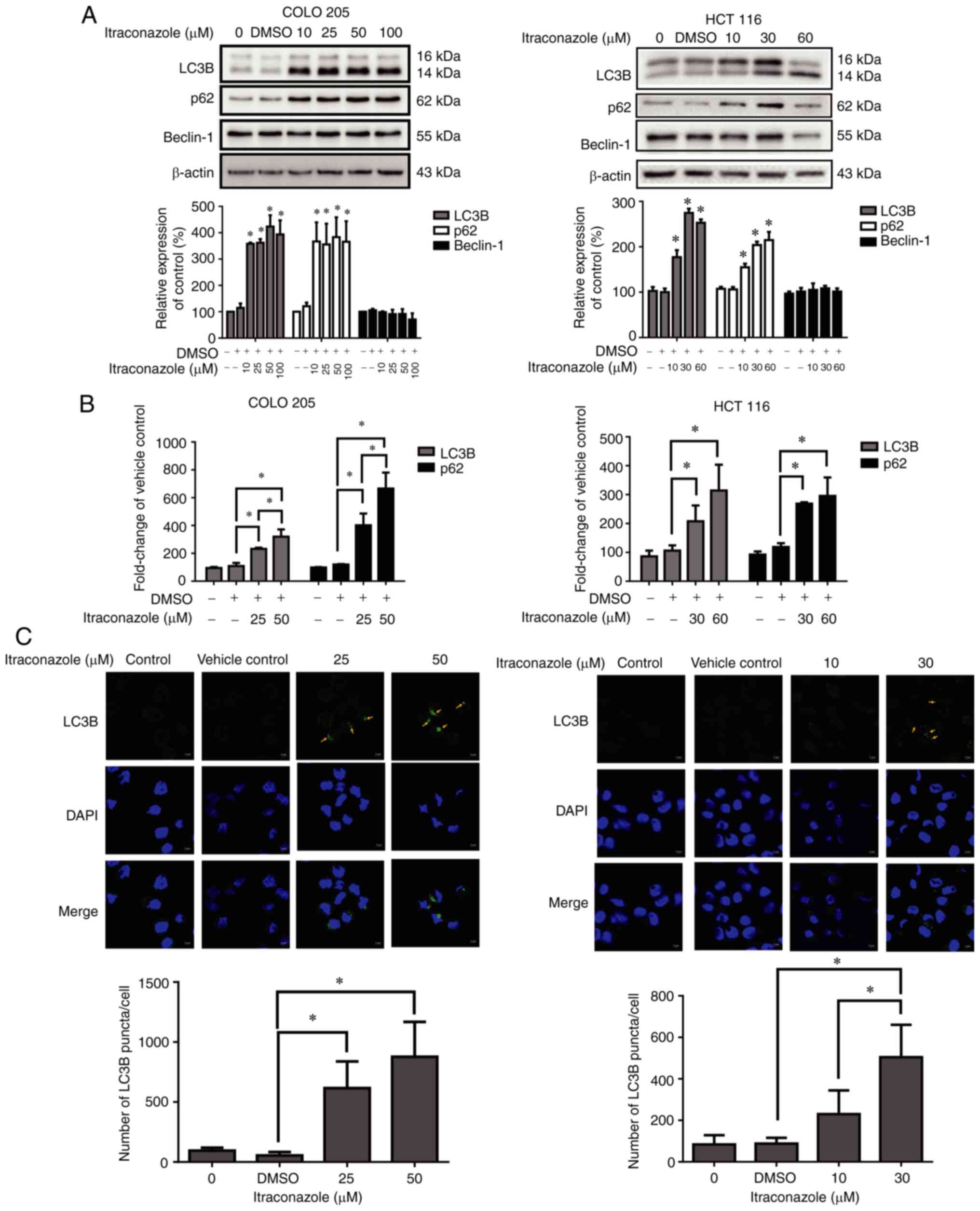
Itraconazole induces autophagy in
colon cancer
Autophagic cell death plays an important role in the
treatment of cancer (31);
therefore, the present study investigated the effect of
itraconazole on inducing autophagy in colon cancer cells. The
results demonstrated that the expression levels of the
autophagy-related proteins, LC3B and p62, significantly increased
in COLO 205 and HCT 116 cells following treatment with itraconazole
for 24 h (P<0.05; Fig. 3A), and
the mRNA expression levels of LC3B and p62 significantly increased
following treatment with itraconazole, in a concentration-dependent
manner (P<0.05; Fig. 3B). In
addition, the number of LC3B puncta/cell increased following
treatment with itraconazole (P<0.05; Fig. 3C). Taken together, these results
suggest that itraconazole can induce autophagy in colon cancer
cells.
