Introduction
TETs, including thymoma and thymic carcinoma (TC),
are the primary neoplasms of epithelial cells in the thymic gland
of the anterior mediastinum. Thymoma is a low-grade malignant tumor
associated with myasthenia gravis and other autoimmune diseases,
whereas TC is an aggressive tumor with no known molecular links to
autoimmune diseases (1). The World
Health Organization (WHO) histopathological classification system
distinguishes thymoma from TC and neuroendocrine tumors of the
thymus (NECTT), and divides thymoma into five categories (types A,
AB, B1, B2 and B3) based on the morphology of epithelial cells and
the lymphocyte-to-epithelial cell ratio (2). To date, various staging systems have
been proposed for TETs. The Masaoka-Koga classification is
currently the most widely accepted clinical staging system, which
is based on local invasion and tumor metastasis (3). The new TNM stage classification system,
now in the 8th edition, can be used for all TETs, and considers not
only tumor characteristics, but also lymph nodal and metastatic
characteristics (4).
Treatment of TETs may involve surgery, radiation
and/or chemotherapy, and optimal treatment depends on the clinical
stage. The resectability of a tumor influences patient prognosis,
and surgery remains the mainstay of treatment for TETs. Surgical
resection and/or postoperative radiotherapy is appropriate for most
patients with completely resected TETs. Multimodality therapy
involving surgery, chemotherapy and radiotherapy appears to
increase the rate of complete resection and survival in advanced
TETs (5–7); one-third of patients that present with
advanced-stage or relapsing tumors require administration of
chemotherapy, and systemic review recommends that the most popular
and active regimens are cisplatin-anthracycline and
cisplatin-etoposide combinations (8,9).
However, metastatic and inoperable refractory/recurrent TETs,
particularly TC, are associated with poor prognosis (10). The rarity of these tumors makes
clinical trials difficult, and the development of novel drugs is
slow (11). Thus, focusing on
specific molecular alterations may provide a clearer understanding
of TETs and aid the development of targeted therapies.
In the past decade, attempts have been made to
characterize TETs at the molecular level, and the aberrant
expression of EGFR (12), VEGF
(13), insulin-like growth factor 1
receptor (14) and programmed cell
death protein 1/programmed cell death 1 ligand 1 (15) has been shown to contribute to the
tumorigenesis of TETs. However, the rarity of these tumors hampers
the development of targeted therapy (11). Only a few targeted drugs have
progressed to phase-II trials, including mTOR (16) and KIT tyrosine kinase inhibitors
(17). Therefore, the identification
of novel biological targets is important for the introduction of
molecular profiling-directed therapies in clinical practice
(11).
Our previous study included the genome-wide
screening of aberrantly-methylated CpG islands in thymoma and TC
using the Infinium® Human Methylation 450K BeadChip
(Illumina, Inc.), and identified 92 CpG islands that were
significantly hypermethylated in TC compared with thymoma.
Furthermore, four cancer-related genes (GHSR, GNG4, HOXD9
and SALL3) were selected, for which promoter methylation was
examined in 46 TETs and 20 paired thymic tissue sample using
bisulfite pyrosequencing. The results revealed that promoter
methylation of these genes was significantly higher in TC than in
thymoma and normal thymic tissue. In addition, relapse-free
survival (RFS) was significantly worse in tumors with higher DNA
methylation than for those with lower DNA methylation levels in all
TET samples (18).
GHSR is aberrantly hypermethylated in a
number of cancer types (including lung, breast, prostate,
pancreatic and colorectal cancer, as well as glioblastoma and B
cell chronic lymphocytic leukemia), and its methylation levels may
be used to discriminate between cancer and normal tissue (19). GHSR is a receptor of ghrelin that
regulates functional processes, such as hormone secretion, the
energy balance and gastric acid release (20). GHSR encodes a member of the G
protein-coupled receptor family that has two known transcripts,
native GHSR1a and variant GHSR1b. GHSR1a is the primary functional
receptor for the endogenous ligand ghrelin. The splice variant
GHSR1b is not activated by ghrelin or growth hormone secretagogues,
and thus, it currently not known whether the receptor is functional
(21). The splice variant of native
ghrelin, In-1 ghrelin may be acylated by ghrelin-O-acyltransferase
(GOAT) to facilitate binding with GHSR1a. Since it shares the
initial 13 amino acids with native ghrelin, In-1 ghrelin may also
activate GHSR1a (22).
The aim of the present study was to systematically
investigate the presence of different components of the ghrelin
system (native ghrelin, GOAT and GHSR1a) and splicing variants
(In-1 ghrelin and GHSR1b) in thymoma and TC, in order to compare
their expression with that in adjacent thymic samples and to
validate the results at the protein level using
immunohistochemistry (IHC). The association between the DNA
methylation of GHSR and the mRNA expression of ghrelin-GHSR
family members was also investigated.
Materials and methods
Patients and samples
The present study was conducted using a
retrospective, observational design. In total, 58 TET tissue
samples were obtained from patients with histologically confirmed
TET who underwent surgery at Tokushima University Hospital
(Tokushima, Japan) between January 1990 and January 2018 (median
age, 62 years; mean age, 59.86±11.87; range, 28–84 years). In
addition, 17 paired adjacent thymic tissue samples were obtained
from the patients. The patient characteristics are displayed in
Table SI. Tissue samples were
snap-frozen and stored at −80°C until the isolation of DNA and RNA.
Tumor specimens were characterized using the Masaoka-Koga staging
system (3) and WHO histological
classification (23) (Tables SII and SIII). The present study was approved by
the Ethics Committee of the University of Tokushima (Tokushima,
Japan; approval no. 3759), and was conducted according to the
Declaration of Helsinki. Written informed consent was obtained from
all patients.
RNA extraction
RNA was extracted from frozen tissue using the
RNeasy Mini kit (Qiagen GmbH) according to the manufacturer's
instructions. The quantity and purity of nucleic acid were assessed
using a spectrophotometer (NanoDrop-1000; Thermo Fisher Scientific,
Inc.) and diluted to 1.0 µg/μl; 1 µl RNA from each sample was used
for reverse transcription.
Reverse transcription-quantitative
(RT-q) PCR
Total RNA was reverse transcribed to cDNA using
iScript Reverse Transcription Supermix for RT-qPCR (Bio-Rad
Laboratories, Inc.), and qPCR was performed using Sso Advanced
Universal SYBR-Green Supermix (Bio-rad Laboratories, Inc.) and the
96-well format Applied Biosystems 7500 Real-Time PCR System (Thermo
Fisher Scientific, Inc.) according to the manufacturers'
instructions. Temperature and duration of reverse transcription
were as follows: Priming for 5 min at 25°C, reverse transcription
for 20 min at 46°C, and inactivation for 1 min at 95°C. The
thermocycling condition used for qPCR consisted of an initial step
at 95°C for 30 sec, followed by 40 cycles of denaturation (95°C for
15 sec) and annealing/extension (60°C for 1 min). The sequences of
the specific primers for ghrelin, In-1 ghrelin, GHSR1a, GHSR1b and
GOAT were obtained from a previous study by Luque et al
(24). The mRNA levels of β-actin
were used as an internal control for normalization. All primer
sequences for qPCR are shown in Table
SIV. The relative mRNA expression levels were calculated using
Human Thymus Total RNA (Takara Bio, Inc.) as a normal thymus
control. Relative gene expression was calculated using the
2−ΔΔCq method (25).