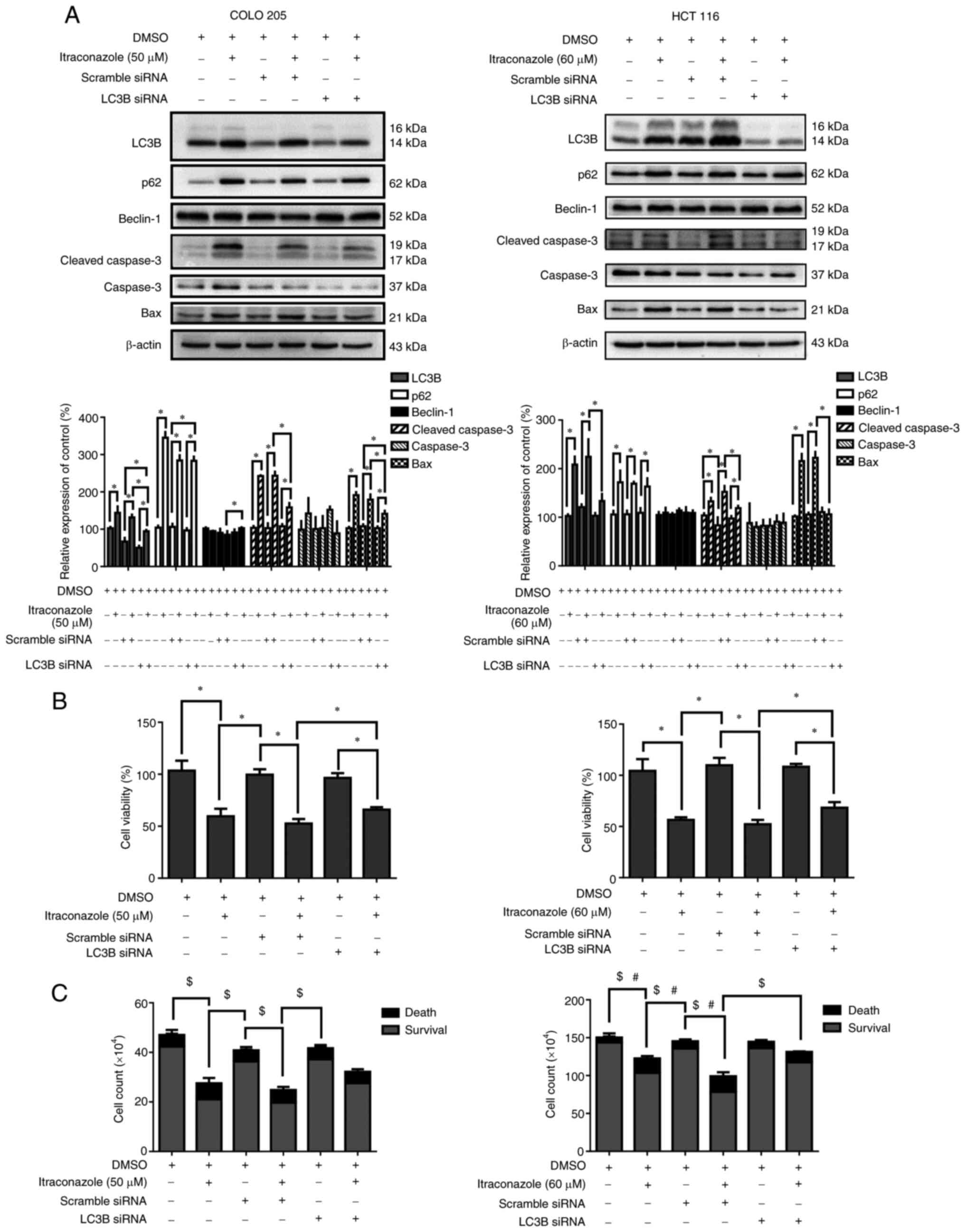
Itraconazole-induced autophagy
increases cell death in colon cancer cells
A previous study has reported that itraconazole can
induce both apoptosis and autophagy in colon cancer cells (32). The role of autophagy in
itraconazole-induced cell death remains controversial; thus, the
present study used autophagy siRNA targeting LC3B to determine the
role of autophagy in itraconazole-induced cell death. The results
demonstrated that the expression levels of the autophagy-related
proteins, LC3B and p62, decreased following transfection of COLO
205 and HCT 116 cells with LC3B siRNA (P<0.05; Fig. 4A). Furthermore, the expression levels
of the apoptosis-related proteins, cleaved caspase-3 and Bax, also
decreased (P<0.05; Fig. 4A). The
viability of cells transfected with LC3B siRNA was assessed via the
CCK-8 and trypan blue exclusion assays. The results demonstrated
that cell viability decreased following treatment with itraconazole
and transfection with the scrambled siRNA. Notably, these effects
were reversed following transfection with LC3B siRNA (P<0.05;
Fig. 4B and C). Collectively, these
results suggest that autophagy plays an important role in
itraconazole-induced cell death.