IHC
Formalin-fixed paraffin-embedded (FFPE) sections
(thickness, 3 µm) were stained using the Envision system (ChemMate
Envision kit; Dako; Agilent Technologies, Inc.) according to the
manufacturer's instructions. After dewaxing in xylene and
rehydration in a descending ethanol series, antigen retrieval was
performed by heating in Dako Real Target Retrieval Solution, pH 9
(Dako; Agilent Technologies, Inc.) at >120°C using a pressure
chamber (2100 Retriever; Aptum Biologics Ltd.) for 20 min. Tissue
samples were incubated overnight at 4°C with the following primary
antibodies: Rabbit Anti-GHS-R1A (1:500; cat. no. H-001-62; Phoenix
Pharmaceuticals, Inc.) and Rabbit Anti-GHS-R1B (1:400; cat. no.
H-001-61; Phoenix Pharmaceuticals, Inc.). The tissue samples were
incubated with ready-to-use peroxidase labelled polymer secondary
antibody (EnVision+ Dual Link System-HRP; cat. no. K4063; Dako;
Agilent Technologies, Inc.) at room temperature for 1 h. The
proportion and intensity of GHSR1a and GHSR1b staining were scored
independently by BT, KKo and SS. Staining intensity was scored as
follows: Negative, 0; weak, 1; moderate, 2; and strong, 3.
Proportion score was defined as follows: ≤30% positive cells, 0;
31–70% positive cells, 1; and ≥70% positive cells, 2. The staining
score was defined as the sum of the proportion and intensity
scores, and a score ≥4 indicated strong staining for the GHSR
proteins.
Statistical analysis
Descriptive results are expressed as the mean ±
standard deviation. The Shapiro-Wilk test was applied to assess the
normality of numerical datasets. When data were normally
distributed, expression between the tumor tissues was assessed
using an unpaired sample t-test, while differential expression
between tumor tissues and adjacent thymic tissues was assessed
using a paired sample t-test. Multiple comparisons were evaluated
using one-way ANOVA followed by Dunnett's post hoc test. When data
exhibited non-normal distribution, expression between the tumor
tissues was assessed using the Mann-Whitney U test, and
differential expression between tumor tissues and adjacent thymic
tissues was assessed using the Wilcoxon signed-rank test. Multiple
comparisons were assessed using the Kruskal-Wallis test followed by
Dunn's post hoc test. Methylation-dependent expression analysis and
gene expression correlation analysis were carried out using
Pearson's correlation coefficient when datasets were normally
distributed, while Spearman's rank correlation coefficient was
applied for non-normally distributed datasets. Categorical data
were assessed using Fisher's exact test. RFS analyses were
performed using the Kaplan-Meier method and compared with the
log-rank (Mantel-Cox) test. High- or low-expression groups were
assigned based on median values. Statistical analyses were
performed using GraphPad Prism 5.00 (GraphPad Software, Inc.) and
SPSS 25.0 (IBM Corp), and P<0.05 was considered to indicate a
statistically significant difference.
Results
Patient characteristics
In total, 58 patients (25 male and 33 female; mean
age 59.86±11.97 years) diagnosed with TETs were included in the
present study. Of these, 13 patients also had myasthenia gravis
(Table SI). According to the WHO
histological classification, 41 samples were thymoma, 13 were TC
(including a single case of TC combined with type B2 thymoma) and 4
were NECTT. Tumor characteristics are displayed in Tables SII and SIII.
In all TET cases (n=58), the median follow-up time
was 5.25 years (range, 0.36–22.41 years); 2 patients died from
their tumors, 1 from another disease and 1 from an unknown cause.
Furthermore, 18 patients experienced recurrence: Pleural
dissemination in 10, lung metastasis in 6, lymph node metastasis in
3 and multiple organ metastasis in 2 (Table SI). In thymoma cases (n=41), the
median follow-up time was 5.43 years (range, 0.75–22.41 years); 1
patient died from another disease and 1 from an unknown cause. In
addition, 7 patients experienced recurrence: Pleural dissemination
in 5 and lung metastasis in 2. Clinicopathological characteristics
of the patients and corresponding DNA methylation, RNA expression,
and protein expression levels are provided in Table SV.
Relative expression of ghrelin system
components in TETs and paired thymic tissue samples
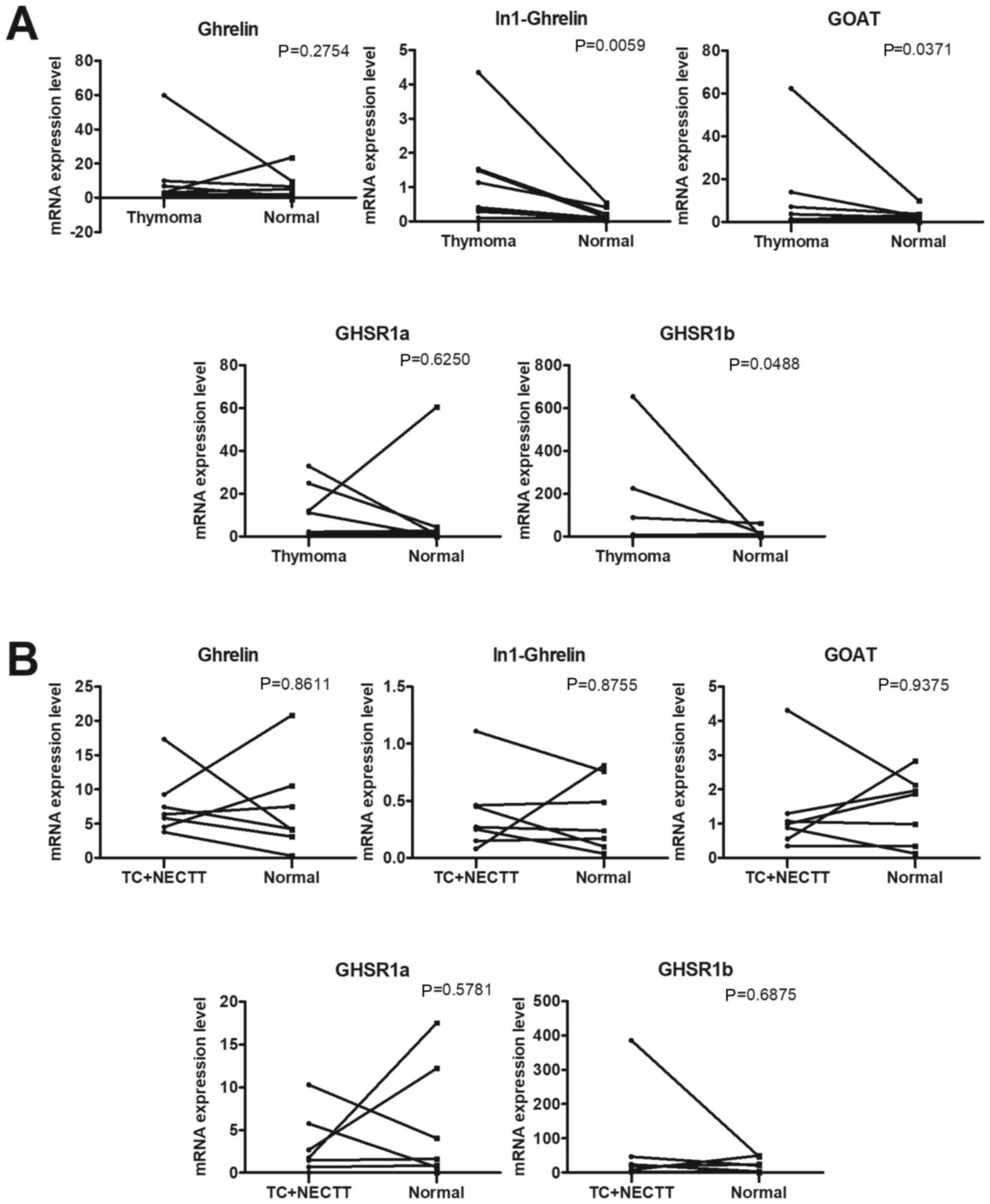
The mRNA expression of ghrelin system components in
thymoma (Fig. 1A) and TC (Fig. 1B) tissue was compared with that in
matched thymic tissue samples. The RT-qPCR results revealed that
expression of the splicing variant ligand In-1 ghrelin and variant
receptor GHSR1b were significantly higher in thymoma than in
adjacent thymic tissue samples (P=0.0059 and P=0.0488,
respectively; Wilcoxon signed-rank test). By contrast, no
significant differences were observed in the expression of the
native ligand ghrelin and native receptor GHSR1a between thymoma
and adjacent thymic tissue. The expression of the
ghrelin-activating enzyme GOAT was significantly higher in thymoma
tissue than in adjacent thymic tissue samples (P=0.0371; Wilcoxon
signed-rank test). On the other hand, no significant changes were
noted in the expression of five components of the ghrelin-GHSR
family in TC + NECTT (Fig. 1B).