Discussion
The results of the present study demonstrated that
itraconazole increased the 5-year survival rate of patients with
late-stage colon cancer. Furthermore, treatment with itraconazole
decreased TKT expression and induced apoptosis in vitro.
Taken together, these results suggest that autophagy plays an
important role in itraconazole-induced cell death. Itraconazole is
a well-known medication for treating fungal infection and giving
new indications to the FDA proofed drug is safe and cost-effective
(33,34). Notably, itraconazole is associated
with clinical outcomes and survival rates in patients with advanced
gastric cancer (35). The results of
the present study suggest that itraconazole may be a promising
adjuvant therapeutic agent for colon cancer, based on the analyses
of medical records in Taiwan.
As an FDA-approved antifungal drug, itraconazole has
been demonstrated to inhibit the proliferation of gastric and liver
cancer cells (36,37). Itraconazole targets AMP-activated
protein kinase activation in the Hedgehog pathway, and inhibition
of this signaling pathway can significantly decrease cell
proliferation (32). Itraconazole
induces cell cycle arrest at G1 and G2 phases
in non-small cell lung cancer and also induces apoptosis (38). Alterations in the energy metabolism
of cancer cells may be due to impaired mitochondrial function
(39). Given that the glycolytic
pathway serves as an intermediate and signaling complex, key
enzymes in the signaling network, such as TKT, may be used as
promising therapeutic candidates (40). The results of the present study
demonstrated that itraconazole decreased cell viability and induced
cell apoptosis, and in the cell cycle, induced subG1
phase and G1 arrest. Furthermore, cleaved caspase-3
expression increased, while TKT expression decreased following
treatment with itraconazole. Collectively, these results suggest
that itraconazole inhibits the proliferation and induces the
apoptosis of colon cancer cells.
Autophagy plays a distinct role in tumor
progression. In cancer cell biology, disruption of
autophagy-related gene expression can enhance spontaneous tumor
progression and increase the expansion of hepatitis B virus-induced
premalignant lesions (41).
Furthermore, autophagy-related genes, such as mTOR, class I PI3K
and AKT, activate oncogenes to inhibit autophagy and enhance tumor
formation (42). The results of the
present study demonstrated that itraconazole increased the
expression levels of the autophagy-related proteins, LC3B and p62,
and LC3B puncta formation. Notably, p62 expression increased
following treatment with itraconazole, and autophagy was also
enhanced. A recent study demonstrated that p62 expression can serve
as a predictor for drug-induced autophagic cell death (43). Furthermore, p62 depletion suppresses
the recruitment of LC3B to autophagosomes, which increases the
basal level of LC3B in cells overexpressing p62 (44). Taken together, these results suggest
that p62 expression is associated with autophagic activity.
It is well-known that autophagy is an evolutionarily
conserved intracellular recycling system; thus, with increased
intracellular oxidative stress and organ damage, autophagy begins
to self-degrade the injured organelle, producing more ATP to
maintain cell function and survival (31). Given that 3-MA can induce undesired
cytotoxicity on COLO 205 cells, it is difficult to identify the
exact role of autophagy on itraconazole-induced apoptosis (Fig. S1); thus, the present study used
siRNA to silence LC3B expression and identify the role of autophagy
in itraconazole-treated colon cancer cells. The results
demonstrated that decreased autophagy significantly increased cell
viability compared with the itraconazole-only group. Taken
together, these results suggest that itraconazole can induce both
apoptosis and autophagy to inhibit the proliferation of colon
cancer cells.
In the present study, the effect of itraconazole on
the inhibition of colon cancer cell proliferation was reflected by
an increased 5-year survival rate. However, the present study is
not without limitations. First, itraconazole is not the standard
treatment for colon cancer (45);
thus, the number of patients with colon cancer treated with
itraconazole was extremely small. Increasing the sample size of the
database will increase the reliability of the data to confirm the
clinical outcome of itraconazole treatment. Secondly, the present
study only performed in vitro experiments to determine the
effects of itraconazole on colon cancer cells. Thus, prospective
studies will perform in vivo experiments to determine
whether itraconazole directly inhibits colon cancer growth in mice.
Currently, clinical trials assessing the effects of itraconazole on
patients with colon cancer are ongoing. Thirdly, HCT 116 cell
proliferation was inefficient in the matrix gel, thus the colony
formation assay was unable to be performed to determine whether
itraconazole induces apoptosis in both COLO 205 and HCT 116 cells
(data not shown). Alternatively, the present study performed
Annexin V/PI staining analysis to determine the effect of
itraconazole on the apoptosis of colon cancer cells. Lastly, the
associations between TKT, autophagy and apoptosis are highly
complicated, involving gene-gene interactions and a specific
sequence of gene expression. Thus, further studies are required to
determine the potential role of itraconazole-induced autophagy in
protecting cell survival or promoting cell death.
To the best of our knowledge, the present study was
the first to assess the in vitro effects of itraconazole on
inhibiting the proliferation of colon cancer cells. The results
presented here offer a potential mechanism of action of
itraconazole in inducing apoptosis and autophagy; thus,
itraconazole may be used as a therapeutic target for the treatment
of colon cancer.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was partly supported by the
Kaohsiung Veterans General Hospital (grant nos. VGHKS109-D04-1 and
VGHKS109-180).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author upon reasonable
request.
Authors' contributions
PWS, YMC, and SJY designed the experiments. CLL, ECL
and HSH performed the experiments and molecular biology experiment
results statistics. CHY and CLC acquired and analyzed the data.
PWS, YMC, CLL and SJY drafted the initial manuscript. HSH and SJY
confirmed the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Review Board of Kaohsiung Veterans General Hospital (IRB no.
KSVGH18-CT10-07) and performed in accordance with the tenets of the
Declaration of Helsinki (1975) and its later amendments (2013). The
requirement for written informed consent was waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Miller KD, Siegel RL, Lin CC, Mariotto AB,
Kramer JL, Rowland JH, Stein KD, Alteri R and Jemal A: Cancer
treatment and survivorship statistics, 2016. CA Cancer J Clin.
66:271–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Marisa L, de Reyniès A, Duval A, Selves J,
Gaub MP, Vescovo L, Etienne-Grimaldi MC, Schiappa R, Guenot D,
Ayadi M, et al: Gene expression classification of colon cancer into
molecular subtypes: Characterization, validation, and prognostic
value. PLoS Med. 10:e10014532013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Färkkilä N, Torvinen S, Sintonen H, Saarto
T, Järvinen H, Hänninen J, Taari K and Roine RP: Costs of
colorectal cancer in different states of the disease. Acta Oncol.
54:454–462. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aiello P, Sharghi M, Mansourkhani SM,
Ardekan AP, Jouybari L, Daraei N, Peiro K, Mohamadian S, Rezaei M,
Heidari M, et al: Medicinal plants in the prevention and treatment
of colon cancer. Oxid Med Cell Longev. 2019:20756142019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang Yp, Hu Lf, Zheng Hf, Mao Cj, Hu Wd,
Xiong Kp, Wang F and Liu Cf: Application and interpretation of
current autophagy inhibitors and activators. Acta Pharmacol Sin.
34:625–635. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Onorati AV, Dyczynski M, Ojha R and
Amaravadi RK: Targeting autophagy in cancer. Cancer. 124:3307–3318.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Islam MA, Sooro MA and Zhang P: Autophagic
regulation of p62 is critical for cancer therapy. Int J Mol Sci.
19:14052018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lauren P: The two histological main types
of gastric carcinoma: Diffuse and so-called intestinal-type
carcinoma. An attempt at a histo-clinical classification. Acta
Pathol Microbiol Scand. 64:31–49. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jin S, Wei J, You L, Liu H and Qian W:
Autophagy regulation and its dual role in blood cancers: A novel
target for therapeutic development (Review). Oncol Rep.
39:2473–2481. 2018.PubMed/NCBI
|
|
10
|
Huang F, Wang BR and Wang YG: Role of
autophagy in tumorigenesis, metastasis, targeted therapy and drug
resistance of hepatocellular carcinoma. World J Gastroenterol.