Overall, the data indicate that In-1 ghrelin, GHSR1b and GOAT
expression were significantly higher in thymoma tissues than in
thymic-adjacent tissues.
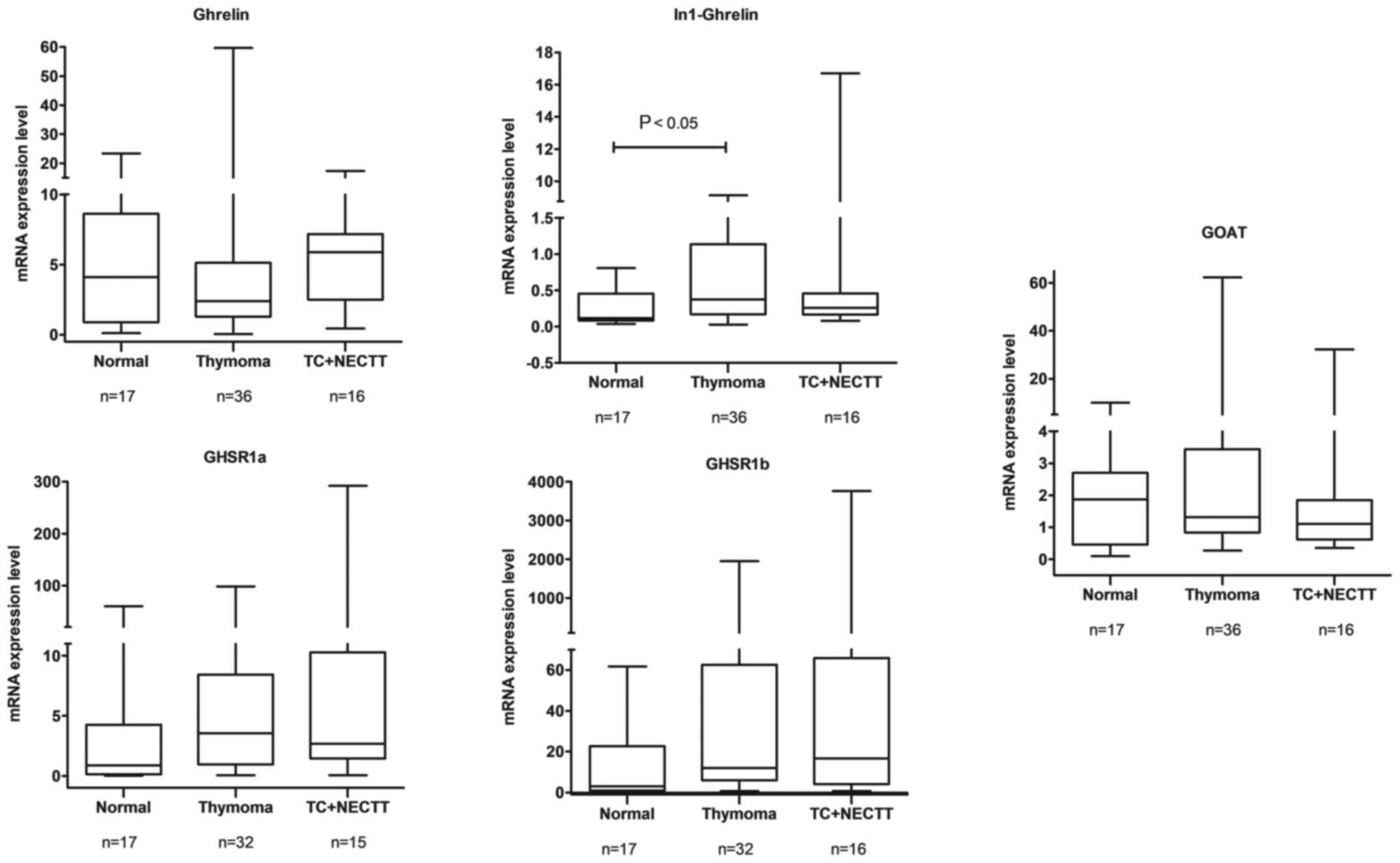
Expression of ghrelin system
components in the thymus, thymoma and TC + NECTT
The differential quantitative expression of ghrelin
family transcripts in the thymus, thymoma and TC + NECTT is
displayed in Fig. 2. The median
expression rates of In-1 ghrelin in the thymus, thymomas and TC +
NECTT were 0.120, 0.3750 and 0.260, respectively. The median
expression rate of the variant ligand In-1 ghrelin was
significantly higher in thymoma than in the thymus (P=0.0170;
Kruskal-Wallis test). The corresponding expression rates of the
variant receptor GHSR1b in the normal thymus, thymomas, and TC +
NECTT were 3.020, 11.93 and 16.67, respectively, and the median
expression rate of GHSR1b was slightly higher in thymoma than in
the thymus (P=0.0547; Kruskal-Wallis test). Furthermore, no
significant differences were observed in the expression of the
native ligand ghrelin, native receptor GHSR1a and GOAT. Therefore,
expression analysis of ghrelin system components in TETs indicated
that In-1 ghrelin was upregulated in thymoma tissues, but not in
TC. Also, there was a tendency for GHSR1b to be expressed at a
higher level in thymoma.
Expression of ghrelin system
components at different tumor stages
The expression of each of the five transcripts was
investigated at different tumor stages using the Masaoka-Koga
staging system. The stages were stratified into two distinct groups
[early (stage I+II) and advanced (stage III+IV)], and expression
was compared. The results indicated that the median expression
levels of In-1 ghrelin in the early and advanced stages of TET
were, respectively: i) 0.3650 and 0.3150 for In-1 ghrelin; ii)
3.045 and 4.370 for ghrelin; iii) 3.445 and 3.510 for GHSR1a; iv)
13.37 and 14.36 for GHSR1b; and v) 1.320 and 1.095 for GOAT. These
differences were not statistically significant (Fig. S1). Collectively, the data indicate
that the expression of ghrelin system constituents did not
significantly differ between TET stages.
RFS analysis of patients with TETs and
different mRNA levels of ghrelin, In-1 ghrelin, GHSR1a, GHSR1b and
GOAT
The levels of ghrelin, In-1 ghrelin, GHSR1a, GHSR1b
and GOAT were divided into high- or low-expression groups based on
their median values, and RFS analysis was conducted. Although no
significant differences were observed between the groups, there was
a tendency for tumors with slightly higher expression levels of the
variant ligand In-1 ghrelin, variant receptor GHSR1b and GOAT to be
associated with poor prognosis in patients with TETs (Fig. S2).