24:4643–4651. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xiang Y, Zhao J, Zhao M and Wang K:
Allicin activates autophagic cell death to alleviate the malignant
development of thyroid cancer. Exp Ther Med. 15:3537–3543.
2018.PubMed/NCBI
|
|
12
|
Stepanova NG and Demcheva MV: Formation of
a pentose phosphate cycle metabolite, erythrose-4-phosphate, from
initial compounds of glycolysis by transketolase from the rat
liver. Biokhimiia. 52:1907–1913. 1987.(In Russian). PubMed/NCBI
|
|
13
|
Warburg O: On the origin of cancer cells.
Science. 123:309–314. 1956. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Qin Z, Xiang C, Zhong F, Liu Y, Dong Q, Li
K, Shi W, Ding C, Qin L and He F: Transketolase (TKT) activity and
nuclear localization promote hepatocellular carcinoma in a
metabolic and a non-metabolic manner. J Exp Clin Cancer Res.
38:1542019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ricciardelli C, Lokman NA, Cheruvu S, Tan
IA, Ween MP, Pyragius CE, Ruszkiewicz A, Hoffmann P and Oehler MK:
Transketolase is upregulated in metastatic peritoneal implants and
promotes ovarian cancer cell proliferation. Clin Exp Metastasis.
32:441–455. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chao YK, Peng TL, Chuang WY, Yeh CJ, Li
YL, Lu YC and Cheng AJ: Transketolase serves a poor prognosticator
in esophageal cancer by promoting cell invasion via
epithelial-mesenchymal transition. J Cancer. 7:1804–1811. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu LH, Yang JH, Zhang DT, Zhang S, Wang L,
Cai PC, Zheng JF and Huang JS: The TKTL1 gene influences total
transketolase activity and cell proliferation in human colon cancer
LoVo cells. Anticancer Drugs. 18:427–433. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cao L, Hong W, Cai P, Xu C, Bai X, Zhao Z,
Huang M and Jin J: Cryptotanshinone strengthens the effect of
gefitinib against non-small cell lung cancer through inhibiting
transketolase. Eur J Pharmacol. 890:1736472021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bhatia A, Kanish B, Badyal DK, Kate P and
Choudhary S: Efficacy of oral terbinafine versus itraconazole in
treatment of dermatophytic infection of skin-A prospective,
randomized comparative study. Indian J Pharmacol. 51:116–119. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pantziarka P, Sukhatme V, Bouche G, Meheus
L and Sukhatme VP: Repurposing drugs in oncology
(ReDO)-itraconazole as an anti-cancer agent. Ecancermedicalscience.
9:5212015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Choi CH, Ryu JY, Cho YJ, Jeon HK, Choi JJ,
Ylaya K, Lee YY, Kim TJ, Chung JY, Hewitt SM, et al: The
anti-cancer effects of itraconazole in epithelial ovarian cancer.
Sci Rep. 7:65522017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu R, Li J, Zhang T, Zou L, Chen Y, Wang
K, Lei Y, Yuan K, Li Y, Lan J, et al: Itraconazole suppresses the
growth of glioblastoma through induction of autophagy: Involvement
of abnormal cholesterol trafficking. Autophagy. 10:1241–1255. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rudin CM, Brahmer JR, Juergens RA, Hann
CL, Ettinger DS, Sebree R, Smith R, Aftab BT, Huang P and Liu JO:
Phase 2 study of pemetrexed and itraconazole as second-line therapy
for metastatic nonsquamous non-small-cell lung cancer. J Thorac
Oncol. 8:619–623. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Antonarakis ES, Heath EI, Smith DC,
Rathkopf D, Blackford AL, Danila DC, King S, Frost A, Ajiboye AS,
Zhao M, et al: Repurposing itraconazole as a treatment for advanced
prostate cancer: A noncomparative randomized phase II trial in men
with metastatic castration-resistant prostate cancer. Oncologist.