Comparative DNA methylation and gene
expression analysis
Methylation-dependent expression analysis was
performed to clarify whether the expression of ghrelin family
components was associated with the methylation of GHSR. Our
previous study reported the hypermethylation of GHSR in
thymoma and TC tissue relative to adjacent thymic tissue samples
(18). The present results showed
that mRNA expression of the variant ligand In-1 ghrelin and variant
receptor GHSR1b was positively associated with GHSR
methylation (ρ=0.4007 and ρ=0.4259, respectively; Spearman's rank
correlation coefficient) in thymoma tissue, whereas that of native
ghrelin, the native receptor GHSR1a and GOAT was not (Fig. S3A and Table I). Furthermore, the expression of
ghrelin, In-1 ghrelin, GHSR1a, GHSR1b and GOAT was not associated
with GHSR methylation in TC + NECTT tissue (Fig. S3B and Table I). Overall, these data indicate that
the expression of variant ligand In-1 ghrelin and variant receptor
GHSR1b is dependent on GHSR methylation in thymoma
tissues.
 | Table I.Association between GHSR methylation
and expression levels of ghrelin system components in thymic
epithelial tumors. |
Table I.
Association between GHSR methylation
and expression levels of ghrelin system components in thymic
epithelial tumors.
|
| Thymoma | TC + NECTT |
|---|
|
|
|
|
|---|
| Tumor Gene | P-value | ρ value | P-value | ρ value |
|---|
| Ghrelin | 0.5292 | −0.1321 | 0.9044 | −0.03396 |
| In1-Ghrelin | 0.0472a | 0.4007 | 0.4664 | 0.2038 |
| GHSR1a | 0.5007 | 0.1412 | 0.8168 | −0.06821 |
| GHSR1b | 0.0380a | 0.4259 | 0.7806 | 0.07864 |
| Ghrelin
O-acyltransferase | 0.2880 | 0.2212 | 0.5933 | 0.1501 |
Correlation analysis of gene
expression levels
The relationships among the quantitative mRNA
expression levels of each of the five transcripts of the ghrelin
system (ghrelin, In-1 ghrelin, GHSR1a, GHSR1b and GOAT) were
subsequently determined using Spearman's rank correlation
coefficient. The results revealed a positive correlation between
In-1 ghrelin/GOAT (ρ=0.6232) in thymoma tissue. Similarly,
correlations were observed between GHSR1b/GHSR1a as well as
GHSR1a/GOAT, with ρ=0.5158 and ρ=0.3944, respectively (Fig. S4 and Table IIA). In TC + NECTT tissue samples,
positive correlations were noted between In-1 ghrelin/GOAT,
GOAT/GHSR1b and GHSR1b/GHSR1a (ρ=0.6824, ρ=0.5676 and ρ=0.6571,
respectively) (Fig. S5; Table IIB).
 | Table II.Correlation analysis of the
expression levels of ghrelin system components, in thymoma, TC and
NECTT. |
Table II.
Correlation analysis of the
expression levels of ghrelin system components, in thymoma, TC and
NECTT.
| A, Thymoma |
|---|
|
|---|
| Transcript | Ghrelin | In1-ghrelin | GOAT | GHSR1a | GHSR1b |
|---|
| Ghrelin |
| −0.2146 | −0.2576 | −0.03045 | −0.07823 |
| In1-Ghrelin |
|
| 0.6232c | 0.2270 | 0.2518 |
| GOAT |
|
|
| 0.3944a | 0.2051 |
| GHSR1a |
|
|
|
| 0.5158b |
| GHSR1b |
|
|
|
|
|
|
| B, TC +
NECTT |
|
|
Transcript | Ghrelin |
In1-ghrelin | GOAT | GHSR1a | GHSR1b |
|
| Ghrelin |
| 0.3353 | 0.09706 | −0.2143 | −0.04118 |
| In1-Ghrelin |
|
| 0.6824b | −0.07143 | 0.3765 |
| GOAT |
|
|
| 0.1786 | 0.5676a |
| GHSR1a |
|
|
|
| 0.6571b |
| GHSR1b |
|
|
|
|
|
GHSR1a and GHSR1b protein expression
analysis
GHSR1a and GHSR1b protein expression was identified
in 20 TET tissue samples (including 14 thymoma and 6 TC tissue
samples) and 3 thymic tissue samples. The staining of both proteins
was cytoplasmic. Fig. 3A-a and B-a
shows GHSR1a and GHSR1b staining, respectively, in the cytoplasm of
epithelial cells but not lymphocytes in the normal thymus. Fig. 3A-b and B-b shows strong staining of
GHSR1a and moderate staining of GHSR1b, respectively, in type AB
thymoma. Fig. 3A-c and B-c shows
weak staining of GHSR1a and strong staining of GHSR1b,
respectively, in the type B2 thymoma. Fig. 3A-d and B-d shows strong staining of
GHSR1a and GHSR1b, respectively, in TC. The tissue samples were
grouped into weakly- and strongly-stained groups by their protein
immunoreactivity, and the mRNA expression levels were compared. The
mRNA expression levels of both GHSR1a and GHSR1b were higher in the
strongly-stained group (mean GHSR1a, 8.35±23.76; mean GHSR1b,
19.61±1381.5) than in the weakly-stained group (mean GHSR1a,
1.71±29.2; mean GHSR1b, 9.71±184.5) (Fig. 3C and Table III). Furthermore, the relationship
between the expression of GHSR1a and GHSR1b, and clinical findings
(sex, age, tumor stage, pathology type and myasthenia gravis), was
assessed. Table III shows that
variant GHSR1b protein was higher in TC (P=0.018; Fisher's exact
test) and in men (P=0.018; Fisher's exact test). GHSR1a protein
expression was not correlated with any of the assessed clinical
findings. In addition, RFS analysis indicated that high expression
of GHSR1b was not associated with prognosis (P=0.0624; Fig. S6).