18:163–173. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang X, Wei S, Zhao Y, Shi C, Liu P, Zhang
C, Lei Y, Zhang B, Bai B, Huang Y and Zhang H: Anti-proliferation
of breast cancer cells with itraconazole: Hedgehog pathway
inhibition induces apoptosis and autophagic cell death. Cancer
Lett. 385:128–136. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hsieh CY, Su CC, Shao SC, Sung SF, Lin SJ,
Yang YH and Lai EC: Taiwan's national health insurance research
database: Past and future. Clin Epidemiol. 11:349–358. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin CC, Lai MS, Syu CY, Chang SC and Tseng
FY: Accuracy of diabetes diagnosis in health insurance claims data
in Taiwan. J Formos Med Assoc. 104:157–163. 2005.PubMed/NCBI
|
|
28
|
Edge SB and Compton CC: The American joint
committee on cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zonoobi E, Saeedfar K, Pourdowlat G,
Masjedi MR and Behmanesh M: The study of IL-10 and IL-17A genes
expression in patients with different stages of asthma: A
case-control study. Tanaffos. 17:146–154. 2018.PubMed/NCBI
|
|
30
|
Runwal G, Stamatakou E, Siddiqi FH, Puri
C, Zhu Y and Rubinsztein DC: LC3-positive structures are prominent
in autophagy-deficient cells. Sci Rep. 9:101472019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yun CW and Lee SH: The roles of autophagy
in cancer. Int J Mol Sci. 19:34662018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Deng H, Huang L, Liao Z, Liu M, Li Q and
Xu R: Itraconazole inhibits the Hedgehog signaling pathway thereby
inducing autophagy-mediated apoptosis of colon cancer cells. Cell
Death Dis. 11:5392020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Whitburn J, Edwards CM and Sooriakumaran
P: Metformin and prostate cancer: A new role for an old drug. Curr
Urol Rep. 18:462017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bellmann R and Smuszkiewicz P:
Pharmacokinetics of antifungal drugs: Practical implications for
optimized treatment of patients. Infection. 45:737–779. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lan K, Yan R, Zhu K, Li W, Xu Z, Dang C
and Li K: Itraconazole inhibits the proliferation of gastric cancer
cells in vitro and improves patient survival. Oncol Lett.
16:3651–3657. 2018.PubMed/NCBI
|
|
36
|
Wang W, Dong X, Liu Y, Ni B, Sai N, You L,
Sun M, Yao Y, Qu C, Yin X and Ni J: Itraconazole exerts anti-liver
cancer potential through the Wnt, PI3K/AKT/mTOR, and ROS pathways.
Biomed Pharmacother. 131:1106612020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hu Q, Hou YC, Huang J, Fang JY and Xiong
H: Itraconazole induces apoptosis and cell cycle arrest via
inhibiting Hedgehog signaling in gastric cancer cells. J Exp Clin
Cancer Res. 36:502017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Alhakamy NA and Md S: Repurposing
itraconazole loaded PLGA nanoparticles for improved antitumor
efficacy in non-small cell lung cancers. Pharmaceutics. 11:6852019.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zheng J: Energy metabolism of cancer:
Glycolysis versus oxidative phosphorylation (Review). Oncol Lett.
4:1151–1157. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gill KS, Fernandes P, O'Donovan TR,
McKenna SL, Doddakula KK, Power DG, Soden DM and Forde PF:
Glycolysis inhibition as a cancer treatment and its role in an
anti-tumour immune response. Biochim Biophys Acta. 1866:87–105.
2016.PubMed/NCBI
|
|
41
|
Qu X, Yu J, Bhagat G, Furuya N, Hibshoosh
H, Troxel A, Rosen J, Eskelinen EL, Mizushima N, Ohsumi Y, et al:
Promotion of tumorigenesis by heterozygous disruption of the beclin
1 autophagy gene. J Clin Invest. 112:1809–1820. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Choi AMK, Ryter SW and Levine B: Autophagy
in human health and disease. New Engl J Med. 368:651–662. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chu CW, Ko HJ, Chou CH, Cheng TS, Cheng
HW, Liang YH, Lai YL, Lin CY, Wang C, Loh JK, et al: Thioridazine
enhances P62-mediated autophagy and apoptosis through
wnt/beta-catenin signaling pathway in glioma cells. Int J Mol Sci.
20:4732019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bjørkøy G, Lamark T, Brech A, Outzen H,
Perander M, Overvatn A, Stenmark H and Johansen T: p62/SQSTM1 forms
protein aggregates degraded by autophagy and has a protective
effect on huntingtin-induced cell death. J Cell Biol. 171:603–614.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Watanabe T, Muro K, Ajioka Y, Hashiguchi
Y, Ito Y, Saito Y, Hamaguchi T, Ishida H, Ishiguro M, Ishihara S,
et al: Japanese society for cancer of the colon and rectum (JSCCR)
guidelines 2016 for the treatment of colorectal cancer. Int J Clin
Oncol. 23:1–34. 2018. View Article : Google Scholar : PubMed/NCBI
|