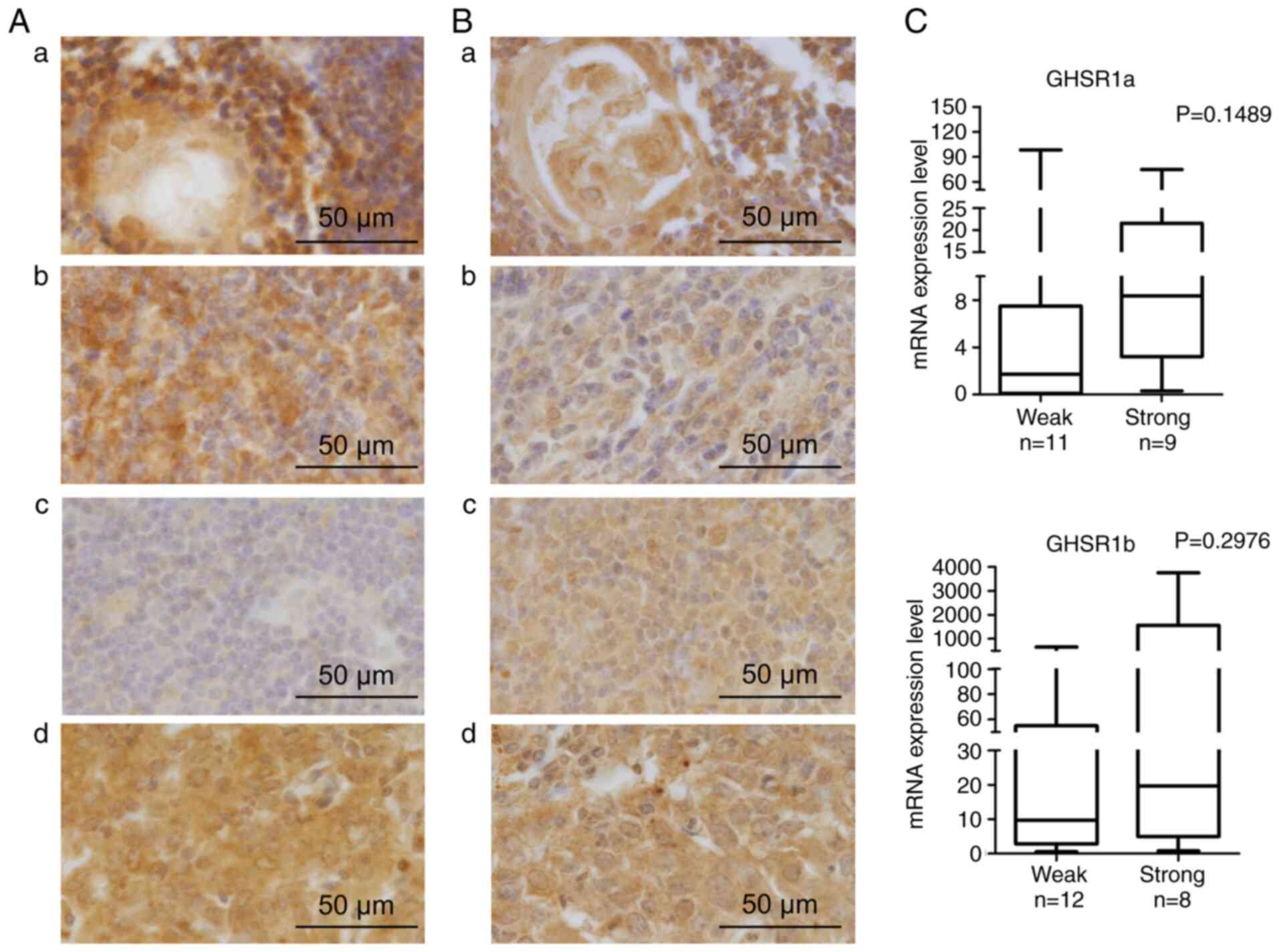 | Figure 3.Protein expression analysis. (A)
Representative images of GHSR1a protein expression. (a) Thymic
tissue: Dense aggregates of lymphocytes in the cortical area were
not stained; epithelial cells of medullary area were stained. (b)
Type AB thymoma: Cytoplasm of tumor cells was strongly stained, and
lymphocytes were not stained (intensity score, 3; proportion score,
2). (c) Type B2 thymoma: Cytoplasm of the tumor cells was not
stained (intensity score, 1; proportion score, 1). (d) TC:
Cytoplasm of the tumor cells was strongly stained (intensity score,
3; proportion score, 2). (B) Representative images of GHSR1b
protein expression. (a) Medullary area of thymus: Lymphocytes in
the medullary area were not stained, epithelial cells were stained.
(b) Type AB thymoma: Cytoplasm of the tumor cells was weakly
stained, and lymphocytes were not stained. (intensity score, 2;
proportion score, 1). (c) Type B2 thymoma: Cytoplasm of the tumor
cells was strongly stained (intensity score, 3; proportion score,
1). (d) TC: Cytoplasm of the tumor cells was strongly stained
(intensity score, 3; proportion score, 2). (C) Association between
GHSR protein expression and corresponding mRNA expression. Analyses
were performed using the Mann-Whitney U test. GHSR, growth hormone
secretagogue receptor; TC, thymic carcinoma. |
 | Table III.Association between GHSR1a and GHSR1b
immunoreactivity and clinicopathological characteristics in
patients with thymic epithelial tumors. |
Table III.
Association between GHSR1a and GHSR1b
immunoreactivity and clinicopathological characteristics in
patients with thymic epithelial tumors.
|
| GHSR1a
immunoreactivity (n=20) | GHSR1b
immunoreactivity (n=20) |
|---|
|
|
|
|
|---|
| Clinicopathological
characteristics | Weak (n=11) | Strong (n=9) | P-value | Weak (n=12) | Strong (n=8) | P-value |
|---|
| Sex,
female/male | 8/3 | 6/3 | 0.769 | 11/1 | 3/5 | 0.018 |
| Mean age ±SD,
years | 56.5±9.48 | 61±13.04 | 0.178 | 7/5 | 3/5 | 0.650 |
| Masaoka-Koga
staging system, stages I and II/III and IV | 10/1 | 6/3 | 0.285 | 11/1 | 5/3 | 0.255 |
| WHO histological
classification, thymoma/thymic carcinoma | 9/2 | 5/4 | 0.336 | 11/1 | 3/5 | 0.018a |
| Mean GHSR1a mRNA
expression ±SD | 1.71±29.2 | 8.35±23.76 | 0.1489 | NA | NA | NA |
| Mean GHSR1b mRNA
expression ±SD | NA | NA | NA | 9.71±184.5 | 19.61±1381.5 | 0.2976 |
| Myasthenia gravis,
positive/negative | 7/4 | 7/2 | 0.642 | 7/5 | 7/1 | 0.325 |
Discussion
Since the discovery of canonical ghrelin as an
intrinsic ligand for GHSR, research has been conducted to elucidate
its functions and pathophysiological roles. Though, the local
expression of ghrelin and GHSR has been examined in different types
of tumor tissue, the role of ghrelin as a proliferative or
inhibitory factor in cancers remains controversial (26–31).
Ghrelin has been shown to promote the proliferation of normal cell
lines, though its function in cancer cell lines is still being
debated. In in vitro studies, the ghrelin-GHSR axis promoted
cancer cell migration and metastasis through several different
downstream pathways (32–35). Diversified components of the
ghrelin-GHSR axis might show opposing effects in cancer progression
depending on the pathophysiological nature of the cancer type.
The current study aimed to comprehensively elucidate
the role of ghrelin components (the native ligand ghrelin, native
receptor GHRS1a, splicing variant ligand In-1 Ghrelin, variant
receptor GHSR1b and key enzyme GOAT) in TETs. The results
demonstrated that the variant ligand In-1 ghrelin and variant
receptor GHSR1b were significantly upregulated in thymoma tissue,
and no significant differences were observed in the expression of
native receptor ghrelin and native receptor GHSR1a between thymoma
and adjacent thymic tissue. On the other hand, in TC + NECTT, no
significant differences were noted in the expression of ghrelin
system components between tumor tissue and matched thymic tissue
samples. Supporting evidence prior to the present study indicated
that splicing variant In-1 ghrelin was significantly upregulated in
all types of pituitary adenoma tissue and gastroenteropancreatic
neuroendocrine tumors (36,24). Likewise, upregulation of In-1 ghrelin
in prostate and breast cancer was associated with the increased
aggressiveness of the disease (37–39).
GHSR1b was also found to be upregulated in hormone-related tumors,
such as colorectal cancer, breast cancer, adrenocortical tumors and
lung carcinoids (34,40–42).
Accumulating evidence shows that In-1 ghrelin and GHSR1b play as
oncogenic role in hormone-related tumors. Thymoma is a low-grade
malignant tumor of the thymic epithelium with immature T cell
migration associated with autoimmune diseases. However, TC is a
malignant tumor with evidently atypical cells of an invasive nature
without immature T cell migration and autoimmune disease (1,6).
Thymoma, but not TC, is a functioning tumor (6). In the present study, the variant ligand
In-1 ghrelin and variant receptor GHSR1b were upregulated in
thymoma, but not in TC tissue, which suggests that a switch in
expression to the variant ligand and receptor from the native
ligand and receptor of the ghrelin system contributes to the
tumorigenesis of thymoma, but not TC.
In the present study, the co-expression of GHSR1a
and GHSR1b was observed in thymoma and TC tissue samples. The
upregulated co-expression of GHSR1a and GHSR1b was detected in
growth hormone-secreting pituitary adenomas and
adrenocorticotrophic hormone pituitary adenomas. By contrast, the
expression of both receptors was significantly downregulated in
non-functioning pituitary adenomas (36). In a human erythroleukemic cell line
(HEL), the upregulation of GHSR1b was shown to stimulate cellular
proliferation via an autocrine pathway (43). Until recently, GHSR1b was considered
a non-functional orphan receptor due to the truncation process
during alternative splicing, where it loses its ability to bind
with its ligand ghrelin. Subsequent studies revealed that
overexpression of GHSR1b attenuated GHSR constitutive signaling by
heterodimerizing with GHSR1a (44).
In non-small cell lung cancer, GHSR1b was found to heterodimerize
with Neurotensin Receptor 1, which also belongs to the G-protein
coupled receptor superfamily, and to exert growth-promoting effects
by modulating the transcription of downstream target genes
(45).
In the present study, although the result was not
significant, there was a tendency towards higher expression of
variant ligand In-1 ghrelin, variant receptor GHSR1b and the
acylation enzyme GOAT in association with worse prognosis in
patients with TET than those with lower expression levels.
Rincón-Fernández et al (39)
reported that the high expression of In-1 ghrelin in breast cancer
samples was correlated with shorter RFS. Therefore, it may be
hypothesized that the expression of In-1 ghrelin and GHSR1b could
be used as a prognostic tool in thymoma. To confirm the mRNA
expression results of ghrelin, In-1 ghrelin, GHSR1a and GHSR1b, the
protein expression of these components in TETs and the thymus was
assessed using IHC. Although commercially-available antibodies
against GHSR1a and GHSR1b were used (which can discriminate between
each protein), distinct antibodies against ghrelin and In-1 ghrelin
could not be found. However, the mRNA expression levels of GHSR1a
and GHSR1b were higher in the strongly-stained group than in the
weakly-stained group. These results confirmed that mRNA expression
of GHSR1a and GHSR1b was roughly correlated with protein
expression. Variant GHSR1b protein expression was higher in TC and
in male patients. Thus the mRNA and protein expression of GHSR1b
may promote the malignant behavior of the tumor.
GOAT is a key enzyme for the acylation of ghrelin
(22). The present study revealed
that the expression of GOAT was significantly upregulated in
thymoma, but not in TC + NECTT tissue samples and a strong positive
correlation was observed between GOAT and In-1 ghrelin expression
in both thymoma and TC + NECTT tissue samples. However, the
expression of GOAT did not correlate with that of native ghrelin.
Similarly, GOAT was upregulated in NET and breast cancer tissue,
and its expression was associated with that of In-1 ghrelin.
Previous studies have reported increases in GOAT expression and its
plasma levels in patients with prostate cancer, which was
correlated with tumor aggressiveness (38,46,47).
Therefore, GOAT may act as a modulator of the elevated expression
of In-1 ghrelin in thymoma.
Epigenetic abnormalities, such as aberrant promoter
hypermethylation, associated with the functional disruption of
genes, are found in all types of tumor (48). Due to the tumor-initiating role of
this epigenetic dysregulation, methylation markers may be
preferable for diagnosing tumors at the earliest stage (49). Hypermethylation of GHSR has
been observed in numerous different tumor types, and is attracting
interest as a pan-cancer marker in clinical practice, with
promising sensitivity and specificity (19,50–54). Our
previous research demonstrated that GHSR was significantly
hypermethylated in thymoma and TC + NECTT tissue compared with
adjacent-healthy thymic tissue samples (18). A previous study by Coppedè et
al (55) examined the
methylation rate of CpG islands of 152-bp located in the
promoter/first exon region of GHSR in 65 surgically-resected
myasthenia gravis-positive thymoma cases, and 43 adjacent thymic
tissue samples, using the methylation-specific high-resolution
melting (MS-HRM) method. The study showed that GHSR
hypermethylation was observed in 18 thymomas (28%) compared with
the healthy thymus (mean, 5.1 vs. 0.2%), and concluded that
GHSR methylation was not a pan-cancer marker. This result is
in contrast to a previous study by Kishibuchi et al
(18), which examined the CpG sites
found downstream of the transcriptional start site of GHSR.
The result showed that all thymoma tissue samples were methylated,
and that methylation level was significantly higher than that in
the adjacent thymic tissue samples, irrespective of myasthenia
gravis status. The results of both aforementioned studies cannot be
easily compared due to the differences in methylation analysis
methodology (pyrosequencing vs. MS-HRM). Taking this into account,
we support the theory that GHSR hypermethylation is the
epigenetic driver to different types of malignancies, and a
promising candidate marker for pan-cancer detection.
The present study showed a positive correlation
between the methylation of GHSR and the expression of
variant ligand In-1 ghrelin and variant receptor GHSR1b in thymoma,
but not in TC tissue samples. Therefore, the DNA methylation of
GHSR may be associated with a shift from native expression
(ghrelin and GHSR1a) to variant expression (In-1 ghrelin and
GHSR1b) in thymoma.
There were some limitations to the present study,
which need to be addressed. TC and NECT case numbers were lower (13
for TC and 4 for NECTT) than those with thymoma because these
tumors are so rare. Due to the limited number of TET samples
collected at Tokushima University, the overall sample size could
not be increased. Thymoma can be divided into 5 types (types A, AB,
B1, B2 and B3) based on the morphology of epithelial cells and the
lymphocyte-to-epithelial cell ratio, and the ratio of lymphocytes
to tumor cells is high in AB, B1 and B2 thymoma (2). Since separate tumor cells could not be
separated from lymphocytes prior to RNA extraction, the presence of
lymphocytes in resected AB, B1 and B2 thymomas may have influenced
the mRNA expression rate. Thymoma is a low-grade malignancy; 58
patients with TET were followed up for 5.25 years (0.36–22.41
years), some of whom died; therefore, overall survival analysis
could not be performed.
In conclusion, our previous studies revealed that
GHSR was significantly hypermethylated in both thymoma and
TC + NECTT tissue samples. To elucidate the role of the
ghrelin-GHSR axis in TETs, the expression of five different
components of the ghrelin system was systematically analyzed in
tumor and normal tissue samples in the present study. The variant
ligand In-1 ghrelin, variant receptor GHSR1b, and acylation enzyme
GOAT were found to be upregulated in thymoma, but not in TC.
Furthermore, the mRNA expression of the variant ligand In-1 ghrelin
and variant receptor GHSR1b was positively associated with the DNA
methylation of GHSR in thymoma tissue. Therefore, the DNA
methylation of GHSR may be associated with a shift from
native expression (ghrelin and GHSR1a) to variant expression
(In-ghrelin and GHSR1b). Moreover, the slightly stronger expression
of In-1 ghrelin, GHSR1b and GOAT was associated with a worse
prognosis in patients with TET than those with weaker expression.
The aberrant DNA methylation of the receptor and strong expression
of the variant ligand and receptor may be related to the aggressive
behavior of thymoma.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported in part by
Grants-in-Aid for Scientific Research (grant no. 20K09178) from the
Ministry of Education, Culture, Sports, Science and Technology,
Japan.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author upon reasonable
request.
Authors' contributions
BT, KKo, HTa and AT conceived and designed the
study. BT, KKo, SS and KM performed all experiments and data
curation, and drafted the manuscript. MT and KKa analyzed and
interpretated the curated data. BT, KKo, SS, YK and NK analyzed and
interpreted patient gene expression data. BT, KKo, SS, HTo and MY
performed the immunohistochemical staining and interpreted the
association between the clinical data and immunoreactivity. All
authors have read and approved the final manuscript. BT and KKo
confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was performed in accordance with
the principles outlined in the Declaration of Helsinki. Following
the approval of all aspects of the present study by the local
Ethics Committee of Tokushima University Hospital (approval no.
3759), formal written consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Tateo V, Manuzzi L, De Giglio A, Parisi C,
Lamberti G, Campana D and Pantaleo MA: Immunobiology of thymic
epithelial tumors: Implications for immunotherapy with immune
checkpoint inhibitors. Int J Mol Sci. 21:90562020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Travis WD, Brambilla E, Nicholson AG,
Yatabe Y, Austin JH, Beasley MB, Chirieac LR, Dacic S, Duhig E, et
al: The 2015 World Health Organization Classification of Lung
Tumors: Impact of genetic, clinical and radiologic advances since
the 2004 classification. J Thorac Oncol. 10:1243–1260. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Detterbeck FC, Nicholson AG, Kondo K, Van
Schil P and Moran C: The Masaoka-Koga stage classification for
thymic malignancies: clarification and definition of terms. J
Thorac Oncol: Cancer. 6:S1710–1716. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lababede O and Meziane MA: The eighth
edition of TNM staging of lung cancer: Reference chart and
diagrams. Oncologist. 23:844–848. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kondo K: Therapy for thymic epithelial
tumors. Gen Thorac Cardiovasc Surg. 62:468–474. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shimosato Y, Mukai K and Matsuno Y: Tumors
of the mediastinum. AFIP Atlas of Tumor Pathology, Series 4. Armed
Forces Institute of Pathology; Washington, DC: 2010
|
|
7
|
Kondo K and Monden Y: Therapy for thymic
epithelial tumors: A clinical study of 1,320 patients from Japan.
Ann Thorac Surg. 76:878–885. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Berghmans T, Durieux V, Holbrechts S,
Jungels C, Lafitte JJ, Meert AP, Moretti L, Ocak S, Roelandts M, et
al: Systemic treatments for thymoma and thymic carcinoma: A
systematic review. Lung Cancer. 126:25–31. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Girard N, Ruffini E, Marx A, Faivre-Finn C
and Peters S: Thymic epithelial tumours: ESMO Clinical Practice
Guidelines for diagnosis, treatment and follow-up. Ann Oncol.
26:v40–v55. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Scorsetti M, Leo F, Trama A, D'Angelillo
R, Serpico D, Macerelli M, Zucali P, Gatta G and Garassino MC:
Thymoma and thymic carcinomas. Crit Rev Oncol Hematol. 99:332–350.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kelly RJ, Petrini I, Rajan A, Wang Y and
Giaccone G: Thymic malignancies: from clinical management to
targeted therapies. J Clin Oncol. 29:4820–4827. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Suzuki E, Sasaki H, Kawano O, Endo K,
Haneda H, Yukiue H, Kobayashi Y, Yano M and Fujii Y: Expression and
mutation statuses of epidermal growth factor receptor in thymic
epithelial tumors. Jpn J Clin Oncol. 36:351–356. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cimpean AM, Raica M, Encica S, Cornea R
and Bocan V: Immunohistochemical expression of vascular endothelial
growth factor A (VEGF), and its receptors (VEGFR1, 2) in normal and
pathologic conditions of the human thymus. Ann Anat. 190:238–245.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Girard N, Teruya-Feldstein J, Payabyab EC,
Riely GJ, Rusch VW, Kris MG and Zakowski MF: Insulin-like growth
factor-1 receptor expression in thymic malignancies. J Thorac
Oncol. 5:1439–1446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Weissferdt A, Fujimoto J, Kalhor N,
Rodriguez J, Bassett R, Wistuba II and Moran CA: Expression of PD-1
and PD-L1 in thymic epithelial neoplasms. Mod Pathol. 30:826–833.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zucali PA, De Pas T, Palmieri G, Favaretto
A, Chella A, Tiseo M, Caruso M, Simonelli M, Perrino M, et al:
Phase II study of everolimus in patients with thymoma and thymic
carcinoma previously treated with cisplatin-based chemotherapy. J
Clin Oncol. 36:342–349. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Thomas A, Rajan A, Berman A, Tomita Y,
Brzezniak C, Lee MJ, Lee S, Ling A, Spittler AJ, et al: Sunitinib
in patients with chemotherapy-refractory thymoma and thymic
carcinoma: an open-label phase 2 trial. Lancet Oncol. 16:177–186.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kishibuchi R, Kondo K, Soejima S, Tsuboi
M, Kajiura K, Kawakami Y, Kawakita N, Sawada T, Toba H, et al: DNA
methylation of GHSR, GNG4, HOXD9 and SALL3 is a common epigenetic
alteration in thymic carcinoma. Int J Oncol. 56:315–326.
2020.PubMed/NCBI
|
|
19
|
Moskalev EA, Jandaghi P, Fallah M,
Manoochehri M, Botla SK, Kolychev OV, Nikitin EA, Bubnov VV, von
Knebel Doeberitz M, et al: GHSR DNA hypermethylation is a common
epigenetic alteration of high diagnostic value in a broad spectrum
of cancers. Oncotarget. 6:4418–4427. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kojima M and Kangawa K: Ghrelin: Structure
and Function. Physiol Rev. 85:495–522. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Howard AD, Feighner SD, Cully DF, Arena
JP, Liberator PA, Rosenblum CI, Hamelin M, Hreniuk DL, Palyha OC,
et al: A receptor in pituitary and hypothalamus that functions in
growth hormone release. Science. 273:974–977. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Seim I, Herington AC and Chopin LK: New
insights into the molecular complexity of the ghrelin gene locus.
Cytokine Growth Factor Rev. 20:297–304. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Travis WD, Brambilla E, Burke AP, Marx A
and Nicholson AG: introduction to the 2015 World Health
Organization classification of tumors of the lung, pleura, thymus,
and heart. J Thorac Oncol. 10:1240–1242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Luque RM, Sampedro-Nuñez M, Gahete MD,
Ramos-Levi A, Ibáñez-Costa A, Rivero-Cortés E, Serrano-Somavilla A,
Adrados M, Culler MD, et al: In1-ghrelin, a splice variant of
ghrelin gene, is associated with the evolution and aggressiveness
of human neuroendocrine tumors: Evidence from clinical, cellular
and molecular parameters. Oncotarget. 6:19619–19633. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin T-C and Hsiao M: Ghrelin and cancer
progression. Biochim Biophys Acta Rev Cancer. 1868:51–57. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Volante M, Allia E, Fulcheri E, Cassoni P,
Ghigo E, Muccioli G and Papotti M: Ghrelin in fetal thyroid and
follicular tumors and cell lines: expression and effects on tumor
growth. Am J Pathol. 162:645–654. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cassoni P, Papotti M, Ghè C, Catapano F,
Sapino A, Graziani A, Deghenghi R, Reissmann T, Ghigo E, et al:
Identification, characterization, and biological activity of
specific receptors for natural (ghrelin) and synthetic growth
hormone secretagogues and analogs in human breast carcinomas and
cell lines. J Clin Endocrinol Metab. 86:1738–1745. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bai RX, Wang WP, Zhao PW and Li CB:
Ghrelin attenuates the growth of HO-8910 ovarian cancer cells
through the ERK pathway. Braz J Med Biol Res. 49:e50432016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Díaz-Lezama N, Hernández-Elvira M,
Sandoval A, Monroy A, Felix R and Monjaraz E: Ghrelin inhibits
proliferation and increases T-type Ca2+ channel
expression in PC-3 human prostate carcinoma cells. Biochem Biophys
Res Commun. 403:24–29. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cassoni P, Allia E, Marrocco T, Ghè C,
Ghigo E, Muccioli G and Papotti M: Ghrelin and cortistatin in lung
cancer: Expression of peptides and related receptors in human
primary tumors and in vitro effect on the H345 small cell carcinoma
cell line. J Endocrinol Invest. 29:781–790. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lin TC, Liu YP, Chan YC, Su CY, Lin YF,
Hsu SL, Yang CS and Hsiao M: Ghrelin promotes renal cell carcinoma
metastasis via Snail activation and is associated with poor
prognosis. J Pathol. 237:50–61. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dixit VD, Weeraratna AT, Yang H, Bertak D,
Cooper-Jenkins A, Riggins GJ, Eberhart CG and Taub DD: Ghrelin and
the growth hormone secretagogue receptor constitute a novel
autocrine pathway in astrocytoma motility. J Biol Chem.
281:16681–16690. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Waseem T, Javaid ur R, Ahmad F, Azam M and
Qureshi MA: Role of ghrelin axis in colorectal cancer: A novel
association. Peptides. 29:1369–1376. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Duxbury MS, Waseem T, Ito H, Robinson MK,
Zinner MJ, Ashley SW and Whang EE: Ghrelin promotes pancreatic
adenocarcinoma cellular proliferation and invasiveness. Biochem
Biophys Res Commun. 309:464–468. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ibáñez-Costa A, Gahete MD, Rivero-Cortés
E, Rincón-Fernández D, Nelson R, Beltrán M, de la Riva A, Japón MA,
Venegas-Moreno E, et al: In1-ghrelin splicing variant is
overexpressed in pituitary adenomas and increases their aggressive
features. Sci Rep. 5:87142015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hormaechea-Agulla D, Gahete MD,
Jiménez-Vacas JM, Gómez-Gómez E, Ibáñez-Costa A, L-López F,
Rivero-Cortés E, Sarmento-Cabral A, Valero-Rosa J, et al: The
oncogenic role of the In1-ghrelin splicing variant in prostate
cancer aggressiveness. Mol Cancer. 16:1462017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gahete MD, Córdoba-Chacón J,
Hergueta-Redondo M, Martínez-Fuentes AJ, Kineman RD, Moreno-Bueno
G, Luque RM and Castaño JP: A Novel Human Ghrelin Variant
(In1-Ghrelin) and Ghrelin-O-Acyltransferase Are Overexpressed in
Breast Cancer: Potential Pathophysiological Relevance. PLoS One.
6:e233022011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Rincón-Fernández D, Culler MD, Tsomaia N,
Moreno-Bueno G, Luque RM, Gahete MD and Castaño JP: In1-ghrelin
splicing variant is associated with reduced disease-free survival
of breast cancer patients and increases malignancy of breast cancer
cells lines. Carcinogenesis. 39:447–457. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jeffery PL, Murray RE, Yeh AH, McNamara
JF, Duncan RP, Francis GD, Herington AC and Chopin LK: Expression
and function of the ghrelin axis, including a novel preproghrelin
isoform, in human breast cancer tissues and cell lines. Endocr
Relat Cancer. 12:839–850. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Barzon L, Pacenti M, Masi G, Stefani AL,
Fincati K and Palù G: Loss of growth hormone secretagogue receptor
1a and overexpression of type 1b receptor transcripts in human
adrenocortical tumors. Oncology. 68:414–421. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Herrera-Martínez AD, Gahete MD,
Sánchez-Sánchez R, Salas RO, Serrano-Blanch R, Salvatierra Á,
Hofland LJ, Luque RM, Gálvez-Moreno MA, et al: The components of
somatostatin and ghrelin systems are altered in neuroendocrine lung
carcinoids and associated to clinical-histological features. Lung
Cancer. The components of somatostatin and ghrelin systems are
altered in neuroendocrine lung carcinoids and associated to
clinical-histological features. Lung Cancer. 109:128–136. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
De Vriese C, Grégoire Fo, De Neef P,
Robberecht P and Delporte C: Ghrelin is produced by the human
erythroleukemic HEL cell line and involved in an autocrine pathway
leading to cell proliferation. Endocrinology. 146:1514–1522. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Leung PK, Chow KB, Lau PN, Chu KM, Chan
CB, Cheng CH and Wise H: The truncated ghrelin receptor polypeptide
(GHS-R1b) acts as a dominant-negative mutant of the ghrelin
receptor. Cell Signal. 19:1011–1022. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Takahashi K, Furukawa C, Takano A,
Ishikawa N, Kato T, Hayama S, Suzuki C, Yasui W, Inai K, et al: The
neuromedin U-growth hormone secretagogue receptor 1b/neurotensin
receptor 1 oncogenic signaling pathway as a therapeutic target for
lung cancer. Cancer Res. 66:9408–9419. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gómez-Gómez E, Jiménez-Vacas JM,
Carrasco-Valiente J, Herrero-Aguayo V, Blanca-Pedregosa AM,
León-González AJ, Valero-Rosa J, Fernández-Rueda JL,
González-Serrano T, et al: Plasma ghrelin O-acyltransferase (GOAT)
enzyme levels: A novel non-invasive diagnosis tool for patients
with significant prostate cancer. J Cell Mol Med. 22:5688–5697.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Herrera-Martínez AD, Gahete MD,
Sánchez-Sánchez R, Alors-Perez E, Pedraza-Arevalo S, Serrano-Blanch
R, Martínez-Fuentes AJ, Gálvez-Moreno MA, Castaño JP, et al:
Ghrelin-O-Acyltransferase (GOAT) enzyme as a novel potential
biomarker in gastroenteropancreatic neuroendocrine tumors. Clin
Transl Gastroenterol. 9:1962018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jones PA and Baylin SB: The fundamental
role of epigenetic events in cancer. Nat Rev Genet. 3:415–428.
2002. View
Article : Google Scholar : PubMed/NCBI
|
|
49
|
Brena RM, Plass C and Costello JF: Mining
methylation for early detection of common cancers. PLoS Med.
3:e4792006. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ordway JM, Budiman MA, Korshunova Y,
Maloney RK, Bedell JA, Citek RW, Bacher B, Peterson S, Rohlfing T,
et al: Identification of Novel High-Frequency DNA Methylation
Changes in Breast Cancer. PLoS One. 2:e13142007. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Daugaard I, Dominguez D, Kjeldsen TE,
Kristensen LS, Hager H, Wojdacz TK and Hansen LL: Identification
and validation of candidate epigenetic biomarkers in lung
adenocarcinoma. Sci Rep. 6:358072016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Nakagawa T, Matsusaka K, Misawa K, Ota S,
Takane K, Fukuyo M, Rahmutulla B, Shinohara KI, Kunii N, et al:
Frequent promoter hypermethylation associated with human
papillomavirus infection in pharyngeal cancer. Cancer Lett.
407:21–31. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Verlaat W, Snijders PJF, Novianti PW,
Wilting SM, De Strooper LM, Trooskens G, Vandersmissen J, Van
Criekinge W, Wisman GB, et al: Genome-wide DNA methylation
profiling reveals methylation markers associated with 3q gain for
detection of cervical precancer and cancer. Clin Cancer Res:.
23:3813–3822. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Coppede F, Stoccoro A, Lazzarotti A,
Spisni R and Migliore L: Investigation of GHSR and GHRL methylation
in colorectal cancer. Epigenomics. 10:1525–1539. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Coppedè F, Stoccoro A, Nicolì V, Gallo R,
De Rosa A, Guida M, Maestri M, Lucchi M, Ricciardi R, et al:
Investigation of GHSR methylation levels in thymomas from patients
with Myasthenia Gravis. Gene. 752:1447742020. View Article : Google Scholar : PubMed/NCBI
|